Abstract
A captive, 5-y-old, female, Indian blue peafowl (Pavo cristatus) was submitted for postmortem examination and diagnostic work-up following 24 h of open-mouth breathing, lethargy, and drooping of wings. Grossly, the trachea was diffusely, dorsoventrally flattened with irregular walls and significant narrowing of the lumen. There was no evidence of trauma in the surrounding skin, subcutaneous tissue, or esophagus. Microscopically, tracheal cartilage was diffusely replaced by immature bone tissue with wide medullary cavities containing thin, immature, and disorganized trabeculae covered by abundant osteoclasts. The proximal tibiotarsus and femur were osteopenic, with thin compact cortical bone, multifocally distended osteonic canals, and increased osteoclastic activity and fibrosis on endosteal surfaces of affected trabeculae. Concurrent tracheal collapse and osteopenia have not been reported previously in an Indian blue peafowl, to our knowledge.
The Indian blue peafowl (Galliformes, Phasianidae, Pavo cristatus) originates from South Asia. Indian blue peafowl are the most globally distributed of the 3 peafowl species, and are kept as exotic pets, housed in zoologic institutions, or live in the wild as semi-feral to feral populations. 13
Longitudinal collapse of the trachea causing respiratory signs and mortality in avian species is rarely reported in the literature.7,16 Most reported cases of trachea stenosis in pet, aviary, and zoo birds are associated with damage to the trachea and narrowing of the lumen subsequent to intratracheal procedures, traumatic events, or post-surgical extraction of foreign bodies.5–7,11,12,17
Here we describe the clinical and pathologic findings of fatal tracheal collapse in an osteopenic captive Indian blue peafowl. The carcass of a 5-y-old, female, Indian blue peafowl was submitted to the California Animal Health & Food Safety Laboratory System (CAHFS), Turlock branch (University of California–Davis, Davis, CA, USA) for postmortem examination and diagnostic work-up. The bird was submitted by a hobby aviculturist and belonged to a small muster of 3 peacocks and 1 peahen with peachicks. All birds were housed in an outdoor enclosure with a chain-link fenced perimeter. Birds were fed homemade chicken scratch, Exotic Gamebird Maintenance (Mazuri) with a calcium:phosphorus ratio of 2:1, a mix of fruit and vegetables, and ad libitum fresh drinking water. There was no record of prior vaccinations or medications. The owner reported a 24-h clinical course before death. Clinical signs included open-mouth breathing, lethargy, and drooping of wings. There was no history or evidence of a previous traumatic event occurring before the onset of the signs and death. No evidence of illness was observed in the other 4 adult peafowl.
At autopsy, the peahen weighed 2.71 kg and was in good postmortem and body condition, evidenced by well-fleshed pectoral musculature. The only significant macroscopic finding was longitudinal, dorsoventral flattening of the trachea, particularly severe in the middle third, resulting in marked narrowing of the tracheal lumen. The tracheal rings were irregularly shaped. Long bones were grossly unremarkable and resisted bending attempts. Representative tissue samples from cerebrum, cerebellum, cervical vertebrae, spinal cord, peripheral nerves, nasal passage, infraorbital sinus, trachea, heart, lungs, liver, kidneys, skeletal muscles, proventriculus, gizzard, intestines, ovary, oviduct, proximal tibiotarsus, and femur were collected in 10% buffered formalin (pH 7.2) and fixed for 24–48 h. Tissues with an osseous component were demineralized (Decal decalcifier; StatLab) for 4–5 h (trachea) or overnight (nasal passage and bones). All tissues were processed by standard histologic procedures to produce H&E-stained sections. In addition, 4-µm thick sections of trachea were stained with Masson trichome (StatLab) and toluidine blue (LabChem).
Swabs from the infraorbital sinus, trachea, lung, heart, liver, and ovary were cultured on 5% sheep blood agar (Hardy) and MacConkey agar plates (Hardy) and incubated with 7% CO2 at 37°C for 48 h. A swab from the trachea was also cultured anaerobically on Brucella blood agar and phenylethyl alcohol blood agar plates (Anaerobe Systems) at 37°C for 2–5 d. No bacterial respiratory pathogens were isolated.
An oropharyngeal swab was collected for detection of avian influenza A virus (avian IAV) by reverse-transcription quantitative real-time PCR (RT-qPCR). Additional tracheal swabs were collected and submitted for tests for gallid herpesvirus 1 (syn. infectious laryngotracheitis virus, ILTV), infectious bronchitis virus (IBV), and Mycoplasma gallisepticum (MG) by qPCR and RT-qPCR, previously reported in captive peafowl.2–4 Avian IAV, IBV, ILTV, and MG were not detected. For general viral screening, homogenized tracheal tissue was processed and examined by transmission electron microscopy (TEM), as described previously 1 ; results were negative.
Microscopically, in transverse tracheal sections, collapsed cartilage rings (Fig. 1A) were replaced by bone tissue with widened medullary cavities containing disorganized, thin, immature trabeculae with jagged borders, abundant osteoclasts, and hematopoietic cells (Fig. 1B). In Masson trichome–stained transverse sections of the collapsed trachea, blue-stained, immature bone replaced most of the ring cartilage tissue (Fig. 1C). Toluidine blue–stained transverse tracheal sections had scant, small metachromatic areas of remaining cartilage. The nasal passage and sinus walls had microscopic changes similar to those observed in the trachea. Sections of femur and proximal tibiotarsus had thin trabeculae, abundant osteoclasts, and small amounts of fibrous tissue diffusely covering the endosteal surfaces of thin compact redundant cortical bone, and distended resorption canals (Fig. 1D).
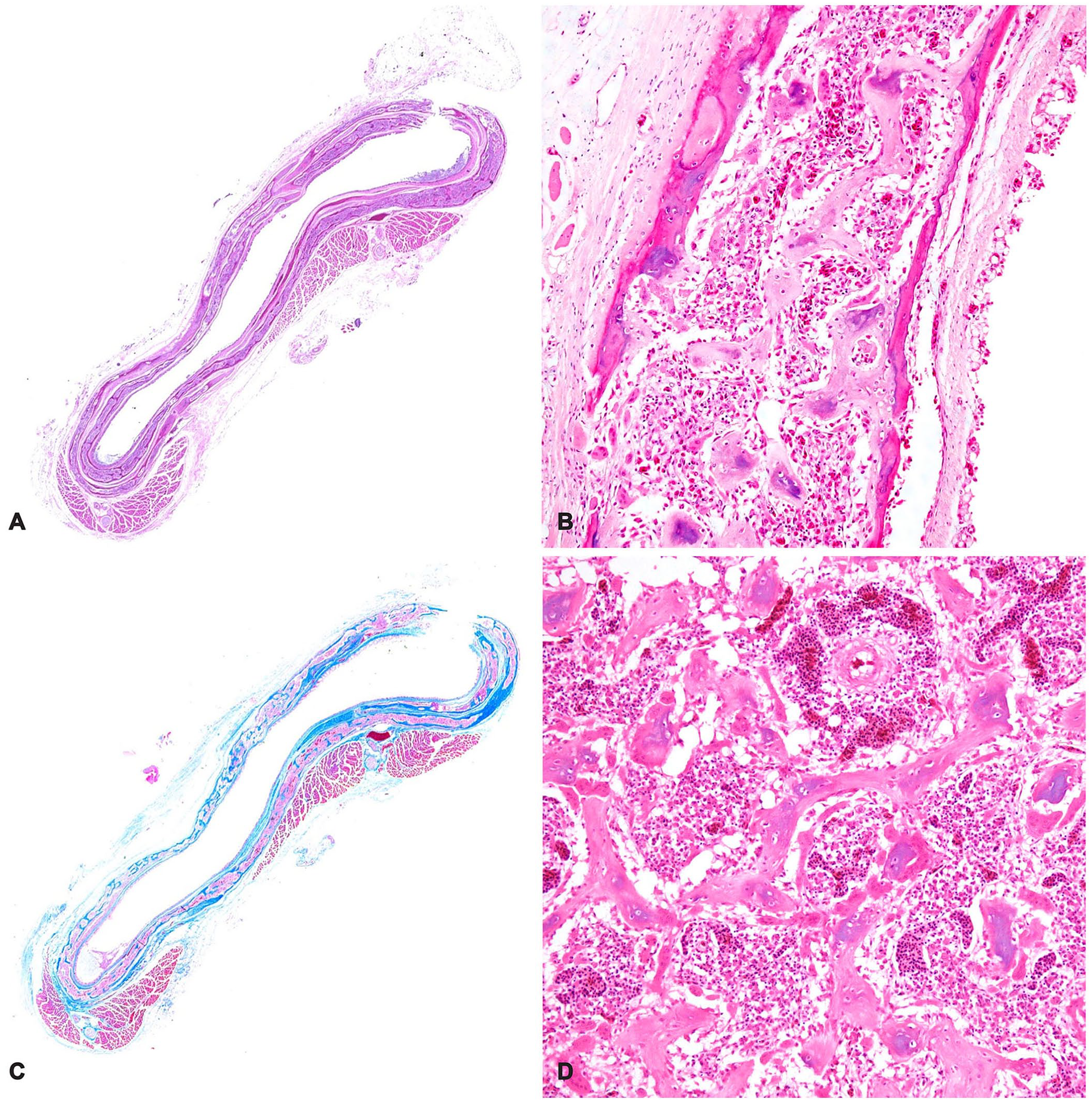
Histologic findings of the collapsed trachea from a captive Indian blue peafowl (Pavo cristatus) with osteopenia.
Fibrous osteodystrophy and osteoporosis are conditions triggered by unbalanced diets causing increased bone resorption in avian species.14,19 Fibrous osteodystrophy is noted in young birds, caused by excessive phosphorus in the diet, and noted microscopically as thin cortical bone, and replacement of trabecular bone by fibroblasts and collagen bundles. Osteoporosis, a multifactorial entity that affects caged layers, is seen as a decrease of normal mineralization of structural bone, producing fragility and susceptibility to fractures. Osteoporosis is particularly triggered when diets with an imbalanced calcium:phosphorus normal ratio are fed, but is also influenced by other dietary factors such as phytates or vitamin D levels. 19 Despite the absence of spontaneous fractures and lack of bone weakness at autopsy, both proximal tibiotarsus and femur in our case had histologic changes suggestive of osteopenia. Significant bone resorption with abundant osteoclasts but unremarkable proliferation of fibrous tissue was observed. Although osteopenia is typically noted in old laying hens, chronically inadequate dietary calcium can lead to osteoporosis and lead to similar lesions in young laying birds, as may have happened in our case. Potential nutritional deficiencies contributing to osteopenia and tracheal collapse were not elucidated in our case, and serum of this animal for calcium and phosphorus determination was not available. A genetic component for the tracheal collapse cannot be ruled out.
We found 2 reports of focal or diffuse tracheal collapse associated with ossification of cartilage in bird species different from peafowls searching in Google, PubMed, CAB Direct, Web of Science, and Scopus using the search terms “trachea”, “collapse”, and “birds”.8,16 Collapse of the proximal trachea was described in a 5-y-old, pet Pekin duck (Anas platyrhynchos) with a 75% reduction in the trachea lumen. The duck suffered acute respiratory distress episodes with head-shaking and post-exertional collapse for 6 mo before euthanasia. The trachea was microscopically characterized by the replacement of tracheal cartilage by mature bone with the medullary cavity containing hematopoietic cells and adipocytes, with fibrosis in the lamina propria. 8 Multiple dyspneic episodes were noted for 2 y before death in a 13-y-old, zoo-housed, yellow-crowned amazon parrot (Amazona ochrocephala). Grossly, severe narrowing of the tracheal lumen was noted due to dorsoventral flattening. Microscopically, the tracheal collapse was associated with diffuse ossification and osteopetrosis of tracheal rings, and there was extensive replacement of tracheal cartilage by compact bone. 16 Bones were not evaluated in either reported case of tracheal collapse, although radiographic evidence of osteopenia was noted in the yellow-crowned amazon parrot.8,16
We found no agents associated with respiratory disease by histology, bacteriology, TEM, or molecular tests of the trachea; however, the possibility of a pathogenic agent that was no longer present or was undetected cannot be ruled out. In our case, there were no gross or microscopic changes indicative of trauma in the surrounding region that could have contributed to tracheal collapse in this bird. Gross appearance of the trachea somewhat resembled lesions caused by bordetellosis (turkey coryza), 15 which was included as one of the first differential diagnoses in this case, especially after noting reported seropositivity against Bordetella avium in captive peafowl from 3 zoologic institutions in Michigan, USA. 10 However, the lesions observed in the trachea and nasal walls of our case were not compatible with this bacterial disease, and there was no evidence of Bordetella spp. growth in aerobic culture from the trachea.
The peafowl was in good body condition. A brief period of respiratory signs and lethargy was reported before death, which contrasts with recurrent episodes of respiratory distress in the adult yellow-crowned amazon parrot and the Pekin duck, reported with diffuse and focal tracheal collapse, respectively.8,16 Ossification of tracheal cartilage rings may be noted normally with age in avian species, promoting rigidity and resistance to collapse,9,18 but the diffuse immature ossification and remarkable osteoclast proliferation noted microscopically in the trachea in our case likely reduced elasticity and prevented the tissue stability necessary to avoid tracheal collapse.
Footnotes
Acknowledgements
We thank the staff of CAHFS Turlock, San Bernardino, and Davis branches for excellent technical assistance in this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
Funding was provided by the California Animal Health and Food Safety Laboratory System, School of Veterinary Medicine, University of California–Davis.
