Abstract
Background:
End-of-life care is a driver of increasing healthcare costs; however, palliative care interventions may significantly reduce these costs. Economic evaluations that measure the incremental cost per quality adjusted life years (QALY) are warranted to inform cost-effectiveness of the intervention relative to a comparator and permit evaluation of investment against other therapeutic interventions. Evidence from the literature up to 2011 indicates a scarcity of cost-utility studies in palliative care research.
Aim:
This literature review evaluates economic studies published between 2011 and 2019 to determine whether the methods of economic evaluations have evolved since 2011.
Design and Data Sources:
A literature search was completed using CENTRAL, OVID MEDLINE, EMBASE and other sources for publications between 2011 and 2019. Study characteristics, methodology and key findings of publications that met the inclusion criteria were reviewed. Quality of studies were assessed using indicators developed by authors of the previous literature review.
Results:
46 papers were included for qualitative synthesis. Among them only 6 studies conducted formal cost-effectiveness evaluations-of these 5 measured QALYs and 1 employed probabilistic analyses. In addition, with the exception of 1 costing analysis, all other economic evaluations undertook a healthcare payer perspective. Quality of evidence were comparable to the previous literature review published in 2011.
Conclusion:
Despite the small increase in the number of cost-utility studies, the methods of palliative care economic evaluations have not evolved significantly since 2011. More probabilistic cost-utility analyses of palliative care interventions from a societal perspective are necessary to truly evaluate the value for money.
Keywords
Introduction
Globally, end-of-life care is known to consume a large proportion of healthcare resources. In the United States, Medicare spends 27% to 30% of its total budget on care for people in their last year of life. 1 Likewise, estimates from the United Kingdom indicate approximately 20% of hospital bed days to be taken up by end-of-life care. 2 Canada is no exception to this observed trend with the cost of dying said to range from $10,000 to $40,000. This is largely due to an end-of-life care approach that focuses on curative treatments or disease focused interventions rather than symptom management. 1 Regardless, in Ontario (one of Canada’s most populous provinces) 51.9% of all decedents receive some form of palliative care in the last year of life. 3 While families bear substantial caregiving and out-of-pocket costs, a lot of these end-of-life costs are absorbed by the Canadian healthcare system. 1 As a result, the sustainability of publicly funded healthcare interventions, especially for terminally ill patients, has been under question.
Fortunately, there is evidence that indicates that palliative care services can significantly reduce the healthcare costs of end-of-life care through reduced hospital admission, length of stay, visits to intensive care unit (ICU) and inappropriate diagnostics or interventions.1,4 Palliative care seeks to prevent and treat the suffering of both patients with serious medical illness and their families through a multidisciplinary approach. Through services such as better symptom control, coordination of care, and improved communication between healthcare professionals and the patient and family, palliative care aims to enhance the end-of-life journey for terminally ill patients and their caregivers.5,6 A palliative care team may consist of specialty-trained clinicians, registered nurses, social workers chaplains and caregivers. The care can be provided in a variety of settings-hospital and home being the most popular ones. 5 Owing to ageing population, growing demands are being placed on palliative care services. However, due to the highly competitive funding environment, palliative care must compete with other therapeutic interventions for finite resources. Henceforth, policy makers have become increasingly interested in the cost associated with end-of-life care and the cost-effectiveness of palliative care. 5
Theoretically, economic evaluations in healthcare can be completed in one of 4 ways: cost-utility analysis, cost-effectiveness analysis, cost-benefit analysis or cost-minimization analysis.7,8 As recommended by several guidelines, cost-utility analysis is the gold standard for economic evaluations in healthcare decision-making. Its use of Quality-Adjusted-Life-Years (QALYs), which provides a common metric, permits comparative assessment of healthcare interventions across therapeutic areas. 9 However, the appropriateness of using QALYs as the outcome measure of palliative care effectiveness has been debated. QALYs take into account both the quality of life, and the quantity of life generated by the intervention. However, measuring quantity of life generated from palliative care in terminally ill patients is arguably incompatible. 9
Consequently, cost-benefit analysis has been popular in evaluating palliative care. 10 Literature suggests an imperfect overlap between what is measured in QALY scores and what is measured under resource utilization. For example, medical intervention can result in higher QALY score or lower utilization of non-healthcare resources such as leisure and caregiver time or lower utilization of healthcare resources such as ICU, hospitalization and emergency visits. Therefore, cost-benefit analysis has proven to be instrumental in evaluating the economic impact of palliative care by measuring both the benefits and costs of treatment in the same currency. 10 Though cost-benefit analysis is scrutinized for seemingly quantifying the value of human life in monetary terms, it does simplify the difficult nature of measuring the effectiveness of an intervention among terminally ill patients. 11 Subsequently, in the past, vast majority of studies evaluating palliative care has been comparative costing studies based on resource utilization.
A literature review published in 2014 identified only 1 formal cost-effectiveness evaluation among 46 economic evaluations published up to 2011 indicating the scarcity of cost-utility analyses in this therapeutic area. 6 Remaining available research consisted of aforementioned costing studies. The review revealed the use of relatively small observational studies and varied methodological approaches to assess the impact of palliative care interventions on costs separately from direct patient important outcomes. However, since then, there have been calls to include QALYs in the economic evaluation of palliative care.7,9,12 Despite criticisms of the limited gain in life years, end-of-life care can generate QALYs from changes in quality of life in the last days of life. 12 Therefore, in order to effectively allocate scarce resources across healthcare, cost-utility analysis is now the recommended method to evaluate the economic implications of palliative care interventions. Additionally, Smith et al (2014) reported direct costs, from the provider or third-party payer perspective, to be the focus of all 46 studies included in the review. Informal caregivers and out-of-pocket costs are central in the care for terminally ill individuals. Therefore, ignoring informal care in economic evaluations can bias cost-effectiveness research. 13
This paper will update the previous literature review of cost and cost-effectiveness of palliative care by assessing the quality of economic studies published between 2011 and 2019 and evaluating the evolution of methods since 2011 or the lack thereof.
Methods
The inclusion criteria for this review were patients (adult or children) that are terminally ill with any progressive life-limiting disease and is expected to have prognosis of less than 1-year survival. All variations, frequencies and administration (e.g. by clinician, by nurse) of hospice or palliative services in any setting (e.g. hospital-based, home-based) were included in this review. Palliative care comparators are disease focused and target the underlying illness. The term “usual care” is commonly used to capture this type of end-of-life care. Usual care involves acute therapies, and aggressive life-sustaining treatments (e.g. cardiopulmonary resuscitation, intubation and mechanical ventilation support). Studies that compare 2 palliative care services (e.g. early versus late or inpatient versus home-based) were also included in this review. Studies without a comparator were excluded. Costing studies, cost-effectiveness or cost benefit analyses were included. Studies that only looked at resource utilization without costing or clinical outcomes without costs were excluded. Randomized controlled trials, cohort studies (retrospective, prospective, and quasi), before and after studies and modeling studies were included in this review. Case studies, case series, study protocols, reviews, editorials, descriptive or qualitative studies, non-comparative quantitative studies and burden of illness studies were excluded.
Literature search of both electronic databases and grey literature were conducted up to August 31, 2019. Electronic search was completed using Cochrane Central Register of Controlled Trials (CENTRAL) and the OVID interface for MEDLINE Epub Ahead of print, In-Process & Other Non-Indexed Citations, Ovid MEDLINE (R) Daily and Ovid MEDLINE (R) (1946 to present) and EMBASE (1974 to 2019 August 31). The search strategy comprised of controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), EMTREE and keywords. The search was kept broad in order to capture all publications pertinent to the research question. The main search concepts grouped together were “end of life care” AND “cost/cost effectiveness.” See
A single reviewer independently screened the titles and abstracts of studies generated by the indicated search strategy. Duplicates were removed and studies were excluded based on following criteria: wrong population (e.g. not terminally ill, diabetes), wrong study type (e.g. qualitative, non-comparative), wrong intervention (e.g. radiotherapy, chemotherapy) and wrong outcomes (e.g. no costs). Corresponding abstracts that seemed eligible or required further evaluation was included for full-text review. Full texts were also screened independently to select studies that met the inclusion criteria. Using a standard data extraction form, the reviewer independently extracted data from the included studies which were randomly spot checked by a second reviewed. Only studies published between 2011 and 2019 were included in this literature review.
For the purpose of this review, the quality indicators reported in the literature review by Smith et al (2014) was used to assess the quality of the included studies. 6 This checklist was adapted from existing evaluation guidelines.14-19 Drawing on the Cochrane Handbook, Drummond’s guidelines for economic submissions, the CONSORT statement, and the Delphi list, a set of 31 indicators suitable for evaluating diverse set of papers was customized. Refer to publication by Smith et al. (2014) for complete details of the criteria. 6 Briefly, the 31 indicators cover core 6 categories: study description, sample selection and size, measurement, reporting, analysis and conclusion. Microsoft excel spreadsheet was used to evaluate the studies with 3 possible scores: 0 (poor), 2 (incomplete or unclear) or 4 (good). In cases where the indicators were not applicable, it was excluded from the final calculation. An overall mark, ranging from 0 (lowest quality) to 1 (highest quality) was calculated for each of the 6 core categories.
Results
Study Selection
A total of 3284 unique records were returned from the initial database search. After 936 duplicates were removed, a total of 2348 unique records were screened and 2187 were excluded. The full text of 161 papers were assessed and 46 papers were selected for qualitative analysis. Details of the screening process and reasons for exclusion are shown Figure 1.
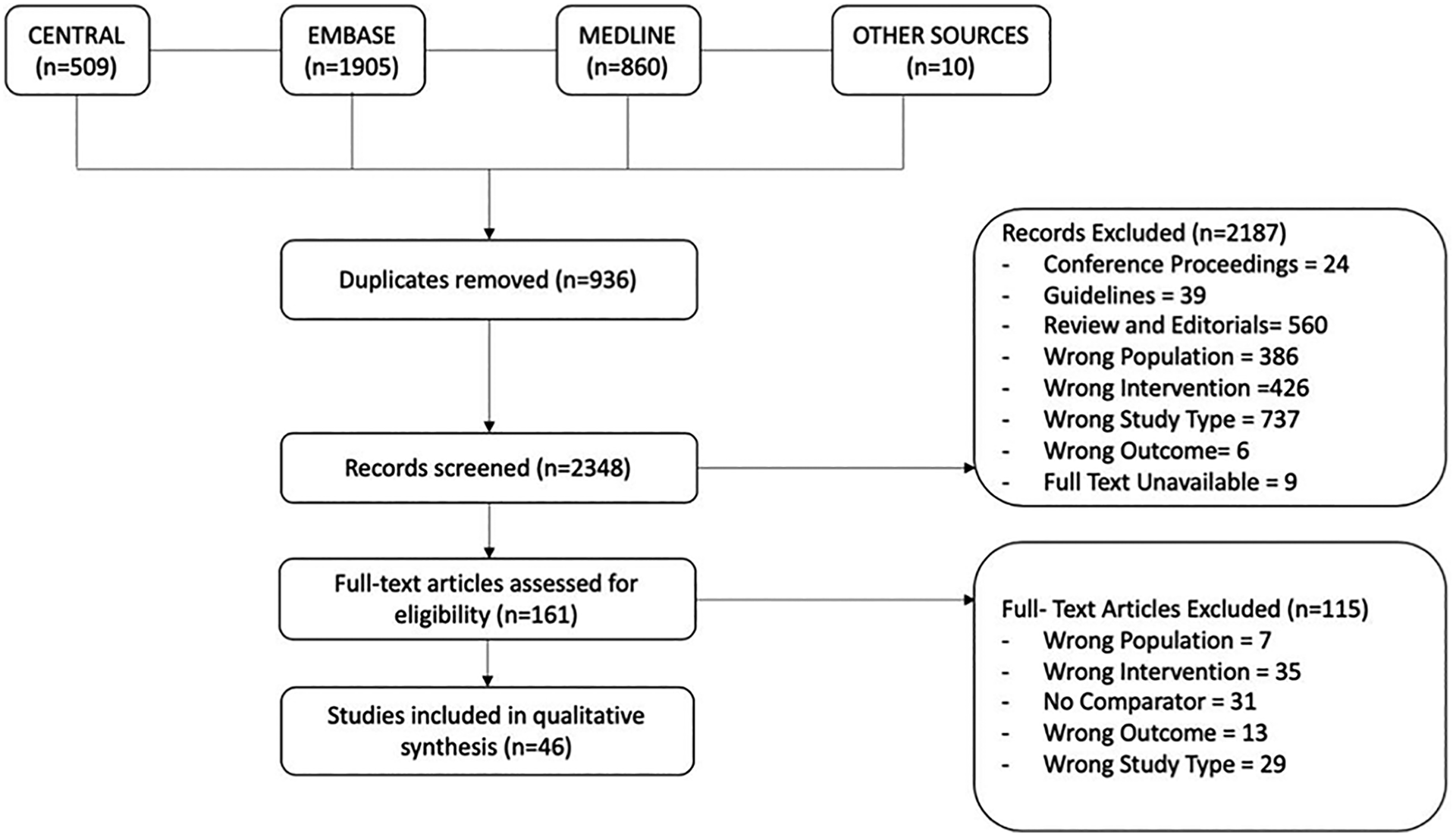
PRISMA flow diagram.
Study Characteristics
The key characteristics of the 46 included papers are outlined in Table 1.20-66 The publications can be categorized into 6 different study types-6 before and after studies, 2 cross sectional studies, 4 modeling studies, 6 prospective studies, 3 randomized controlled trials (RCTs), and 25 retrospective cohort studies. The studies were also assessed for primary progressive diagnoses. Palliative patients of all disease types were involved, but cancer, heart failure, dementia and end-stage renal and liver disease were the most common diagnoses among the patient population included in these economic evaluations.
Characteristics of Included Studies (n = 46).
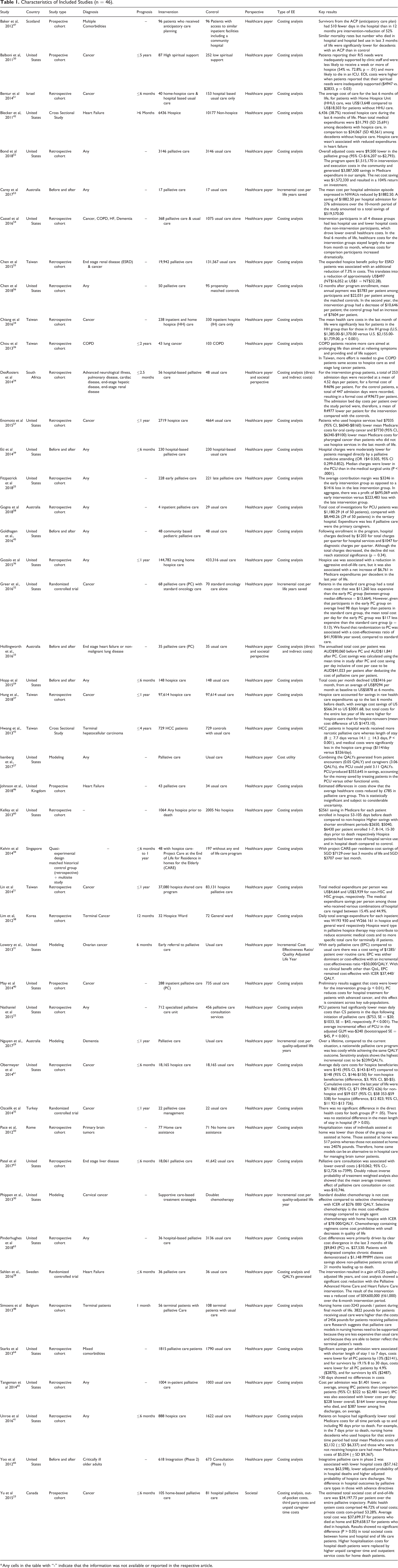
* Any cells in the table with “-” indicate that the information was not available or reported in the respective article.
The literature evaluates a variety of palliative care interventions including hospital-based, home-based, nursing home hospice care and community based palliative care relative to usual care. While most studies analyzed palliative care interventions against disease focused or acute care (termed “usual care”), 6 studies made comparisons across 2 palliative care interventions,20-25 such that either different types of care (e.g. palliative care unit versus consultation), different locations (e.g. hospital versus home) or different methods of administration (e.g. early versus late) were evaluated, and 1 study compared end of life care between lung cancer and chronic obstructive pulmonary disease patients. 26 Among the 46 included publications, all studies evaluated costs from a healthcare payer perspective with the exception of 1 study that evaluated the costs from a societal perspective. 23
Additionally, the studies were also characterized for the type of economic evaluation employed. While all the studies completed a costing, analysis based on resource utilization, only 6 studies met the criteria of a formal cost-effectiveness study. Of these, QALYs generated by palliative care were measured in 5 studies27-31 and 1 study reported the economic impact of palliative care in incremental cost per life years saved. 32 Of the 46 studies, all focused only on direct hospital costs, with the exception of 3.23,33,34 This includes, but is not limited to healthcare professional fees, procedure costs, imaging, diagnostic tests, laboratory tests and blood analysis, medication and length of stay in the hospital, emergency room or intensive care unit. In addition, 3 studies considered system-level costs such as overhead fees32-34 and 1 study included patient out-of-pocket and caregiver time. 23 Finally, the country in which the economic evaluations were based on is important to note as the health care system and associated fees differ across nations. The studies included in this literature review were predominantly based on data from the United States (25). The remaining studies were based on data from Australia (4), Taiwan (6), Canada (1), Israel (1), South Africa (1), Sweden (1), Turkey (1), United Kingdom (1), Scotland (1), Singapore (1), Korea (1), Rome, (1), and Belgium (1).
Study Quality
Cohort studies are often scrutinized for the increased risk of variations in demographic and clinical characteristics across study groups. However, it’s important to note that more than 60% of the included cohort studies used propensity score matching to control for selection bias. The 6 before and after studies were of moderate quality in all indicators except for the analysis section where the details of statistical analysis were poorly reported.
Costing Studies
Consistent with the previous literature review, key findings of 43 out of 46 studies indicate palliative care to have lower costs compared to usual care. However, ∼ 6% of reviewed studies suggest otherwise. A retrospective study from the United States that compared nursing home hospice care to usual care found hospice care to be associated with a reduction in aggressive end-of-life care, but it was also associated with a net increase of $6,761 in Medicare expenditures per patient in the last year of life. 36 Similarly, a retrospective study from Taiwan that evaluated hospice care relative to usual care found costs for the last year of life to be higher for hospice users than for those using usual care ($68,273.38 SD $54,115.67 vs. $66,800.28 SD $55,416.09; p = 0.989). This increase in cost was attributed to lengthy stay in hospice care. 37 Lastly, a study by Chou et al. (2013), found that the cost of hospice and non-hospice care for patients with COPD was $135, 483.50 and $117,530.00 respectively (p = 0.141) and for lung cancer patients the costs were $67,233.50 and $65,027.00 respectively (p = 0.764). With the exception of these 3 studies, all other costing studies found palliative care to be cost saving. Though not all studies report on statistical significance (p-value less than 0.05); 11 studies reported the observed cost savings to be statistically significant (p < 0.05)20,24,26,28,38-44 and 5 studies reported the observed cost savings to be statistically insignificant (p > 0.05).35,45-48 Across studies, the cost savings associated with palliative care interventions relative to usual care were largely driven by reduced resource utilization such as decreased hospital admissions and readmissions, reduced length of stay, and avoidance of aggressive treatments.
Cost Effectiveness Studies
Since the focus of this literature review is more on the use of QALYs in palliative care economic evaluations and the use of formal cost-effectiveness studies, key results from publications that met this criterion were assessed more closely. Amongst the 46 studies, there were 6 formal cost effectiveness studies of varying quality (Table 2).
Formal Cost-Effectiveness Studies (n = 6).

Perspective of Economic Evaluation
In addition to the type of economic evaluations completed, the perspective under which the intervention was evaluated was also of interest to this literature review. All the studies comparing palliative care to usual care were conducted from a healthcare payer perspective with the exception of 1 cohort study evaluating home-based palliative care against hospital-based palliative which was completed with a societal perspective. 23 This costing analysis estimated the total societal cost per patient over 6 months of palliative care to be $34,197.73 CAD. 46.72% of the cost was attributed to public health system costs and 53.28% was out-of-pocket costs. The average total cost of home-based and hospital-based palliative care showed no statistically significant difference (p > 0.05). Higher hospitalization costs for hospital-based palliative care patients were neutralized by the high unpaid caregiver time and outpatient service costs among home-based palliative care patients. 23 A similar study that evaluated inpatient hospice care and home hospice care reported significantly less costs in the home-hospice group ($1,385-$1,370 USD versus $2,155-$1,739, p < 0.001). 24 However, this was expected as the costing analysis was conducted from a healthcare payer perspective.
Discussion
Main Findings
In the previously published literature review by Smith et al. (2014), there were 5 RCTs, 2 prospective cohort studies, 34 retrospective cohort studies, and 3 before and after studies. 6 The distribution of study types has not evolved much since 2011. For example, the majority of included studies (54%) were still retrospective cohort studies. However, up until 2011, there was only 1 formal cost-effectiveness study conducted to evaluate palliative care interventions. 49 Though patient and caregiver outcomes were measured on Palliative Care Outcome Scale and Zarit Carer Burden Inventory, respectively; the results from that 1 cost-effectiveness study were not presented in QALYs. Between 2011 and 2019, there have been 6 cost-effectiveness studies evaluating the value for money of palliative care interventions. Among them, only 5 studies measure the effect of the intervention in QALYs and only 4 of the 5 performed a sensitivity analysis (Table 2). Amongst the studies that investigated cost-effectiveness only 1 study evaluated the uncertainty in evidence using probabilistic analyses as recommend by some guidelines (UK and Canada). In addition, it is important to note that all 5 studies that evaluated cost-effectiveness in QALYs found palliative care interventions to be less expensive than standard care. In contrast, the study by Greer et al. found palliative care to be more cost effective unless it was in the patient’s final 30 days of life. 32
The quality of costing studies is also varied. Even though most cohort studies do undertake propensity score matching to decrease the risk of selection bias, this is not a significant evolution in methods since 2011. As Smith et al (2014) also reports that majority of the cohort studies in their review also controls for selection bias this way. Finally, a key finding of the previous literature review was the lack of focus on informal care or out-of-pocket costs. Since palliative care impacts patients as well as caregivers, societal perspective is of high value in the economic evaluation of this intervention. However, since 2011, there has only been 1 study that evaluated palliative care from a societal perspective which contrary to the majority of economic evaluations from a healthcare payer perspective did not find that palliative care significantly reduced costs compared to usual care. 23 Regardless, when discounting study type and methods, most economic evaluations consistently found palliative care to be associated with lower costs relative to a comparator group. However, further investigation from a societal perspective is required to capture all relevant costs and consequences.
Limitations
This was a comprehensive review of the literature that investigated the financial implications of palliative care relative to a comparator. While the review does summarize the key findings of economic evaluations since 2011, it was rather designed to address the change in characteristics and methods of economic evaluations. Therefore, this review was limited to the defined scope of work as an assessment that focuses on the evolution of economic evaluation methods since 2011. Consequently, a meta-analysis of cost and outcomes from the literature was not completed.
In addition, a limitation of a literature review of this nature is the availability of these studies using the methods outlined in the methodology. Specifically, 9 articles were excluded because they did not have a full-text available. It is possible that these studies could have influenced the overall results.
Lastly, the quality of evidence was assessed using a tool adapted from various evaluations guidelines. Although they are drawn from credible guidelines, the 6 core categories and the 31 indicators within them is one of many approaches that could’ve been taken to assess the quality of evidence. Since this review aimed to update the literature review by Smith et al. (2014), the included studies were evaluated using the same quality assessment tool developed by Smith and colleagues. However, taking a different approach to appraising the quality of evidence may have resulted in a more precise assessment as the used tool only allowed 3 possible scores (0, 2 and 4). Moreover, the quality of the papers was judged by a single individual. Although measures were taken to minimize interpretation bias by having randomized checks completed by a second reviewer, a revision of quality assessment with a panel of reviewers will strengthen the study results and ensure consistency.
What This Study Adds
As for future directions, more formal cost-effectiveness studies are warranted in evaluating the economic impact of palliative care interventions. Though there have been 6 studies completed since 2011, the generalizability of results is limited due to the variability in healthcare reimbursement models in different countries. Additionally, 5 of the 6 cost-utility studies did not employ probabilistic analyses. Utilization of probabilistic distribution in modeling studies and bootstrap regression models in trial-based economic evaluations can significantly enhance the quality of economic analysis and meet the current guidelines of pharmacoeconomic evaluations.
Lastly, 2 studies that compared home hospice care to hospital-based hospice care provided a perfect example of how different the results of an economic evaluation can be depending on the perspective in which the analysis is completed.23,50 While the evaluation from the payer perspective found cost of care to be significantly less for home-based palliative care group, the evaluation from societal perspective found no significant difference in cost of care between hospital and home-based hospice care. Given the vital role informal caregivers play in the end-of-life care of terminally ill patients, future economic evaluation must consider both direct and indirect cost and QALYs.
Conclusion
Economic evaluation of palliative care is challenging but also a necessity given the disproportional share of healthcare expenditure for patients at the end-of-life. While the use of QALYs in measuring outcomes in palliative care has been debated in the past, recent guidelines for pharmacoeconomic evaluations calls for the use of cost-utility analysis. For that reason, a literature review was completed to examine whether methods of palliative care economic evaluations have evolved since the deficiency in cost-utility analysis was last reported in 2011. Despite the small increase in cost-utility studies since 2011, the methods of palliative care economic evaluations have not evolved significantly. Aligned with the guidelines for the economic evaluation of healthcare programs in several countries (e.g. Canada, UK and the US), researchers are encouraged to compare palliative care interventions in terms of costs and QALYs from a payer (e.g. Ministry of Health) or a societal (e.g. also taking into account productivity losses or out of pocket expenditures) perspective.67,68 To truly evaluate value for money and improve decision making, it is also very important that researchers take into account in their analyses the different types of uncertainty inherent to the economic evaluation according to best practices (e.g. sampling uncertainty for trial-based economic evaluations, parameter uncertainty when conducting model-based economic evaluations).53-55,67,69-71 Finally, researchers should follow reporting guidelines for economic evaluations of healthcare programs. 72
Supplemental Material
Supplemental Material, sj-pdf-1-ajh-10.1177_10499091211011138 - Economic Evaluation of Palliative Care Interventions: A Review of the Evolution of Methods From 2011 to 2019
Supplemental Material, sj-pdf-1-ajh-10.1177_10499091211011138 for Economic Evaluation of Palliative Care Interventions: A Review of the Evolution of Methods From 2011 to 2019 by Anna Parackal, Karishini Ramamoorthi and Jean-Eric Tarride in American Journal of Hospice and Palliative Medicine®
Supplemental Material
Supplemental Material, sj-pdf-2-ajh-10.1177_10499091211011138 - Economic Evaluation of Palliative Care Interventions: A Review of the Evolution of Methods From 2011 to 2019
Supplemental Material, sj-pdf-2-ajh-10.1177_10499091211011138 for Economic Evaluation of Palliative Care Interventions: A Review of the Evolution of Methods From 2011 to 2019 by Anna Parackal, Karishini Ramamoorthi and Jean-Eric Tarride in American Journal of Hospice and Palliative Medicine®
Footnotes
Authors’ Note
Concept: Parackal, Tarride. Interpretation and analysis of data: All authors. Drafting of manuscript: Parackal, Ramamoorthi. Critical revision of manuscript for important intellectual content: All authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
