Abstract
Patients with idiopathic pulmonary fibrosis (IPF) occasionally experience acute exacerbations after surgery for lung cancer. Several recent studies have revealed a prophylactic effect of perioperative pirfenidone treatment on postoperative acute exacerbations of IPF in patients with lung cancer. A 75-year-old woman consulted with her pulmonologist because of an IPF shadow detected by follow-up chest computed tomography 2 months after surgical treatment of biliary cancer. Another 7 months later, chest computed tomography showed a 23- × 14-mm nodule located in the right lower lobe with high accumulation of fluorodeoxyglucose detected by positron emission tomography, resulting in a radiological diagnosis of primary lung cancer with IPF. We administered perioperative pirfenidone treatment followed by right lower lobectomy using uniportal video-assisted thoracoscopic surgery after attaining a pathological diagnosis of adenocarcinoma. The patient developed no acute exacerbations of IPF during the postoperative period, and she had no recurrence of lung cancer for 15 months after surgery. We successfully used a combination of perioperative antifibrotic medication and minimally invasive surgery after lung cancer surgery in a patient with IPF.
Introduction
Idiopathic pulmonary fibrosis (IPF) is the most common type of idiopathic interstitial pneumonia, described pathologically as “usual interstitial pneumonia.” IPF frequently requires invasive treatment, including thoracic surgery for lung cancer, because patients with IPF have a 3.5- to 7.3-times higher risk of developing lung cancer.1–3 On its own, IPF can be fatal, with a median survival of 3 to 4 years after diagnosis. Life-threatening acute exacerbations of IPF (AE-IPF) reportedly occur in about 20% of patients with IPF who undergo surgery for lung cancer, resulting in a mortality rate of ≥50%.4–6 A retrospective nationwide investigation conducted by the Japanese Association for Chest Surgery revealed that among patients with IPF, AE-IPF occurred in 5.0% of those who underwent wedge resection, 10.5% of those who underwent segmentectomy or lobectomy, and 20.0% of those who underwent bi-lobectomy or pneumonectomy. 7 The gradual increase in the risk of AE according to the volume of lung removed should be brought to clinicians’ attention. The West Japan Oncology Group (WJOG) recently reported the safety and efficacy of perioperative pirfenidone, an oral antifibrotic agent that reduces AEs in patients with IPF after surgery for lung cancer. 8 We therefore adopted a perioperative pirfenidone protocol at our institution for patients with lung cancer and IPF who require pulmonary resection. We herein report the successful use of this perioperative pirfenidone protocol and minimally invasive uniportal video-assisted thoracoscopic surgery (U-VATS) for lobectomy and lymph node dissection in a patient with lung cancer and IPF. The patient experienced a quick and successful recovery without AE-IPF.
Case presentation
A 75-year-old woman with hypertension and IPF underwent cholecystectomy for carcinoma of the gallbladder. Follow-up chest computed tomography (CT) showed a shadow indicative of IPF in both the right and left lower lobes (Figure 1(a)). During follow-up with serial CT examinations, a nodule was detected adjacent to the IPF shadow in the right lower lobe (Figure 1(b)). This nodule gradually enlarged over 6 months (Figure 1(c), (d)). Fluorodeoxyglucose-positron emission tomography showed high accumulation of fluorodeoxyglucose in the nodule, with a maximum standardized uptake value of 5.67 (Figure 2); there were no other findings consistent with malignancy. Laboratory analysis revealed elevated concentrations of cytokeratin 19 fragment, carcinoembryonic antigen, sialylated carbohydrate antigen Krebs von den Lungen 6, pulmonary surfactant protein D, and pulmonary surfactant protein A (Table 1). A preoperative pulmonary function test showed a normal vital capacity of 2.01 L (percent predicted: 94.8%), a normal forced vital capacity of 2.06 L (percent predicted: 105%), and a normal forced expiratory volume in 1 second/forced vital capacity ratio of 83.5%. However, the patient’s carbon monoxide diffusing capacity was moderately reduced at 7.51 mL/minute/mmHg (percent predicted: 53.9%). The radiologic differential diagnoses of the nodule included a localized malignant tumor, such as primary lung cancer, or an oligo-metastasis of gallbladder carcinoma. Pirfenidone was orally administered at a dose of 600 mg/day for the first 2 weeks, followed by 1200 mg/day. Surgery was performed after at 2 weeks of 1200-mg/day administration.
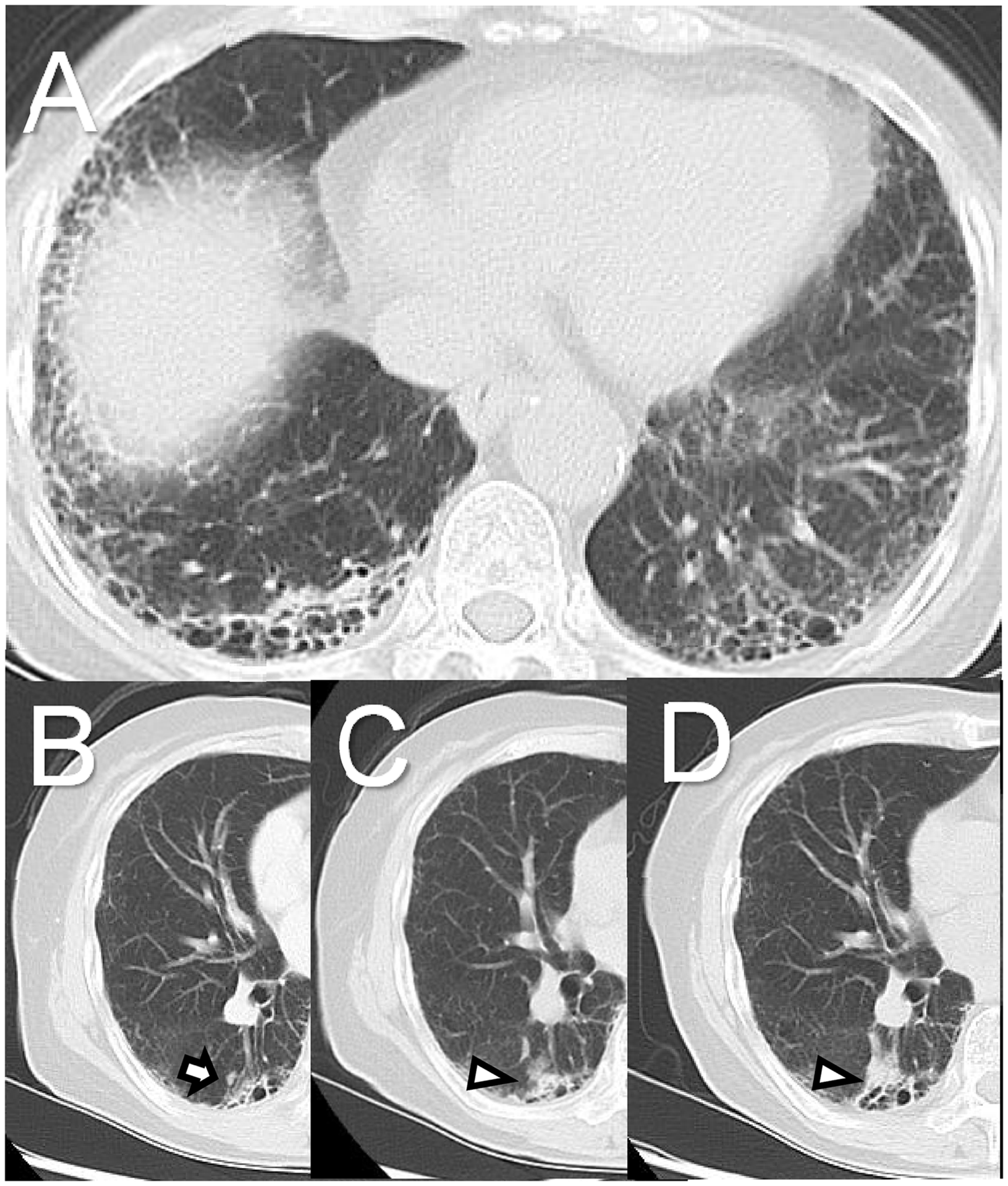
Preoperative chest computed tomography. (a) Subpleural- and basal-predominant honeycombing and reticulation were observed. (b) A tiny nodule was present adjacent to the fibrotic shadow in the right lower lobe (arrow). (c) Three months later, the nodule had enlarged (arrowhead). (d) A further 3 months later, the nodule had continued to enlarge and reached the convergence of a peripheral pulmonary artery and vein and the fibrotic area (arrowhead).

Preoperative fluorodeoxyglucose-positron emission tomography showed a nodule with high accumulation of fluorodeoxyglucose.
Preoperative laboratory analysis results.

*Elevated beyond the upper end of the reference range.
Under general anesthesia and using single-lung ventilation, we performed right lower lobectomy and lymph node dissection using U-VATS (Additional File Video 1). The patient’s postoperative course was excellent. Pirfenidone was restarted at 1200 mg/day on postoperative day 1 and continued until postoperative day 30. Pathologic examination of the resected specimen showed an area of adenocarcinoma measuring 30 mm, with an invasive component measuring 25 mm, located in segments 6, 9, and 10. The pathologic stage of the adenocarcinoma was T1cN0M0 Stage IA3 (Figure 3). A usual interstitial pneumonia pattern was noted in the background of the tumor (Figure 4). Fifteen months after U-VATS, chest CT showed no recurrence of the tumor and no evidence of AE-IPF.

Macroscopic and histopathologic findings of the resected specimen. (a) Adenocarcinoma is shown inside the red outline. (b, c) Adenocarcinoma, borderline atypical epithelium, and reactive hyperplasia were observed continuously (hematoxylin and eosin staining).

Histopathological examination of the resected specimen showed subpleural distribution of fibrosis and honeycombing in the right lower lobe.
Discussion
We successfully averted postoperative AE-IPF using perioperative pirfenidone administration 8 combined with minimally invasive but curative surgery to perform right lower lobectomy and group 2 lymph node dissection using U-VATS in a patient with lung cancer and IPF. The use of U-VATS potentially damages only a single intercostal region, and the use of a single small incision reportedly minimizes postoperative thoracic pain and maximizes the cosmetic advantage. The safety and efficacy of U-VATS compared with multiportal VATS have been confirmed by propensity-matched procedures, 9 prospective randomized trials, 10 and systematic reviews and meta-analyses. 11 Ye et al. 12 reported that U-VATS is less invasive than three-port VATS based on assessment of cytokines such as interleukin 6. Additionally, a meta-analysis by Harris et al. 13 demonstrated that U-VATS was associated with a statistically significantly shorter duration of chest tube drainage, shorter hospital stay, and lower overall morbidity than multiportal VATS.
Postoperative AE-IPF is often life-threatening. The Japanese Association for Chest Surgery performed a retrospective nationwide investigation, and the principal investigators Sato et al. 14 developed a risk calculation and scoring system for patients with interstitial pulmonary disease undergoing pulmonary resection for lung cancer. 14 They noted that AEs occurred in 164 (9.3%) of 1763 patients, with a mortality rate of 43.9%. These AEs were the leading cause of 30-day mortality. 7 A phase II clinical trial conducted by the WJOG (WJOG 6711L) showed that the perioperative use of pirfenidone, an antifibrotic agent, seems to prevent AE-IPF. 8 Moreover, a phase III clinical trial by North East Japan Study Group (NEJ034) is ongoing to confirm the preventive effect of the pirfenidone protocol against AE-IPF after lung cancer surgery. At present, the use of a minimally invasive procedure, such as U-VATS lobectomy with lymph node dissection, may be a good choice for patients with both lung cancer and IPF; however, the superiority of less invasive procedures on the preventive effect is not yet supported by evidence-based medicine. An accumulation of this evidence is necessary to clarify this issue.
In conclusion, our patient underwent U-VATS lobectomy and lymph node dissection for non-small cell lung cancer and successfully avoided AE-IPF with a perioperative pirfenidone protocol. Although the additional preventive effect of U-VATS with the pirfenidone protocol against the development of AE-IPF after lung cancer surgery has not been confirmed scientifically, minimally invasive surgery may be feasible and have a crucial role in lung cancer surgery for patients with IPF.
Footnotes
Ethics
The institutional ethics board waived the requirement for ethics approval because of the nature of this study (case report).
Declaration of conflicting interest
There is no conflict of interest.
Funding
This research received no specific grant from any funding agency.
Contributions
SA, TM, KN, and FI decided the treatment policy. IS performed the pathological diagnosis. SA, TM, KN, JT, and IS drafted the manuscript.
Availability of data and material
The data that support the findings of this study are available from the corresponding author.
Supplemental Material
Supplemental material is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
