Abstract
Emphysematous gastritis is a rare, serious infection with very few reported cases in veterinary species. Gas-producing bacteria are most often implicated in both animals and humans. Here, we describe a case of emphysematous gastritis in a 7-y-old dog that was diagnosed with ileus of 12 d, chronic pancreatitis, and exocrine pancreatic insufficiency. Histologically, the gastric submucosa was markedly expanded by emphysema, and the mucosa had multifocal necrosis, hemorrhage, and gram-positive bacilli. Anaerobic culture of the stomach wall yielded 3+ growth of Clostridium perfringens, Enterococcus faecalis, and Escherichia coli. PCR typing identified the C. perfringens isolate as type C, further characterized as a beta2 toxin–producing strain. Fluorescence in situ hybridization using a C. perfringens probe identified bacilli within the gastric mucosa and submucosa. C. perfringens type C causes enteritis necroticans in humans and necrohemorrhagic enteritis in livestock when levels of endogenous trypsin are low or dietary levels of trypsin inhibitors are high. The exocrine pancreatic insufficiency in our case likely allowed C. perfringens type C to exert cytotoxic effects on the gastric mucosa and contributed to the unique emphysematous gastritis.
A 7-y-old, castrated male Weimaraner dog was presented to the Texas A&M Veterinary Medical Teaching Hospital (College Station, TX, USA) because of icterus and a 9-d history of hyporexia that progressed to anorexia. Initial bloodwork revealed severe cholestatic hepatopathy (alkaline phosphatase: 1,383 U/L [RI: 24–147 U/L]; total bilirubin: 42.8 μmol/L [RI: 0–13.7 μmol/L]) and severe neutrophilia (42.3 × 109/L [RI: 3–11.5 × 109/L]) with a left shift (band neutrophils: 0.46 × 109/L [RI: 0–0.3 × 109/L]). On physical examination, the dog was icteric, sarcopenic, and had peripheral lymphadenopathy. The dog was hospitalized for testing, supportive treatment for biliary disease, and placement of a nasogastric tube for enteral nutrition.
On day 3 of hospitalization, contrast-enhanced computed tomography (CT) revealed a thick, heterogeneous, irregularly marginated pancreas with non–contrast-enhancing regions in the right lobe, consistent with pancreatic necrosis; wispy soft-tissue streaking within the cranial abdominal mesentery, consistent with steatitis; extrahepatic bile duct obstruction; mild gallbladder and gastric wall thickening; and multiple lymphadenopathy. Fine-needle aspirate samples from the pancreas, liver, and a sternal lymph node were evaluated cytologically, revealing neutrophilic and histiocytic inflammation of all organs and necrosis within the pancreatic sample. Abdominal fluid analysis showed a neutrophilic exudate; anaerobic and aerobic culture did not yield bacterial growth. Serum canine pancreatic lipase immunoreactivity was within its RI; thus, pancreatitis was not suspected as the sole etiology for the patient’s clinical course. Given a concern for an infectious etiology—because of the mixed inflammation in numerous sampled visceral organs—we tested serum, whole blood, and feces for infectious agents on day 4 of hospitalization. Heterobilharzia americana was detected via fecal PCR testing, and H. americana eggs were seen on fecal sedimentation. The dog had a mildly positive titer (1:100) for Neospora caninum, was seronegative for Toxoplasma gondii, and negative for Hepatozoon americanum/canis via whole-blood PCR testing and for Histoplasma capsulatum via urine antigen detection.
Treatment was initiated for H. americana (fenbendazole, praziquantel) via esophagostomy tube, as well as gastrointestinal (GI) prokinetics (because of the development of ileus), pantoprazole (because of regurgitation), and clindamycin (as an empirical agent against protozoal infection). Given progressive paralytic ileus, enteral feeding was discontinued because of persistent regurgitation, and, by day 9 of hospitalization, the ileus became refractory to medication and necessitated gastric decompression. A central IV line was placed, and partial parenteral nutrition (amino acids and lipids) was begun. A GI blood panel was performed to investigate the potential of pancreatic insufficiency, given the changes observed on CT and the positive Heterobilharzia PCR result. The GI panel revealed low trypsin-like immunoreactivity (3.2 µg/L [RI: 10.9–50 µg/L]), consistent with exocrine pancreatic insufficiency (
Throughout hospitalization, cholestatic hepatopathy and hyperbilirubinemia continued to worsen in this dog, raising further concern for progression of biliary tract obstruction. Because of ileus, several liters of gastric fluid were repeatedly removed during decompression, and, on day 11 of hospitalization, the gastric fluid was dark red and malodorous. Given the lack of improvement despite extensive supportive care, the dog was euthanized after 12 d of hospitalization.
An autopsy was performed within one hour after euthanasia. The mucous membranes and subcutaneous and visceral adipose tissue were moderately icteric, and the common bile duct was dilated and obstructed. The omentum was diffusely firm, thickened, and compacted tightly around the stomach and pancreas, creating a mass-like effect in the right cranial abdomen. The pancreas was diffusely pale-tan and firm. The stomach contained a moderate amount of dark-red, opaque fluid. All areas of the mucosa had numerous coalescing, dark-red, roughened, and slightly depressed regions (
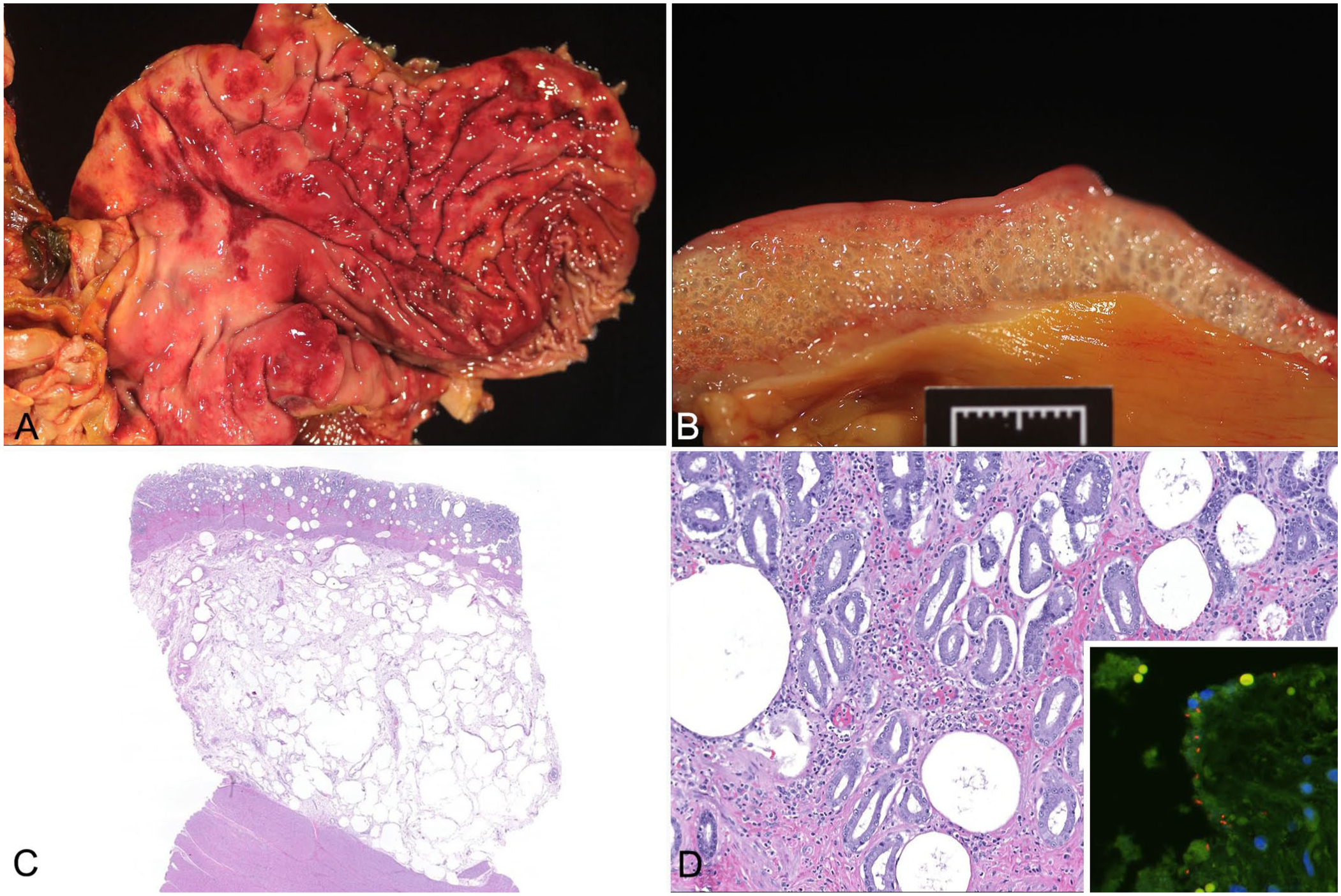
Clostridium perfringens type C–associated emphysematous gastritis in a dog.
Histologically, the gastric submucosa was diffusely and markedly expanded by emphysema, with lesser edema extending multifocally into the mucosa (
Tissue from the stomach was used to inoculate media for anaerobic cultures at the Texas A&M Clinical Microbiology Laboratory. For the anaerobic culture, Brucella agar and CNA plates were inoculated and incubated at 37°C under anaerobic conditions that were generated using an anaerobe jar and an anaerobic gas sachet (Mitsubishi Gas). Culture plates were inspected daily for growth. Relative quantification of the culture growth was established by noting the quadrant of the culture plates that contained growth (e.g., 3+ growth meant growth in 3 of 4 quadrants of the original culture plate). Bacteria were identified using matrix-assisted laser-desorption/ionization–time-of-flight mass spectrometry with FlexControl v.3.4.206.94 and MBT Compass 4.1 (Bruker). Anaerobic culture of the stomach tissue yielded 3+ growth of Clostridium perfringens, Enterococcus faecalis, and Escherichia coli, and 2+ growth of Enterococcus faecium and Veillonella parvula.
Three to 5 C. perfringens isolates were submitted to the Texas Veterinary Medical Diagnostic Laboratory for further testing, similar to the approach used for antimicrobial susceptibility testing of isolates. Total nucleic acids were extracted from the C. perfringens isolates (MagMAX 96 viral RNA isolation kit, Kingfisher Flex magnetic particle processor; ThermoFisher). Typing of the C. perfringens isolate was performed by real-time PCR (rtPCR) using primers and probes targeting the alpha toxin (cpa); beta toxin (cpb); epsilon toxin (etx); iota toxin (ia); enterotoxin (cpe), and beta2 toxin (cpb2) genes8,19 (Path-ID qPCR master mix; Life Technologies). The rtPCR testing detected cpa, cpb, and cpb2 genes. The C. perfringens cpe gene and the netF toxin gene were not detected by rtPCR testing. 21
Additional testing was pursued to demonstrate C. perfringens within the examined tissue sections. Fluorescence in situ hybridization (FISH) for C. perfringens with a C. perfringens 16S rRNA probe, Cperf191 (3′-GTAGTAAGTTGGTTTCCTCG), as previously developed and validated, 5 was performed on formalin-fixed, paraffin-embedded, unstained tissue sections from the stomach. FISH was performed using an automated stainer. After deparaffinization in xylene and hydration in ethanol, slides were hybridized with an oligo probe diluted to 5 ng/µL in hybridization buffer with formamide for 6 h. After hybridization, the slides were extensively washed in buffer, mounted with antifade solution containing 4′,6-diamidino-2-phenylindole, and examined under an epifluorescence microscope. Positive bacilli were detected within the mucosa and rarely in the submucosal emphysema (Fig. 1D, inset). Additionally, the sensitivity of the Cperf191 probe was confirmed using 3 additional institutional cases of proven C. perfringens infection, all of which tested positive.
The detection of the cpa and cpb toxin genes classified the C. perfringens isolate as type C. The detection of the cpb2 gene further characterized the C. perfringens isolate as a beta2 toxin–producing strain. The culture and molecular results, along with observed necrohemorrhagic and emphysematous gastritis, were indicative of C. perfringens infection. 18 We additionally demonstrated intralesional C. perfringens using FISH. Hence, this was a case of C. perfringens type C–associated emphysematous gastritis in a dog. We retrieved no cases of canine C. perfringens type C–associated emphysematous gastritis in a search of Google Scholar, PubMed, CABI Direct, Web of Science, and Scopus, using search terms “Clostridium perfringens type C”, “emphysematous”, “gastritis”, or “gastroenteritis”, and “dog” or “canine” as of 2025 Nov 20, suggesting that this condition has not been reported previously in dogs.
Emphysematous gastritis is a serious, often fatal condition with few reports in humans and even fewer documented cases in veterinary species. Gas-forming bacteria, often seen in histologic section and/or cultured from gastric tissue or fluid, lead to an accumulation of gas within the gastric wall. In humans, responsible bacteria include E. coli, C. perfringens, Pseudomonas aeruginosa, Staphylococcus aureus, Sarcina ventriculi, and Enterobacter species, and mixed growth is common.1,11,13 Across isolated case reports in cats, dogs, and horses, bacteria cultured from gastric tissue or fluid include Clostridium septicum, 2 Staphylococcus felis, 15 E. faecalis, 15 and Sarcina spp. 9 Bacteria were within histologic sections of emphysematous gastritis in a few reports in dogs, cats, and horses, but bacterial culture was not performed.6,12,15,20 In our case, C. perfringens was considered the most significant causative agent for the dog’s emphysematous gastritis given the identified toxin genes; Enterococcus faecalis and E. faecium, E. coli, and V. parvula were likely commensal isolates acting as opportunistic pathogens.
In humans, risk factors for developing emphysematous gastritis include previous abdominal surgery, alcohol abuse, pancreatitis, renal failure, and conditions that delay gastric emptying.1,11,13 Given the fewer cases in animals, risk factors are not well understood. However, 2 cats developed emphysematous gastritis concurrently with acute pancreatitis.6,12 A single case of emphysematous gastritis in a dog had concurrent emphysematous hepatitis following elective ovariohysterectomy. 10 Given the paucity of cases, it is not possible to associate risk factors with emphysematous gastritis in dogs and cats, but the cases with pancreatitis and previous abdominal surgery are instructive, given that these are known risk factors in humans. In our case, chronic pancreatitis and clinical ileus likely contributed to the gastritis, given that both are documented risk factors in humans with emphysematous gastritis.
The bacterial culture of C. perfringens type C from the stomach in our case is unusual. The more commonly isolated C. perfringens strain in dogs with GI disease is C. perfringens type A, with additional production of enterotoxin or NetF toxin.16,21 Although less frequently reported, C. perfringens type A has been isolated from dogs with diarrhea with production of beta2 toxin with or without enterotoxin. 14 C. perfringens type C is most commonly known for causing necrotizing enteritis in neonatal piglets, lambs, calves, goats, and foals, and enteritis necroticans in humans.3,4,16 Microscopically, the submucosa is expanded by hemorrhage, edema, emphysema, and variable amounts of inflammatory cells. 3 C. perfringens type C is linked to ingestion of trypsin inhibitors through diet, such as colostrum, or physiologic endogenous trypsin deficiency, resulting from protein-deficient diets or pancreatic disease.16,17 In health, beta toxin, which is produced by C. perfringens type C, is degraded by trypsin, and healthy degradation is lost in the presence of trypsin inhibitors or the lack of endogenous trypsin. 16 Additional C. perfringens toxins with trypsin sensitivity include beta2 toxin, 7 which was also identified in the strain in our case.
The pancreatic disease in association with gastric C. perfringens type C infection in our case is intriguing. We hypothesize that the pancreatic disease was the catalyst for this rare infection and resulting disease manifestation. Chronic pancreatitis and extensive peripancreatic fibrosis contributed to extrahepatic bile duct obstruction and the development of ileus. The dog received empirical clindamycin once in the hospital, likely causing intestinal dysbiosis. 22 The ileus also contributed to delayed gastric emptying, which, along with pancreatic disease, are risk factors for developing emphysematous gastritis in humans.1,11,13 These conditions likely led to colonization of the stomach by C. perfringens type C and other bacteria, which manifested as emphysematous gastritis. We hypothesize that duodenogastric reflux occurred in our case, given the persistent ileus and regurgitation, which would introduce trypsin into the gastric environment. However, this dog had developed EPI as a result of chronic pancreatitis, which led to endogenous trypsin deficiency. Hence, any refluxed trypsin activity in the gastric environment would be minimal, which could have allowed trypsin-sensitive beta toxins from C. perfringens type C to exert cytotoxic effects on the gastric mucosa, contributing to the emphysematous gastritis. Our case adds dogs to the list of species susceptible to C. perfringens type C infections. We hypothesize that the known mechanism for type C infections in other species (i.e., loss of trypsin inhibition) can be applied to dogs as well.
Footnotes
Acknowledgements
We thank the personnel of the Small Animal Emergency, Critical Care, and Internal Medicine services of the Veterinary Medical Teaching Hospital, Pathology Diagnostic Laboratory, Gastrointestinal Laboratory, and Clinical Microbiology at Texas A&M University, and the Molecular Diagnostics section of Texas Veterinary Medical Diagnostic Laboratory, College Station branch, for their clinical contributions and technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
