Abstract
Acute liver injury (ALI) is a recognized adverse event with adeno-associated virus (AAV)-based gene therapies, including delandistrogene moxeparvovec, an AAVrh74-based gene therapy for Duchenne muscular dystrophy. Progression of ALI to acute liver failure (ALF) is rare. In clinical trials, ALF was defined as an international normalized ratio ≥1.5 with encephalopathy and/or ascites, occurring <26 weeks since product/treatment exposure, and in the absence of identified preexisting liver disease as assessed by the treating investigator. The two cases presented here represent the only known instances of ALF following delandistrogene moxeparvovec to date, both resulting in fatal outcomes. Both cases occurred in nonambulatory patients (ages 15 and 16 years). Both patients exhibited abrupt elevations in aminotransferases from baseline approximately 4 weeks after delandistrogene moxeparvovec administration. Over the subsequent weeks and despite interventions, aminotransferases, including gamma-glutamyl transferase (GGT), declined concurrently with a continued rise in total bilirubin, consistent with severe hepatocellular injury and a harbinger of ALF. An interdisciplinary expert panel was convened and concluded that no single biomarker or clinical feature consistently predicted the rapid progression of severe ALI to ALF. Advisors recommended basing treatment intervention for severe ALI on the rate and magnitude of changes from baseline in liver biomarkers (e.g., 2–3× increase in alanine aminotransferase within ≤1 week). For these severe cases, most advisors recommended intravenous methylprednisolone as the initial treatment whereas typical ALI may be managed with oral corticosteroid adjustment or may resolve with time, as seen in the clinical trial experience. Advisors hypothesized that delandistrogene moxeparvovec-related ALI is T-cell-mediated and discussed the potential of additional immunosuppression. Advisors also recommended enhancing baseline evaluation for hepatic comorbidities with lipid profiling, transient elastography, and abdominal ultrasound to further study potential risk factors for ALI progressing to ALF. Finally, there was strong support for generating additional real-world evidence and conducting prospective clinical trials to inform clinical management of ALI in practice.
Keywords
INTRODUCTION
Delandistrogene moxeparvovec is a recombinant adeno-associated virus (AAV)-based gene transfer therapy approved for use in patients aged ≥4 years with a confirmed DMD gene mutation in the United States and select other countries.1–8 The therapy utilizes a rhesus isolate AAV serotype 74 (rAAVrh74) vector 9 and MHCK7 promoter/enhancer 10 designed to drive the expression of the delandistrogene moxeparvovec micro-dystrophin transgene in cardiac and skeletal muscle, including the diaphragm. 11 Clinical studies have demonstrated long-term functional stabilization or attenuation of disease progression in treated patients, accompanied by a safety profile generally manageable with appropriate monitoring.12–17 Reported serious adverse events (AEs) associated with delandistrogene moxeparvovec include nausea, vomiting, increased transaminases, rhabdomyolysis, myocarditis, immune-mediated myositis, and acute liver injury (ALI; defined as gamma-glutamyl transferase [GGT] >3× upper limit of normal [ULN], glutamate dehydrogenase [GLDH] >2.5 ULN, alkaline phosphatase [ALP] >2× ULN, alanine aminotransferase [ALT] >3× baseline when baseline is elevated; Box 1).13–21 Given the occurrence of ALI following AAV-based gene therapies,22–25 treatment with delandistrogene moxeparvovec is recommended to be deferred in patients with acute liver disease until resolution or adequate control is achieved. Patients with preexisting hepatic impairment, chronic liver conditions, or acute liver disease (e.g., viral hepatitis) may be at increased risk for serious hepatic AEs and warrant careful evaluation prior to infusion. 13
Box 1. ALI and ALF Definitions
GGT >3× ULN GLDH >2.5 ULN ALP >2× ULN ALT >3× baseline when baseline is elevated
International normalized ratio ≥1.5 Encephalopathy and/or ascites <26 weeks since exposure to product/treatment No identified preexisting liver disease as assessed by the treating investigator
*Definition as described in delandistrogene moxeparvovec clinical trials; ALF, acute liver failure; ALI, acute liver injury; ALP, alkaline phosphatase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase; GLDH, glutamate dehydrogenase; INR, international normalized ratio; ULN, upper limit of normal.
Overall, while most genetically eligible patients with Duchenne muscular dystrophy (DMD) tolerate therapy well, ALI, a known risk with AAV gene-based therapy including delandistrogene moxeparvovec, has commonly been reported following treatment. In clinical trials, 62 of 156 patients (42%) treated with delandistrogene moxeparvovec experienced ALI (including Study 101 cutoff date, June 8, 2023; Study 102 cutoff date, September 29, 2023; Study 103 and Study 301 Part 1 cutoff date, January 15, 2024). Although commonly observed, a majority of the patients were asymptomatic. Nausea and vomiting are typically reported; two patients exhibited jaundice, including one patient with right upper quadrant tenderness to palpation (without asterixis, lethargy, or somnolence). Progression of ALI to ALF is rare. We present the only two cases known (to the sponsor and authors) to date of ALF following delandistrogene moxeparvovec, both occurring in nonambulatory patients with DMD approximately 3 months post-treatment with fatal outcomes. In clinical trials of delandistrogene moxeparvovec, ALF was defined as an international normalized ratio (INR) ≥1.5 with encephalopathy and/or ascites, occurring within 26 weeks since exposure to product/treatment, and in the absence of identified preexisting liver disease as assessed by the treating investigator (Box 1). We also present recommendations from a series of interdisciplinary expert committee panels that reviewed these two cases and discussed approaches for earlier ALI detection, considerations for ALI and ALF management, and risk stratification. The panels were comprised of globally recognized medical specialists, each bringing extensive domain expertise and decades of clinical and academic leadership in neuromuscular medicine (some with AAV gene therapy experience), hepatology, immunology, transplant medicine, pathology, cardiology, and hematology.
CLINICAL COURSE OF ALF
Case 1
The first case of fatal ALF following delandistrogene moxeparvovec occurred in a 16-year-old male (body weight, 72.9 kg) with DMD (exon 22 deletion) who lost independent ambulation at 12 years of age and was treated in the commercial setting. In addition to hypogonadism and osteoporosis, his medical history included severe neuromuscular restrictive lung disease (forced vital capacity, 33% predicted) and obstructive sleep apnea requiring chronic nocturnal bilevel positive airway pressure (BiPAP). For DMD management, he initially received daily deflazacort starting at age 6, which was transitioned to daily prednisone at age 13, prompted by insurance challenges, and changed to intermittent prednisone (100 mg/day) twice weekly at age 14 (2 years before gene therapy). Beginning the day before delandistrogene moxeparvovec infusion (Day −1), prophylactic prednisone (60 mg/day, ∼0.82 mg/kg/day) was added on days he did not receive his scheduled intermittent dose (Fig. 1A).
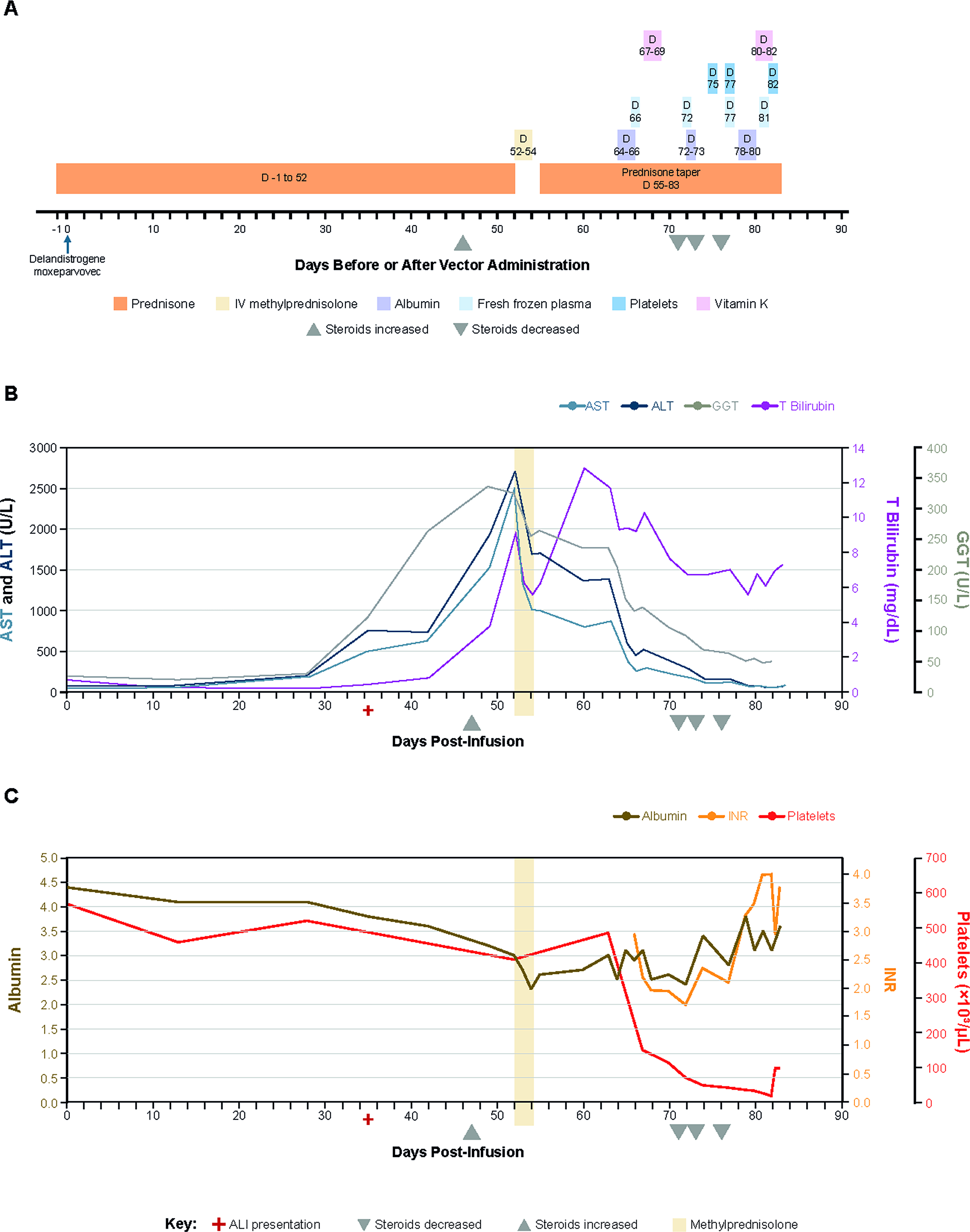
Hepatic biomarker trends in Case 1 following delandistrogene moxeparvovec administration. ALI presentation and management are shown.
The patient received delandistrogene moxeparvovec (9.31 × 1015 total vg) (Day 0). Routine laboratory analysis on Day 13 was unremarkable for any abnormalities (Table 1). On Day 28, the patient attended a routine clinical visit, at which time he reported 1 week of congestion and cough, occasionally with darker colored sputum, without fever. Chest X-ray demonstrated “mild peri-bronchial thickening” attributed to a likely viral process, but no pneumonia. Aspartate aminotransferase (AST) and ALT levels were increased to 174 U/L (2.8×; baseline 62 U/L) and 196 U/L (2.8×; baseline 69 U/L), respectively; gamma-glutamyl transferase (GGT) level was 29 U/L (baseline, 25 U/L) (Fig. 1B, Table 1). ALP was 58 U/L. Platelets were 519 × 10³/μL, and white blood cell (WBC) count was 12.2 × 103/μL.
Case 1 Laboratory Results
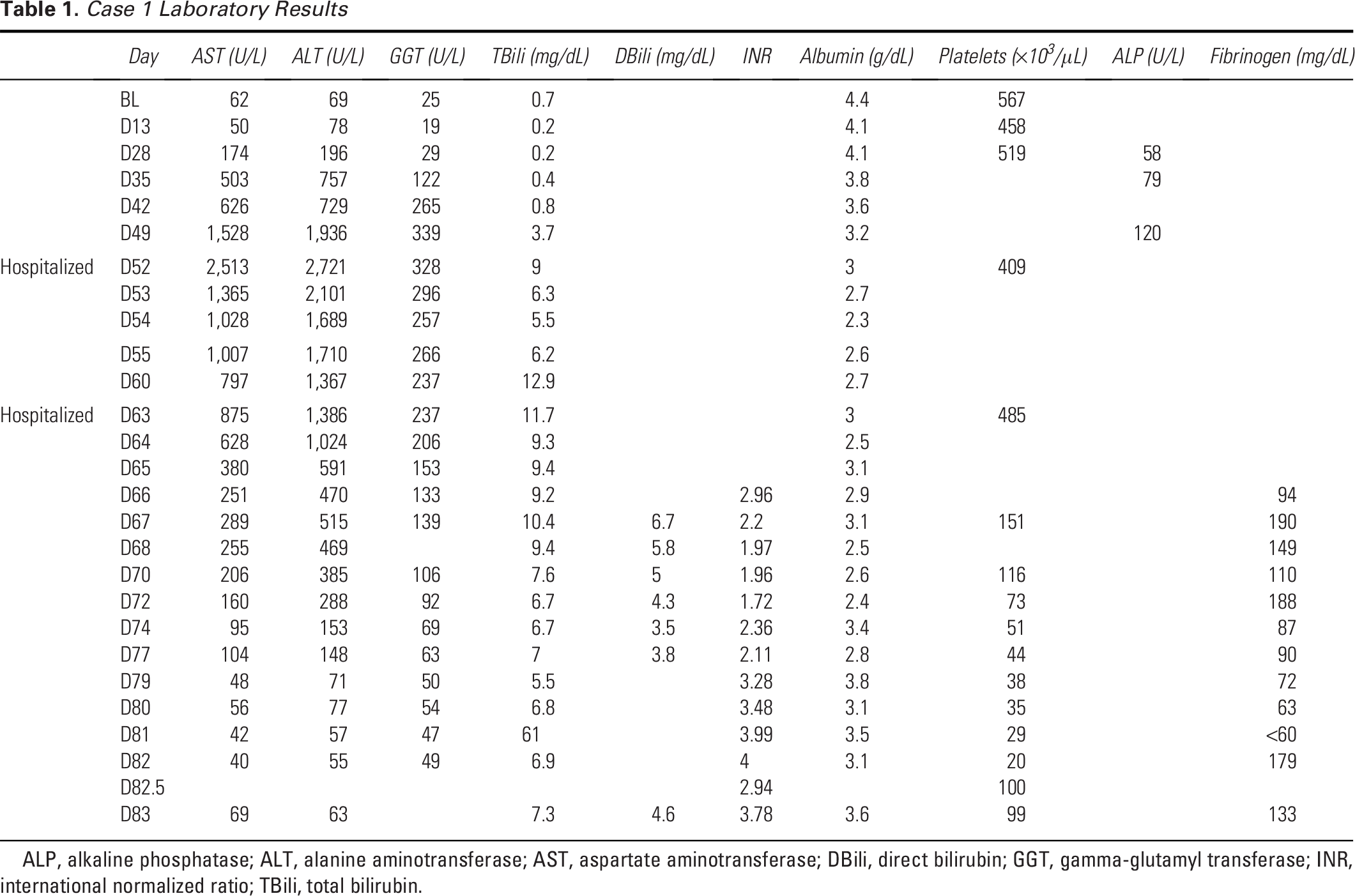
ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; DBili, direct bilirubin; GGT, gamma-glutamyl transferase; INR, international normalized ratio; TBili, total bilirubin.
The following week (Day 35), AST and ALT levels further increased to 503 U/L (8.1×) and 757 U/L (11×), respectively, and GGT level increased to 122 U/L (4.9×); total bilirubin was 0.4 mg/dL (baseline, 0.7 mg/dL). ALP was normal at 79 U/L. Additional follow-up laboratory monitoring on Day 42 showed further increase in transaminases with a GGT of 265 U/L (10.6×); bilirubin remained within normal limits (0.8 mg/dL). Albumin was mildly low at 3.6 g/dL (normal range, 3.7–5.6). On Day 46, prednisone was increased from 60 mg/day to 90 mg/day (∼1.23 mg/kg/day) on days intermittent prednisone for DMD was not given (Fig. 1A). On Day 49, ALT level was 1,936 U/L (28.1×), GGT level was 339 U/L (13.6×), albumin was borderline low at 3.2 g/dL, and total bilirubin was elevated at 3.7 mg/dL. ALP remained normal at 120 U/L.
On Day 52, the patient was advised to go to the emergency department (ED) due to reports of worsening back pain, nausea, malaise, and dark urine. WBC count was 10.6 × 103/μL, and platelets were 409 × 103/μL. Urinalysis showed small red blood cells (RBCs), moderate bacteria with epithelial cells and mucus, urinary bilirubin, and ammonium biurate crystals. The patient’s ALT was 2,721 U/L (39.4×), GGT was 328 U/L (13.1×), albumin was low at 3.0 g/dL, and total bilirubin was 9.0 mg/dL. The patient was admitted for a 3-day pulse of intravenous (IV) methylprednisolone given on Days 52–54 while liver biomarkers were closely monitored (Fig. 1B,C; Table 1). Blood sampling was clinically challenging during this phase of the patient’s course, and contemporaneous coagulation parameters were therefore not available.
Following the 3-day pulse, transaminases trended down (Day 54: ALT, 1,689 U/L; GGT, 257 U/L; and total bilirubin, 5.5 mg/dL) while albumin remained low (2.3 g/dL). The patient was discharged home on oral prednisone 100 mg daily (∼1.37 mg/kg/day). The patient reported abdominal pain and constipation Day 55 post with ALT (1,710 U/L), GGT (266 U/L), and total bilirubin (6.2 mg/dL). On Day 60, ALT and GGT levels slightly trended down (1,367 U/L and 237 U/L, respectively), while total bilirubin peaked at 12.9 mg/dL (10.8× ULN), and sodium and albumin were low (125 and 2.7 g/dL, respectively).
On Day 63, the patient returned to the ED with intermittent lower abdominal pain, urinary frequency, and nausea. Vital signs were notable for tachycardia (heart rate 155 bpm), attributed to discomfort and possible dehydration; the patient was afebrile and hemodynamically stable. The patient was admitted, and laboratory examinations revealed the following: WBC count, 33 × 103/μL; platelets, 485 × 10³/μL; sodium, 123 mmol/L; ALT, 1,386 U/L (20.1×); AST, 875 U/L; GGT, 237 U/L; and albumin, 3.0 g/dL [Grade 1 hypoalbuminemia]) (Fig. 1B,C; Table 1). Urinalysis showed cloudy, amber urine with a large amount of blood, many RBCs, moderate bacteria, presence of nitrites, and elevated glucose and protein. Despite initial concern for urinary tract infection (UTI), subsequent bacterial culture results later ruled out UTI. Abdominal imaging showed a mild stool burden without obstruction. Troponin was normal. Management included IV ceftriaxone and ketorolac, sodium supplementation, and supportive fluids.
On Day 64, the patient remained hospitalized with ongoing liver dysfunction and electrolyte abnormalities. He was switched to cephalexin for treatment of presumed UTI. Despite sodium correction efforts, a subsequent value of 120 mmol/L indicated persistent hyponatremia. Liver biomarkers remained markedly elevated (Fig. 1B,C; Table 1). Albumin was 2.5 g/dL, consistent with synthetic liver dysfunction. Management included IV 25 g of 25% albumin and salt tablets.
On Day 65/66, findings were consistent with ALF,26,27 evidenced by ascites on renal ultrasound and pleural effusions and an elevated INR of 2.96. Hyponatremia (127 mmol/L) and hypoalbuminemia (2.9 g/dL) persisted. Salt tablets, IV albumin, and furosemide were administered. While transaminases trended down (ALT, 591 U/L; AST, 380 U/L; and GGT, 153 U/L), total bilirubin remained elevated at 9.4 mg/dL. CT of the abdomen demonstrated an unremarkable appearance of the liver.
Over the next few days, the patient developed worsening dyspnea and increased pleural effusion with increased BiPAP needs and was transferred to the pediatric intensive care unit (PICU). Laboratories showed persistent liver dysfunction (ALT peaked at 515 U/L, total bilirubin, 10.4 mg/dL) and hypoalbuminemia (as low as 2.5 g/dL). Sodium remained low (nadir 128 mmol/L. Imaging revealed moderate ascites, diffuse edema, and bilateral pleural effusions. Coagulation was impaired (fibrinogen, 94 mg/dL), prompting administration of fresh frozen plasma (FFP) and vitamin K. Echocardiogram demonstrated normal cardiac function. IV ceftriaxone, albumin, and furosemide were administered. Effusions improved with diuresis, but the patient experienced severe pain and anasarca.
From Days 69–71, the patient was transferred out of the PICU with improved respiratory status and chest imaging, though abdominal pain persisted. Prednisone was tapered (from 100 mg/day to 60 mg/day) due to concerns about delayed wound healing. Liver function remained impaired (total bilirubin, 7.6 mg/dL), and albumin remained low (2.6 g/dL). Sodium stayed below normal (131 mmol/L), and platelets declined to 116 × 10³/μL by Day 70. A new skin tear with fluid leakage was noted and attributed to subcutaneous edema. Supportive care continued with diuretics, albumin, and antibiotics.
On Days 72–81, the patient remained clinically fragile with persistent liver dysfunction, coagulopathy, and progressive thrombocytopenia (platelets declined from 73 × 10³/μL to 29 × 10³/μL). Liver enzymes trended down (ALT decreased from 288 U/L to 57 U/L), but total bilirubin remained elevated (peak 6.8 mg/dL). INR rose to 3.99, and fibrinogen dropped below 60 mg/dL, indicating worsening synthetic function. Sodium was normalized; albumin fluctuated, requiring repeated IV supplementation. Diuretics were adjusted, and FFP and platelets were intermittently transfused. The previously identified skin tear demonstrated yellow drainage, ultimately attributed to bilirubin, though with concern for possible purulence initially, topical mupirocin was applied. Imaging showed persistent ascites without abscess. Due to ongoing concerns for wound healing, prednisone dosing was reduced from 60 mg daily, then 40 mg daily, and ultimately 20 mg daily. Epstein–Barr virus and cytomegalovirus (CMV) PCRs were negative, with positive CMV IgM/IgG. Hepatology and infectious disease teams were consulted.
On Days 82 and 83, the patient experienced further rapid clinical decline. On Day 82, the platelet count dropped to 20 × 10³/μL (Grade 4 thrombocytopenia), and INR was 2.94; liver dysfunction persisted (total bilirubin, 6.9 mg/dL). That evening, the patient acutely decompensated with desaturation, agitation, forced eye deviation, and unresponsiveness. Computed tomography (CT) confirmed a large, right and smaller, left intracranial hemorrhage. The patient was intubated and transferred to the PICU; care was transitioned to comfort measures. On Day 83 (Week 12), labs showed persistent liver injury and coagulopathy (platelets 99 × 10³/μL after transfusion; bilirubin, 7.3 mg/dL, INR 3.78). Supportive care was continued. Later that day, the patient was transitioned to comfort care and passed away due to an intracranial hemorrhage secondary to ALF.
With consent from the patient’s family, gross and microscopic autopsy examinations were conducted. Comprehensive postmortem examination revealed multisystem pathology consistent with progressive organ failure. The cardiovascular system showed multiple areas of myocardial fibrosis, remote infarction, myocyte hypertrophy, fat infiltration, and focal arterial intimal proliferation. In the central nervous system, there was an acute hemorrhage in the right and left frontal lobes with intraventricular and subarachnoid extension, along with cerebral edema. The respiratory system exhibited diffuse bronchopneumonia with hyaline membrane formation, patchy pulmonary hemorrhage, and edema, alongside ascites and pleural effusions. Additional findings included hypocellular bone marrow with left-shifted granulopoiesis and reduced megakaryocytes, cutaneous changes consistent with Cushingoid features and striae, and scattered ulcerations on the abdomen, flanks, and thighs. There was lymphoid depletion in the spleen and lymph nodes, and lipid depletion in the adrenal glands, reflecting systemic stress and chronic illness.
Liver pathology showed distorted lobular architecture and areas of steatosis (Fig. 2). Higher magnification revealed a marked ductular reaction and periportal injury, with relative preservation of centrilobular hepatocytes, although many appeared injured. Macrovesicular steatosis was prominent in viable hepatocytes. Notably, inflammatory infiltrates were minimal. Masson trichrome and reticulin staining demonstrated periportal collapse but minimal-to-no significant fibrosis (Fig. 2), a common feature in pediatric ALF indicative of rapid, substantial injury such that the chronic changes of fibrosis were absent, and collapse of lobule architecture is observed.
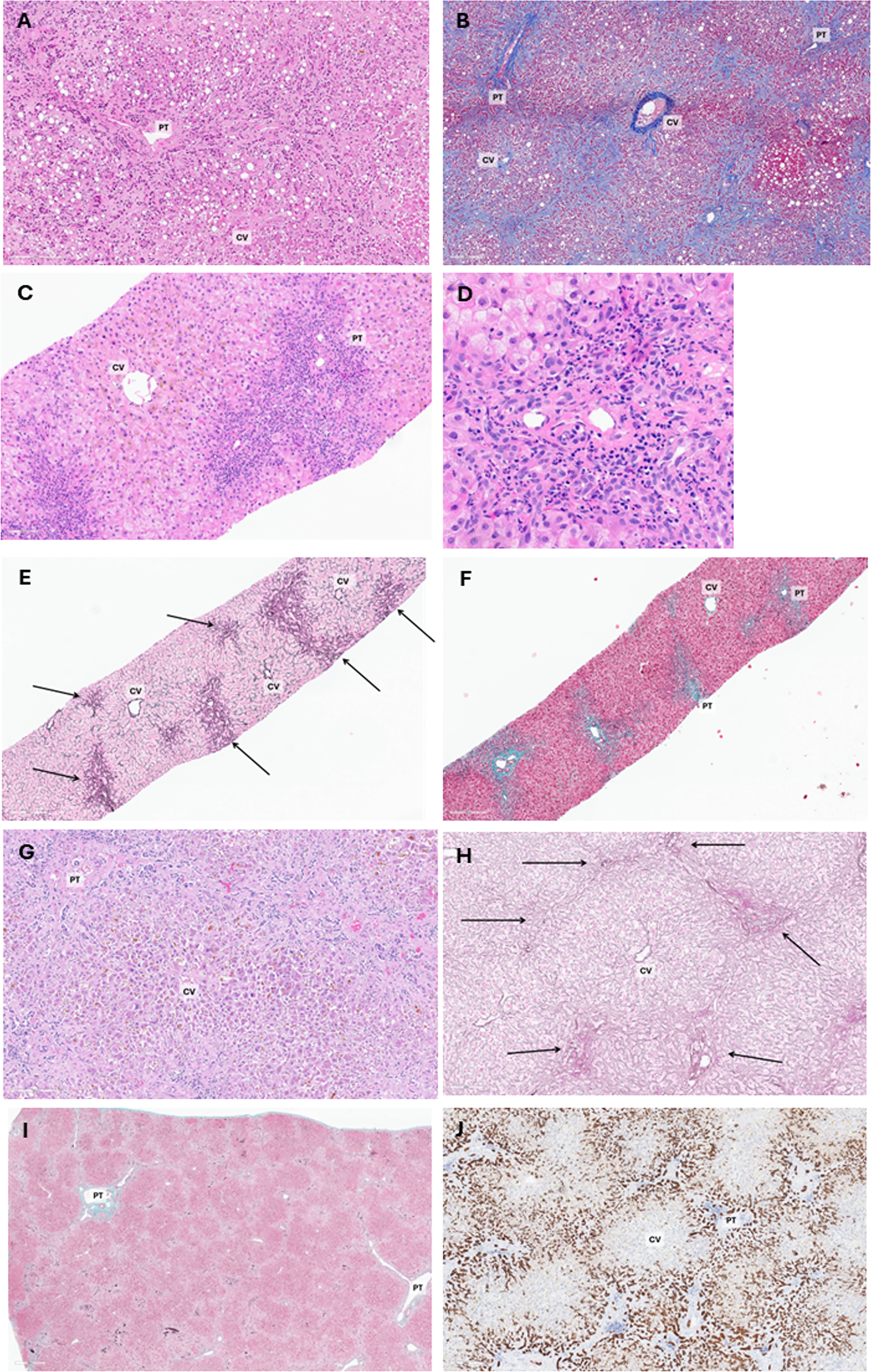
Liver histology showing periportal collapse, marked ductular reaction, and cholestasis without significant fibrosis.
Case 2
The second case of fatal ALF following delandistrogene moxeparvovec occurred in a 15-year-old male (body weight, 54 kg) in Part B of the ENVISION clinical trial (NCT05881408). The patient was diagnosed with DMD (frameshift deletion of exons 46–50) and also lost ambulation at 12 years of age. The patient’s relevant medical history includes alpha-1 antitrypsin type MZ genotype, history of four fractures and treatment with zoledronic acid for osteoporosis. The patient also had a history of resolved myocarditis, a single troponin I elevation, and self-limiting chest pain. Cardiac magnetic resonance imaging performed almost 1 week prior to the infusion was interpreted as having no significant change from prior imaging that showed late gadolinium enhancement, which may be associated with fibrofatty replacement, in the lateral free wall with normal left ventricular dimension and normal ejection fraction of 66%. His DMD was being treated with deflazacort 24 mg daily (∼0.45 mg/kg/day), and other chronic medications included concomitant treatment with lisinopril, carvedilol, testosterone, famotidine, quetiapine, and guanfacine (Fig. 3A). Prior to the delandistrogene moxeparvovec infusion (Day −1), prophylactic prednisone (60 mg/day; ∼1.11 mg/kg/day) was added, per label recommendations.
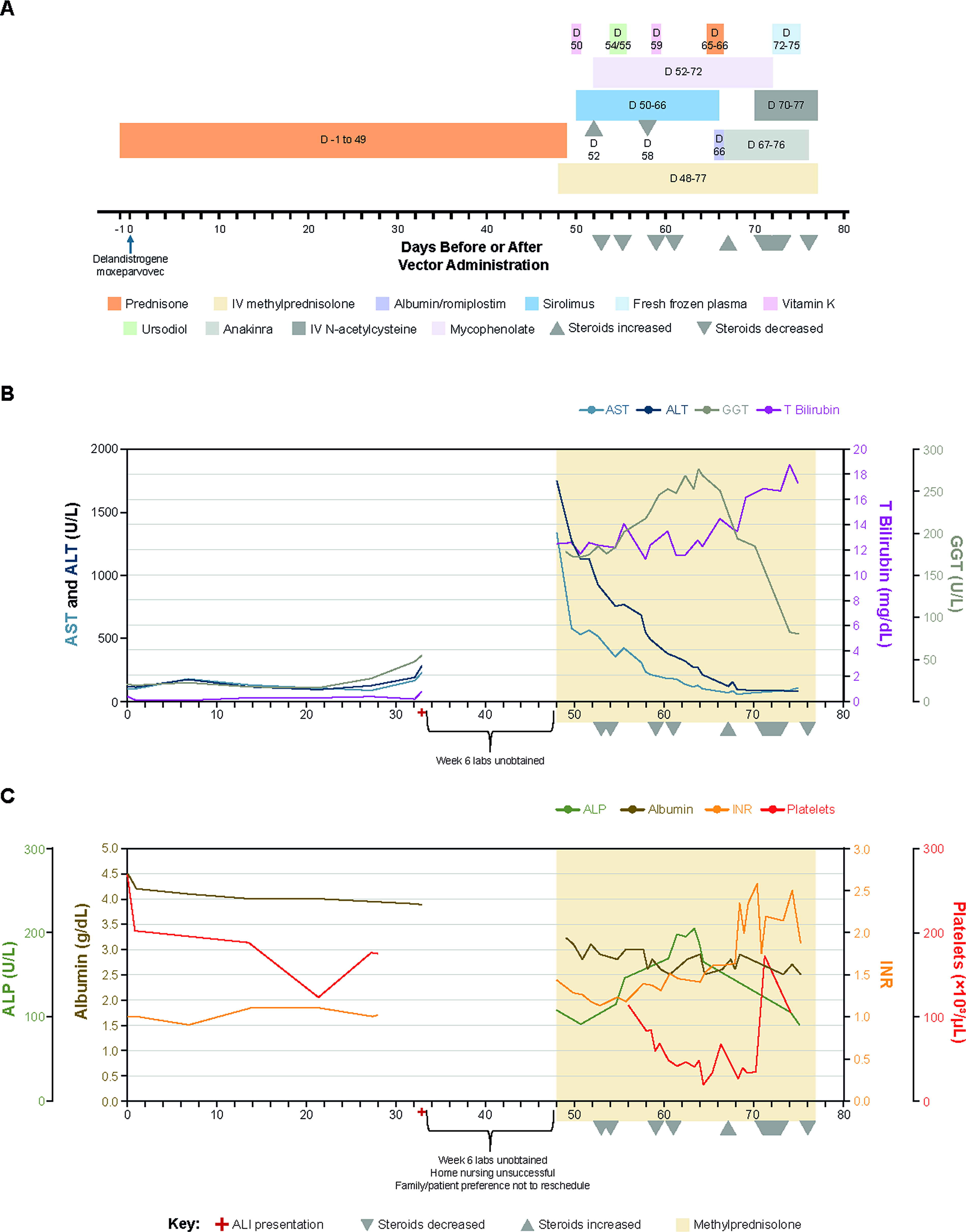
Hepatic biomarker trends in Case 2 following delandistrogene moxeparvovec administration. ALI presentation and management are shown.
On Day 0, the patient received delandistrogene moxeparvovec (7.18 × 1015 total vg). Routine labs on Day 22 showed stable ALT at 96 U/L (baseline, 119 U/L) and AST at 107 U/L (baseline, 101 U/L) (Fig. 3B, Table 2). Total bilirubin and albumin levels were within normal limits (0.3 mg/dL and 4.0 g/dL, respectively). Platelet count was mildly reduced at 123 × 10³/μL; GGT and GLDH (test available for DMD clinical trials but not commercially available at the time) were normal. Day 28 laboratories showed the following: ALT, 127 U/L; AST, 89 U/L; GGT, 28 U/L; total bilirubin, 0.4 mg/dL; platelets, 176 × 10³/μL; and complement component 4 (C4), 15 mg/dL.
Case 2 Laboratory Results
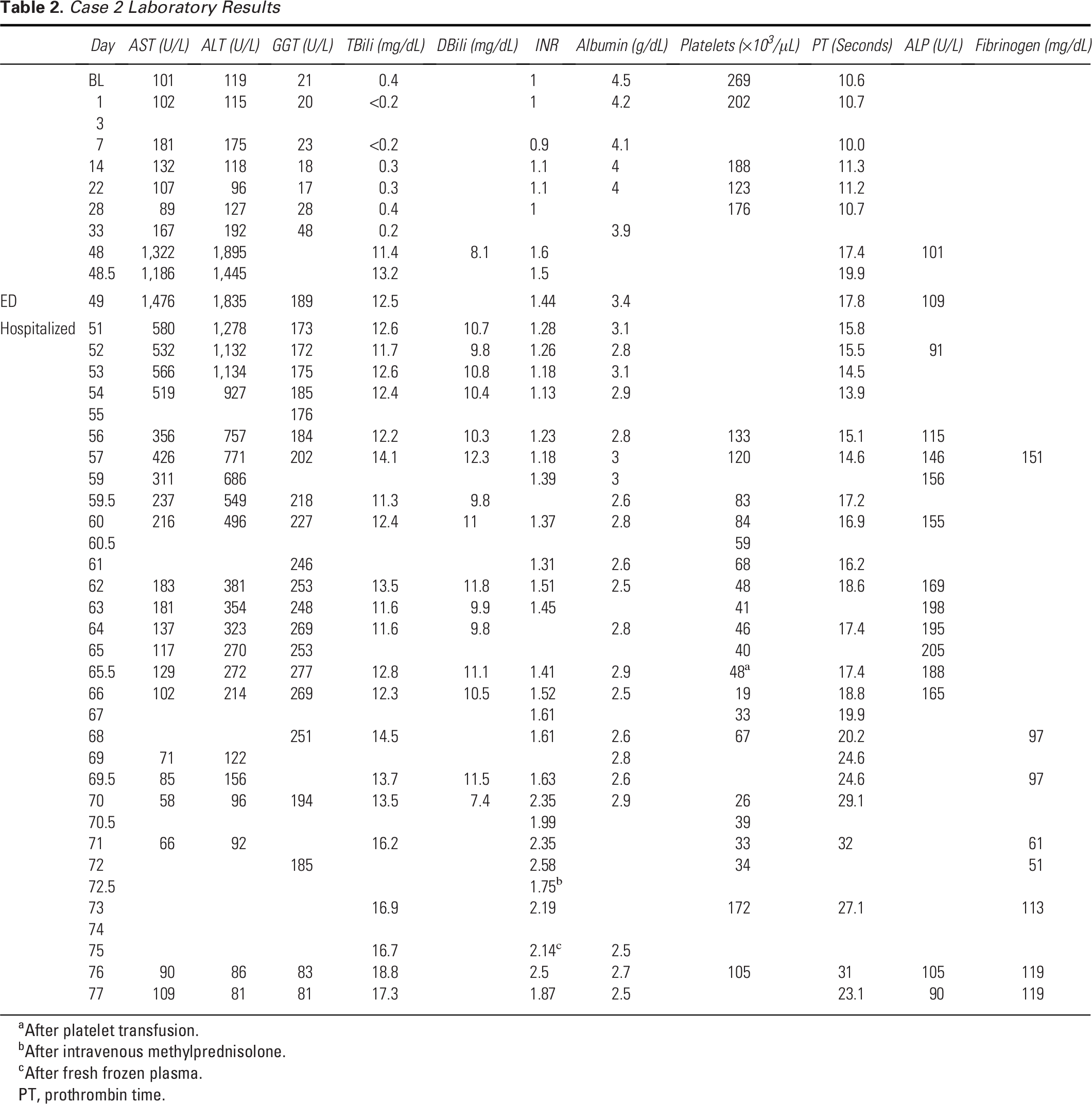
After platelet transfusion.
After intravenous methylprednisolone.
After fresh frozen plasma.
PT, prothrombin time.
On Day 33, GGT levels increased to 48 U/L (2.3×; baseline 21 U/L; reference range, 0–42 U/L); ALT, AST, total bilirubin, and albumin levels remained near baseline levels (Fig. 3B; Table 2). Around this time (on an unspecified date), the patient complained of abdominal pain, but it was not reported to the study team for 1 week. Home phlebotomy services were unable to obtain the scheduled blood samples per protocol during postinfusion Week 6 (Days 34–47).
On Day 48, the patient presented to the ED with mild epigastric pain, dark urine, and jaundice, without emesis or chest pain. Laboratory evaluation revealed elevated ALT (1,895 U/L; 15.9×), AST (1,322 U/L; 13.1×), and GGT (197 U/L; 9.4×). Total bilirubin was 11.4 mg/dL (28.5×; baseline, 0.4 mg/dL). Coagulation parameters were abnormal, with INR 1.6 and prothrombin time 17.4 s. There were no signs of ascites or encephalopathy. Urinalysis showed cloudy, amber urine with marked glucosuria (500 mg/dL) and bacteriuria (+1).
On Day 49, the patient was admitted to the ICU for close monitoring and management of progressing ALI. Pulse dosing of methylprednisolone was initiated (Fig. 3A). A viral work-up was also underway. Right upper quadrant ultrasound interpretation showed diffusely increased hepatic echogenicity, consistent with fatty infiltration (similar to that observed nearly 4 years prior). The gallbladder was described as contracted without peri-cholecystic fluid and biliary ductal dilation. Echocardiogram revealed normal ventricular size and systolic function, with no pericardial effusion or significant changes from prior studies. Hepatobiliary laboratory findings showed the following: AST, 1,476 U/L (14.6×); ALT, 1,835 U/L (15.4×); GGT, 189 U/L (9×); and total bilirubin 12.5 mg/dL (31.3×). Albumin was decreased (3.4 g/dL), and INR remained elevated (1.44) although improved since last measurement (Fig. 3B). Coagulation factors were notable for low factor VII (20%; normal range, 57–139%), low factor X (50%; normal range, 73–128%), and borderline-low factor V (60%; normal range, 60–138%), concerning for synthetic liver dysfunction and vitamin K deficiency. Electrolytes and blood gases showed low ionized calcium (1.12 mmol/L), elevated venous oxygen saturation (93.7%), and elevated pO2 (66.9 mmHg). Inflammatory markers were markedly elevated: interleukin-8 (IL-8; known to be elevated in sepsis and cholestasis,28,29) 155.0 pg/mL and IL-10 (anti-inflammatory cytokine responding to elevated interferon [IFN] 30 ) 10.0 pg/mL. Sirolimus (1 mg TID then 1 mg daily thereafter) was initiated on Day 50 to treat a presumed adaptive immune response causing ALI.
On Day 51, the patient remained clinically stable; troponin was normal, albumin was slightly lower, and INR was lower (1.28). Total bilirubin increased to 12.6 mg/dL (31.5×), and jaundice improved. Transaminases remained elevated but were trending down (ALT, 1,278 U/L [10.7×] and AST, 580 U/L [5.7×]). Albumin continued to trend down at 3.1 g/dL, and total protein was low at 5.4 g/dL; GGT was lower at 173 U/L (8.2×) (Fig. 3B,C; Table 2). Sodium was within normal limits (sodium, 138 mmol/L), and calcium was low at 7.8 mg/dL. First sirolimus trough level was subtherapeutic at 4.1 ng/mL (target, 10–12 ng/mL, a higher range similar to that used in liver allograft acute T-cell-mediated rejection, given the severity of injury). The patient was transferred to the liver transplant service.
On Day 52, the patient developed mild hyponatremia and thrombocytopenia (sodium, 136 mmol/L); platelet count was 140 × 103/μL. Liver biomarkers remained elevated (Fig. 3B,C; Table 2). Total bilirubin was 11.7 mg/dL, with direct bilirubin 9.8 mg/dL (32.7×). Albumin continued to drop to 2.8 g/dL, as did total protein to 5.1 g/dL. INR was lower at 1.26. ALP dropped to 91 U/L. Sirolimus dose was increased to 2 mg daily, and daily IV methylprednisolone was tapered to 750 mg following completion of the 5-day pulse at 1 g/day. Mycophenolate was started for additional T-cell modulation.
On Day 53, total bilirubin trended up at 12.6 mg/dL while liver enzymes remained stable (AST, 566 U/L; ALT, 1,134 U/L; and GGT, 175 U/L). Sirolimus trough level was 8.7 ng/mL. On Day 55, ursodiol was added for ongoing total bilirubin elevation (12.4 mg/dL on Day 54) and the attempt to more aggressively wean IV methylprednisolone was slowed down due to an increase in GGT (185 U/L). Sirolimus trough level was 13.1 ng/mL.
On Day 56, a liver biopsy showed marked ductular reaction with periductular neutrophils and marked lobular cholestasis with no significant ongoing hepatitis. Albumin remained low at 2.8 g/dL, total bilirubin remained elevated at 12.2 mg/dL, and persistent mild thrombocytopenia (platelet count, 133 × 103/μL) was noted. On Day 57/58, the sirolimus trough level was supratherapeutic at 17.2. Laboratory findings continued to show persistent liver dysfunction and mild thrombocytopenia (Table 2). Ultrasound showed a slight increase in echogenicity of liver parenchyma, which might have reflected hepatic steatosis versus other hepatic pathology.
On Days 58–60, IV methylprednisolone was decreased to 250 mg due to a decline in ALT and AST (686 U/L and 311 U/L, respectively); sirolimus was decreased to 1 mg daily because of high trough values, and the dosage of mycophenolate mofetil was increased to 1,000 mg twice daily. Factor VII remained low at 24% (normal range, 57–139%) despite vitamin K replacement, while factor VIII was markedly elevated to 798% (range, 49–191%), concerning for hepatocyte synthetic dysfunction. Platelets declined further to 59 × 103/μL. Immature platelet fraction was high to 18.2% (range, 1.6–6.1%), which was discussed as potentially a consumptive thrombocytopenia.
On Day 62, laboratory values were as follows: ALT, 381 U/L; AST, 183 U/L; GGT, 253 U/L; total bilirubin, 13.5 g/dL; platelets, 48 × 103/μL; and albumin, 2.5 g/dL. On Day 64, von Willebrand factor antigen and activity were extremely elevated outside the measurable range with values > 1,000% (range, 35–197%) and > 390% (range, 47–151%), respectively. On Day 65, laboratory values trended down (ALT, 272 IU/L; AST, 129 IU/L; and total bilirubin, 12.8 mg/dL). Oral prednisone was decreased to 120 mg/day. Cytokine panel revealed IL-6 was 11.9 pg/mL (range, 0–3.5 pg/mL), IL-8 was 1,220 pg/mL (range, 0–10.0 pg/mL), and IL-13 was 40.3 pg/mL (range, 0–2.5 pg/mL). Echocardiogram showed trivial pericardial effusion. There were no schistocytes on the peripheral blood smear. Terminal complement complex (sC5b9) remained within normal limits (evaluated on Days 50, 51, 65, and 70) as did ADAMTS13 (evaluated on Days 49 and 55).
On Days 66/67, platelets decreased further to 19 × 103/μL, and romiplostim was started to promote platelet production. Anakinra was also started given the elevated cytokine results. The patient complained of intermittent abdominal discomfort felt to be gastrointestinal reflux +/− constipation. Prednisolone was increased back to 250 mg/day. On Day 68, laboratories were as follows: platelets, 67 × 103/μL; fibrinogen, 97 mg/dL; GGT, 251 U/L; total bilirubin, 14.5 mg/dL; albumin, 2.6; INR, 1.61; and ferritin, 2,248 ng/mL (ULN, 82.8 ng/mL). The patient received IV caspofungin (70 mg once) and IV ampicillin (2,000 mg).
The patient was transferred back to the ICU on Day 69 because of severe abdominal pain and altered mental status. The patient reported improvement with pain following administration of nalbuphine and found to have a normal neurologic and mental status examination. Abdominal CT showed fat stranding around the duodenum and jejunum (i.e., duodenitis), which was potentially steroid-related, cystitis, ascites, and a small pleural effusion; the biliary system could not be evaluated well. He was treated for a suspected UTI. On Day 71, ascites was more evident on ultrasound and clinical examination (complaints of abdominal pain).
On Days 72/73, chest X-ray showed worsening pulmonary edema and pleural effusion. The patient was intubated because of worsening fluctuating mental status, with increased ammonia (peak of 118 µmol/L), although not enough for the hepatology team to assume encephalopathy was the only etiology. Sepsis was also considered as a possible etiology of altered mental status given the sudden elevation in WBC count. There was consideration for liver transplantation. However, the patient was not considered a candidate because of concern that the etiology of liver failure could not be definitively isolated to the liver, as the team raised the possibility of recurrence in an allograft, and because the individualized prognosis in this case was assessed as poor, with transplantation anticipated to prolong suffering and compromise quality of life. INR increased to 2.58. The patient was treated with one dose of IV methylprednisolone 1 g, with a plan to decrease to 500 mg daily, and INR dropped to 1.75. Bleeding and oozing around the endotracheal tubbing and IV sites were noted.
On Days 74–76, the patient remained sedated, intubated, and treated with N-acetylcysteine, epinephrine for hypotension, continuous renal replacement therapy (CRRT) to correct fluid balance, insulin drip for hyperglycemia, platelets, FFP, and a blood transfusion for anemia (hemoglobin, 7.3 g/dL). Bleeding continued from the oropharynx. Blood cultures were negative, and broad-spectrum antibiotics were discontinued. Ammonia level was in the 40 s despite minimal stool output. Fluid overload prompted initiation of furosemide for diuresis.
On Day 77, the patient developed a systemic inflammatory response characterized by hyperammonemia, coagulopathy, hyperbilirubinemia, encephalopathy, myocarditis with arrhythmia, and fluid overload. Clinical deterioration continued despite intensive medical support, including escalation of vasoactive therapy, electrolyte repletion, and transfusion support. Imaging with abdominal CT revealed bowel ischemia with pneumatosis, though no free air or confirmed perforation was present. The patient became oliguric and increasingly fluid overloaded, requiring continuation of CRRT. Hemodynamic instability persisted, and the patient was not considered a surgical candidate. After multidisciplinary evaluation and discussion with the family, it was determined that further interventions were unlikely to reverse the clinical trajectory. The family elected to prioritize comfort and compassionate end-of-life care. The patient passed away due to complications of progressive acute decompensation with uncontrollable hemodynamic instability resulting from liver failure. 31
With consent from the patient’s family, gross and microscopic autopsy examinations were conducted. Autopsy findings included small stature, adrenal insufficiency due to chronic steroid use and small liver size with signs of liver failure. No significant abnormalities were found in the brain on gross examination, and cultures from the spleen and lung showed no fungal or bacterial growth.
Liver pathology showed a similar pattern of injury as that observed in the premortem biopsy, with progression of periportal collapse, ductular reaction, and lobular cholestasis (Fig. 2). Only a few scattered portal lymphocytes were seen. Gomori trichrome staining again showed no significant fibrosis.
EXPERT PANEL RECOMMENDATIONS
Compounding mechanistic explanations of ALI
Advisors debated whether ALI and ALF exist on a spectrum; most suggested that a secondary insult may differentiate fatal cases. One proposed two-hit hypothesis, which included an initial endothelial or coagulation insult that may be compounded by progressive microvascular injury. Parallels were drawn to the fatal X-linked myotubular myopathy AAV gene therapy cases in which the patients who had predisposing underlying liver disease due to a disease-inherent cholestatic phenotype were similarly resistant to all therapy. 32 The resistance was hypothesized to be cytokine-driven, prompting trials of immunomodulatory agents, including anakinra. 33
Advisors also proposed a role for T cell-mediated liver injury as the leading hypothesis for ALI following treatment with delandistrogene moxeparvovec as described by others. 23 Although there are no data examining T-cell immunity or direct toxicity from the micro-dystrophin transgene product for either patient, a recent case report of a 10-year-old nonambulatory patient with DMD who had presented with ALI after receiving delandistrogene moxeparvovec showed evidence of CD3+ T-lymphocytes in liver biopsy. 34 In this model, following hepatocyte transduction, proteosomal processed capsid proteins are presented by major histocompatibility complex (MHC) class I molecules, resulting in clearance of the transduced cells by CD8+ T cells. 23 Antigen-presenting cell uptake of AAV vectors can also result in activation of CD4+ T helper cells. In Study 101, T cell responses to AAVrh74 peptides (i.e., IFNγ enzyme-linked immunospot assay [ELISpot]) were detected at varying timepoints post-dosing (2–12 weeks). 17 This mechanism is consistent with the striking periportal injury noted at autopsy as the periportal hepatocytes are likely the highest transduced cells. Complement-mediated injury was hypothesized to not be implicated, given the late evolution of the ALI compared with complement-mediated injury that typically occurs early after dosing, as well as the lack of laboratory studies indicative of complementation activation.
Advisors noted the need for future analyses of demographics, genetic factors, hepatic variables, trends, and liver conditions compared with the entire treatment population to uncover potential mechanistic explanations. Because of the lack of evidence for CD8 activation in the liver tissue in both cases, further immune studies elucidating the mechanism of injury are also warranted. Advisors described the value of investigating CD8+ T cell responses to AAV capsid proteins. Advisors also described the value of using tools as well as human leukocyte antigen (HLA) typing and tissue spatial transcriptomics for hypothesis-generating work (e.g., ELISpot) as it is possible that they may have a rare HLA-type or some other host factor that makes them prone to ALF.
Identify early indicators of ALI and define populations at elevated risk for ALF
Demographic and medical history similarities
Advisors reviewed the demographic and medical histories of each patient and discussed several hepatic risk factors that were present. Both patients were of similar age and nonambulatory (Table 3). Although both weighed ≥50 kg, no relationship has been observed between liver clinical safety biomarkers and delandistrogene moxeparvovec vector genome exposure. 35 Other commonalities in their past medical history included ADHD, hypogonadism, as well as some level of steatosis noted on ultrasound of the liver. As with most patients with DMD, both had a history of chronic steroid use and evidence of osteoporosis or osteopenia.
Comparison of Fatal ALF Cases following Delandistrogene Moxeparvovec

Administered as standard of care for DMD.
A1AT, MZ, alpha-1 antitrypsin type MZ; ADHD, attention-deficit/hyperactivity disorder; ALT, alanine aminotransferase; COD, cause of death; DMD, Duchenne muscular dystrophy; FFP, fresh frozen plasma; GGT, gamma-glutamyl transferase; ICH, intracranial hemorrhage; IV, intravenous; MMF, mycophenolate mofetil; NAC, N-acetylcysteine; PRBC, packed red blood cells; rAAVrh74, recombinant adeno-associated virus rhesus isolate serotype 74; US, ultrasound; vg, vector genome.
Each patient also had a distinct hepatic risk factor considered by the committee. In Case 1, advisors considered an early intercurrent respiratory illness—presumed viral and clinically improving several weeks after a caregiver-reported fever—as a potential contributing factor, noting that viral infections can be accompanied by transient aminotransferase elevations. Convalescent serologic testing was of interest to the experts, but could not be pursued because stored serum was unavailable. During this interval, there were no clinical features of coagulopathy, cholestasis, or ALF, and laboratory findings were limited to elevated aminotransferases; coagulation testing was not performed. Advisors considered that earlier assessment in the overall course, when concern for liver function emerged, might have offered additional context regarding evolving hepatic involvement.
In Case 2, the patient had an alpha-1 antitrypsin Pi*MZ genotype, which has been associated with increased susceptibility to liver disease in the presence of additional hepatic stressors (e.g., metabolic dysfunction, intercurrent illness, and inflammatory stress).24,36–38 Although not part of the expert committee discussions, clinical experience indicates heterogeneous clinical outcomes among PiMZ recipients, consistent with a context-dependent risk profile.
Advisors also reviewed liver pathology, which showed that both patients had periportal collapse with ductular reaction, lobular cholestasis, and minimal-to-absent fibrosis (Fig. 3), suggestive of acute liver damage. There was no evidence of chronic liver injury and no significant inflammatory infiltrates, which may have been due to several weeks of intensive immunosuppression or an alternate mechanism of liver failure.
Both patients also showed hematologic abnormalities, including thrombocytopenia with high immature platelet fraction and coagulopathy with markedly low fibrinogen, probably due to a combination of consumption and synthetic dysfunction. Neither case exhibited increases in RBC schistocytes to suggest thrombotic microangiopathic processes, such as disseminated intravascular coagulation, though both had profound coagulopathy, including severe hypofibrinogenemia. This hematologic profile was distinct from typical gene therapy responses.
The advisors did not identify a single hepatic risk factor in either patient that could predict progression from ALI to ALF. They also acknowledged that the events that progressed to liver failure may be the most severe end of a spectrum, including ALI, or may be a separate pathophysiologic process altogether. They hypothesized that multiple compounding variables may have contributed to ALF (Box 2), many of which are common, interrelated features of the typical disease course to DMD in advanced stages. For example, while low bone mineral density in DMD is not a direct hepatic risk factor, it is reflective of chronic corticosteroid exposure, malnutrition, and ambulatory status, all attributed to advanced disease. These upstream conditions may be associated with reduced physiologic reserve, including potential vulnerability to hepatic stressors. Advisors also discussed potential mitigation strategies for specific risk factors (e.g., acute illness prevention).
Box 2. Potential Hepatic Risk Factors
Chronic corticosteroid use Low bone density Metabolic syndrome Steatosis Ambulatory status Medications metabolized by CYP3A4 Acute illness Alpha-1 antitrypsin type MZ genotype
Hepatic biomarker trends
Review of hepatic biomarker levels following delandistrogene moxeparvovec administration showed a rapid rate of rise in ALT over baseline followed by increasing GGT with subsequent total bilirubin and INR elevations in both patients, and both met the criteria for Hy’s law. Case 1 had actively prolonged INR without FFP while the INR for Case 2 was initially mildly abnormal but trending upwards throughout the course.
Advisors also reviewed hepatic biomarker trends in ALI cases that did not progress to ALF. Most of these events resolved with appropriate management, including corticosteroid dose modification based on GGT ≥ 150 U/L and/or other clinically significant liver function abnormalities (e.g., total bilirubin > 2× ULN) following infusion. 1 Although the time of onset (ALT increase) in both ALF fatalities was similar to those observed in the non-progressing ALI cases, declining ALT/AST with persistently elevated or declining GGT with elevated total bilirubin were seen in both fatal ALF cases consistent with progressive hepatocyte burnout and periportal collapse (Fig. 4). Furthermore, INR is generally not elevated, and total bilirubin elevations are rare (and lower in magnitude) in ALI in contrast with the persistent synthetic dysfunction observed with the ALF cases. Likewise, the hematological abnormalities of thrombocytopenia and hypofibrinogenemia were not observed in non-progressing ALI cases. Refractory hyponatremia also appears to be a worrisome sign.
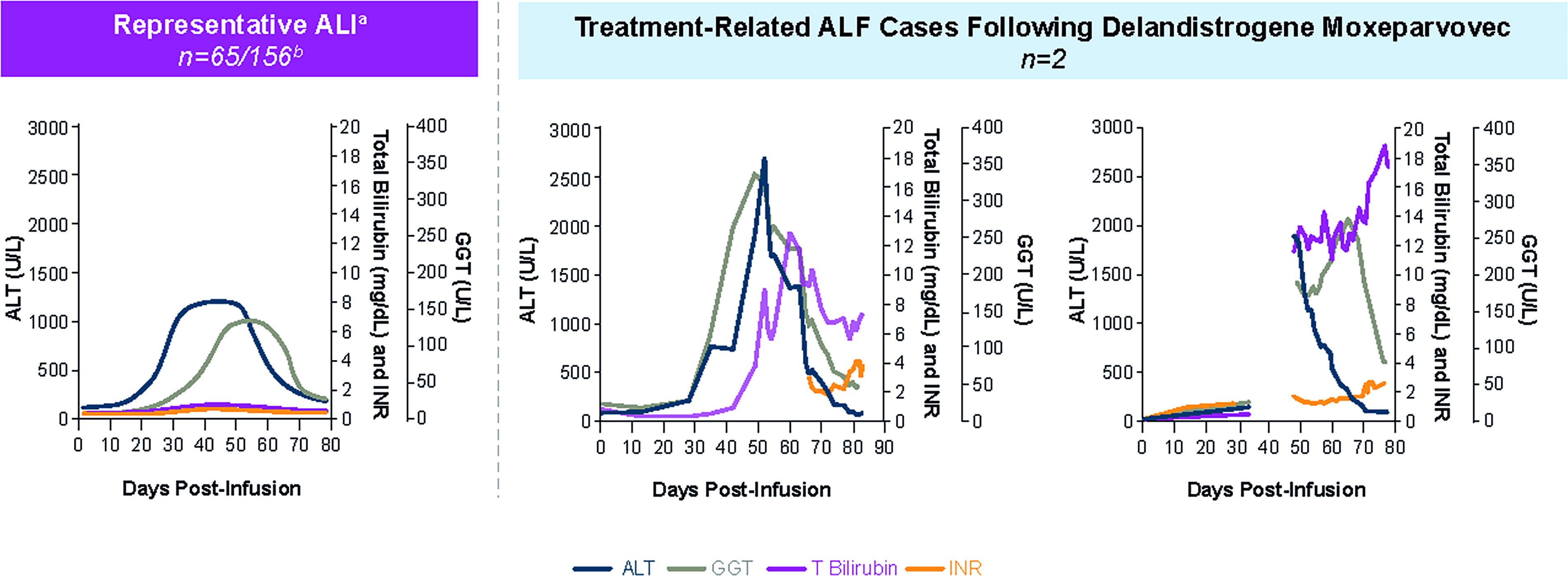
Representative hepatic biomarker trends in ALI compared with the fatal ALF cases.
Advisors noted that a late finding of declining aminotransferases, along with GGT, with a simultaneous continued rise in total bilirubin as an ominous sign in both fatal ALF cases, highlighting the severity of the hepatic injury and resulting in irreversible hepatocyte injury. Advisors noted that once ALF has been identified, modalities to support liver function should be considered (including evaluation for possible transplant if clinically appropriate).
Optimized clinical management approaches for ALI
Advisors considered a variety of ALI management strategies, including current oral prednisone dose, increasing current oral prednisone dose, IV methylprednisolone, IV immunoglobulin (IVIG), sirolimus, a combination of immunosuppressants, and other immunosuppressant (e.g., tacrolimus). Prednisone has been widely used both proactively and reactively in response to aminotransferase elevations that occur after treatment with AAV-based gene therapies. In addition, the use of IVIG in two pediatric cases of ALI following treatment with delandistrogene moxeparvovec was recently described. 22 Ursodiol, in addition to corticosteroids have also been used for management of ALI with hyperbilirubinemia management following delandistrogene moxeparvovec. 39 Prophylactic sirolimus and tacrolimus have also been trialed in other studies to limit AAV gene therapy-induced hepatotoxicity.40,41 In a patient with DMD treated with delandistrogene moxeparvovec who was hospitalized with ALI (peak GGT, 529 U/L), both sirolimus (1 mg daily with a goal trough of 4–6 ng/mL) and two doses of basiliximab (10 mg) were administered as induction therapy following failure of first-line methylprednisolone and sub-optimal response to the second-line IVIG therapy. 42
While some advisors initially supported increasing oral prednisone dose and switching to IV methylprednisolone when response is poor, the majority supported immediate escalation to IV methylprednisolone in cases with rapid, substantial aminotransferase elevations to mitigate the inflammatory injury response. The initial switch to IV methylprednisolone should be initiated within days of the first evidence of a change in GGT. Advisors also supported earlier initiation of IV methylprednisolone at lower hepatic thresholds (discussed below), particularly if aminotransferase elevations appear rapid and substantial (e.g., ALT or GGT nearly doubling in 1 week). Advisors emphasized the need for real-world data and investigator protocols to inform practice.
Strategies to prevent and mitigate ALI and ALF
Potential approaches for earlier detection of ALI
Overall, advisors highlighted the need for earlier intervention before or when approaching hepatic biomarker thresholds. They recommended lowering the current ALI treatment thresholds that would trigger intervention. Because baseline ALT and AST are often elevated in patients with DMD, GGT, and GLDH have typically been used to monitor liver toxicity. Advisors also did not recommend using an absolute value, such as GGT ≥150 U/L, as a threshold for intervention as all agreed rapid/substantial changes require immediate treatment. Advisors agreed that GGT thresholds should be based on patients’ baseline values as they may help mitigate assay variability given the wide range of ULN values between laboratories and the age-dependent differences in ULN thresholds. Some advisors suggested intervening before or when approaching hepatic biomarker thresholds (e.g., GGT 2× baseline value or total bilirubin >ULN). Finally, they emphasized the importance of increased frequency of aminotransferase monitoring in the setting of abnormalities/treatment to ensure that the intervention is having an impact.
Potential approaches for capturing risk
Advisors recommended consideration of enhancing baseline liver characterization with lipid panels, transient elastography (e.g., FibroScan), and ultrasound. Potential for evaluating steatosis risk was also discussed, particularly in nonambulatory patients as they often experience significant weight gain and elevated body mass index, 43 moderate-to-severe hepatic steatosis has been reported in approximately two-thirds of these patients. 44 It was also noted that standard of care corticosteroid treatment for DMD can contribute to insulin resistance and steatosis risk. 44 However, there was general consensus among the advisors that there is insufficient data at present to label steatosis as a risk factor for ALF although its role in an overall cumulative constellation of multiple risk factors needs to be further considered.
Potential approaches for improved ALI prevention
Advisors considered three different prophylactic immunosuppression strategies for the prevention of ALI: (1) increasing corticosteroid regimen without adding additional prophylactic agents, (2) current corticosteroid regimen 1 plus prophylactic sirolimus, and (3) increased corticosteroid regimen plus addition of either prophylactic sirolimus (mTOR inhibitor) or tacrolimus (calcineurin inhibitor). Although no prophylactic immunosuppressive regimen has been demonstrated to significantly reduce AAV-related hepatotoxicity, these options were selected as corticosteroids and sirolimus are the most commonly used immunosuppressive agents in systemic AAV gene therapy programs. 33 Moreover, sirolimus targets T-cells, inhibiting their activation and proliferation, and hepatotoxicity following AAV-based gene therapy is hypothesized to be at least in part driven by an adaptive immune response to the viral capsid (largely T-cell-mediated). 45 Indeed, a case report of ALI in a patient following delandistrogene moxeparvovec showed moderate mixed portal inflammation (predominantly T-cells), 34 a feature noted in another case of ALF following AAV-based gene therapy. 46
Most advisors preferred addition of prophylactic sirolimus to the current corticosteroid regimen 1 over escalating corticosteroid doses, though some advisors remained if the addition of prophylactic sirolimus would have a significant clinical effect. Higher steroid exposure was deemed excessive due to safety and tolerability risks related to high doses of corticosteroids, particularly in older and heavier patients; behavioral side effects, weight gain, and psychiatric symptoms were also noted. Advisors recommended avoiding prolonged use of 2 mg/kg/day corticosteroid dosing; pulse IV methylprednisolone was proposed as a more effective and tolerable escalation strategy. For steroid-naïve patients, initiation of prophylactic corticosteroids 7 days prior to infusion was recommended as described in delandistrogene moxeparvovec prescribing information. 1
Sirolimus regimen recommendations included initiating sirolimus 1–2 weeks prior to infusion with a target trough of 2–4 ng/mL with several advisors considering a wider target trough range of 2–8 ng/mL. Advisors also considered maintaining sirolimus for 8–12 weeks postinfusion, given the timing of AAV-associated ALI and monitoring trough levels every 2 weeks unless clinical changes warrant more frequent checks. Advisors recommended discontinuing based on clinical and laboratory parameters. Some advisors initially supported universal prophylaxis. However, most ultimately favored trialing prophylactic immunosuppression in high-risk subgroups first due to limited mechanistic data and potential for overtreatment. Recent evidence of the potential for increased transgene expression with prophylactic therapy would favor early treatment for all patient ages independent of other risk factors.
SUMMARY
While no individual risk factor was identified to consistently predict ALF, advisors hypothesized that there were likely multiple compounding variables. Advisors suggested enhancing baseline liver characterization (e.g., lipid panels, transient elastography, and ultrasound) to better understand risk factors of developing ALF. Advisors recommended lowering thresholds for hepatic biomarkers that prompt additional treatment and emphasized the use of IV methylprednisolone for cases involving rapid, significant changes in liver biomarkers, aiming to mitigate inflammatory injury. They also strongly recommended increased monitoring to ensure the efficacy of the intervention. Advisors noted that declining aminotransferases in conjunction with a continued rise in total bilirubin was suggestive of substantial hepatic injury that may be concerning for progression to ALF.
Of the >1,000 patients treated with delandistrogene moxeparvovec as of October 2025, the two cases presented here represent the only ones to experience drug-related ALF. Liver injury is a known risk associated with AAV-based gene therapies.22–24 While the mechanistic underpinnings of gene therapy-related ALI are currently incompletely defined, T-cell-mediated liver injury is thought to play a major role. 23 This is supported by positive impact of sirolimus in suppressing ALI following AAV gene therapy. 41 It is possible that addressing T-cell activity with sirolimus may impact the likelihood of developing ALI or reduce the severity of ALI if it occurs.
There was a general preference for adding prophylactic sirolimus 1–2 weeks prior to delandistrogene moxeparvovec infusion as a second agent to the current corticosteroid regimen for ALI prevention rather than increasing prophylactic corticosteroid intensity. The safety profile of sirolimus was also discussed as being the least likely to cause additional AEs. Most advisors did not ultimately support universal prophylaxis in the absence of a benefit to gene therapy efficacy (e.g., improved transduction) due to the safety profile in younger populations. Most advisors favored trialing prophylactic immunosuppression in high-risk subgroups first due to limited mechanistic data and potential for overtreatment. To this end, the U.S. Food and Drug Administration (FDA) has approved the evaluation of an enhanced immunosuppressive regimen designed to mitigate the risk of ALI and ALF in nonambulatory patients receiving delandistrogene moxeparvovec. Patients will receive sirolimus as part of the immunosuppressive regimen in Cohort 8 of the ENDEAVOR study (NCT04626674), a two-part, open-label, multi-cohort Phase 1 b study assessing the transduction, expression, and safety of delandistrogene moxeparvovec in patients with DMD. Additionally, the delandistrogene moxeparvovec label has been updated to include a black box warning for ALI and ALF, consistent with other AAV-delivered gene therapies, as well as the exclusion of nonambulant patients. 1
While these considerations encompass broad, extensive domain expertise, and decades of clinical and academic leadership, advisors expressed the need for additional studies to better address risk stratification, analyze the utility of prophylactic immunosuppression, and define additional strategies in ALI management.
AUTHORS’ CONTRIBUTIONS
P.B.S.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). C.P.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). T.D.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). C.A.C.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). J.A.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). A.D.S.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). C.G.B.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). J.S.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). B.J.B.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). A.V.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). J.F.B.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). S.M.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). B.J.S.-J.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). B.J.W.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). M.G.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). K.E.G.W.: Conceptualization and writing—original draft (lead) and writing—review and editing (equal). S.M.: Participated/contributed in one or more expert committee panel discussions (equal), conceptualization and writing—original draft (lead), and writing—review and editing (equal). D.A.: Participated/contributed in one or more expert committee panel discussions (equal), conceptualization and writing—original draft (lead), and writing—review and editing (equal). C.M.M.: Participated/contributed in one or more expert committee panel discussions (equal) and writing—review and editing (equal). J.R.M.: Conceptualization and writing—original draft (lead) and writing—review and editing (equal).
Footnotes
ACKNOWLEDGMENTS
The authors thank Diana Bharucha-Goebel, MD of Children’s National Hospital, for her participation and contribution to the expert committee findings, and Vannary Chhay, PharmD, of Sarepta Therapeutics, Inc., for support with the expert committee meetings. Medical writing and editorial support were provided by Marjet D. Heitzer, PhD, of 360 Medical Writing, in accordance with Good Publication Practice (GPP) 2022 guidelines (![]() ).
).
FUNDING INFORMATION
The expert committee meetings and article development were funded by Sarepta Therapeutics, Inc.
AUTHOR DISCLOSURE
P.B.S. reports being a consultant for Novartis, Biogen, Roche, Sarepta Therapeutics, Inc., argenx, Alexion, UCB, Catalyst, Grifols, and CSL Behring, and is receiving grants/research support from Novartis, Biogen, Solid, Pfizer, Sarepta Therapeutics, Avidity, Dyne, UCB, Abcuro, Arcellx, Astellas, Epicrispr Biotechnologies, Arcellx, Arthex, Edgewise, AMO Pharma, NMD Pharma. C.P. participates on an advisory board and is a consultant for Biogen, Sarepta Therapeutics, Inc., AveXis/Novartis Gene Therapies, Genentech/Roche, and Scholar Rock; serves as a speaker for Biogen; and is a Principal Investigator of studies sponsored by AveXis/Novartis Gene Therapies, AMO, Astellas, Biogen, CSL Behring, FibroGen, PTC Therapeutics, Pfizer, Sarepta Therapeutics, Inc., and Scholar Rock. T.D. has been a consultant for Sarepta Therapeutics, Inc. until 8/2025. C.A.C. has been a consultant for Sarepta Therapeutics, Inc. J.A. has no relevant disclosures. Alan D. Salama has no relevant disclosures. C.G.B. is a noncompensated member of Sareta Therapeutics, Inc., scientific advisory board. J.S. is a consultant/advisor for Boehringer Ingelheim, Capricor Therapeutics, Catalyst, Dyne, Immunoforge, Medpace, NS Pharma, Sarepta Therapeutics, Inc., Secretome, Solid, and Wave; serves on the DSMB for Sardocor; has received research funding from Ametris. B.J.B. has no relevant disclosures. A.V. has a consultancy/advisory role with AMO Pharma, AveXis, Biogen, Edgewise Therapeutics, FibroGen, Novartis, Pfizer, PTC Therapeutics, Sarepta Therapeutics, Inc., UCB Pharma, Catalyst, and Scholar Rock; has received research funding from AMO Pharma, Capricor Therapeutics, Edgewise Therapeutics, FibroGen, Muscular Dystrophy Association, Novartis, Parent Project Muscular Dystrophy, Pfizer, RegenxBio, and Sarepta Therapeutics, Inc., and has other relationship(s) with MedLink Neurology for editorial services. J.F.B. is a consultant for Alexion, Argenx, Audentes, AveXis/Novartis, Biogen, CSL Behring, Cytokinetics, Dyne, Edgewise, Entrada, Fibrogen, Genentech/Roche, ITF, Janssen, Marathon, Momenta, NS Pharma, Percheron, PTC Therapeutics, RegenxBio, Sarepta Therapeutics, Inc., Scholar Rock, Takeda, and WaVe, is a speaker for AveXis and Biogen, is a medical advisory council member for Cure SMA, and is a site investigator for clinical trials with Alexion, Astellas, AveXis/Novartis, Biogen, Biohaven, Catabasis, CSL Behring, Cytokinetics, Fibrogen, Genentech/Roche, Ionis, Italfarmaco, Lilly, Janssen, Pfizer, PTC Therapeutics, Sarepta Therapeutics, Inc., Scholar Rock, Summit, and WaVe. S.M. has received compensation for Advisory Board participation from Dyne, Novartis, and Sarepta Therapeutics, Inc. She has received personal compensation as a consultant for Avidity Biosciences. She has received research funding from Dyne, Genentech/Roche, Pfizer, and Sarepta. B.J.S.-J. has no relevant disclosures. B.J.W. is a consultant for Sarepta Therapeutics, Inc. M.G. is an employee of F. Hoffmann-La Roche Ltd and may have stock options. K.E.G.W. and D.A. are employees of Sarepta Therapeutics, Inc. and may have stock options. S.M. was an employee of Sarepta Therapeutics, Inc. at the time of the expert committee meetings and article development and may have stock options. J.R.M. receives compensation from Sarepta as a medical advisor. C.M.M. reports grants from Avidity, Capricor Therapeutics, Catabasis, Edgewise Therapeutics, Epirium Bio, Italfarmaco, Pfizer, PTC Therapeutics, Santhera Pharmaceuticals, Solid Biosciences, and Sarepta Therapeutics, Inc., and has a consultancy/advisory role with Biomarin, Capricor Therapeutics, Catalyst, Edgewise Therapeutics, Italfarmaco, NS Pharma, PTC Therapeutics, F. Hoffmann-La Roche Ltd, Santhera Pharmaceuticals, Solid Biosciences, and Sarepta Therapeutics, Inc. He has received honoraria from Edgewise Therapeutics, PTC Therapeutics and Sarepta Therapeutics, Inc. C.G.B., B.J.B., S.M., and B.J.S.-J. did not receive compensation for participation in the expert committee meetings.
