Abstract
A 2-y-old, intact female, mixed-breed dog was presented to the veterinary hospital with abdominal distension, anemia, and lethargy following a chronic history of nonspecific gastrointestinal signs. CBC and serum biochemistry revealed moderate nonregenerative anemia with neutrophilia, hypoalbuminemia, hyperglobulinemia, hypoglycemia, decreased urea and creatinine, and hypercholesterolemia. Abdominal radiographs and ultrasound revealed a large heterogeneous mesenteric mass and ascites. Abdominocentesis confirmed septic peritonitis with filamentous bacteria. Fine-needle aspiration of the mass yielded pyogranulomatous inflammation and hyphae. An exploratory laparotomy revealed a large cranial abdominal mass with granulomas present throughout the abdominal cavity. Due to the poor prognosis and disseminated disease, the owner elected euthanasia. Postmortem and histologic examinations detected intralesional mycetomas and bacterial colonies within the mesenteric masses. 16S ribosomal RNA gene PCR and sequencing using formalin-fixed, paraffin-embedded sections identified Nocardia yamanashiensis, Nocardioides cavernae, and Nocardioides zeicaulis. Fungal culture, PCR, and sequencing confirmed Scedosporium apiospermum. Our report highlights the importance of molecular methods in conjunction with culture and histologic findings for diagnosing coinfections caused by infrequent etiologic agents. Additionally, we provide a comprehensive literature review of Scedosporium apiospermum infections in dogs.
A 2-y-old, 12.8-kg, intact female, mixed-breed dog was presented to the Auburn University Small Animal Veterinary Teaching Hospital Emergency Service (Auburn, AL, USA) with a 1-wk history of a distended abdomen, anemia, and lethargy. The dog had chronic nonspecific gastrointestinal signs that included vomiting, hyporexia, and progressive weight loss unsuccessfully managed with famotidine (1 mg/kg PO q24h, 7 d). The patient was up-to-date with vaccinations (as per the owner) and was not being treated with any immunosuppressants including corticosteroids.
On presentation, the patient was quiet but alert, with a 39.3°C body temperature (RI: 38.3–39.2°C), tachypnea (44 bpm, RI: 18–34 bpm), and pale-pink and moist oral and conjunctival mucous membranes. CBC revealed a moderate normocytic, normochromic, nonregenerative anemia (Hct 0.22 L/L; RI: 0.38–0.59 L/L) with mild anisocytosis and slight polychromasia, and marked leukocytosis (47.6 × 109/L, RI: 5.1–17.4 × 109/L) characterized by mature neutrophilia (35.7 × 109/L; RI: 2.6–10.4 × 109/L) with a left shift (5.2 × 109/L; RI: 0.0–0.3 × 109/L), monocytosis (2.8 × 109/L; RI: 0.2–1.2 × 109/L), and a slight toxic change in neutrophils. Serum biochemistry showed decreased albumin (11.7 g/L; RI: 30.0–43.0 g/L), glucose (2.77 mmol/L, RI: 4.22–6.44 mmol/L), urea (3.0 mmol/L; RI: 3.2–12.1 mmol/L), creatinine (26 μmol/L; RI: 44–141 μmol/L), and increased globulin (62 g/L; RI: 20–43 g/L) and cholesterol (9.4 mmol/L; RI: 3.4–8.7 mmol/L). A large peripancreatic mass with several other masses and effusion were found during an abdominal ultrasound. Abdominocentesis confirmed an exudate (total solids: 51 g/L; cells: 54.7 × 109/L); the differential cell count was ~72% nondegenerate neutrophils, 21% macrophages, and 8% lymphocytes.
A direct smear of the abdominal fluid yielded very rare filamentous rod-shaped bacteria that were found intracellularly and in a few small extracellular mats and microcolonies. Macrophages were variably vacuolated and occasionally contained intracytoplasmic phagocytized debris. Fine-needle aspiration of the largest mass revealed numerous degenerate neutrophils and epithelioid macrophages surrounding dense mats of fungal organisms that were pleomorphic and varied from round-oval yeast (sometimes with narrow-based budding) to elongated, 2.5–4-μm, variably septate hyphae or pseudohyphae with occasional bulbous ends and thin colorless walls or capsules and granular blue-pink internal structures (Fig. 1A). Exploratory laparotomy confirmed widespread peritoneal granulomas. A large, firm, irregular mass encompassed most of the cranial abdomen, including the entire small intestinal tract and mesentery, greater curvature of the stomach and omentum, and head of the spleen. Given the extension of the lesions in conjunction with a poor prognosis of an intra-abdominal fungal infection, the owner elected euthanasia.
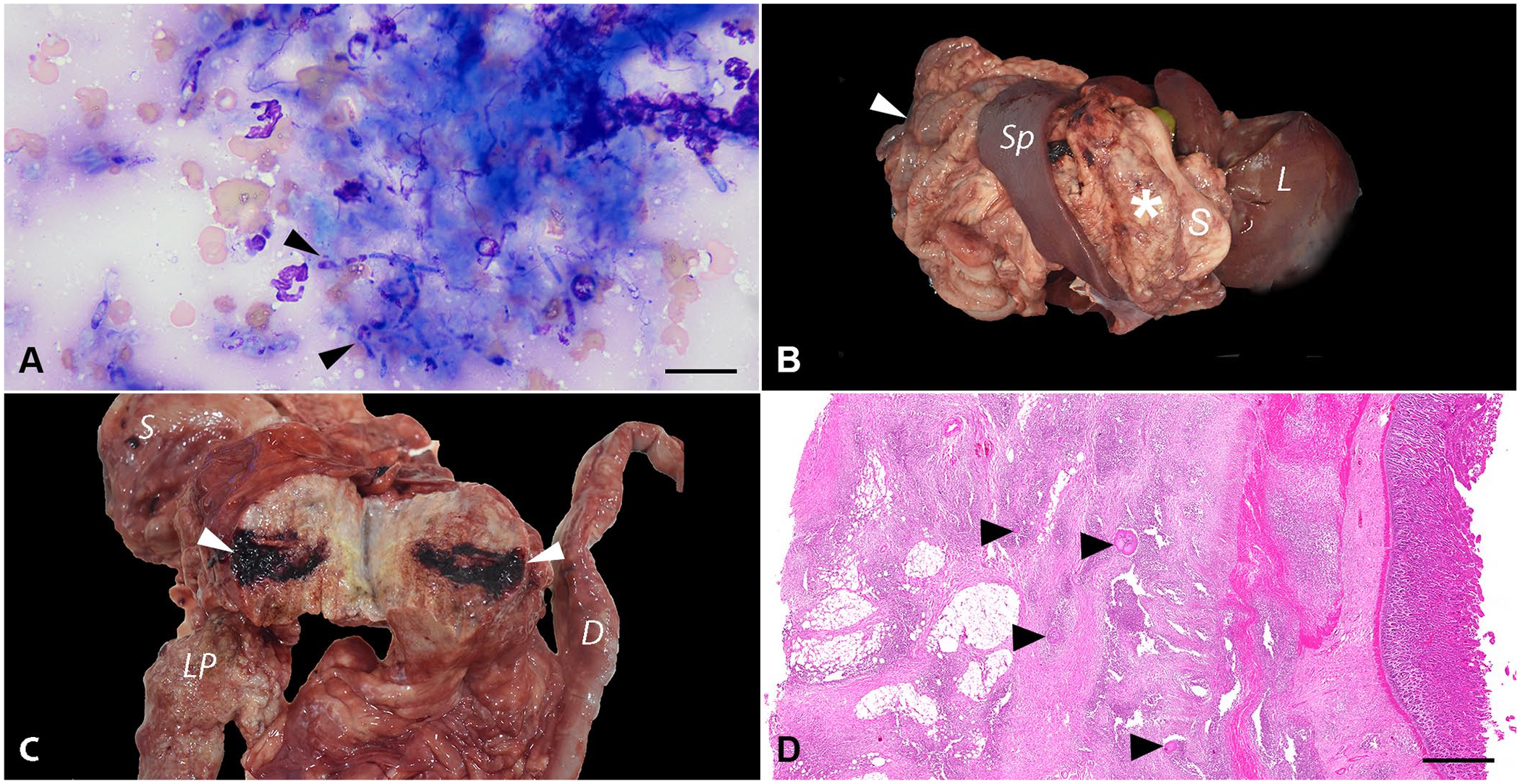
Systemic nocardiosis, nocardioidiosis, and scedosporiosis coinfection in a 2-y-old mixed-breed dog.
On postmortem examination, the dog was in poor nutritional condition (body condition score 2 of 9). The peritoneal space contained ~100 mL of dark-red fluid. The peritoneum was diffusely thickened, with dark-red discoloration and extensive fibrous adhesions encompassing the liver, pancreas, intestine, stomach, and mesentery. An 11 × 12 × 6.5-cm, tan, firm mass effaced the greater omentum between the greater gastric curvature and the left pancreatic lobe (Fig. 1B). On cut surface, the mass was granular, with 0.1–0.3-cm, yellow necrotic foci, and extensive hemorrhagic areas (Fig. 1C). Additional masses were present throughout the abdominal wall. The intestines were diffusely adhered and markedly effaced by similar masses found in the peritoneum, with the intestinal serosa diffusely dark-red and covered with fibrin. The liver adhered to the body wall. White-to-beige, round, firm, 2–4-cm nodules were on the left liver lobes.
On histologic examination, the serosal surfaces of the gastrointestinal tract and the adjacent mesenteries were markedly expanded and effaced by a plethora of reactive fibroblasts amid collagen bundles admixed with scattered large, clear, well-delineated vacuoles. The tissue was obscured by extensive, pyogranulomas composed of a core of neutrophils, epithelioid macrophages, and fewer plasma cells and lymphocytes surrounding central mats of radiating filamentous bacteria (sulfur granules) and central mats of large, non-pigmented fungi (mycetoma; Figs. 1D, 2A). Fite and Gram stains highlighted 1-µm thick, filamentous, variably gram-positive, acid-fast bacteria and occasional 1-µm gram-positive cocci bacteria (Fig. 2B, 2C). The fungal hyphae were septate, 2–6-µm wide, irregularly branching, with 4–7-µm wide terminal bulbous dilations (Fig. 2D), highlighted with Gomori methenamine silver stain (Fig. 2E). In the remaining gastrointestinal tissue, necrotic debris, edema, and hemorrhage were scattered. Histologic findings were similar within the intestinal serosa and lymph nodes. Other histopathologic findings included marked pulmonary congestion and edema, focal lymphoplasmacytic and histiocytic interstitial pneumonia, eosinophilic and granulomatous hepatic capsulitis, moderate periportal lymphoplasmacytic hepatitis, and cholestasis. The eosinophilic and pyogranulomatous inflammation extended to the retroperitoneal and perirenal adipose tissue and the renal capsule.
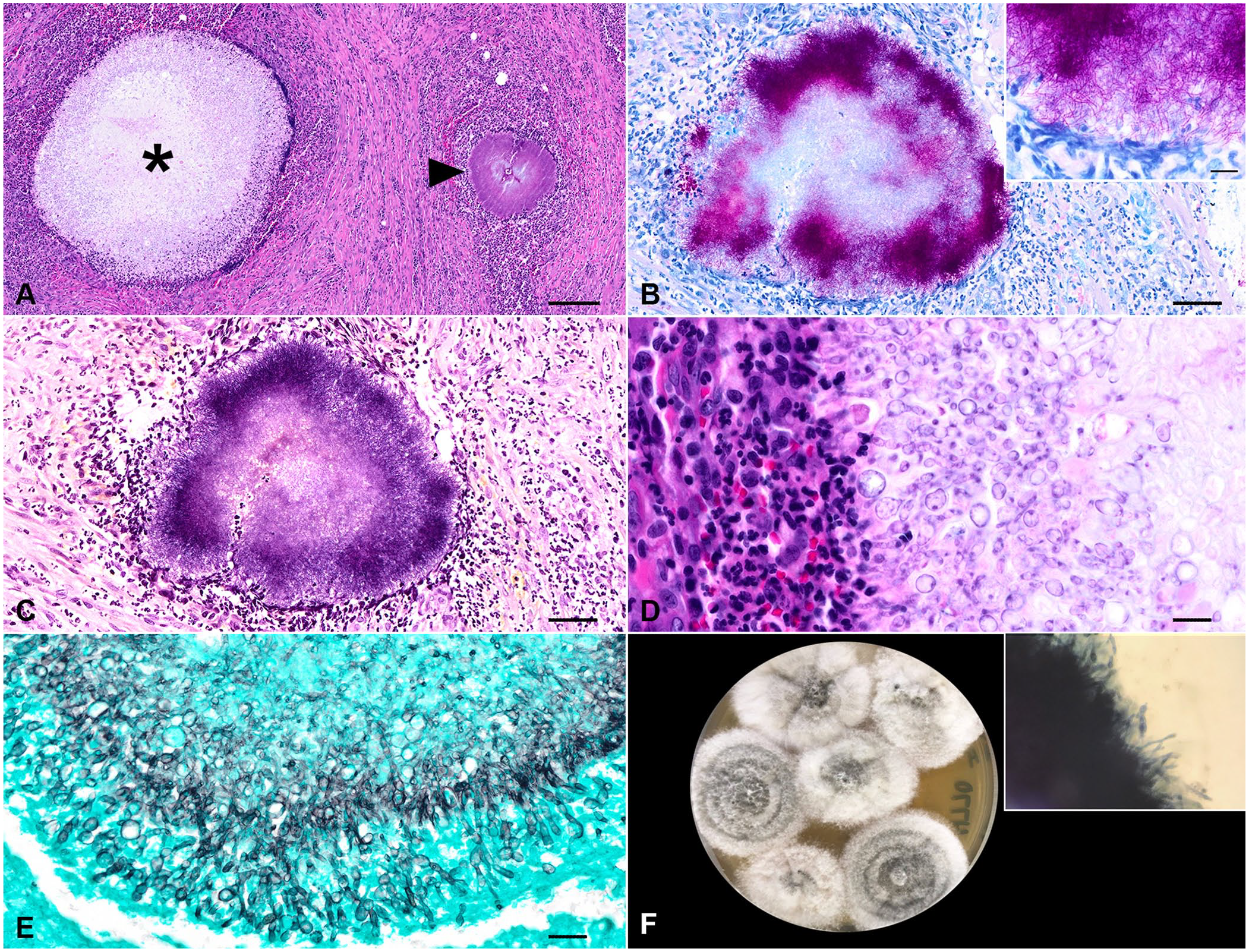
Photomicrographs of the mesenteric mass.
The aerobic culture from mesenteric tissue yielded a heavy growth of mold. The mesenteric tissue plated to Sabouraud dextrose agar (BD Difco), inhibitory mold agar (IMA; BD BBL), and mycobiotic agar (BD BBL) yielded, by day 4, growth of a mold that started white and cottony, with the growth on IMA turning a light-gray with age and having a tan reverse (Fig. 2F). Use of KOH with Parker ink revealed copious branching, septate, hyaline hyphae (Fig. 2F inset). The fungal culture was submitted for identification to the Fungus Testing Laboratory at the University of Texas Health Science Center (San Antonio, TX, USA). Phenotypic characterization and DNA sequencing of 3 genetic loci were used: internal transcribed spacer (ITS) of the rDNA, and partial β-tubulin (TUB) and calmodulin (CAM) genes. Amplification PCR of all target loci was performed with negative and positive controls following validated and published procedures.7,41
BLASTn (https://blast.ncbi.nlm.nih.gov/Blast.cgi) searches were performed. Top matches with sequences of type (CBS 117407) or reference (FMR-8619) strains in BLASTn searches were considered significant. ITS, CAM, and TUB are considered good markers for delimiting Scedosporium species, and ITS and CAM are considered optimal markers. 40 Top matches were with Scedosporium apiospermum type strain CBS 117407T, with 100% base pair match for ITS (GenBank LC739048) and 96.75% for TUB (LC739115) and with the reference strain FMR-8619, with 99.22% match for CAM (GenBank AJ890192). Formalin-fixed, paraffin-embedded sections of the lymph nodes were submitted to the Animal Disease Diagnostic Laboratory at Purdue University (West Lafayette, IN, USA) for subgenic 16S ribosomal DNA PCR and next-generation sequencing, followed by analysis with Geneious (Dotmatics) and BLAST, using a published method. 24 BLASTn searches showed 100% identities with sequences of the following type strains: Nocardia yamanashiensis (GenBank NR_117395.1), Nocardioides cavernae (NR_156135.1), and Nocardioides zeicaulis (NR_148831.1).
Nucleotide data from our study are available in GenBank under the following accessions: Nocardia yamanashiensis (PP565356), Nocardioides cavernae (PP565357), Nocardioides zeicaulis (PP565358), S. apiospermum (calmodulin PP621216, β-tubulin PP621217). Overall, the ancillary molecular assays in combination with gross and histologic findings confirmed systemic coinfection of Nocardia yamanashiensis, Nocardioides cavernae, Nocardioides zeicaulis, and S. apiospermum. Our histologic findings that supported Nocardia infection were the dense mats of Fite-positive, gram-positive filamentous bacteria as reported previously. 34 The histologic findings that support Scedosporium were the thin (3–5-µm wide), non-pigmented, irregularly branching, septate hyphae with bulbous dilations. 51
Nocardia spp. are in the class Actinomycetes, order Mycobacteriales, family Nocardiaceae. 50 French veterinarian Edmond Nocard first described the bacteria within this genus in 1888 on the island of Guadeloupe while studying bovine farcy, a condition causing granulomatous inflammation, abscesses, and pulmonary contention (sic) in cattle now attributed to Nocardia farcinica. 15 Nocardia spp. are saprophytic bacteria that are gram-positive, catalase-positive, partially acid-fast, non-motile aerobes that form characteristic beaded filamentous branches.15,50 Found worldwide, Nocardia spp. are environmental bacteria involved in plant decomposition, with pathogenic species isolated from house dust, garden soil, beach sand, swimming pools, and tap water. 56 Nocardiosis is a suppurative-to-granulomatous, localized, or disseminated opportunistic infection affecting humans and many species of animals that is observed more frequently in immunocompromised hosts.46,50
Various Nocardia spp. are pathogenic in the dog, including: N. abscessus, N. asteroides, N. brasiliensis, N. caviae, N. cyriacigeorgica, N. otitidiscaviarum, and N. veterana. 56 Clinical manifestations of primary nocardiosis are varied and depend on the route of exposure, which can include local cutaneous trauma or inhalation of contaminated particles. 28 Young outdoor dogs frequently develop subcutaneous abscesses secondary to foreign body migration or puncture wounds. 56 Once inoculated, nocardiosis can remain localized (either respiratory or cutaneous-subcutaneous) or have systemic hematogenous spread to other organs. 28 Disseminated or systemic nocardiosis usually develops in young and immunosuppressed dogs. 56 The chronic use of immunosuppressive medications increases the risk of infection. 56 Initially beginning as pulmonary nocardiosis, dogs exhibit peracute clinical signs such as tachypnea, hemoptysis, hypothermia, collapse, and death, with the dissemination of the organisms from the lung into other organ systems leading to signs such as lethargy, fever, decreased appetite, and signs related to the location of infection. 46 The organs involved most frequently in disseminated nocardiosis are skin and subcutaneous tissues, kidneys, liver, spleen, lymph nodes, CNS, eye, bone, and joints. 46 Treatment options for infections that remain localized include surgical debridement of abscesses and fistulous tracts in combination with antimicrobials, including sulfonamides, and discontinuing any immunosuppressive therapy. 46 The prognosis is poor for disseminated nocardiosis, and treatment options are limited. 46
In our case, 16S rRNA PCR and next-generation sequencing allowed the detection of 3 species of opportunistic bacteria, 2 of them apparently without previously documented medical relevance. Molecular methods, rather than culture, are a reliable source of species identification through sequence analysis of 16S rRNA PCR products. 46 Other molecular methods, such as DNA-DNA hybridization and matrix-assisted laser desorption/ionization–time-of-flight mass spectrometry (MALDI-TOF MS), allow the differentiation of closely related species.4,53 Nocardia yamanashiensis has been isolated sporadically from subcutaneous lesions in humans.2,22,35 Additionally, we identified 2 species belonging to the genus Nocardioides, a genus in the class Actinomycetes, order Propionibacteriales, family Nocardioidaceae. 49 Nocardioides spp. are gram-positive, non–acid-fast, catalase-positive, aerobic, and mesophilic bacteria with a ubiquitous distribution. 23 Nocardioides spp. have been isolated from environmental sources including soil, plants, and aquatic environments.18,23,49 Specifically, Nocardioides cavernae was first isolated from a soil sample from a karst cave in Xingyi County in Guizhou province, southwestern China 18 ; Nocardioides zeicaulis was retrieved from the stem tissue of a healthy maize plant. 23 Nocardiosis refers strictly to the infections caused by Nocardia spp. 15 and, to date, there are no medically relevant species or strains within the Nocardioides genus. 49 Given the detection of 2 species within the Nocardioides genus involved in infectious processes in our case, we propose the term “nocardioidiosis” to define infection caused by these bacteria.
Fungi belonging to the Scedosporium spp. complex are versatile and widely distributed organisms that are important human and veterinary pathogens. 12 Scedosporium spp. cause cutaneous, subcutaneous, or disseminated infections in both immunocompetent and immunocompromised hosts, either as primary or opportunistic pathogens.10,40 The route of infection is through inoculation by minor penetrating trauma, through exposed wounds, or inhalation. 10 Scedosporiosis is an infection caused by Scedosporium spp., and sporadic infections in dogs have been attributed to Allescheria boydii, 21 Monosporium apiospermum, 52 Pseudallescheria boydii,1,3,13,32,44,54,55 Scedosporium inflatum, 42 S. apiospermum,5,6,8,9,11,20,25,33,36–39,43,45,48,51,57 and S. prolificans.14,19,47 However, the taxonomy of Scedosporium spp. is rather complex given that it has been subjected to continuous changes since the twentieth century. 10 Such changes within the nomenclature are somewhat confusing and worth clarification when retrieving canine scedosporiosis cases from the literature to avoid considering 2 names as different etiologic agents.
Allescheria boydii has been renamed Pseudallescheria boydii. 31 Monosporium apiospermum was renamed S. apiospermum, 10 and S. inflatum and S. prolificans are now known as Lomentospora prolificans. 30 Scedosporium is the anamorph genus of the teleomorphic Pseudallescheria in the family Microascaceae. 26 Molecular phylogenies published in the last decade led to comprehensive changes in fungal taxonomy and nomenclature and the abolition of the dual nomenclature that was originally based on the anamorph/teleomorph concept of fungi.16,26 This led to the comprehensive revision of the Scedosporium/Pseudallescheria genus, and Scedosporium was adopted as the current name for the genus following the one name/one fungus concept. A comprehensive phylogenetic assessment of the Microascaceae led to the exclusion of S. prolificans from Scedosporium and adoption of Lomentospora prolificans, which is the older name of S. prolificans. 30 The genus Scedosporium includes the S. apiospermum complex (S. angustum, S. apiospermum, S. boydii, S. ellipsoideum, S. fusarium), S. aurantiacum, S. cereisporium, S. dehoogii, S. desertorum, and S. minutisporum.16,17,26,30 Thus, the term scedosporiosis refers to the infections caused exclusively by Scedosporium spp.
S. apiospermum infection has been reported in dogs; affected animals were 8-mo-old to 10-y-old (Table 1). Clinical presentations include unilateral rhinitis, superficial keratitis, cutaneous or subcutaneous infections, localized skeletal infections, and disseminated or localized visceral infections (Table 1). The prognosis for localized infections is favorable after antifungal treatment with voriconazole, itraconazole, terbinafine, or ketoconazole in combination with surgical debridement.6,8,9,25,51 The prognosis is guarded-to-poor in cases with disseminated visceral infection. 11 However, extended remission after surgical removal in combination with systemic antifungal therapy has been reported in cases of visceral disseminated infection.48,51,57 In our case, the extension of the lesions made surgical treatment nonviable; thus, euthanasia was elected and medical treatment was not considered.
Documented cases of Scedosporium apiospermum infections in dogs.
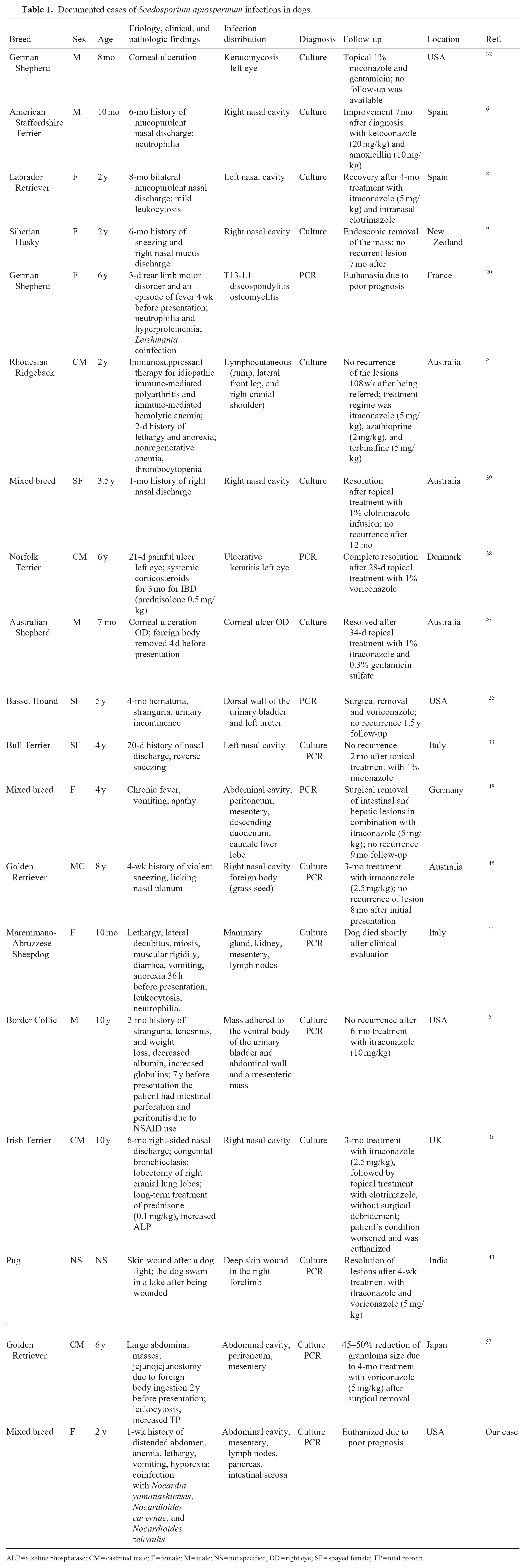
ALP = alkaline phosphatase; CM = castrated male; F = female; M = male; NS = not specified, OD = right eye; SF = spayed female; TP = total protein.
The diagnosis of scedosporiosis is achieved through culture in combination with molecular testing.11,33,45,51 Because the identification of the fungus based only on morphology is unreliable, ancillary testing using MALDI-TOF MS and DNA sequencing of phylogenetically informative targets such as ITS, CAM, and TUB is recommended for S. apiospermum identification.27,29,57 As found in our case, S. apiospermum colonies are wooly, with gray-brown to gray-white margins, and branching, septate, hyaline hyphae. 57 The reported histologic features align with our findings, with the description of mycetomas containing 3–5-μm wide, non-pigmented, parallel-walled, irregularly branching, septate hyphae with terminal bulbous dilations. 51
In the documented cases of S. apiospermum intra-abdominal infection, the agent was inoculated through penetrating trauma directly into the peritoneal space without inflammatory lesions in the ventral subcutis or abdominal wall. 57 Specifically, the 2 case reports of intra-abdominal infection reported previous intestinal perforation and peritonitis or jejunostomy due to a foreign body as the inciting causes.51,57 In our case, there was no evidence of previous abdominal surgery or traumatic injury; the patient had not been spayed. However, we strongly suspect that penetrating trauma through the abdominal wall is the most likely origin of infection in our case, in the absence of primary enteric disease.
The use of corticosteroids at immunosuppressive doses to treat immune-mediated conditions, including polyarthritis and inflammatory bowel disease, has been identified to increase the risk for opportunistic lymphocutaneous and ocular infection caused by S. apiospermum in dogs.5,38 The patient in our case was not under chronic treatment with immunosuppressive drugs, including corticosteroids, according to the owner. Viral comorbidities were unlikely given that the patient was up-to-date with vaccinations, according to the owner. Histologic findings within the liver and kidneys revealed the extension of the peritoneal inflammation within the capsule, retroperitoneal, and perirenal adipose tissue. Viral inclusions or suppurative infiltrates within the airways were not found. However, the absence of viral inclusions does not rule out a viral coinfection. Ancillary testing for viral infections was not performed in our case and this is a limitation of our study. CBC and serum biochemistry findings reported in canine scedosporiosis are nonspecific and include neutrophilia, hyperglobulinemia, hyperproteinemia, thrombocytopenia, nonregenerative anemia, and increased alkaline phosphatase activity.5,20,32,36,51 Neutrophilia, nonregenerative anemia, and hyperglobulinemia were the clinicopathologic findings reported in our case, due to chronic peritonitis. Leishmania coinfection was documented in a S. apiospermum case of T13-L1 discospondylitis and osteomyelitis in a dog. 20
The coinfection by ubiquitous saprobic and opportunistic infectious agents is infrequent and most likely derived from their inoculation during penetrating trauma. The extension of the lesion and immunologic status of the affected host are determinant factors. Nocardiosis and scedosporiosis are well-documented, rare infections in dogs. However, the presence of Nocardioides bacteria, historically lacking medical relevance, poses a diagnostic challenge. Thus, molecular methods are essential in these cases.
Footnotes
Acknowledgements
We thank Lisa Parsons, Lisa Jolly, and Keisha Snerling from the Auburn University College of Veterinary Medicine (AUCVM) Histology Laboratory for their technical assistance and slide preparation. We thank Stephen Gulley for his technical assistance during the postmortem evaluation.
Declaration of conflicting interests
Eric J. Fish is the owner of EJF Veterinary Consulting. The remaining authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
