Abstract
During the COVID-19 pandemic, there were months of delay in deploying community pharmacies for the National COVID-19 Vaccination Programme in Ireland. We aimed to explore what may have accounted for this delay between 15th December 2020 (publication of policies) and 14th June 2021 (commencement of community pharmacy–based vaccination). We carried out a multi-method qualitative case study that involved engaging with 11 stakeholders, reviewing 246 documents, and conducting semi-structured interviews with 11 policy elites. Using reflexive thematic analysis, we developed three themes. The first provides evidence that the delay was, in part, due to operational barriers related to the logistical and safety aspects of the Programme. The second, on the other hand, presents a perspective that the delay was unreasonable as it was based on perceived deficits in community pharmacies. Finally, the third highlights the inability of the pharmacy profession to influence health policy due to a lack of strategic and cohesive leadership and the dominance of the medical profession. Overall, we argue that the delay can be explained by a complex interplay between technical, socio-political, institutional, and regulatory factors, underpinned by a chronic lack of strategic direction for pharmacy in the Irish health system.
Keywords
Introduction
During the COVID-19 pandemic, continual calls were made by pharmacy agencies and pharmacists globally to involve community pharmacies (CPs) in COVID-19 vaccination programmes to increase coverage (Costa et al., 2022; Maidment et al., 2021; Paudyal, Fialová, et al., 2021; Sousa Pinto et al., 2021). Indeed, global evidence showed that CPs had an extensive vaccination track record (FIP, 2016; MacDougall et al., 2016), experience in dealing with previous pandemics (Schwerzmann et al., 2017), and the capability to deal with the increased healthcare demand posed by COVID-19 (Cadogan & Hughes, 2021; Costa et al., 2022).
CPs were swiftly leveraged as COVID-19 vaccination outlets within the first quarter of 2021 in numerous high-income countries, including Canada, France, Italy, Switzerland, the United Kingdom, and the United States (BCPHA, 2021; Costantino et al., 2022; PGEU, 2021; Piraux et al., 2022; Stämpfli et al., 2021). For instance, CPs in the United Kingdom started delivering COVID-19 vaccination in January 2021 (PGEU, 2021) and over 1.7 million vaccines were already administered by March 2021 (Wickware, 2021). CPs in many of the European countries, including France, Switzerland and the United Kingdom, had already been involved in vaccination programmes (e.g., influenza), prior to the COVID-19 pandemic (FIP, 2016; PGEU, 2021). Thus, the pre-existing infrastructure may have been a contributory factor to the swift introduction of COVID-19 vaccination.
In Ireland, however, there were months of delay before CPs were engaged as outlets for COVID-19 vaccination. This is puzzling as CPs in Ireland have been providing influenza vaccination since the winter of 2011/12, and the service was subsequently expanded to include herpes zoster and pneumococcal polysaccharide vaccines (Henman, 2020).
The National COVID-19 Vaccination Programme (NCVP) policies published in December 2020 outlined the vaccination role of CPs (DoH, 2021a, 2021b). Nevertheless, they were only activated in June 2021 upon campaigning by the profession and after mass vaccination centres (MVCs) and general practices (GPs) which were the other planned vaccination outlets, had already been made operational (Murphy & Heaney, 2021). This article seeks to explore what accounted for the delay in involving CPs in the NCVP in Ireland between 15th December 2020 (publication of the NCVP policies) and 14th June 2021 (commencement of CP-based COVID-19 vaccination).
Drawing on stakeholder engagement, documentary analysis, and elite interviews, this multi-method qualitative case study brings to light a highly technical yet politicised implementation process. Situating the polarised perspectives of interviewees that attributed the delay to either rational or irrational decision-making, this analysis shows that the delayed implementation was underpinned by the persistent structural marginalisation of pharmacy in the Irish health policy landscape.
In the following section, we summarise some key findings of the literature on health policy implementation. This is followed by an overview of our research methodology. Subsequently, we present the case study problem and empirical analysis, while the final section provides a discussion of the analysis and conclusions.
Background
Policy implementation concerns “what develops between the establishment of an apparent intention on the part of [the] government to do something, or to stop doing something, and the ultimate impact in the world of action” (O’Toole, 2000, p. 266). Policy implementation is far from a purely technical and apolitical process. It is shaped by “top-down” factors (for example, government capacity, political drivers, and resource availability), and “bottom-up” factors (for example, the ideas, interests, and influences of actors involved in implementation) (Buse et al., 2023; Campos & Reich, 2019). Successful implementation requires political leadership and careful management of the stakeholders involved, including interest groups, public officials and ministries of finance. Unsurprisingly, due to its complexity and political nature, policy implementation gaps are common globally (Hudson et al., 2019).
Most health policy studies focus on technical issues with far fewer analyses dedicated to health politics (Erasmus et al., 2014). The few existing studies on health policy implementation in Ireland suggest that implementation gaps are the result of both top-down and bottom-up factors, particularly political and stakeholder leadership, financial and technical concerns, and institutional path dependence (the idea that decisions are constrained by previously established institutional structures and policies [Wilsford, 1994]).
The implementation of health system reform projects toward universal care in Ireland, including the 10-year Sláintecare programme produced by a cross-party parliamentary committee in 2017, has failed repeatedly due to poor planning, competing healthcare interests, a lack of political accountability, and resistance from stakeholders who benefit from the status quo (Connolly, 2023). A case study of the implementation of the 2001 Primary Care policy in Ireland found that the gap between policy formulation and implementation can be attributed to a lack of political will and perceptions of resource availability and system capability (Kelly et al., 2016). The absence of dentistry leadership in the health system and stakeholder engagement also led to failures in implementing oral health policies (McAuliffe et al., 2022). Destabilising crises can present windows of opportunity for health system changes (Asensio & Popic, 2019; Mauro & Giancotti, 2021; Pierson, 1996). For instance, COVID-19 accelerated certain elements of the Sláintecare programme, such as the introduction of independent health identifiers (Burke et al., 2021). However, crises can also stimulate reform efforts without ensuring their implementation. For example, the introduction of a proposed universal health insurance system including free GP care in Ireland in the aftermath of the Great Recession (2011–2016) was stymied by competing political ideologies, financial concerns, and the perceived administrative unviability of the plan (Devitt, 2022).
Research exploring the policy processes, including implementation, shaping the practice of (community) pharmacy is lamentably scarce internationally (Traulsen & Almarsdóttir, 2005). This research lacuna is surprising given that CPs are influenced by complex political, legislative, economic, and historical factors. In Ireland, CPs have been facing commercial precarity induced by austerity-related funding cuts (IPU, 2017, 2018), delayed contract negotiations with the government for a new service and remuneration model (IPU, 2022b), an overly stringent regulatory regime (Lynch et al., 2022; Lynch & Kodate, 2020), the lack of a pharmacy professional body and senior management level representation in the health system (Lynch, 2014; O’Sullivan & McCarthy, 2022), professional rivalries with GPs (IMO, 2015), and workforce retention issues (IPU, 2019, 2022c). Some of these challenges are shared by pharmacists in other countries. For instance, while pharmacists in the United Kingdom are represented by a professional body and chief pharmacists, they similarly face issues around professional rivalries, financial pressures, and workforce shortages (Andalo, 2017; Connelly, 2023; CPE, 2024).
Regarding the role of CPs in vaccination programmes, evidence suggests that political, regulatory, and resource hindrances may prevent the participation of CPs in vaccination programmes, despite clear benefits for population health (Burson et al., 2016; Paudyal, Fialová, et al., 2021). To date, no research has explored the delayed policy implementation of the COVID-19 vaccination role of CPs in Ireland.
Methodology
To understand the delayed involvement of CPs in the NCVP, we conducted a retrospective health policy analysis. A multi-method qualitative case study was undertaken, involving stakeholder engagement, documentary analysis, and semi-structured elite interviews (Sheikh et al., 2011; Yin, 2009).
We situated our research in the Big Q qualitative paradigm, which privileges both qualitative research techniques and values (Madill & Gough, 2008). We were guided by the philosophy of critical realism, which postulates the existence of a mind-independent reality while recognising that socio-cultural contexts shape our understanding of it (Bhaskar, 1975, 1979). This informed our research design, which emphasised reflexivity and drew on multiple qualitative methods to interrogate the research question from different angles. Regarding the former, the lead researcher AK, a pharmacist qualified in Ireland and a researcher with an interdisciplinary background in public health and sociology, kept a reflexive journal (Supplemental Material A) to explore how his social positions, influenced the research process (Braun & Clarke, 2022).
Our work was guided by stakeholder engagement to ensure research relevance and enhance rigour (Campus Engage, 2022). A panel of 11 stakeholders from CPs, pharmacy policy, pharmacy regulation, medicine, and population health research advised on the research design, suggested relevant documents and interviewees, assisted in two interview pilots, and informed data interpretation (Supplemental Material B).
Organisational documents, such as policies, reports, and letters, are used to construct and convey intents, narratives, and practices (Silverman, 2011). Key bodies involved in the policy implementation were identified from a preliminary review of policy documents: Government of Ireland, Houses of the Oireachtas (Ireland’s national parliament), Department of Health (DoH), Health Service Executive (HSE), High-level Task Force for COVID-19 Vaccination, Irish Institute of Pharmacy, Irish Pharmacy Union (IPU), National Immunisation Advisory Committee, National Public Health Emergency Team, and Pharmaceutical Society of Ireland (PSI). We retrieved documents related to the NCVP using the search terms “pharmac,” “COVID,” and “vaccine” from the websites of these organisations. This was supplemented by reference link chaining and suggestions from the stakeholder panel. The IPU also shared some archived materials. Additionally, freedom of information (FOI) requests were used to access documents not in the public domain as they could add further context to health policy analysis (Burke et al., 2018). FOI requests were submitted to the DoH, HSE, and PSI, three key public bodies involved in the implementation, to gain access to email correspondences, meeting minutes, and internal documents related to the implementation process. Subsequently, we identified 246 relevant documents (Supplemental Material C). Using NVivo, the documents were analysed to clarify the implementation timeline, identify key policy actors, and inform the interview topic guide and theme development (Supplemental Material D).
Elite interviews can elicit rich insider insights into policy processes (Marshall & Rossman, 2011). Using purposive sampling, we identified key elites involved in the NCVP implementation from AK’s professional knowledge and documentary analysis. This was supplemented by snowball sampling where interviewees were asked to suggest people to interview. AK’s “insider” position as a community pharmacist supported access to pharmacy elites for interviews.
Aided by a piloted topic guide, 11 semi-structured interviews were conducted on Zoom, lasting on average 1 hour each. The topic guide (see Supplemental Material E) includes questions to probe interviewees’ thoughts on various aspects of the policy implementation process (Howlett, 2019), including perceptions on the NCVP, factors and barriers to involving CPs in the NCVP, key policy actors and their influence, procedures for policy deliberation, and instruments that were needed for successful policy implementation. Where relevant information outside the scope of the topic guide was given, it was further explored and collected. Interviewees were also asked to suggest documents and other interviewees for the study. All but one interviewee consented to audio-recording. In that instance, only interview notes were taken. The interviews were transcribed verbatim and the transcripts pseudonymised.
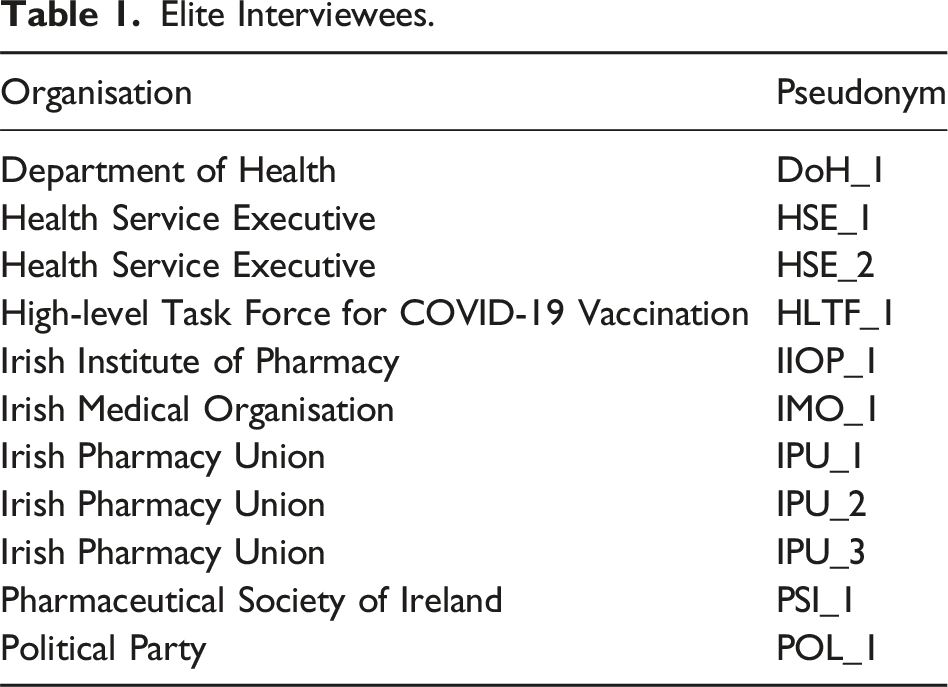
Elite Interviewees.
Responding to the call for health policy analysts to engage with how their “power and positions influence the knowledge they generate” (Walt et al., 2008, p. 308), we used reflexive thematic analysis to analyse our data. We selected this approach as it conceptualises analysis as powered by researchers’ qualitative sensibility, which is the capability to connect data with the wider literature and social contexts (Braun & Clarke, 2022).
We undertook reflexive thematic analysis on NVivo to generate patterns of meanings across the interview data, while drawing on insights from stakeholder engagement and documentary analysis. The analysis shifted between inductive (to explore semantic meanings) and deductive (to interrogate latent meanings) modes. An audit trail is available in Supplemental Material F, and the analysis follows the 15-point guidance for good reflexive thematic analysis (Braun & Clarke, 2022).
Ethics approval was obtained from the Department of Sociology at Trinity College Dublin. All interviewees were requested to complete a written informed consent form before the interviews.
Analysis
In Ireland, the High-level Task Force on COVID-19 Vaccination was established by the Irish Government on 10th November 2020 to support the DoH (which provides strategic policy directions) and HSE (the implementation arm of the former) in rolling out the NCVP. On 15th December 2020, the NCVP Strategy and Implementation Plan were published (DoH, 2021a, 2021b). They outlined a three-phase vaccination roll-out approach, namely, Initial Roll-out, Mass Ramp-up, and Open Access. When more doses became available, CPs, GPs, and MVCs were expected to be involved in the latter two phases.
The NCVP documents outlined that CPs, owing to their successful annual flu vaccination programme, should be utilised to enhance vaccination capacity (DoH, 2021a). Pharmacists in Ireland had been providing various vaccination services for a decade, including flu, herpes zoster, and pneumococcal polysaccharide vaccines (Henman, 2020). Evidence demonstrates that CP-based flu vaccination services in Ireland helped to increase vaccine uptake, especially in those at risk of severe flu symptoms, and was positively received by the public (Finnegan, 2018; PSI, 2015). In addition, the PSI (the pharmacy regulator) made several regulatory amendments to temporarily enhance the authority of pharmacists during COVID-19 to ensure pharmaceutical care continuity, such as increasing the number of repeats and quantity of medicines that can be supplied (Lynch & O’Leary, 2021). Triggered by the pandemic, pharmacists were also authorised to administer flu vaccines offsite from pharmacy premises and to children aged 6 months and older (PSI, 2021a).
However, months of delays in deploying CPs for the NCVP ensued, despite continued advocacy by the IPU (a trade association that represents CPs’ commercial and professional interests). The NCVP Initial Roll-out phase commenced on 29th December 2020 in nursing homes (RTÉ, 2020), followed by the Mass Ramp-up phase on 16th January 2021 in MVCs for priority healthcare workers which was then expanded to priority population groups (O’Brien, 2021). GPs subsequently joined forces on 8th February 2021 (Murray & Dwyer, 2021). However, it was not until 31st May 2021 when the Minister for Health Stephen Donnelly announced the commencement of CP-based COVID-19 vaccination (Houses of the Oireachtas, 2021). CPs started vaccinating people over 50 years old with Janssen vaccines on 14th June 2021 (Murphy & Heaney, 2021).
Figure 1 presents the policy implementation timeline, which was constructed from the documentary analysis (see Supplemental Material G for full details). Policy implementation timeline. CPs, community pharmacies; GOV, government; GPs, general practices; HSE, Health Service Executive; IPU, Irish Pharmacy Union; IT, information technology; MoH, Minister of Health; NCVP, National COVID-19 Vaccination Programme; NIAC, National Immunisation Advisory Committee; PSI, Pharmaceutical Society of Ireland.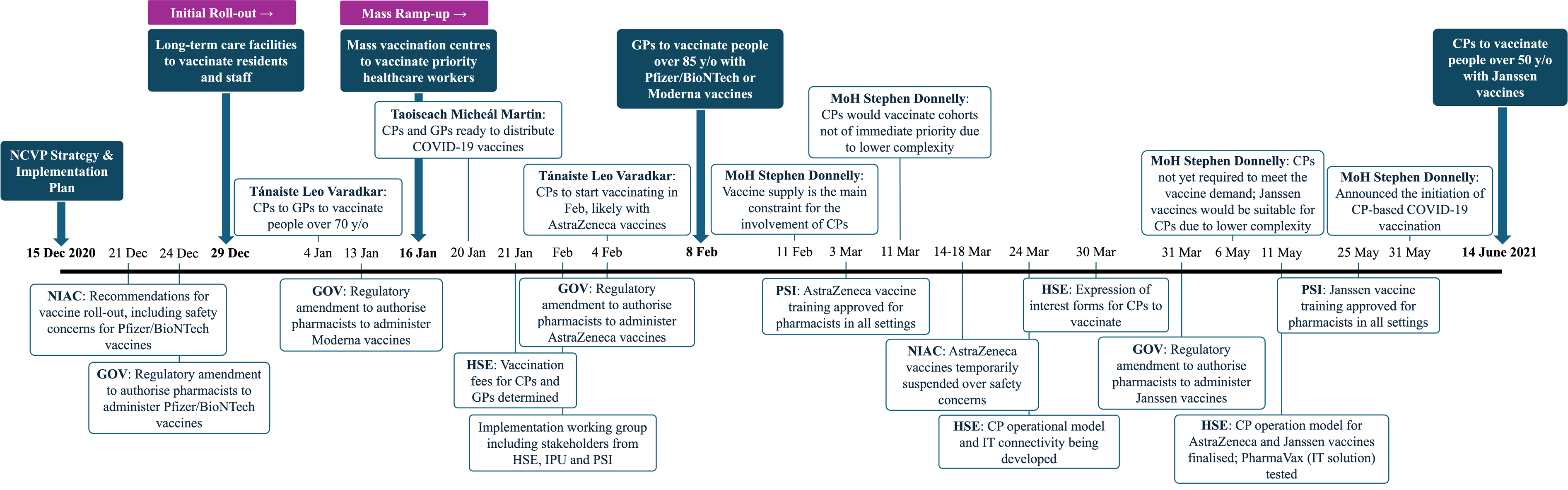
A Tripartite Analytical Output
Three themes were generated to explain the delayed deployment of CPs in the NCVP, two of which each had two supporting sub-themes (Figure 2). Each theme serves as a central organising concept for the data. Thematic map. Dark blue, themes; light blue, sub-themes.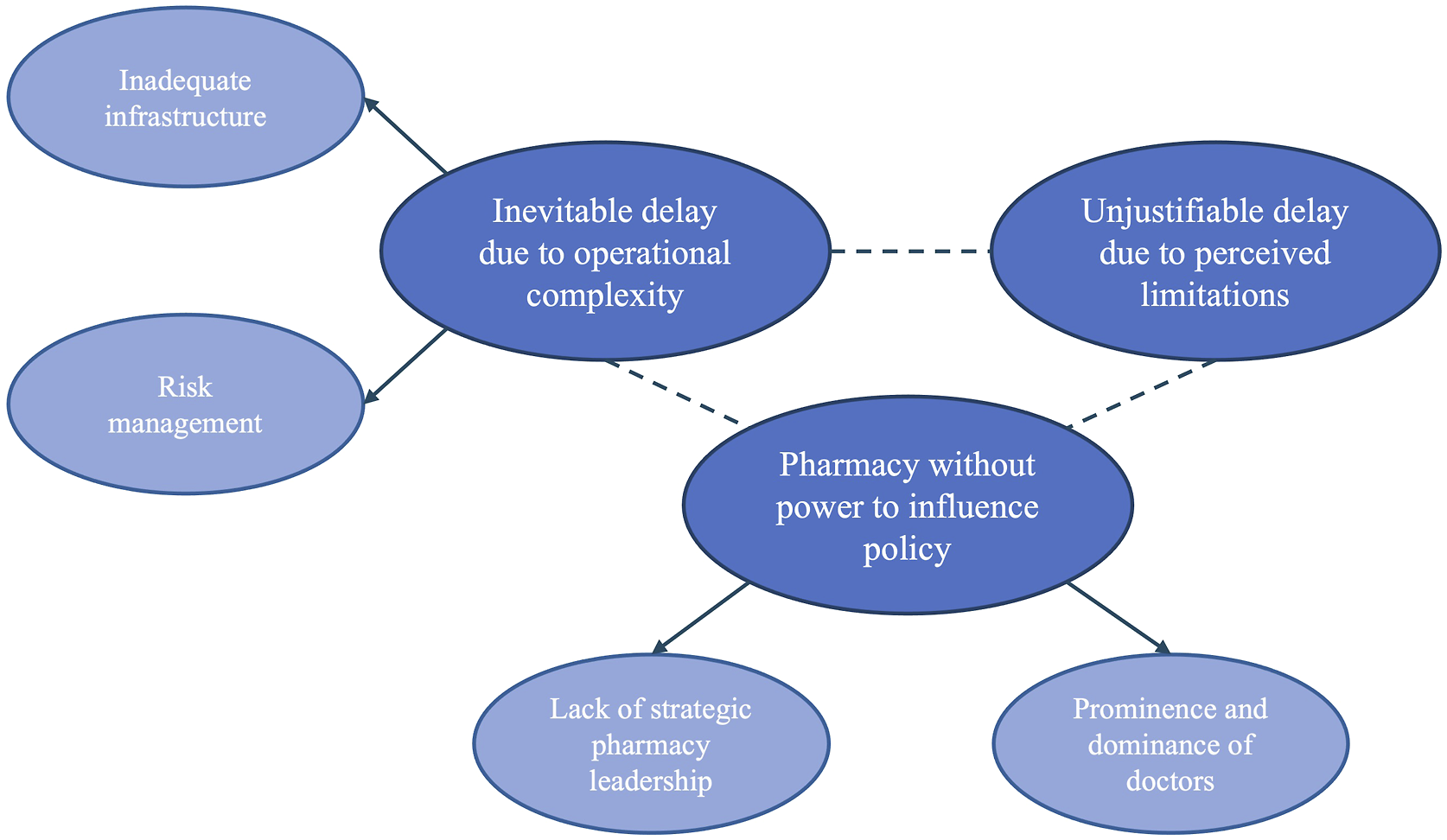
Inevitable Delay Due to Operational Complexity
This theme presents the view that the delayed involvement of CPs in COVID-19 vaccination was inevitable, given the substantial efforts required to overcome various operational obstacles. It supports a previous analysis in Ireland, which found that perceived resource and capability limitations hindered health policy implementation (Kelly et al., 2016).
Several elites acknowledged the value of community pharmacists, particularly their “expertise, relationships, knowledge and facilities” (HSE_2), in supporting the large-scale national programme at speed. We didn't involve community pharmac[ies] as a public relations exercise … We involve[d] them because they could play a key role. (HSE_2)
CP involvement was, however, confronted with a multitude of complex operational barriers, including resource, infrastructure, and regulatory considerations, the addressing of which required laborious multi-sectoral collaboration. In February 2021, the HSE stated that a working group with senior clinical representatives (from the DoH, HSE, IPU, and PSI) was engaged in supporting CP-based COVID-19 vaccination (Bowers, 2021) and some elites stated that all decisions regarding the role of CPs were made logically and rationally. It unfolded in the way [it] kind of needed to. (PSI_1) There was no bias in any direction against one channel or another, it’s just brought in on a rational basis, taking account of all the factors that need[ed] to be considered. (HLTF_1)
The following two sub-themes illustrate that concerns about infrastructure and safety partly explain the delay.
Inadequate Infrastructure
Vaccine supply scarcity and uncertainty (DoH, 2021c) were significant operational considerations in the early phase of the NCVP: “[I]t was like gold dust, every drop was important” (HSE_1). In Ireland, a step-wise approach was taken where outlets that could be efficiently leveraged to deliver vaccines to at-risk cohorts were prioritised (DoH, 2021b).
As MVCs could efficiently deliver vaccines at a high capacity (DoH, 2021a), they received immediate resource and planning priority around logistics, infrastructure, and staffing, “rather than driving additional costs” through other outlets (IPU_1). Moreover, the NCVP operated on a cohort sequencing approach that prioritised vaccination for people at a higher risk of exposure, mortality, or morbidity (DoH, 2021c). Since GPs could identify at-risk patients from their routine patient records and were equipped with an information technology (IT) system that could be “leveraged and locked onto COVAX [the Irish electronic data system for COVID-19 vaccination records]” (HSE_1), they were efficiently brought on stream in February 2021 when more vaccines became available.
On the other hand, CPs were perceived to have inadequate infrastructure, capacity, and experience to deliver COVID-19 vaccination on top of their core function of medication dispensing. It was understood from the beginning that COVID-19 vaccination “wasn’t for every pharmacy” (HSE_1) and, consequently, CPs did not receive similar planning priority as GPs (DoH_1).
Indeed, research shows that operational planning around staffing, space, safety, workflow, training, supply chain, and storage can be challenging for CP-based COVID-19 vaccination programmes (Burson et al., 2016; Maidment et al., 2021). Furthermore, pharmacies were already under higher workload pressure due to COVID-19 (Maidment et al., 2021). These considerations were important for the initial roll-out of Pfizer/BioNTech vaccines (the first COVID-19 vaccine available in Ireland), which were well-recognised as logistically challenging to store and administer.
Moreover, CPs were perceived as unable to identify at-risk patients for vaccination. The IT systems in CPs, which “had no [significant] integration with the health system,” also imposed technical challenges to connect with the COVAX system to record individuals’ COVID-19 vaccination history and for vaccination fees reimbursement (HSE_1). Indeed, IT infrastructure is a recognised barrier hindering CPs from providing vaccination services in many European countries (Henman, 2020; Paudyal, Fialová, et al., 2021; Turcu-Stiolica et al., 2021). To overcome this operational barrier, the IPU led the development of an IT solution called PharmaVax jointly with the DoH and HSE, which took a few months to build.
From this perspective, then, due to various operational issues that rendered significant management necessary, “it just happened in that order” that CPs in Ireland came on stream later than others (HSE_2). Within all the competing priorities to keep moving forward, when was the right time? (HSE_1)
Risk Management
Globally, there has been widespread mis-/dis-information about COVID-19 vaccines (OECD, 2021). Thus, a precautionary approach to vaccine roll-out was taken to ensure public trust, where rigorous evaluation was conducted based on the emerging vaccine safety profiles and the (perceived) capability of CPs to manage the associated risks. It’s difficult decision-making territory at the time … The whole thing was not to take chances with the vaccines even though we’re looking at a global pandemic. (PSI_1) Our knowledge of the vaccines kept changing and our knowledge of the disease kept changing, so [the] guidance kept changing. (HSE_2)
In response to reports of anaphylaxis and logistical challenges associated with the Pfizer/BioNTech COVID-19 vaccines, the National Immunisation Advisory Committee recommended an initial roll-out in facilities with immediate access to medical teams in late December 2020, followed by continued roll-out in other planned facilities in January 2021 in the absence of unanticipated issues (RCPI, 2020). Thus, it follows that the initial roll-out was restricted to MVCs and subsequently expanded to GPs.
Risks associated with CP-based COVID-19 vaccination were reported to be more of a concern. While CPs in Ireland have been authorised to administer various vaccines since 2011 (Henman, 2020), vaccination was not considered a core competency of pharmacists in Ireland by some elites (DoH_1; HSE_1, 2). This is because, unlike doctors, not all pharmacists receive training in administering injections like vaccines during their standard training; they need to obtain additional certifications (PSI, 2021b). Concerns were also raised as to whether CPs had sufficient space to accommodate post-vaccination observation while ensuring social distancing (DoH_1; HLTF_1). Additionally, AstraZeneca vaccines, which were deemed more appropriate for use in CPs due to ease of storage and preparation, faced scrutiny and temporary suspension in light of safety reports (see Supplemental Material G).
Therefore, robust regulatory mechanisms were deemed necessary to assure the quality and safety of CP-based COVID-19 vaccination. To mitigate operational risks, the HSE required CPs interested in providing COVID-19 vaccines to complete an “expression of interest” form issued on 30th March 2021, where they were required to evaluate if they had the facilities and competencies to provide such service (HSE, 2021). This was notably not required of GPs.
Furthermore, under the current legal framework, doctors were able to administer COVID-19 vaccines after completing relevant training. In contrast, individual statutory instruments were necessary to amend the Medicinal Products (Prescription and Control of Supply) Regulations to authorise pharmacists (and other professionals like nurses, midwives, and paramedics) to administer each specific COVID-19 vaccine brand. To provide CP-based vaccination services, pharmacists must undertake a range of training courses depending on their vaccination experience, including modules on specific vaccines (PSI, 2021b). The regulator PSI was also required to individually review and approve each of the COVID-19 vaccine training modules designed by the HSE National Immunisation Office (see Supplemental Material H). If you are looking to expand your scope of delivery of services, on behalf of the state, you have to be in a position to show and demonstrate the quality assurance around that. (HSE_1)
Numerous elites articulated that the regulatory regime, albeit potentially perceived as “over-engineered” (PSI_1), was necessary to ensure patient safety (HSE_1, 2; PSI_1). A CP-based flu vaccination safety incident in 2011 resulted in an incident review report that recommended that rigorous governance and training must be in place for “any such expansion in the scope of practice of a healthcare professional” (PSI, 2012, p. 4). However, some elites articulated that the PSI addressed all the regulatory barriers as efficiently as possible (HSE_2; PSI_1). The Medicinal Products (Prescription and Control of Supply) Regulations were also swiftly amended and approved by the Minister for Health to enable pharmacists to administer COVID-19 vaccines when they became available (Supplemental Material G). Indeed, the PSI was found to demonstrate agility during the pandemic by rapidly introducing regulatory changes to expand the professional capacity of pharmacists, including the authority to extend prescription validity (Lynch & O’Leary, 2021).
Unjustifiable Delay Due to Perceived Limitations
This theme offers an opposing view to the previous one, where the delay was seen as unjustifiable and attributed to perceived inadequacy in the capability of CPs to deliver COVID-19 vaccination. Due to the potential of CPs as accessible and trusted vaccination outlets, several elites—especially those from the IPU—perceived the decision not to deploy CPs earlier as unjustifiable and “hard to understand” (POL_1). Indeed, reflecting the international evidence on CPs’ high accessibility and public trust (Isenor et al., 2016; Maidment et al., 2021; Pantasri, 2022; Wubishet et al., 2021), research shows that over 50% of individuals in Ireland can access a CP within 1 km and 85% within 5 km (IPU, 2022a). CP-based flu vaccination has also increased uptake, including in those receiving the vaccine for the first time and those in an at-risk category (PSI, 2015). Furthermore, community pharmacists are the most trusted profession in Ireland (Ipsos, 2023) and the majority of the public would like CPs to provide more services (IPU, 2023).
While the roll-out of CP-based COVID-19 vaccination globally was impeded by a lack of political support (Maidment et al., 2021), promises that CPs in Ireland would be used as vaccination channels were publicly and repeatedly made by the Minister for Health Stephen Donnelly, Tánaiste Leo Varadkar, and Taoiseach Micheál Martin (see Supplemental Material G). However, pharmacists felt that they were being strung along with platitudes and the IPU had “no information other than what is in the media” (IPU, 2021). Where clarification was sought from the Minister for Health, DoH, or HSE, the reasons given for the delay were perceived as unclear, disingenuous, or even “bullshit and obfuscation” (IPU_3). This led to feelings of distrust, disrespect, and frustration. I’ve raised this a number of times … [and the response] was kind of vague. (POL_1)
Operational barriers identified by officials that supposedly delayed the involvement of CPs (as discussed in the previous theme) were viewed as unreasonable. Indeed, the capability of CPs to adapt services to meet the demands posed by COVID-19 was evident both in Ireland and internationally (Costa et al., 2022; Maidment et al., 2021; Pantasri, 2022). Therefore, questions as to why operational concerns, such as capacity and cold storage, applying to CPs did not also delay the involvement of GPs were raised (IPU_1, 2). In the same vein, the ad-hoc “expression of interest” mechanism enacted by the HSE to ensure the quality and safety of CP-based vaccination, which was not applied to GPs, was also seen as unfair (HSE, 2021). Without any evidence, it was taken and a given that the service could be delivered by one set of healthcare professional[s], and it couldn’t be delivered by the other. (IPU_2)
As discussed, the roll-out of NCVP was based on priority groups to ensure at-risk populations received COVID-19 vaccines first. Since CPs do not routinely collect or have access to comprehensive patient records to identify patients in priority groups for COVID-19 vaccination, like GPs do, several alternatives were apparently proposed by the IPU, including accessing information like age and diagnoses via the HSE system for patients with community drug schemes (IPU_1, 3). Nonetheless, these proposals were reportedly rejected by the HSE. An elite also mentioned that the operational guidance for CP-based vaccination, which was reportedly drafted in conjunction with the HSE National Immunisation Office and was similar to that for GPs, faced a significant delay in formal approval by the HSE (IPU_3).
Additionally, the regulatory mechanisms governing the vaccination scope of pharmacists were perceived to have been (exceedingly) bureaucratic. This phenomenon is common globally where the scope of CP-based COVID-19 vaccination is hindered by regulatory frameworks (Burson et al., 2016; Costa et al., 2022). Evidence shows that community pharmacists in Ireland generally also perceive the PSI as “demanding higher standards of service delivery and infrastructure than what is considered necessary or proportionate” (Lynch & Kodate, 2020, p. 214).
Due to the persistent lack of clarity and urgency given to the role of CPs, the IPU resorted to coordinated media and political campaigning with MKC Communications, a public relations firm, to leverage their influence (MKC, 2022). This was seen as a key factor resulting in the eventual involvement of CPs. I know that campaigning made the HSE quite uncomfortable because it led to politicians asking them questions … I believe without all of that campaigning, the HSE [would] just have ignored us until it was all over. (IPU_3)
Pharmacy Without Power to Influence Policy
As policy processes are inevitably influenced by a range of actors, political ideologies, and vested interests (Buse et al., 2023; Grindle & Thomas, 1991; Kingdon & Stano, 1984), this theme reflects the broader socio-political context that contributed to the diminished influence of CPs in the health system, including on the issue of COVID-19 vaccination. Medical politics, it’s often said, puts partisan politics in the shade. (POL_1) It’s a messy kind of world of politics with a small p. (PSI_1)
In the past few decades, healthcare systems around the world have been subjected to various forms of market-based, pro-competitive reforms (Maarse, 2006; van de Ven et al., 2013). This includes the primary healthcare sector, which has been increasingly privatised in the hope of increasing efficiency and affordability (Mercille, 2019; Rico et al., 2003).
As the only European country in which the public health system provides free primary care to a minority of residents, Ireland’s primary care system has been heavily reliant on private contractors like CPs and GPs since the 1970s (Devitt, 2021). Nonetheless, several elites highlighted how the commercial viability of CPs and GPs in Ireland depends on service remuneration from the state (HSE_1; IMO_1; IPU_2). This has resulted in competitive contract negotiations with the state for funding and, to some extent, professional territorialism and rivalries as each side tries to protect its professional and financial interests (Henman, 2020). Similar rivalries have been evidenced in international research, where the introduction of new pharmacy services like vaccination has faced opposition from GPs in Australia, Canada, New Zealand, and Wales (Evans et al., 2016; Gauld et al., 2021; Hattingh et al., 2016; Moore et al., 2014; Valiquette & Bédard, 2015). Professionals still have to fight for their space in decision-making. All professionals, (pauses) I would say. (HSE_2) GPs are expected to take the [financial] risk to provide a public service … as pharmac[ies do] too … The whole model is all a bit dodgy. (IMO_1)
Historically, pharmacists in Ireland—unlike doctors—have lacked influence in health policy. For example, initiatives like the Future Pharmacy Practice report (PSI, 2016), which promotes the expansion of pharmacists’ roles, failed to gain policy traction (HSE_1). More recently, despite the past Minister for Health Simon Harris’ promise of increased pharmacy investment and statement that CPs could be the “shopfront of Sláintecare” (a reform programme towards universal healthcare) in 2019 (Quann, 2019), his office proposed cuts in dispensing fees that would cost the sector over €50 million several months later (Cullen, 2019). While this was rolled back upon strong opposition, it demonstrates how policy decisions do not always align with political promises.
In this institutional and political context, it is perhaps less surprising that CP struggled to make its voice heard on the issue of NCVP: “We had no leverage to extract what had been promised to us” (IPU_3). This is supported by two sub-themes: one focuses on the lack of influence of the pharmacy profession, while the other addresses the significant power held by the medical profession within Irish health policy.
Lack of Strategic Pharmacy Leadership
In Ireland, the lack of central, strategic, and cohesive leadership has impeded the role advancement of pharmacy: “Pharmacy has shot itself in the foot” (IIOP_1). First, most elites agreed that the lack of a central pharmacy voice in the DoH and HSE may have impeded the efficient roll-out of CP-based COVID-19 vaccination. Notably, no pharmacy stakeholder was present on the High-level Task Force on COVID-19 Vaccination (DoH, 2020). Further, the Chief Pharmacy Officer post in the DoH has been vacant since 2013 (Lynch, 2014), in contrast with neighbouring jurisdictions like the United Kingdom (where each of the four countries has such an officer). Evidence also shows that the previous lack of senior dentistry representation at the DoH and HSE has resulted in “political disinterest” in oral health policies in Ireland (McAuliffe et al., 2022, p. 8). Thus, without senior pharmacy leadership, it is possible that “there will never be the vision to properly integrate pharmacy services into the healthcare system” (IPU_3).
Nonetheless, some non-pharmacist elites were unsure about the role a Chief Pharmacist Officer in the DoH or HSE would play in public health (POL_1; IMO_1): “It’s not about divvying up the cake” (IMO_1). In July 2022, the Minister for Health Stephen Donnelly also mentioned that the DoH did not have plans to appoint such an officer (Houses of the Oireachtas, 2022), as there were already qualified pharmacists working in his office. Further, it was maintained that a previous proposal to have pharmacy subsumed under the representation of Health and Social Care Professionals in the HSE was met with pharmacy opposition due to a preference for a specific pharmacy officer (IIOP_1). However, the profession has not been able to campaign for one successfully.
Second, the pharmacy profession in Ireland does not have a professional leadership body (O’Sullivan & McCarthy, 2022) that can promote the role of CPs for the public good without (perceived) undue commercial influence. The PSI serves as the pharmacy regulator that safeguards the public’s interests, while the IPU operates as a trade association representing the business and professional interests of CPs. There is currently no independent body dedicated to the professional interests of CPs to enhance patient care (although the Irish Institute for Pharmacy, at the behest of the PSI, manages the Continuous Professional Development of the profession with limited autonomy and resources). This contrasts with some other health professions, such as general practitioners, where their professional interests are represented by the Irish College of General Practitioners and their trade interest (and some professional interests) by the Irish Medical Organisation (IMO), while regulation is overseen by the Medicine Council of Ireland. Due to increased commercialism and competition, policymakers may further perceive an erosion in the professionalism of pharmacists (Bradley, 2009; Traulsen & Almarsdóttir, 2005). For instance, the Health (Community Pharmacy Contract Agreement) Regulations to control the density of CPs were revoked in 2002 due to legal cases brought forward by some CPs, creating a pro-competitive sector (Purcell, 2004).
Third, the refusal of the IPU to be part of the primary care team structure proposed in the 2001 “Primary Care: A New Direction” strategy (DoH, 2001)—based on concerns about the potential loss of professional autonomy and that some CPs would financially benefit from co-location with GPs due to increased footfall (Bradley, 2009; Henman, 2020)—was perceived to have diminished the role of CPs in primary care. It was suggested that this may have had implications for the influence of CPs in the policy space. It was a decision that parted the ways in terms of primary care … [The IPU] have been challenged to get themselves back. (HSE_1) We’ve counted ourselves out of every single representation to the health system, and yet we complain. (IIOP_1)
Finally, the straddling between professional and commercial imperatives has given rise to tension within the IPU (and the wider profession), and this lack of cohesion may have limited the effectiveness of its advocacy (Shiffman et al., 2004; Shiffman & Smith, 2007). The IPU seem to think that you can kind of be a trade union, and a professional body, and a supplier of training … They’re riven with rivalries [and] infighting … [and they’re] very much kind of a marriage of convenience between different kinds of stakeholders. (PSI_1)
Such tension also manifested in the context of the NCVP, where diverse interests within the IPU had to be managed by the leadership to ensure the patients’ interests were centred in all decisions: [COVID-19 vaccination] provides professional satisfaction for our staff and it also provides some badly needed revenue at a time when business is difficult. I can’t deny that that’s the case, no more than that is the case [for] GP[s] (IPU_3)
Prominence and Dominance of Doctors
Capitalising on their medical expertise, monopolistic strategies, and access to decision-makers, international evidence shows that the medical profession traditionally holds significant power in influencing health policy (even though such power may be in decline) (Berridge, 1997; Immergut, 1990; Laugesen & Rice, 2003). This phenomenon is also well-documented in Ireland (Barrington, 1987; Wren, 2003). Interestingly, in small and post-colonial countries like Ireland, vested interests such as the medical profession may have easier access to senior policymakers to influence policy processes (Grindle & Thomas, 1991; Walt et al., 2008).
In this study, several elites discussed the strong representation and power of doctors at senior management levels in the DoH and HSE. While this was at times understood to be reasonable due to their clinical expertise—“doctors know best” (POL_1)—the health system has been institutionally orientated to prioritise medical doctors in health service delivery, sometimes to the neglect of other professionals that could be utilised or leveraged. [GPs have] put in the work and built up their offering … Everything flows from that. (PSI_1) The health service tends to go first to GPs … not necessarily just about vaccination. (HSE_1)
A political elite outlined that the IPU “certainly [doesn’t] have the kind of access, influence or voice doctors’ unions have” (POL_1) that allows them to lobby for both professional and profit motives. A lot of the influence would have been from doctors … that was determining the shape of the [NCVP]. (POL_1)
The vested interests of doctors in Ireland can be illustrated by their opposition to policies that advance universal healthcare, such as free GP care (Devitt, 2022). Similarly, GPs may see the role expansion of CPs as a professional encroachment that would also reduce their profits (Bradley, 2009; Henman, 2020). In 2015, Dr Ray Walley, the President of the IMO at the time, opposed CP-led vaccination programmes and stated that the Government should “resource GP services properly[,] not substitute them with inappropriate alternatives like pharmacists” (IMO, 2015).
According to several elites, the initial roll-out of CP-based flu vaccination in 2011 was characterised by explicit opposition by GP representatives and internal resistance by senior policy actors. It was maintained that Dr James Reilly, Minister of Health at the time from Fine Gael who was also a general practitioner and previous IMO president, consolidated the influence of GPs’ interests within the health system (POL_1; IPU_3). Indeed, Fine Gael and Fianna Fáil, the two major centre-right political parties in Ireland, have aligned themselves with vested interests in the health sector in the past (Burke, 2009; Devitt, 2022; Wren, 2003). There were delegations from the IMO arrived out to the PSI offices … and [they said that] people would die in pharmacies because pharmacists aren’t capable of vaccinating, and GPs will not be taking responsibility for [that]. (IPU_3) Quite a battle [to get support] from senior people within the Department and from the Minister for Health [Dr James Reilly] … There would have been very much of [a] view … that you couldn’t take this business away from GPs. (POL_1)
GP opposition to CP involvement in vaccination in Ireland is mirrored in other European contexts. For example, the introduction of a CP-based flu vaccination scheme in England in 2015 was also contentious as GPs argued that it disrupted the established vaccination service, lowered uptake, and led to financial losses for GPs (Kaffash, 2016; McKee, 2016).
In the case of the Irish NCVP, an interviewee maintained that GPs with strong political connections conducted covert lobbying to delay and minimise the role of CPs (IPU_3). The motive was to ensure GPs could maximise profits from the NCVP, which was perceived to align with what subsequently happened: when CPs were allowed to provide COVID-19 vaccination in June 2021, the scope was limited to single-dose Janssen vaccines for individuals over 50 years old, almost all of whom had been given the opportunity to be vaccinated at MVCs or their GPs. While this conjecture remains to be substantiated, it aligns with a multi-dimensional conceptualisation of power in which policy processes can be covertly controlled by powerful, “invisible” actors (Lukes, 2005).
Discussion
Our analysis reveals that the delayed involvement of CPs in the NCVP was shaped by complex relations between policy actors and various socio-political, historical, economic, and operational factors. We argue that the involvement of CPs was delayed because the health system—continually shaped by a lack of cohesive and strategic leadership within the pharmacy profession and the political dominance of the medical profession—lacked a clear and strategic direction for pharmacy. Arguably a case of path dependence, this gave rise to the structural unpreparedness of the Irish health system to swiftly leverage CPs for COVID-19 vaccination, evidenced by the operational issues that supposedly delayed their involvement. Instead, an incremental implementation strategy was enacted (Grindle & Thomas, 1991; Lindblom, 1959) to gradually introduce structural change, like legislative amendments, in the form of institutional layering (Thelen, 2004). In parallel, the delay could also be interpreted as a policy implementation failure (Barrington, 1987) to optimise the health system to deliver life-saving vaccines, although the actual adverse health impact remains unclear.
CPs have proven to be valuable community outlets that can help to increase the uptake of pandemic vaccination. In spite of the logistical, workload, and financial challenges associated with the roll-out of COVID-19, community pharmacists in Australia, Canada, France, Norway, and the United States expressed a willingness to partake in COVID-19 vaccination campaigns (Aarnes & Nilsen, 2023; Durand et al., 2022; Gerges et al., 2023; McCormick et al., 2023; Walliar et al., 2023). Evaluation of CP-based COVID-19 vaccination services in Australia, France, Northern Ireland, and Switzerland also revealed that they are positively received by the public due to convenience, accessibility, and trust (Bui et al., 2023; Garland & Jacklin, 2023; Piraux et al., 2022; Stämpfli et al., 2021).
Like pharmacists elsewhere (Paudyal, Cadogan, et al., 2021), CPs in Ireland demonstrated adaptability in responding to the heightened healthcare demand induced by the pandemic, positioning themselves as promising community outlets for expanding COVID-19 vaccine coverage (Cadogan & Hughes, 2021; Hayden & Parkin, 2020; Lynch & O’Leary, 2021). Despite the existing legislation and infrastructure for CP-based vaccination programmes in Ireland, there was nonetheless a delay in the roll-out of CP-based COVID-19 vaccination due to various operational, regulatory, and political challenges.
Similar challenges are evidenced in the international literature. Research indicates that comprehensive operational planning, leadership and systems coordination across workforce, training, infrastructure, and IT systems were essential for community COVID-19 vaccination programmes led by pharmacists in Taiwan, the United States, and several European countries (Ezeude et al., 2022; Lin et al., 2021; Paudyal, Cadogan, et al., 2021). Regulatory frameworks are also a common hindrance to the introduction or expansion of CP-based vaccination (Burson et al., 2016; Costa et al., 2022). Furthermore, the resistance of the medical profession towards new pharmacy services due to an inadequate understanding of pharmacists’ skills, professional territorialism, and competing financial interests is documented in the literature (Bryant et al., 2009; Hindi et al., 2019; Hughes & McCann, 2003; Muijrers et al., 2003; Zhou et al., 2019) and evident in the context of new vaccination services in Ireland (Moore et al., 2014) and elsewhere (Evans et al., 2016; Gauld et al., 2021; Hattingh et al., 2016).
Future viral epidemics and pandemics are inevitable (Morse et al., 2012; Telenti et al., 2021), and health system actors must “partner, coordinate, develop, and strengthen” (Haldane et al., 2021, p. 1) intersectoral pandemic vaccination planning and response to ensure efficient and equitable vaccine access. With regard to improving pandemic preparedness in Ireland, based on our analysis, we recommend that (1) The legislative and regulatory framework governing the scope of pharmacists’ (vaccination) practice should undergo review to enhance flexibility especially during periods of public health crises; (2) The integration of IT infrastructure between CPs and the broader health system, particularly concerning electronic patient records, should be further developed; and (3) The different pharmacy bodies collectively develop a cohesive leadership vision and engagement strategy, including continued advocacy for senior representation like Chief Pharmacy Officers in the DoH and HSE, to advance pharmacy practice in the public’s best interest.
To our knowledge, this is the first health policy analysis exploring the delayed implementation of CP-based COVID-19 vaccination in Ireland. By drawing on multiple methods, our qualitative case study generated a novel, rigorous, and in-depth analysis of the complex policy process shaping the role of CPs in the NCVP. The lead researcher’s ‘insider’ position as a pharmacist enabled access to pharmacy elites, trust, and rapport building (CUREC, 2020). Since the interviews occurred approximately a year and a half after the implementation, recall bias may be an issue (Silverman, 2011), but this was mitigated by having a diverse pool of elites and supporting data interpretation with documentary sources. Although we believe that the 11 elite interviews conducted had adequate information power to address the research question (Malterud et al., 2016), we concede that our analysis may not have captured the full spectrum of perspectives given not all key policy actors were interviewed. Stakeholder engagement was also a strength of this study. Albeit time-consuming (especially since the stakeholders have busy professional lives), it was a process that enabled the identification of further documents and interviewees.
The interpretation of conflicting narratives of the policy implementation process is also a strength of this study. While contrasting perspectives can initially result in unclear conclusions, in this case, an over-arching factor—the political weakness of pharmacy in Ireland—enabled the reconciliation of seemingly contradictory narratives: the supposed operational challenges partially delayed the involvement of CPs in the NCVP, but they also reflect policy actors’ perception of CPs’ capability as inadequate. Thus, arguments about whether the delay was rational or unfair are not incongruous. We believe that it would be fruitful to extend this case study to a comparative analysis with countries where CP involvement in COVID-19 vaccination also experienced delays—such as Australia where operational challenges are evident (Jackson & Al-Wassiti, 2021; Walliar et al., 2023)—as well as countries where CPs were swiftly engaged—such as the United Kingdom (Wickware, 2021)—to develop cross-case insights with wider applicability (Gilson et al., 2011).
Given the complex policy dynamics between ideas, interests, and institutions that shape the landscape of CPs in Ireland, having a deeper grasp of health politics would better equip the profession to advocate for better health policy (Buse, 2008). While our work has shed some light on some of these dynamics, we hope it will inspire future work in this area.
Supplemental Material
Supplemental Material - What Obstructs Health Policy Implementation? A Multi-Method Qualitative Case Study of the Delayed Deployment of Community Pharmacies in Ireland’s National COVID-19 Vaccination Programme
Supplemental Material for What Obstructs Health Policy Implementation? A Multi-Method Qualitative Case Study of the Delayed Deployment of Community Pharmacies in Ireland’s National COVID-19 Vaccination Programme by Aaron Koay and Camilla Devitt in Qualitative Health Research
Footnotes
Acknowledgments
We sincerely thank the policy elites who participated in the interviews and the Irish Pharmacy Union for facilitating access to archived documents. We also extend our gratitude to the stakeholders who guided the research project, including Dr. Shaakya Anand-Vembar, Prof. Claire Anderson, Dan Burns, Dr. Conor Fitzmaurice, Dr. Matthew Lynch, and Margaret O'Doherty. We are equally grateful to other stakeholders who prefer not to be named.
Author Contributions
AK: conceptualisation; data collection; formal analysis; investigation; methodology; project administration; writing—original draft; and writing—review and editing. CD: conceptualisation; supervision; and writing—review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
