Abstract
Recombinant adeno-associated virus (rAAV) vectors have emerged as a leading viral vector for in vivo gene therapy because they offer significant advantages over other viral vectors, such as stable physicochemical properties, low pathogenicity, low integration risk, and long-term expression of the transgene. In recent years, the number of rAAV products that have received approval for commercial marketing and clinical trials has grown rapidly, bringing hope for the treatment of refractory and rare diseases. However, rAAV products are highly innovative and complex, and the manufacturing processes to produce them are diverse and rapidly improving. Concurrently, the quality control methodologies and technologies are rapidly advancing and evolving. As biotechnology rapidly advances, there is a heightened need for communication between regulatory authorities and entities applying for rAAV products to be used for in-human trials or commercial marketing. Here, we focus on the discussion of chemistry, manufacturing, and control issues such as the control of adventitious viruses in different manufacturing processes and quality control during the biologics license application of rAAV products. It is expected to provide references and suggestions for the manufacturing and quality control of rAAV vectors, thereby accelerating the high-quality development of gene therapy products.
Keywords
INTRODUCTION
Adeno-associated virus (AAV) vectors belong to the Parvoviridae family, and are nonenveloped, have an icosahedral structure, and a size of 20–26 nm. AAV viral vectors consist of a protein shell (a capsid) protecting a small, single-stranded DNA genome of approximately 4.7 kb. The genome is flanked at each of its ends by inverted terminal repeats (ITRs), which mainly serve as the origin of viral replication and encode packaging signals. The cap gene encodes three overlapping structural viral capsid proteins (VP1, VP2, and VP3), and 60 of these proteins are arranged into an icosahedral structure in an approximate molar ratio of 1:1:10 (VP1:VP2:VP3) to form the capsid. The rep gene encodes four proteins that are required for viral replication, which are named according to their molecular weights: Rep78, Rep68, Rep52, and Rep40. In addition, the App gene encodes the assembly-activating protein in an alternate reading frame overlapping with the cap gene. This nuclear protein provides a scaffold for capsid assembly, facilitates the entry of the AAV genome into the viral capsid, and promotes the assembly and maturation of viral particles.1,2
To date, 12 natural AAV serotypes and more than 100 variants have been isolated and studied as gene delivery vectors, 3 which differ in their targeting and infection efficiency for different human tissues. For example, AAV8 has a high infection efficiency for liver tissue cells, while AAV1 and AAV9 more efficiently transduce cardiac and skeletal muscle cells, respectively. Clinical studies have used AAV vectors for treating a variety of genetic diseases, including alpha-1-antitryspin deficiency, Batten disease, Canavan disease, cystic fibrosis, hemophilia A and B, Leber’s congenital amaurosis, lipoprotein lipase deficiency, Pompe disease, Duchenne and limb girdle muscular dystrophies, and acquired diseases such as Alzheimer’s disease, heart failure, Parkinson’s disease, rheumatoid arthritis4–6 ; AAV2, AAV5, AAV6, AAV8, and AAV9 vectors are widely used in clinical trials.
By May 2025, nine rAAV products have been approved for commercial marketing, although two (Glybera and Beqvez) have been withdrawn. The reason for Glybera’s withdrawal from the market is limited patient demand and high costs, while the reason for Beqvez’s withdrawal is the manufacturer’s strategic resource reallocation. Among them, five rAAV products have been approved by both the European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA) and one has been approved by the Chinese National Medical Products Administration (NMPA) (Table 1).
Recombinant Adeno-Associated Virus Products That Have Received Regulatory Approval for Commercial Marketing

*Glybera (withdrawn in 2017), Beqvez (withdrawn in 2023).
AAV, adeno-associated virus; EMA, European Medicines Agency; FDA, Food and Drug Administration; NMPA, National Medical Products Administration.
Here, we summarize the key chemistry, manufacturing, and control (CMC) issues in the applications and communication for regulatory approval of rAAV products applied in China as well as discuss the control strategies of adventitious viruses and regulatory considerations of critical quality attributes (CQAs) control during the biological licensing application of rAAV products.
CMC GUIDELINES FOR ADVANCED THERAPY MEDICINAL PRODUCTS IN CHINA
Research and development of advanced therapy medicinal products (ATMPs) in China are developing rapidly, and the number of marketing and clinical trial applications has continued to grow since 2018. With the rapid development of biotechnologies such as gene delivery vectors and gene editing tools, ATMPs have brought new hope for many rare genetic and common refractory diseases and continue to progress in clinical trials. Over the past few years, a series of technical guidelines have been issued by the NMPA to effectively standardize and guide the research and development, production, and application of ATMPs (Table 2). These CMC guidelines have established a regulatory system that is conducive for the development of ATMPs in China.
Chemistry, Manufacturing, and Control Guidelines for Advanced Therapy Medicinal Products in China

ATMPs, advanced therapy medicinal products; CMC, chemistry, manufacturing, and control; rAAV, recombinant adeno-associated virus.
CMC CHALLENGES AND REGULATORY CONSIDERATIONS FOR RECOMBINANT AAV PRODUCTS
There are numerous challenges in adventitious agent control as well as quality control during the recombinant AAV (rAAV) manufacturing process, leading to inconsistencies in quality and thus affecting the safety and efficacy of products. Based on the current status of CMC research and applications for rAAV products in China, the main challenges are summarized as follows: (1) The diversity of manufacturing processes, including the selection of cell lines and the introduction of helper viruses, makes it difficult to standardize manufacturing protocols across institutions and for the variety of vectors being produced. (2) The varying degrees of viral components introduced by different manufacturing processes make it difficult to establish industry standards for adventitious virus control, introducing variability in the assessment of both products and contaminants. (3) Both the rAAV vectors being produced and the methods for determining the strength and purity of products are improved iteratively and rapidly, and so, there are significant challenges to establish and validate standardized quality control methods. (4) rAAV products contain a variety of complex process- and product-related impurities, such as empty or partially capsids, residual plasmid DNA, and replication-competent AAV (rcAAV). However, the impact of such impurities on the safety of rAAV vectors remains unclear and there are no unified standards for the limits on impurities in a product in international regulatory guidelines. (5) Potency measurement is a necessary part of product characterization testing, comparability studies, and stability protocols, which are used to establish that a product is consistently manufactured for administration during all phases of clinical investigation. However, due to the complexities of the mechanisms of action of gene therapy products, a single biological or analytical assay may not provide an adequate measure of potency, and so, critical inconsistencies between production batches could be missed by protocols that are generally standardized rather than specific to the gene therapy product being evaluated.
Regulatory considerations for adventitious viruses in different manufacturing contexts
The production of rAAV products is a complex process that requires delivery of endogenous AAV genes such as rep and cap, a therapeutic gene expression cassette containing ITRs, and helper virus genes into production cells, which translate viral proteins, encapsidate the therapeutic gene, and secrete rAAV particles. Currently, there are three commonly used manufacturing platforms that have undergone significant development and optimization: (1) The triple-plasmid transient cotransfection system in HEK293 cells. (2) The baculovirus-insect cell system in Sf9 cells. (3) The helper virus production system.
The triple-plasmid transient cotransfection system has good scalability. Suspension cells are suitable for large-scale commercial production, but this process is associated with low transfection efficiency and high production costs. In addition, adherent cells are difficult to scale up, and although the risk of viral contamination in suspension cells is reduced, it is not completely eliminated. The baculovirus-insect cell system also has good scalability and enables high-yield, large-scale production, yet it carries a relatively high risk of viral contamination. The helper virus process has high scalability and supports large-scale production, but it poses a high risk of viral contamination (with the possibility of helper virus or recombinant virus contamination). A comparison of rAAV products manufactured using the three platforms in terms of scalability, regulatory challenges, viral safety risks, and commercial applicability is shown in Table 3.
Comparison of the Three Major Recombinant Adeno-Associated Virus Production Platforms
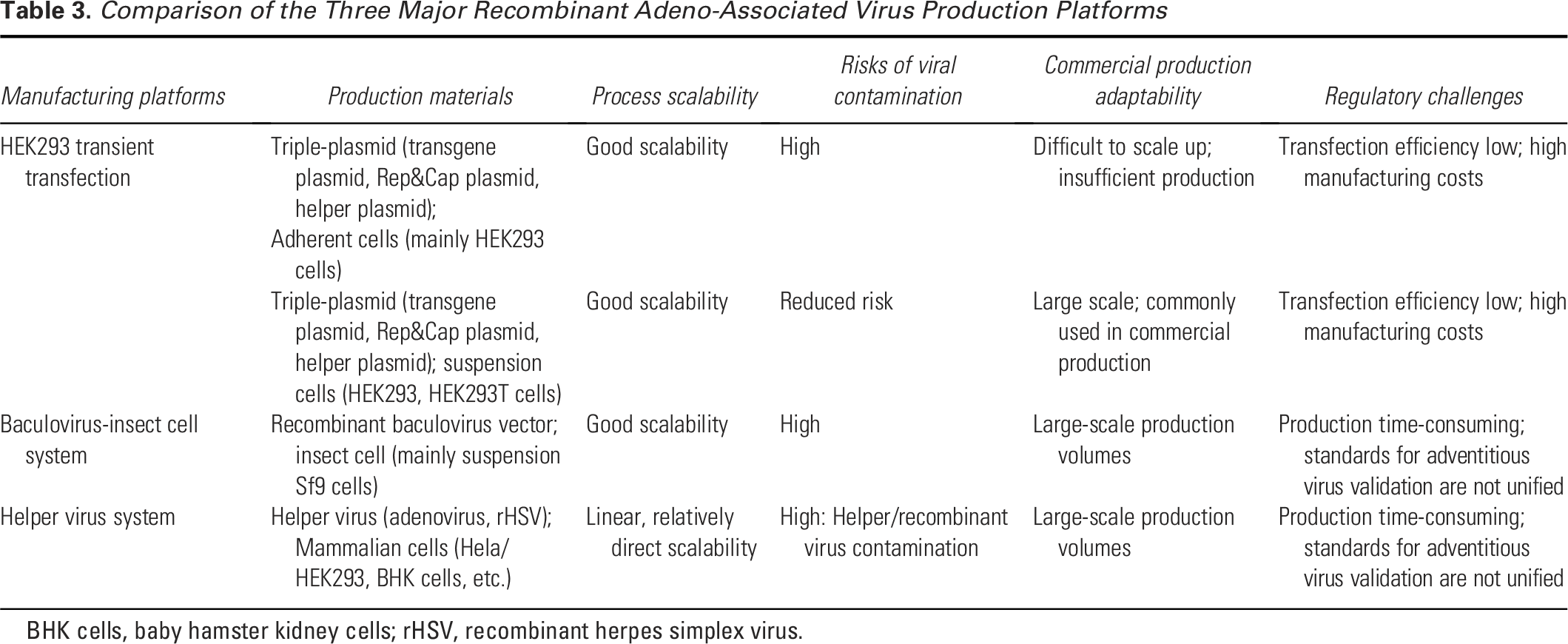
BHK cells, baby hamster kidney cells; rHSV, recombinant herpes simplex virus.
Viral contamination is a potential risk inherent to all AAV products being manufactured. The International Council for Harmonization (ICH) Q5A (R2) regulatory guidelines propose three principal, complementary approaches to control viral contamination of biotechnology products 7 : (1) Selecting and testing cell lines and other raw materials, including media components, for the absence of undesirable infectious viruses. (2) Assessing the risks of each manufacturing process to introduce clear adventitious and endogenous viruses. (3) Testing the product at appropriate steps of production for the absence of contaminating infectious viruses. While the ICH Q5A (R2) guidelines propose an overall strategy for controlling adventitious viruses, there is not yet an international regulatory consensus on the requirements for viral clearance validation of rAAV products and global regulatory authorities have not established requirements for the overall risk control strategy of adventitious viruses in rAAV products produced by different types of manufacturing. We propose control strategies for adventitious viruses in three different rAAV production processes (transient cotransfection platform, baculovirus-insect cell platform, and helper virus platform) and elaborate on the technical requirements. The specific requirements are specified in Fig. 1 and the following content.
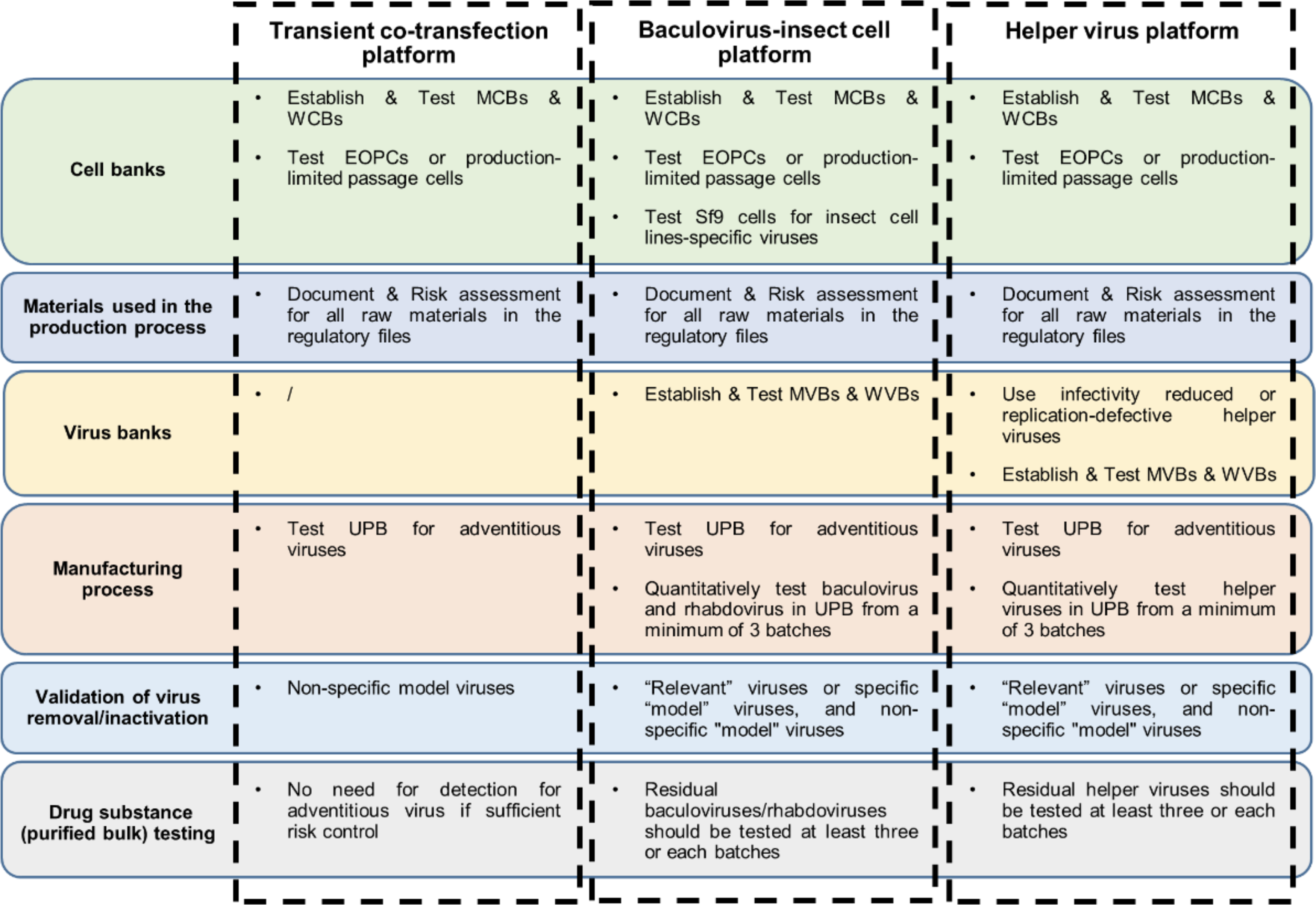
Adventitious-virus control workflows for the main manufacturing platforms. EOPCs, end-of-production cells; MCBs, master cell banks; MVBs, master virus banks; UPB, unprocessed bulk; WCBs, working cell banks; WVBs, working virus banks.
Transient cotransfection platform
Currently, the most widely adopted manufacturing is the triple-plasmid cotransfection of HEK293 cells. HEK293 cells express the adenovirus (AdV) E1a and E1b genes, and three plasmids are transiently transfected into them: (1) A vector plasmid containing the therapeutic gene expression cassette and ITR sequences. (2) A structural plasmid containing rep and cap genes. (3) A helper plasmid containing viral replication genes such as E2A, E4, and VA RNA. The main advantages of the triple-plasmid cotransfection method include a relatively short development cycle in the early stage of the manufacturing process, relatively simple methods needed for plasmid production, preparation, and quality control, and no need for introducing helper viruses, which reduces the risk of adventitious viruses. The main challenges of this process are the construction of plasmids for production, the need to optimize the ratio of the plasmids being transfected, the low transfection and packaging efficiency, and the scaling-up of production. Moreover, the transient expression of the three plasmids can lead to product heterogeneity, such as viral vectors lacking the therapeutic gene cassette (i.e., empty capsids), thus necessitating the establishment of processes to detect and remove impurities. 8 While this method does not intentionally introduce potentially adventitious viruses, they may originate from the production cell line, animal/human-derived raw materials used in the manufacturing process, or contamination during the manufacturing process.
Control of Adventitious Viruses in Cell Banks
HEK293 cells are widely used for manufacturing biological products, and so, the establishment of cell banks that are free of viral contamination has become a widely implemented control process. To minimize the risks of contaminating HEK293 cells, it is recommended to use cells from a clear and definite source and to establish cell banks in standardized manner, such as master cell banks (MCBs) and working cell banks (WCBs). In addition, the end-of-production cells or production-limited passage cells should be tested for contamination.
Control of Adventitious Viruses in Materials
The quality of materials used in the production process may affect the quality, safety, and efficacy of the final product. Specific assessments of the culture media and raw materials should be documented in the regulatory file with respect to identity, purity, sterility, biological activity, and absence of adventitious agents. Typically, chemically defined serum-free media are used in manufacturing rAAV products and the animal-derived components are trypsin and serum. For all animal-derived components, information on the source organism, supplier/vendor, country of origin, infectious agent testing, and stage of manufacture in which the component is used should be documented. This information is collected to mitigate potential concerns regarding transmission of diseases such as bovine spongiform encephalopathy and transmissible spongiform encephalopathy as well as infectious agents. Therefore, a risk-based approach should be adopted to assess the possibility of adventitious agents and other contaminants being introduced by raw materials used in the manufacturing of rAAV products.
Control of Adventitious Viruses in the Manufacturing Process
Viral contaminants from humans or the environment can be introduced into the rAAV product during material addition or transfer steps in the manufacturing process. Thus, it is important to maintain a well-controlled and monitored environment as well as ensure proper use of gowning and aseptic techniques. Concurrently, it is also necessary to tightly control the manufacturing process, select appropriate production steps at which to test for adventitious viruses, and set acceptance criteria. A representative sample of the unprocessed bulk (UPB), which is removed from the production reactor before starting processing steps, represents one of the most suitable levels at which adventitious virus contamination can be detected. Therefore, appropriate testing for viruses should be performed on the UPB.
Validation of Virus Removal/Inactivation
Additional risk mitigation comes from virus clearance validation. Where appropriate, inactivation and/or removal steps can be incorporated into the manufacturing process to further minimize the risk of accidental viral contamination. From the perspective of controlling the risk of viral safety in products, it is necessary to conduct virus clearance validation for all products, including those made with the triple-plasmid cotransfection manufacturing process, which does not intentionally introduce viruses to the cells. This is because the manufacturing facility may not be able to completely prevent the accidental introduction of adventitious viruses or the risk of viral contamination from other methods of production used at the same production site. If the manufacturing process can be designed to include steps at which viruses can be removed and/or inactivated, it can provide further assurance of the product’s viral safety. In addition, if no viruses, virus-like particles, or retrovirus-like particles other than the intended product are found in the cell bank or UPB, virus clearance validation can be evaluated using nonspecific model viruses. If no retrovirus-like particles are detected and the Product-Enhanced Reverse Transcriptase test is negative, then no estimation of retroviral particles per dose is required.
Detection of Adventitious Viruses in Products
As discussed previously, a series of measures should be taken throughout the entire manufacturing process to control the risk of adventitious virus contamination. Overall risk assessment for adventitious virus contamination is considered controllable, and testing for adventitious viruses in drug substance (purified bulk) is not required.
Baculovirus-insect cell platform
In 2002, Urabe and colleagues 9 established the first-generation baculovirus expression vector system, which was successfully applied to the production of AAV1. This system used three types of baculoviruses, each packaged with the therapeutic, rep, or cap gene, and these three baculoviruses need to coinfect a cell to produce AAV particles. The second-generation baculovirus expression vector system reduced the number of baculoviruses required for AAV production from three to two by packaging both the rep and cap genes into one vector and delivering the therapeutic transgene in the other. 10 This system was further improved by stably integrating the rep/cap genes into the genome of the Sf9 cell line and infecting the Sf9 cell line with a single baculovirus carrying the therapeutic gene, which improved the yield of AAV viral vectors that can be produced. 11
Compared with the transient cotransfection manufacturing method, the baculovirus-insect cell system has the advantage of being highly scalable for production. In addition, it often uses the serum-free medium and has been used in the production of various products such as enzymes, glycoproteins, recombinant viruses, and vaccines,12,13 which have shown good safety profiles, making this system easy to implement along with other rAAV production methods. However, because recombinant baculoviruses and insect cells are used in the manufacturing process, this system requires a planned control strategy for adventitious viruses.
Control of Adventitious Viruses in Cell Banks and Materials
The risk control requirements for viral contamination in cell banks and raw materials for the baculovirus-insect cell production system are similar to the transient cotransfection platform. For the baculovirus-insect cell platform, the Sf9 cell line is commonly used, a subclone of SF21AE cells isolated from the ovarian tissue of pupae of the fall armyworm (Spodoptera frugiperda). 14 When testing Sf9 cells for contamination, consideration should be given to detecting specific viruses that have been previously reported (such as nodaviruses) or viruses that may persist in insect cell lines and are known to be infectious to humans. In addition, some Sf9 cells may carry genes of the Spodoptera frugiperda rhabdovirus, and so, rhabdovirus detection specifically should be considered.
Control of Adventitious Viruses in Virus Banking
It is important to establish a virus banking system to maintain consistency in the production of rAAV. A two-tiered virus banking system, a master virus bank and a working virus bank, should be established and tested for factors such as sterility, mycoplasma, and adventitious viruses.
Control of Adventitious Viruses in Manufacturing Process
Compared with the transient cotransfection platform, the in vitro detection of adventitious viruses in the baculovirus-insect cell system is more challenging because baculoviruses in the UPB may interfere with the detection of adventitious viruses. Therefore, it is recommended to use antibodies to neutralize the baculoviruses in the UPB before detecting adventitious viruses. If suitable neutralizing antibodies cannot be obtained or the neutralization effect is unsatisfactory, production control cells should be used for the detection of adventitious viruses. In addition, the ICH Q5A (R2) guidelines indicate that molecular biology methods such as nucleic acid amplification and next-generation sequencing can also be considered for detecting specific viruses. For the purpose of marketing authorization, quantitative detection results of baculoviruses and rhabdoviruses in the UPB from at least three batches of commercially produced vectors must be tested to evaluate the clearance effect of the overall production process on adventitious viruses.
Validation of Virus Removal/Inactivation
In the baculovirus-insect cell manufacturing process, Sf9 cells may contain rhabdoviruses and recombinant baculoviruses are introduced into the production system. Therefore, it is necessary to incorporate virus removal/inactivation steps into the manufacturing steps and conduct validation studies on virus removal/inactivation, as suggested by the ICH Q5A (R2) guidelines.
“Relevant” viruses (i.e., viruses that have been identified or are likely to contaminate the production process) should be used for virus clearance validation studies. When a “relevant” virus is not available or when it is not well adapted to the process evaluation of viral clearance studies, a specific “model” virus (i.e., a virus that is closely related, ideally in the same genus or family, to the known or suspected virus) should be used as a substitute, such as baculoviruses and vesicular stomatitis virus (representing endogenous rhabdoviruses in Sf9 cell lines). In addition, nonspecific “model” viruses with significant resistance to physical or chemical treatments should be considered for validation controls to ensure that the removal/inactivation steps are effective. The selection of the types and number of model viruses should be comprehensively determined based on the quality and characteristics of the packaging/production cells and the manufacturing. Generally, it is recommended to test for at least three viruses with different characteristics to evaluate the virus clearance capacity of the manufacturing process.
Detection of Adventitious Viruses in Products
As discussed previously, a series of measures are taken throughout the entire manufacturing process to control the risk of adventitious viruses in the product. Based on the test results and risk assessment, the absence of residual baculoviruses and rhabdoviruses should be confirmed by testing at least three batches of the drug substance (purified bulk) using an infectivity assay with “relevant” permissive cell lines for sensitive virus detection. Alternatively, molecular methods may be used for testing. Testing for the absence of residual baculoviruses and rhabdoviruses should be performed for each purified bulk preparation unless justified by robust excess clearance.
Helper virus platform
A HeLa cell line stably expressing the helper genes rep and cap as well as the therapeutic gene can be constructed and infected with AdV, enabling it to package rAAV products. 15 Using the suspension-cultivable HeLa S3 cell line, this stable cell production method can be directly scaled up to a 2000 L scale. AAV production using coinfection with the recombinant herpes simplex virus (rHSV) is a very efficient method to generate a large amount of rAAV. In addition to high overall yields up to 1.5 × 105 vg/cell, the apparent increased quality of the rAAV stocks, as measured by an improved viral potency, makes this method very attractive.16,17 Cells, typically the hamster BHK21 cell line or HEK293 and derivatives, 18 are infected with two rHSVs, one carrying the therapeutic gene flanked by AAV ITRs and the second carrying the AAV rep and cap of the desired serotype. Although the application of helper viruses such as AdV and rHSV provides necessary conditions for rAAV replication, it also brings potential risks of adventitious viruses, including the helper viruses themselves. Therefore, it is necessary to establish a multistage risk control strategy.
Control of Adventitious Viruses in Cell Banks and Materials
The requirements for control of viral contamination in MCBs, WCBs, and materials are the same as for the transient transfection platform.
Control of Adventitious Viruses in Virus Banking
The modification of helper viruses is the major risk that must be mitigated by prevention and control measures. Genetic engineering techniques have been developed to attenuate the infectivity of helper viruses or make them replication-defective, which significantly reduces their pathogenicity and autonomous replication ability to lower the risk of contamination in rAAV products. For example, HSV is a human pathogen 19 that is cytotoxic to cells. rHSV vectors are engineered to lack essential viral replication genes, rendering them replication incompetent. As a result, replication-defective rHSVs display significantly reduced cytotoxicity for the producer cells compared with replication-competent HSV. Immediate-early genes encoding infected cell proteins 0, 4, 22, 27, and 47 are commonly deleted from the viral genome (VG) to confer additional safety.20,21 In addition, a helper virus banking system should be established, and characterization of sterility, mycoplasma, and adventitious viruses should be considered.
Control of Adventitious Viruses in Manufacturing Process
It is necessary to consider conducting adventitious virus detection on UPB, and appropriate antibodies should be used to neutralize the helper viruses before detection. In parallel to testing products during the manufacturing process, adventitious virus detection should be performed on production control cells or molecular biology methods such as nucleic acid amplification, and next-generation sequencing can be adopted in accordance with the requirements of the ICH Q5A (R2) guidelines for the detection of specific viruses.
Validation of Virus Removal/Inactivation
Because helper viruses are used in the production process, virus removal/inactivation steps must be included in the production process. Detailed expectations and advice for implementing processes to remove “relevant” viruses are provided in the ICH Q5A (R2) guidelines. Briefly, the process should be demonstrated to remove the “relevant” virus with a high safety margin and should include a well-characterized inactivation step. For clearance of viruses by different steps to be considered additive, the principles of separation must be orthogonal. Because of the well-established differential thermostabilities of AAV and Ad5, heat inactivation can easily provide >5–6 log reduction of Ad5. 22 “Nanofiltration’’ is an additional viral clearance measure that is now routinely incorporated in recombinant protein manufacturing processes. Because of their relatively small size, rAAV vectors can be quantitatively recovered from filters designed for retroviral clearance, and therefore, such a filter should be included in rAAV current good manufacturing practices. 23
Detection of Adventitious Viruses in Products
The requirements for detecting adventitious viruses in products are the same as those for the baculovirus-insect cell production process, with the only difference being the types of viruses to be detected. For the helper virus production process, helper viruses such as rHSV and AdV pose potential contamination risks.
Regulatory considerations for quality control of rAAV
Strength or titer
As vector genomes are the key component needed for the therapeutic effect of an rAAV product, VG titer is the most commonly used CQA to determine dosing. The number of VGs that a patient is dosed with has a high impact on both efficacy and safety 24 : Too low a dose will result in a limited therapeutic effect and too high a dose may result in adverse events caused by overexpression of the transgene. Consequently, how the VG titer is measured is critical to optimize the safety and efficacy of a product. The VG titer is typically assessed by quantitative polymerase chain reaction (qPCR) or droplet digital polymerase chain reaction (ddPCR). In recent years, ddPCR has become the industry standard because it has several advantages over the traditional qPCR, including reduced susceptibility to sample matrix interference, the ability to determine absolute quantification without a standard curve, and superior precision and accuracy. 25
As clinical trials progress, the detection method for determining VG titer may change with the rapid development of technology, however, it is recommended to implement the method change at an early clinical trial phase so that differences in methodology are not a confounding factor. Before implementing any changes in how VGs are determined in clinical trials, a comprehensive risk assessment should be conducted based on the specific circumstances of the method change to ensure that the new method has an equivalent or better ability to control product quality compared with the original method and to guarantee the accuracy of the clinical dosage.
Viral protein purity
Generally, the stoichiometry of the three capsid proteins VP1, VP2, and VP3 is crucial for vector infectivity. 26 Therefore, variations in the capsid–protein ratio are expected to alter the in vivo potency of the rAAV by impacting transduction efficiency. 27 Methods for detecting charge heterogeneity of rAAV capsid proteins mainly include sodium dodecyl sulfate (SDS)–polyacrylamide gel electrophoresis and capillary electrophoresis–sodium dodecyl sulfate (CE-SDS). The CE-SDS method is superior to SDS-polyacrylamide gel electrophoresis in terms of resolution, precision, and robustness. With the continuous advancement of technology, new and improved detection methods may be developed and implemented in manufacturing processes. Therefore, it is encouraged to use detection methods with higher precision and accuracy as they become available for product purity testing.
When assessing the capsid protein purity of AAV843 using the CE-SDS method, in addition to the expected peaks of the VP1, VP2, and VP3 subunits in the CE-SDS profile, an unknown signal peak with a molecular weight slightly smaller than VP3 was detected, which was speculated to be a VP3 variant. The VP3 subunit contains two in-frame start codons (ATG), and when the translation initiation site starts from the weaker second codon, a VP3 variant is generated.28,29 However, how this variant affects the stoichiometry of the proteins required for capsid assembly is not known. It is recommended to formulate a control strategy for quantifying levels of VP1, VP2, VP3, and variants of VP3 at various stages, determine what factors may cause each to be produced or suppressed, and identify their association with product characteristics such as safety, efficacy, and quality control. When necessary, in addition to controlling the total amount of VP1 + VP2 + VP3 in rAAV products, separate control of the content of VP3 molecular variants should also be considered based on a risk assessment of the impact of VP3 variants on viral structure or function.
Size variants
The content of monomers and aggregates in rAAV products is a CQA that reflects the robustness of the production process and the quality characteristics of the product. Currently, there are several methods used to detect monomers and aggregates in rAAV products, such as size-exclusion chromatography (SEC), analytical ultracentrifugation (AUC), and dynamic light scattering. SEC separates solutes of subvisible size (d < 100 nm) based on their hydrodynamic volume. It is also used on high-performance liquid chromatography (HPLC) systems with ultraviolet, fluorescence spectrometer, or differential refractive index detectors to quantify the eluting species 30 and is the industry standard method to assess aggregates due to its simplicity, throughput, and speed. 31
While SEC-based methods are the long-standing industry standard due to their simplicity and high throughput, they are not suitable for quantifying large rAAV aggregates due to issues with filtration, nonspecific interactions, and disaggregation. 31 AUC can assess aggregation, but its low throughput and poor accuracy for determining small aggregate concentrations are limiting factors in process development. Dynamic light scattering shows potential as at-line methods to quantify large aggregates, but its precision is poor and can be inaccurate if the optical properties of the matrix are uncertain. While each method has its own advantages and limitations, it is recommended that comprehensive characterization studies on product monomers and aggregates should be conducted using multiple technical approaches or methods during the research and development phase of rAAV products. During the clinical trial phase, data on monomers and aggregates of each batch, as well as changes during the stability period, should be monitored to formulate the standard limits for monomers and aggregates in each product.
Impurities
rAAV products contain various types of impurities, including those caused by the process and the product. Process-related impurities are derived from the manufacturing process of the materials and components but are not structurally related to the product. Product-related impurities are unintended, nonfunctional products generated from the product components during production or storage. Here, we discuss the quality control considerations for residual host DNA and empty capsid viruses.
Residual Host DNA
Cells that contain viral oncogenes are commonly used to manufacture AAV vectors. Examples include HEK293 cells that contain the AdV E1 gene and HeLa cells that contain human papillomavirus E6 and E7 genes. E1A could induce the immortalization and transformation of cells in cooperation with a viral or cellular oncogene, harboring the putative risk of a circumstantial support or even induction of tumorigenic growth. HPV oncogenes E6 and E7 conspire to induce and maintain cell transformation by interfering with p53 and pRb proteins that control cell cycle, senescence, and apoptosis. NMPA and FDA guidelines require that gene therapy products control for residual host cell DNA and the size of DNA fragments, and recommend that residual DNA be controlled to under 10 ng/dose and the size of residual DNA fragments be controlled below 200 bp. In addition, for known transforming factors in products that have potential safety risks, such as the E1, E6, and E7 genes, residual quantity detection and control should also be conducted. These oncogenic sequences could be quantitatively detected by qPCR or ddPCR. qPCR has a lower accuracy and a narrower dynamic range compared with ddPCR, but ddPCR is more costly. These guidelines can be met by established process optimization strategies when residual host cell DNA is present as a nuclease-sensitive process-related impurity. For example, residual host DNA can be digested into small fragments by nucleases, and removed through subsequent processes such as affinity chromatography, anion exchange chromatography, and ultrafiltration–diafiltration or tangential flow filtration. However, residual nuclease-resistant host cell DNA packaged within AAV capsids is challenging to detect and difficult to remove. Packaged residual DNA impurities derived from vector template and helper sequences were reported to range from 1% to 8% of total DNA in purified vector particles.32,33
Empty Capsid Viruses
rAAV products are generally assessed for three types of capsids: full, intermediate, and empty. While the impact of intermediate and empty capsids on the safety and efficacy of rAAV products is not fully understood, they are generally considered impurities because they are not the intended fully intact vector product. 34 Empty capsids, AAV capsid shells lacking the vector genome, are found at varying levels in clinical-grade AAV vector preparations. 35 A range of empty particles have been observed in AAV production, from less than 50% in cell harvests for wild-type AAV2 to over 98% for some rAAV vectors that package poorly. 36 In addition, some AAV particles are partially packaged with the intended cassette and only contain ITRs and adjacent DNA sequences. These partial and defective AAV particles increase dosing requirements, although the effect may be more pronounced for direct administration to tissues and less so for systemic administration. 37 Furthermore, partial capsids can contain undesired DNA sequences, such as truncated forms of the therapeutic gene, host cell DNA, and/or residual plasmid DNA, and could therefore be a concern for safety due to possible genotoxic or immunogenic effects. 24
Currently, common methods for detecting the empty capsid rate of rAAV products include AUC, ion exchange (IE)-HPLC, and SEC-multiangle light scattering (MALS). The IE-HPLC and SEC-MALS methods have mature platforms, with fast analysis speed and high throughput, but they may not fully distinguish partially packaged capsids from fully packaged ones. Compared with IE-HPLC and SEC-MALS, AUC has a better separation efficiency for fully packaged, partially packaged, and empty capsid viral particles, with a smaller difference between the measured empty capsid rate and the theoretical empty capsid rate, and is widely utilized in industry settings. With the continuous development and progress of technology, new and better detection methods may be gradually applied. Therefore, it is encouraged to prioritize detection methods with high precision and accuracy for the detection and control of the empty capsid rate.
NMPA, FDA, and EMA guidelines all emphasize the necessity of controlling empty capsid viruses, but none of them is able to offer clear requirements for specific detection methods and standard limits. Key considerations for formulating the standard limits of the empty capsid rate include product characteristics, production processes, historical batch testing data, administration methods and doses, and the correlation with clinical safety and efficacy. Similarly, as VG measurement, if the detection method of the empty capsid rate changes during the clinical trial of the product, the impact of the change of the detection method on the safety and efficacy results. Overall, there is a need in the field to reasonably formulate the standard limits of the empty capsid rates in general and for each product.
Potency
Measures of rAAV product potency can include assays to determine the concentration of the product, also referred to as strength or titer, and the functional activity. As discussed previously, for most rAAV products, the VG titer is the dose-determining assay. However, methods must be developed to measure the functional activity of vectors and test for delivery of the vector genome to target cells (infectivity), subsequent expression of the product of the transgene, and the actual functional activity of the transgene product. 38
At the marketing application stage, the potency standard limits should be set based on data accumulated from product development, manufacturing, and clinical results, and revised based on clinical trial data, method variability, batch-to-batch differences, and changes during the stability period. If changes are made to the methods for potency evaluation during the product’s research and development, sufficient risk assessment and research data should be provided in combination with the content, extent, and risk of the changes to determine whether they have an adverse impact on the determination of a product’s potency.
Safety
Quality control tests used to demonstrate the absence of microbial contamination in the vector product are critical to ensure parenteral product safety. Such testing should include assays for sterility, mycoplasma, endotoxin, general safety, and adventitious viruses. Sterility testing must be performed on the drug product, and the sterility assay must be qualified by the assessment of the bacteriostatic and fungistatic activity of the vector product. 38 Endotoxin testing should be performed on the drug product using a validated assay, and acceptance criteria must be appropriate for the planned route of administration. 39
Due to spontaneous recombination events occurring during manufacturing processes, viral vectors could become replication-competent, increasing the risk of immunotoxicity. The mechanism for this is thought to be primarily through nonhomologous recombination events between elements in the vector genome and AAV rep and cap sequences on the packaging plasmids during production. 24 Quality control testing for rcAAV is required for clinical rAAV products, and the recommended method for rcAAV detection is a cell-based protocol that requires multiple rounds of transduction in the presence of Ad5 to amplify any signals from low-abundant virus followed by a qPCR assay for the rep gene. 40 When using this method for rcAAV detection, several key factors must be taken into consideration, such as the selection of a positive control virus, the selection of cocultured cells, and the establishment of standard limits. Specifically, to validate the sensitivity and accuracy of the detection method, it is recommended that the positive control virus be consistent with the serotype of the product. In addition, considering that viruses of different serotypes have different infection characteristics or abilities in different cells, it is encouraged to screen for cell lines that are optimally susceptible to each product.
There is no regulatory consensus on the standard limits for rcAAV. The rAAV product, scAAV2/8-LP1-hFIXco, which has entered clinical trials, uses a combination of cell culture and qPCR for detection, and can control rcAAV ≤1 particle per 2.25 × 106 VG. 41 The vector production systems currently in use have been optimized to control rcAAV ≤1 particle per 1 × 108 VG. 37 These reported values are, in part, a function of the limit of sensitivity of analytical methods for detecting rcAAV, which typically rely on the amplification of AAV rep and/or cap DNA sequences following multiple rounds of passaging a permissive cell line after being transfected with an rAAV product in the presence of an AdV. However, regulatory authorities do not specify a specific numerical threshold value for considering a product to contain rcAAV. Instead, the rationality of establishing standard limits of detection and tolerability for rcAAV is comprehensively judged based on factors such as PCR-based detection methods, accumulated batch data, and clinical safety and efficacy.
CONCLUSIONS
AAV vectors are the leading platform for gene delivery for the treatment of a variety of human diseases, offering great potential to address unmet medical needs. Because viral clearance steps for the production of rAAVs may not achieve the same robustness as for recombinant proteins, the viral safety of the products should be supported by a risk assessment tailored to the product because each of the three well-established rAAV production methods—triple cotransfection, insect-baculovirus, and helper virus—carries specific risks. Risk assessments must consider specific CQAs, and there is a need to identify more relevant CQAs for rAAV products and detect them at higher sensitivity. As technologies improve, gene therapy product manufacturing will continue making iterative improvements and innovative leaps forward, driving the rapid evolution of the field of genetic medicine. Thus, the considerations for detection methods and standard limits of detection and tolerability for CQAs will also continuously evolve to develop better adventitious virus control strategies to make rAAV products safer and effective.
AUTHORS’ CONTRIBUTIONS
J.C. was responsible for the research and writing of the article. W.W., D.L., D.H., and X.J. undertook the revision and proof of the article. W.W. was in charge of the research, writing, and supervision of the article.
Footnotes
FUNDING INFORMATION
This work was supported by the State Key Laboratory of Drug Regulatory Science Project (grant number 2023SKLDRS0138).
AUTHOR DISCLOSURE
The authors declare that they have no conflicts of interest with this work.
