Abstract
Information is limited on the factors that may affect the preputial differential epithelial cell count in healthy intact dogs. Our objectives were to establish RIs of the preputial differential epithelial cell count in dogs and investigate the potential impact of signalment, presence of gross preputial discharge or neutrophils, preputial cleaning, and normal estrogen variation on the differential cell count. We enrolled prospectively 50 client-owned, healthy dogs in our study. Preputial cytology smears were obtained, and we performed a 4-part (parabasal, intermediate, superficial, cornified) differential cell count, before and after gentle preputial cleaning. We found higher counts of fully cornified cells (median: 11–12%) compared to previous studies. The cleaning procedure significantly decreased the intermediate (p = 0.002) and increased the superficial (p = 0.006) cells. Age was positively associated with intermediate (p = 0.007) and negatively with superficial (p = 0.01) and cornified (p = 0.02) cells. The concentration of estrogens was negatively associated with the percentage of parabasal cells (p = 0.04). In healthy dogs, a variable impact may be expected in the preputial differential epithelial cell count, by the cleaning of the prepuce before sampling, the age of the dogs, and the normal variation of estrogen concentration.
Unlike vaginal cytology, which is commonly used in the clinical setting for the evaluation of cycle staging and gynecologic disorders of the bitch, preputial cytology is performed only occasionally, mostly for the assessment of preputial inflammatory and neoplastic conditions.2,3,14 Preputial cytology has also been proposed as a cost-effective tool for the detection of hyperestrogenism in male dogs exposed to endogenous or exogenous estrogen sources.4,5 Preputial mucosa is likely influenced by circulating estrogens in a manner similar to vaginal mucosa, leading to an appreciable increase of superficial cells in cases of persistent hyperestrogenism, which may precede the occurrence of overt feminization syndrome or bone marrow toxicity.5,7,8
Although nucleated squamous cells dominate normal preputial cytology, there is limited information on the RIs and factors (other than excessive estrogens) that may affect the preputial differential epithelial cell count in healthy intact dogs.3,5,13 We hypothesized that by further refining the normal preputial cytologic pattern, the diagnostic utility of this examination in supporting hyperestrogenism would be improved. Therefore, our objectives were to establish RIs of the differential epithelial cell count assessed in preputial cytology of healthy dogs and to investigate the potential impact of factors such as age, breed, body weight, presence of gross preputial discharge or neutrophils, preputial cleaning, and normal estrogen variation on the differential cell count.
We enrolled prospectively in our study client-owned, intact male dogs admitted to a Veterinary Teaching Hospital for preventive health care, elective surgery, or minor dental procedures between September 2020 and March 2022. The study was granted institutional approval (656/15-12-2020), and informed owner consent was required before obtaining the cytologic samples. Dogs were included in the study if they were ≥6-mo-old and were clinically healthy based on history, normal clinical examination findings (including the absence of any appreciable penile, preputial, or testicular lesions or cryptorchidism), and unremarkable CBC (Advia 120; Bayer) and routine serum biochemistry (total protein and creatinine concentrations, and alkaline phosphatase and alanine aminotransferase activities). Dogs with historical evidence of prior hormonal treatment were excluded from the study.
Blood serum estradiol-17β concentrations were measured as a single batch by radioimmunoassay in a commercial diagnostic laboratory (Idexx), and only dogs with concentrations within the laboratory’s RIs for intact male dogs (<91.8 pmol/L [<25 pg/mL]) were eligible for our study. We eventually enrolled 50 male dogs in our study, including 26 purebred and 24 mixed-breed dogs, with an age range of 10 mo–15 y (median: 3 y) and body weights of 3.6–78 kg (median: 20.5 kg).
Before sampling, gross preputial discharge was subjectively graded as 0 (absent), 1 (mild), 2 (moderate), or 3 (copious). Subsequently, 4 preputial cytology smears were prepared from all dogs, by a single experienced clinician (M. Vergou), participating in a 2-y Master of Science program in companion animal internal medicine. Before the initiation of our study, tutorial demonstrations of preputial sampling and smear preparation were performed. A saline-moistened, cotton-tipped applicator was inserted into the prepuce to gently swab the upper preputial mucosa on an ~2-cm2 surface. The collected material was subsequently gently rolled onto 2 glass slides (=pre-cleaning smears). The same procedure was repeated after the prepuce was gently cleaned, using a normal saline-impregnated, non-traumatic gauze to remove any preputial discharge, for the preparation of 2 additional smears (=post-cleaning smears). All smears were air dried, stained with Diff-Quik (Hemacolor; Merck), and their identity masked.
Two preputial cytology smears of sufficient quality (1 pre-cleaning and 1 post-cleaning smear) were evaluated by 2 of the authors (M. Vergou, M.E. Mylonakis). The evaluators, who were anonymized to the identity of the smears, performed a consensus epithelial cell differential, by assessing 100 consecutive cells per smear, using a dual-headed microscope with an embedded ocular micrometer, at 400× magnification (40× objective lens). Neutrophils were also semi-quantified as ≤ or >100 per 100 epithelial cells at 1,000× magnification (100× objective lens).
Epithelial cells were classified based on cytomorphologic features defined for vaginal cytology,2,9,10,12 as follows (Fig. 1): 1) parabasal cells = small round cells (diameter ≤20 μm), with round nuclei and a small amount of cytoplasm; 2) intermediate cells = variably sized, larger than parabasal cells (diameter > 20 μm), with round nuclei and abundant cytoplasm, which may be round, angular, or folded, and may have none or only one cornification line; 3) superficial cells = large cells with small pyknotic nuclei and round, angular, or folded cytoplasm with >1 cornification lines; and 4) cornified cells = similar to superficial cells but with overtly eroded (karyorrhectic or karyolytic) or not-visible nuclei. Any disagreement between the evaluators regarding the identity and classification of cells was settled by reviewing the morphologic criteria specified above.
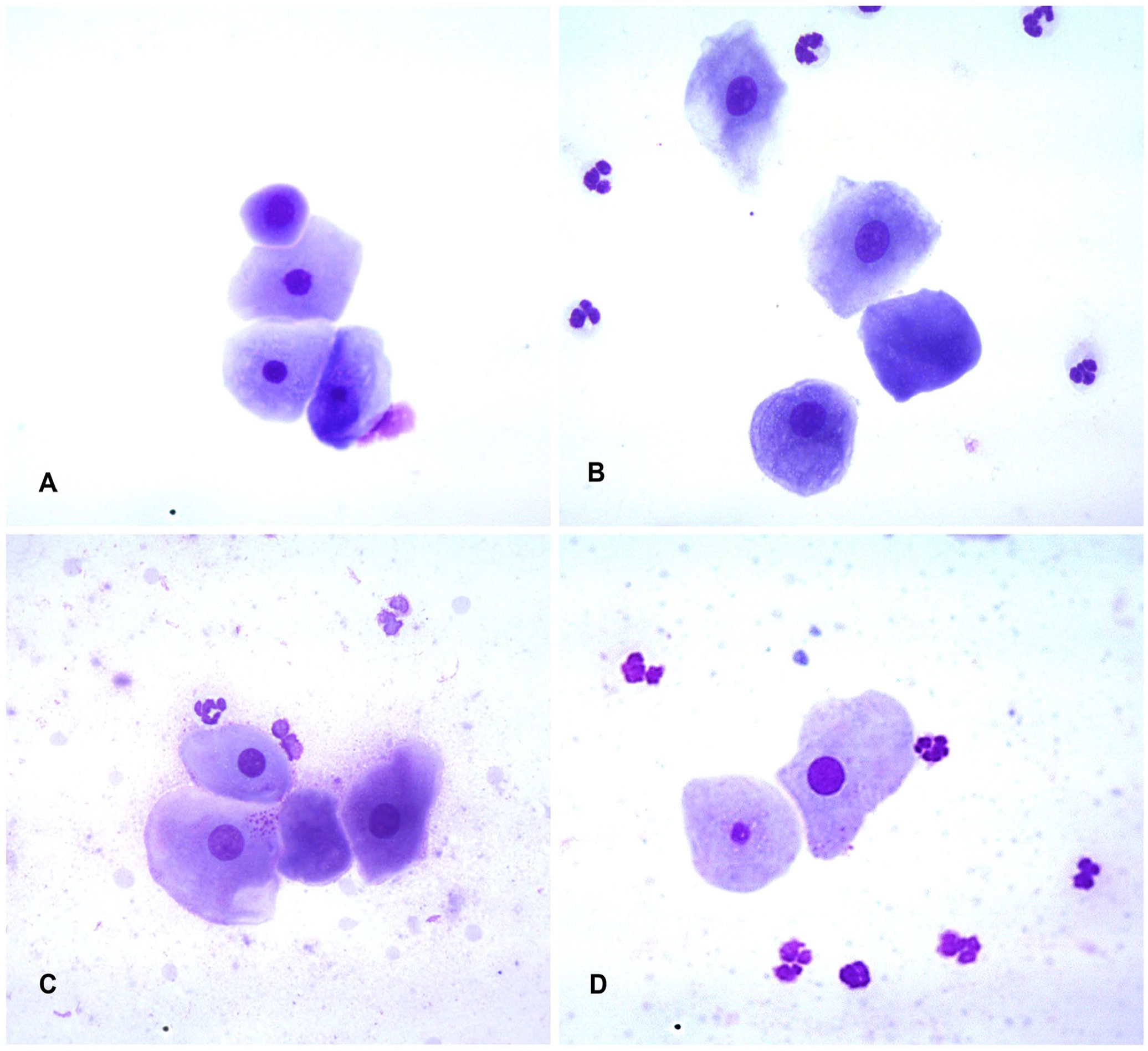
Preputial cytology from a healthy male dog (Diff-Quik, 40× objective lens).
RIs for the percentages of parabasal, intermediate, superficial, and cornified cells were estimated using the “reference intervals” R-package, and CIs were calculated with bootstrapping. 6 Potential differences for variables measured on a continuous scale between groups were assessed with nonparametric statistical methods, including Wilcoxon rank sum test (2 independent samples), Wilcoxon signed-rank test (2 dependent samples), and Kruskal–Wallis test (>2 independent samples). Possible associations between 2 continuous variables were assessed by applying a linear regression model (reported as coefficient and 95% CI). To compare the semi-quantified counts of neutrophils pre- and post-cleaning, a 2 × 2 table was constructed (output expressed as ≤100 or >100) to examine potential associations pre- and post-cleaning. The Pearson chi-square test was assessed, with 2-sided hypothesis specified as the alternative hypothesis. The R programming language (https://www.r-project.org/) was used to construct all tables and to perform all statistical methods. Comparisons were evaluated at the p ≤ 0.05 level of significance.
The cytologic differentiation between intermediate and superficial cells was not always straightforward, especially when cornification lines were not evident, as opposed to the simpler identification of parabasal and cornified cells (Table 1). The cleaning procedure did not affect the parabasal and cornified cells, but the intermediate cells were significantly reduced (p = 0.002) and the superficial cells significantly increased (p = 0.006) post-cleaning compared to pre-cleaning values (Table 2).
Preputial differential epithelial cell counts (pre- and post-cleaning) in 50 clinically healthy, intact dogs.
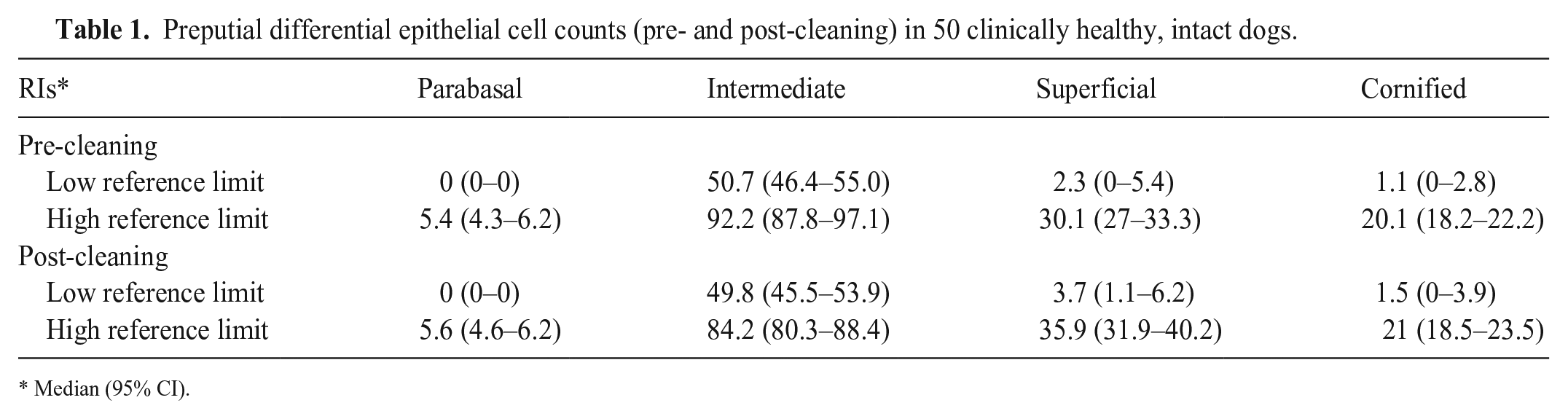
Median (95% CI).
Comparison of the percentages of preputial epithelial cell counts prior to and after cleaning in 50 clinically healthy, intact dogs.
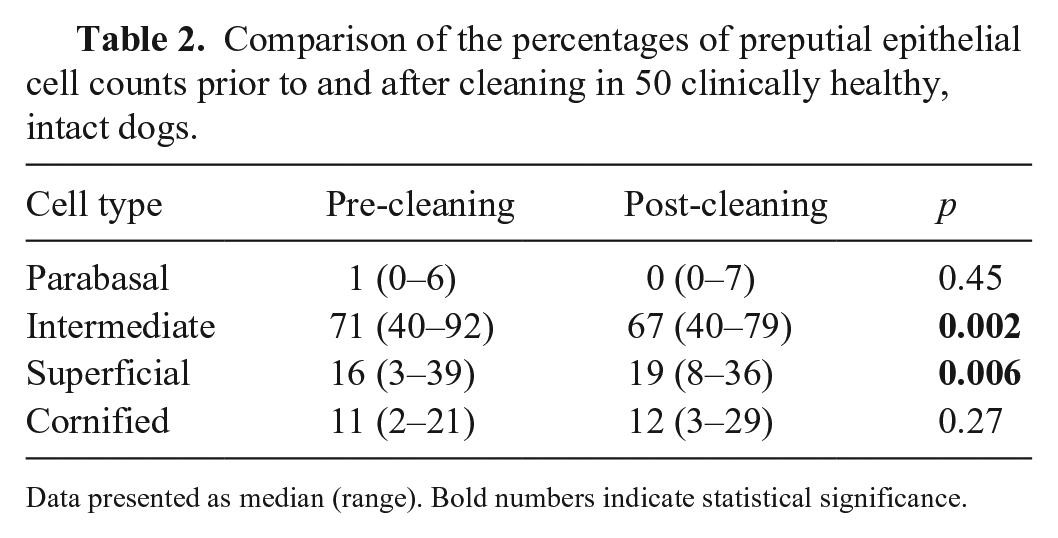
Data presented as median (range). Bold numbers indicate statistical significance.
Age was found to be positively associated with intermediate cells (1.1 [95% CI: 0.31, 1.88]; p = 0.007) and negatively with superficial (–0.67 [–1.2, –0.14]; p = 0.01) and cornified (–0.44 [–0.8, –0.07]; p = 0.02) cells (Table 3). Higher numbers of neutrophils were associated with higher percentages of superficial cells (p = 0.04). The concentration of estrogens was negatively associated with the percentage of parabasal cells (–0.15 [–0.3, –0.003]; p = 0.04). No association was found between age and concentration of estrogens in this canine population. The cleaning procedure significantly decreased neutrophil counts (p = 0.03).
Associations of the percentages of preputial epithelial cell counts with variables in 50 healthy, intact dogs.
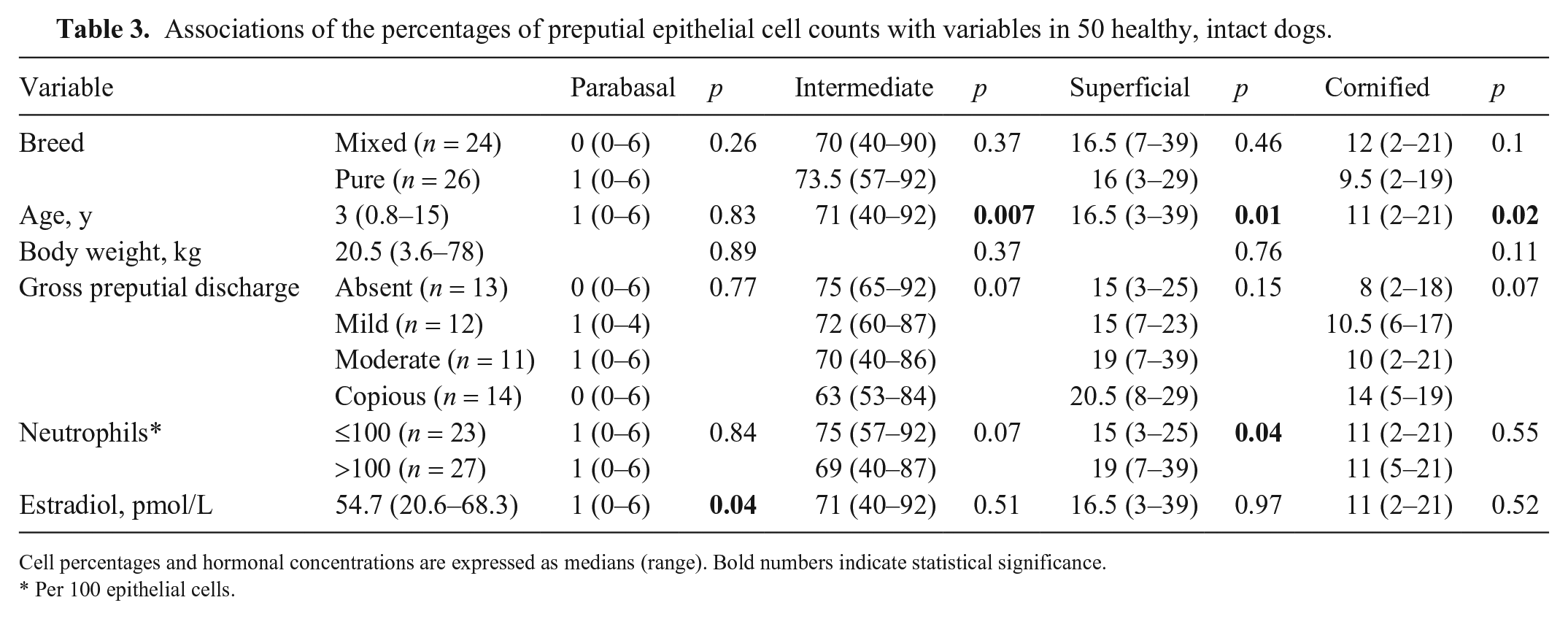
Cell percentages and hormonal concentrations are expressed as medians (range). Bold numbers indicate statistical significance.
Per 100 epithelial cells.
To our knowledge, RIs have not been published previously based on a 4-part differential epithelial cell count of the canine prepuce, looking also into a range of factors potentially affecting the epithelial cell differential. We retrieved no relevant information in a search of Google, PubMed, Web of Science, and Scopus, using the following search terms: “dog” and “cytology” and “prepuce” or “preputial.” Our epithelial cell differential counts were variably impacted by the cleaning of the prepuce before sampling, the age of the dogs, the neutrophilic cytologic component, and the normal variation of estrogen concentration.
Unlike previous studies, which classified epithelial cells into 3 types (parabasal, intermediate, superficial cells), we generated a 4-part differential, by reporting the overtly cornified cells separately from the overall superficial cell population. Our rationale was that discrimination between intermediate and superficial cells may be challenging by optical microscopy. This was highlighted when digital images from canine vaginal material were evaluated by a group of experts in reproduction; a high level of disagreement was found in differentiating intermediate from superficial cells and a high level of agreement in defining cornified cells. 12 We encountered similar difficulty in our study, with the classification of the cells in these 2 categories being sometimes ambiguous. It appears, therefore, that even for experienced raters, a certain level of inaccuracy in defining vaginal (and likely preputial) epithelial cells is to be expected, which may be minimized by quantifying the cornified cells separately. In our study, the disagreement between the evaluators regarding the identity of a subset of cells (mostly between interstitial and superficial) was settled by meticulously reviewing the morphologic criteria for any disputed cell. However, as we did not aim to evaluate the interobserver agreement on cell identification, future studies are warranted to address this issue.
In accord with previous reports, intermediate cells predominated in the epithelial cell differential; however, although parabasal cells were identified uncommonly in our study (median: 0–1%, depending on the mucosal cleaning status), the cells represented the second largest group of epithelial cells (~25%) reported in a previous study. 13 This disagreement may reflect different morphologic criteria applied by the researchers. In our study, by setting a cutoff diameter of ≤20 μm, only a few cells were classifiable as parabasal cells. In addition, in our study, the median percentages of superficial cells were 16.5% and 19%, and that of cornified cells 11% and 12%, pre-cleaning and post-cleaning, respectively. The aggregates of these cell types are substantially higher compared to the 4% of superficial cells reported by one study 5 in 30 healthy dogs, or the 3% reported by another study 13 in 50 healthy dogs. Although it is difficult to explain this discrepancy, it may be partially attributed to the inconsistent classification of superficial non-cornified cells among raters, further underlining the need to refine the expected epithelial cell fractions in normal preputial cytology. It has been suggested that superficial cells >20% on preputial cytology may indicate hyperestrogenism, with sensitivity of 80% and specificity of 98%, 5 although the estrogen cutoffs selected differed substantially between 2 studies5,8 and ranged from 11 to 50 pg/mL. Further studies may be warranted to evaluate the performance of purely cornified preputial cells in predicting hyperestrogenism. Of comparative interest, similar to our study, a 4-part preputial cytology was evaluated in castrated ferrets with adrenocortical disease, and the percentage of cornified cells was higher in those with clinical manifestations of adrenocortical disease compared to healthy ferrets. 11
The cleaning procedure of the prepuce reduced the purulent material, as reflected by the reduced neutrophilic component; furthermore, cleaning did not affect the parabasal and cornified cells, but decreased the intermediate cells and increased the superficial cells. The clinical significance of these changes is rather limited, especially considering that the critical subset of cornified cells was not impacted. Based on these findings, preputial sampling with or without prior cleaning may provide cytologic samples of sufficient quality. Of the signalment variables, unlike breed (mixed vs. purebred) and body weight, which were not associated with the differential epithelial cell count, the age of the dogs was found to be positively associated with intermediate cells and negatively with superficial and cornified cells. Hence, it seems that as age increases, a decline of the superficial and cornified cells may be expected. The cause of this trend is obscure, but a lower epithelial cell proliferation rate in aged dogs may be suspected, although the possibility of a hormonal determinant is rather limited, given that there was no association between age and estrogen concentration in our study. No effect of age on the preputial epithelial cell composition was found in another study. 13 Although gross preputial discharge had no appreciable effect on the preputial differential count, the heavier neutrophilic cytologic component was associated with higher superficial cell percentages. The clinical importance of this association is unclear and likely limited. Finally, estradiol was shown to negatively affect only the parabasal cells, which is not unexpected, considering the vaginal mucosal changes, in which, as the estrogen concentration rises, the parabasal cell count diminishes. 2
We acknowledge several limitations in our study. We recognize that the cohort of dogs recruited to establish RIs for preputial epithelial cells was relatively small. Despite this constraint, we employed rigorous selection criteria to ensure representativeness of the sample and maintain the validity of our findings. To mitigate the impact of the small sample size, we utilized bootstrap estimation. This method is particularly suitable for smaller samples, offering a means of generating a measure of uncertainty around our estimates. Bootstrapping involves resampling from the original data distribution with replacement, permitting us to perform interval estimation in this context and provide valid estimates on the assumption that our sample is representative of the broader population. 1 Although practical and fit-for-purpose, bootstrap estimation may not completely offset the limitation imposed by our small sample size. Another potential limitation was that dogs were considered free from testicular tumors based on palpation only, without ultrasonographic screening. It is possible, therefore, that some of the dogs might have testicular tumors not detectable by digital palpation. However, the possibility of having missed hormonally active masses is unlikely, based on the estrogen concentrations that were well within the RIs of the laboratory. Lastly, only healthy dogs were included in our study, without comparing them with dogs with potential hyperestrogenism. However, our main goal was to reconsider the preputial epithelial cell differential count in normal healthy dogs. Further studies are certainly required to evaluate the diagnostic performance of purely cornified cells in diagnosing hyperestrogenism.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our study was partially funded by Gerolymatos International (Boehringer Ingelheim). No funding sources had any involvement in the study design, collection, analysis, and interpretation of data, in writing reports, and in the decision to submit the article for publication.
