Abstract
Published information about fish botulism is scant. We review here the current literature on fish botulism. Freshwater fish are susceptible to botulism. Only anecdotal evidence exists about possible botulism cases in saltwater fish. With only a few exceptions, the etiology of all cases of fish botulism reported is
Keywords
Botulism is caused by botulinum neurotoxins (BoNTs) produced by BoNT-producing clostridia, including
Most fish botulism cases are produced by
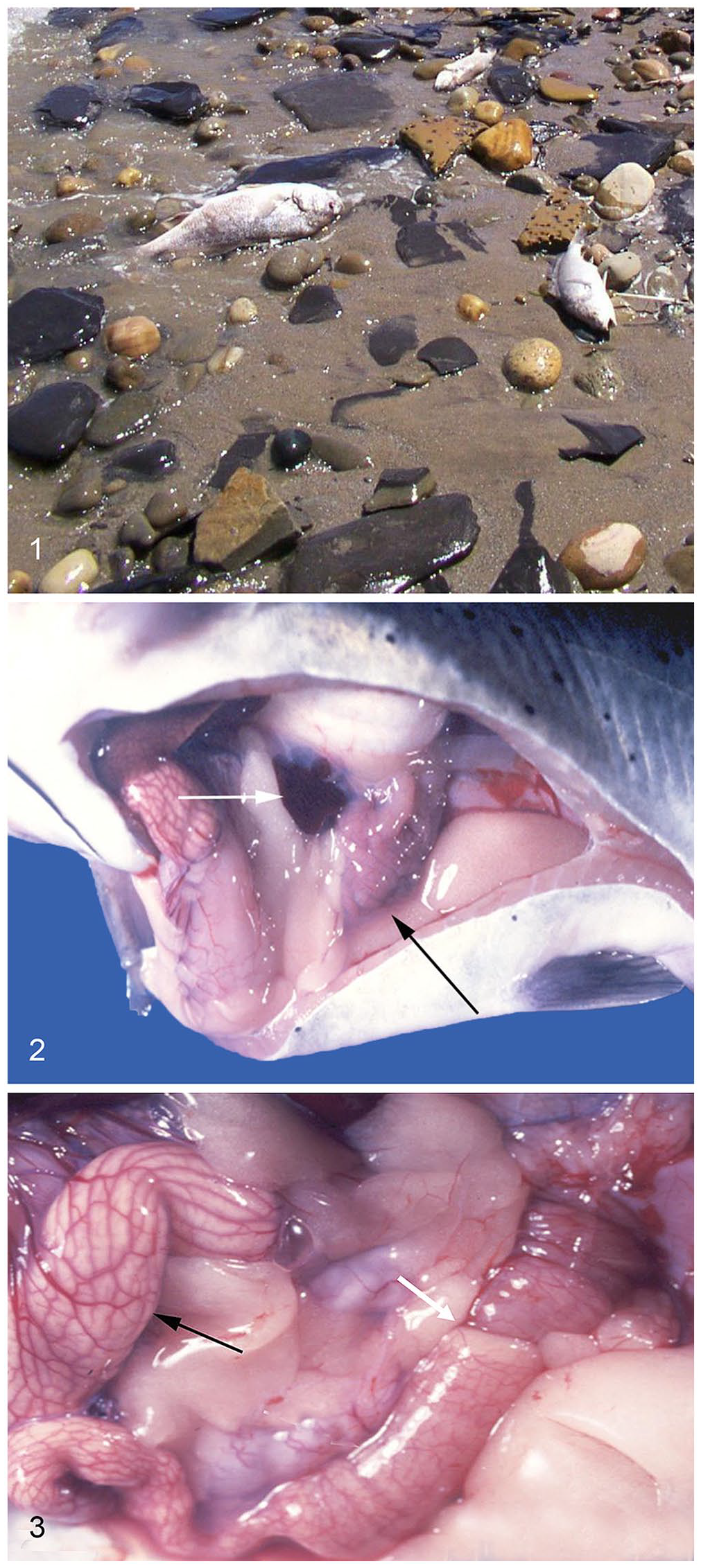
Several dead fish on a beach; part of an outbreak of
Etiology
In North America,
All BoNT subtypes block the release of acetylcholine at the neuromuscular junction and lead to flaccid paralysis. Different types are more prevalent in certain species; for instance, BoNT types A, B, E, and, more rarely F, are mainly responsible for human botulism, whereas toxin types C and D and their mosaic variants are mostly involved in mammalian and avian animal botulism.
34
This is not, however, a strict rule, and exceptions occur. Although
Most cases of fish botulism reported to date have been produced by
Although most cases of fish botulism are produced by
Pathogenesis
The pathogenesis of botulism in fish has not been studied at the molecular level as it has been in other species. However, due to similarities in the neuromuscular system of fish compared to other vertebrates, 52 and the fact that experimental exposure to BoNTs causes similar clinical signs, including flaccid paralysis, 7 it is assumed that the pathogenesis in fish is similar to that in mammalian and avian species.
Experimental infections have demonstrated that several fish species are particularly susceptible to BoNT/E. For instance, in coho salmon inoculated by the oral route, type E toxin was lethal at a dose equivalent to 90 mouse intraperitoneal minimal lethal doses. 17 The median lethal dose in channel catfish fingerlings was 13.7 pg/fish when injected intracoelomically. 7 Zebra fish have variable sensitivity to different BoNTs. The 96-h median immobilizing doses of BoNT/A, BoNT/C, BoNT/E, and BoNT/F for adult male Tübingen strain zebrafish were 16.3, 125, 4.7, and 0.61 pg/fish, respectively. 9
Two mechanisms of intoxication by BoNTs have been proposed for fish. It is thought that fish acquire
Epidemiology
Most cases of botulism reported in fish have been produced by
Sporadic reports of botulism in fish are documented in the scientific literature. In commercial fish production, several large-scale mortality events in salmonid hatcheries have been attributed to type E botulism.5,15,16,27 Outbreaks are most common in juvenile salmonids (including chinook and coho salmon, and steelhead and rainbow trout) raised in earthen bottom ponds. Toxins were detected in sediments, dead fish, and, to a lesser degree, invertebrates found within the ponds.5,16,27 Dead fish are a major source of BoNTs in these epizootics, and cannibalism plays a significant role in the spread of this toxin to other fish. Outbreaks are typically managed by rapidly removing dead fish and transitioning the remaining fish to concrete ponds. 16 Similarly, visceral toxicosis of catfish (VTC) is a form of botulism described in channel catfish. VTC was first recognized in the late 1990s in commercially raised catfish in the southeastern United States.29,54 It predominantly affects food or brood-size fish, and occurs largely in the spring and fall. 29
Sporadic natural outbreaks of type E fish botulism have been documented in freshwater lakes.19,21 The bulk of the published literature regarding mortality events associated with type E botulism has focused on wildlife mortality (specifically waterfowl and shore birds) in the Great Lakes of North America. In these outbreaks, both toxigenic and nontoxigenic strains of
Moribund fish clinically affected by botulism often exhibit changes in behavior (e.g., head bobbing at the surface and inability to maintain a normal position within the water column), which may predispose them to consumption by piscivorous birds that only eat live fish.20,55,56 Experimental studies suggest that susceptibility to type E botulism varies between fish species.55,56 Onset of clinical signs (e.g., hyperpigmentation, behavioral changes) and mortality among evaluated species (round goby, walleye, rainbow trout, yellow trout) vary considerably. For example, at the same dose, mortality ranged from 25% in yellow perch to 92% in round goby. 56
Zoonotic risk of fish botulism
Foodborne type E botulism linked to the consumption of fish has been reported, and it was thus suggested that botulism can be considered a zoonosis. 45 However, the term zoonosis does not apply to these cases of human disease because zoonosis means direct transmission from diseased animals to humans. The cases of human botulism mentioned above resulted from consumption of contaminated food of animal origin, but not from transmission from sick animals (Michel R. Popoff, pers. comm., 2023).
Cases of human botulism type E have been associated with consumption of salted dried ham in France36,44 and Argentina.
48
In those cases, it was suggested that sea salt used for the preparation of ham could have been the source of contamination. However, although
Other cases of type E human botulism have been reported associated with consumption of fish or other seafood.22,35,41,46,53 An outbreak of human type B botulism was reported in France associated with consumption of homemade preserved sardines.11,37 In that outbreak,
Clinical signs
Natural disease
Wild fish with natural botulism are described as being hypersensitive and nervous in the early stages of disease. This progresses to paralysis of fins, loss of equilibrium, swimming on one side, loss of color, gaping of gills, and elevated mortality.5,15,16 In captivity-raised fish, additional reported clinical signs include fish being pushed into screens or to low-flow areas. Muscle fasciculations, quivering barbels, and occasionally eversion of the stomach and exophthalmia have been reported in affected catfish. 29
Experimental disease
Clinical signs in experimental infections are similar to those described above for natural disease, although some subtle variations in clinical signs between fish species were noted. For instance, in fish experimentally infected with
Thus far, there does not appear to be a consensus as to the best method of administration of BoNT/E to reproduce botulism in fish. Some authors administered the toxin orally55,56 in an attempt to replicate what occurs in natural infections, whereas other studies have attempted introducing the toxin via intracoelomic injection.7,8
Pathology
Natural disease
In salmonids that died of botulism, extended gill covers and curved bodies were observed. In these animals, the caudal 10–15 mm of the intestinal tract contained very viscous amber fecal material suggestive of constipation, as described in humans and other animals with botulism. 16
The pathology of VTC was described in detail. 29 Grossly, chyle-rich ascites (Figs. 2, 3), intestinal intussusceptions (Fig. 3), eversion of the stomach into the oral cavity, pale proximal intestine with congested blood vessels (Fig. 3), splenic congestion, and a reticular pattern were observed in the liver. Histologically, there was cerebral, splenic, and hepatic congestion, coupled with splenic lymphoid depletion and perivascular edema, vascular dilation and edema in the gastrointestinal tract, and perivascular edema in the anterior and posterior kidneys.
Experimental disease
In rainbow trout experimentally dosed per os with different doses of BoNT/E, the animals showed dorsal recumbence and opercular abduction,43,44 similar to descriptions in the first experiment of oral exposure to BoNT in fish. 48 The same experimental setting induced progressive dark pigmentation that started as a faint 1–2-cm wide black band just caudal to the pectoral fins and progressively evolved caudally until almost the entire body became black.55,56 Similar results were reported in a separate study with round gobies experimentally exposed to repeated low doses of BoNT/E.55,56 Walleye were in dorsal recumbency with adducted opercular plates and ventrally arched bodies. Yellow perch had abducted opercular plates and darkening of pigmented areas that also progressed to the rest of the body. Loss of color and gaping of the gills was noticed in a field experiment, in which rainbow trout were inoculated with BoNT/E. 5 The mechanism associated with loss of color in fish with botulism is not fully understood. It has been suggested that the chromatophores in fish are innervated, and both the parasympathetic and sympathetic innervation of chromatophores can affect the expansion or contraction of these cells and the degree of pigmentation. 40 It is therefore possible that the changes in pigmentation in BoNT/E-affected fish are related to loss of control over these nerves.
Channel catfish fingerlings developed lesions similar to VTC after experimental intracoelomic inoculation with different doses of BoNT/E, including terminally opened mouths with abduction of the opercula, exophthalmos, ascites, splenic congestion, and intussusception and blanching of the intestine. 7 Zebrafish inoculated intracoelomically with serum of VTC-affected catfish or with BoNT/E had renal tubular necrosis histologically. 8 The gross and microscopic changes observed in some cases of fish botulism seem to be nonspecific and are probably of little diagnostic significance. Something similar occurs in avian and mammalian species in which nonspecific gross findings can be seen occasionally (e.g., nuchal ligament edema in horses). 34
Diagnosis
A presumptive diagnosis of botulism in any animal species is based on clinical signs. However, although highly suggestive, clinical signs alone are not diagnostic and laboratory confirmation is required. As in other species, a botulism outbreak in fish is suspected when there is a rapid increase in mortality associated with flaccid paralysis. Absence of significant gross and microscopic lesions during postmortem examination is supportive of a diagnosis of botulism, although some of the lesions described above in channel catfish with VTC may suggest a diagnosis of this disease. Culture and/or detection of
Diagnostic confirmation requires laboratory detection and identification of the neurotoxin type in animal or feed samples. Despite significant recent developments in methods for the detection of BoNT, mouse bioassay (MBA) remains the gold standard for toxin detection, and it is still the method used most commonly by veterinary diagnostic laboratories. The sensitivity of the MBA is, however, relatively low, and false-negative results are common. Another problem with the MBA is that this test cannot differentiate mosaic types. Finally, the MBA method is time-consuming, laborious, expensive, and presents ethical issues associated with the use of live animals. Very few laboratories throughout the world are equipped to perform the assay. Several alternative laboratory tests to detect BoNTs have been developed, including ELISAs, enzymatic methods based upon BoNT cleavage of synthetic peptides followed by differential detection of the specific product by spectrometry, or cell-based assay.4,6,12,39,42,43,47,49 These techniques, although promising, have, however, not been validated for the diagnosis of fish botulism.
The reported cases of VTC were initially confirmed using a catfish bioassay and mass spectrometric–based endopeptidase methodology, 21 and more recently by bioassay in adult zebrafish. 8 The latter is considered a reliable and practical alternative to rapidly detect BoNT/E in sera from VTC-affected catfish. The preferred samples for MBA in mammals are, in decreased order of sensitivity: gastrointestinal content, liver, and serum, although, if feed suspected to be associated with an outbreak is available, this is the ideal sample to test. However, little information is available in the literature about the sensitivity of testing such samples for the diagnosis of fish botulism.
Because the classic presentation of botulism in wild fish is just en-masse fish mortality, there are several differential diagnoses that should be considered, which vary based on fish species. For example, infection by
Discussion
Subclinical carriage of
The lack of published information about botulism in fish is probably in part associated with the fact that very few laboratories in the world perform routine diagnostic work for this disease and that the current gold standard, MBA, is expensive, laborious, and has low sensitivity. It is therefore possible that at least some cases of botulism in fish go undiagnosed. Another fact that may compound the problem is that the clinical signs and postmortem lesions of botulism are not specific, and a presumptive diagnosis of the disease is not established in many cases. Finally, when mortality occurs in wild fish populations, it is possible that clinical signs are not observed and samples for postmortem examination and ancillary testing are not available.
But even when samples from affected fish are available for laboratory testing, the low sensitivity of the gold standard MBA conspires against a successful diagnostic outcome. As with other species, the low sensitivity of MBA is probably associated with the higher sensitivity of fish to botulism compared to mice. Several other tests for the detection of BoNT, including mass spectrometry, have been used for diagnosis of botulism in humans. 37 Some of these techniques are being validated for use in veterinary medicine and hopefully will be made available to diagnostic laboratories in the not-too-distant future.
To date, only
The reason for this species preference for some types of
Footnotes
Acknowledgements
We thank Ms. Seana Fitisemanu for her work on the references for this paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
