Abstract
Four alpine goats developed diarrhea soon after the owner placed plant clippings believed to be yellow oleander (Thevetia peruviana) into their pen on a suburban property near Palm Desert, CA, USA. A 1-y-old female goat died suddenly ~1 h after eating the plant clippings and was submitted to the San Bernardino Branch of the California Animal Health and Food Safety Laboratory System for postmortem examination. The main autopsy and histopathologic findings were myocardial hemorrhage and necrosis, consistent with cardiac glycoside intoxication. Rumen contents were analyzed by LC-MS/MS; peruvoside, a cardiac glycoside, was detected, but oleandrin, the cardiac glycoside of common oleander (Nerium oleander), was not. An LC–high-resolution MS (LC-HRMS) analysis revealed the presence of peruvoside and neriifolin in the rumen contents and in a tested plant fragment, indicating that the plant was a member of the Thevetia genus. A clipping from the plant fed to the goats and submitted by the owner was identified as yellow oleander, Thevetia peruviana (also known as Cascabela thevetia).
Cardiac glycoside intoxications have been well documented throughout history and continue to be a common cause of plant-related intoxication in livestock around the world.4–6,9,14,18 Several recognizable plant genera and species that contain cardiac glycosides are Digitalis spp. (foxgloves), Nerium oleander (common oleander), Thevetia peruviana (yellow oleander), Convallaria majalis (lily-of-the-valley), Strophanthus spp. (i.e., Kombe arrow poison, common poison rope, climbing oleander), Adonis vernalis (spring adonis, syn. spring pheasant’s eye), and Kalanchoe spp.4,5,7 Most of these plants are quite bitter, and grazing animals tend to avoid them while foraging in regions with more palatable options.7,10,14 However, there are circumstances in which cardiac glycoside–containing plants are presented in a way that makes them unavoidable or tempting to animals and humans. A common form of exposure for livestock and horses is from plant clippings that are either purposely put into a pasture or accidentally mixed in with hay.7,10,14 Also, in times of drought, the evergreen nature of some of these plant species increases their appeal to hungry herbivores.
Many different cardiac glycosides have been identified in various plant species and cultivars. However, most genera contain 1 or 2 characteristic cardiac glycosides, which allows identification of the plant responsible for the intoxication. For instance, Digitalis spp. commonly contain digitoxin and gitoxin, N. oleander contains oleandrin, T. peruviana contains peruvoside and neriifolin, Convallaria spp. contain convallatoxin, Strophanthus spp. contain strophanthidin, Adonis vernalis contains adonitoxin, and Kalanchoe spp. contain bryotoxin A and hellebrigenin (bufadienolides).1,4,5,7,11,13–18
Cardiac glycosides cause intoxication by inhibiting the Na+/K+ ATPase enzyme, leading to an increase in intracellular calcium, which results in alterations in cardiac electric activity, and in turn leads to arrhythmias, ectopic beats, and myocardial damage.1,10,13,14,16–18 Ingestion of a handful of leaves or a few flowers of most cardiac glycoside–containing plants can lead to severe illness and often death in all animals, including humans.
The plant most commonly associated with cardiac glycoside intoxication in animals is N. oleander, although case reports of accidental ingestion and suicidal attempts by humans can be found in the literature involving all of the cardiac glycosides cited above.1,4,6,9,10,14,15,17,18 Information about the intoxication of animals by T. peruviana in the literature is scant and limited to case reports and reviews of human intoxication by yellow oleander–containing weight-loss supplements.4,11,13,14,17 Here we report a cluster of cases of intoxication of goats by T. peruviana.
Four alpine goats developed diarrhea within a few hours after their owner placed yellow oleander (T. peruviana; Fig. 1) clippings into their pen. The goats were part of a small hobby herd on a suburban property near Palm Desert, CA, USA. The goats were the only ruminants on the property and were the only animals affected. The smallest goat who consumed the most trimmings, a 1-y-old female, died within an hour of ingestion and was submitted to the San Bernardino Branch of the California Animal Health and Food Safety (CAHFS) Laboratory System for autopsy. One other goat died 12 h after ingestion of the clippings, despite supportive care, but was not submitted for autopsy. The 2 remaining goats had only mild diarrhea, pale mucous membranes, and lethargy. They received supportive care and recovered within 2 d.
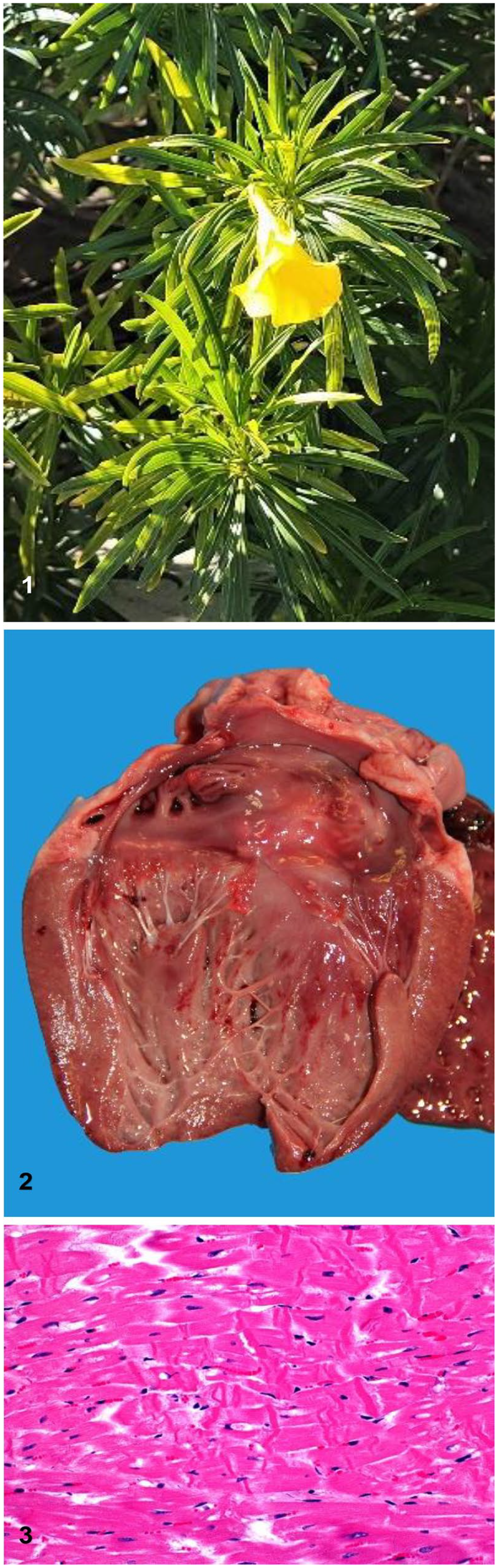
Thevetia peruviana and lesions caused in goats by ingestion of this plant.
An autopsy was performed on the dead goat submitted to CAHFS. Grossly, the carcass was in good nutritional condition, the lungs were diffusely congested and edematous, and stable froth filled the trachea and lower airways. The tracheal mucosa was diffusely bright red. There were subendocardial petechiae and ecchymoses over the left papillary muscle of the heart (Fig. 2). The rumen was distended with a mixture of grain and fragments of dark-green elongated leaves. No other significant gross abnormalities were observed.
Ancillary tests were performed following CAHFS SOPs unless otherwise specified. Samples from lung, liver, kidney, adrenal gland, reticulum, rumen, omasum, abomasum, small intestine, colon, spleen, and the whole brain were collected and fixed by immersion in 10% neutral-buffered formalin for 24–48 h. The brain was then sliced into ~5-mm thick sections, fixed for a further 7 d in fresh formalin, and subsamples were collected from parietal cortex, corpus striatum, thalamus, midbrain at the level of the anterior colliculi, pons, cerebellar peduncles, cerebellum, and medulla at the level of the obex. The tissues were processed routinely for the production of 4-µm thick H&E sections.
Microscopically, there was multifocal, acute, and mostly subendocardial myocardial necrosis and hemorrhage, as well as diffuse pulmonary congestion and edema. The myocardial necrosis was characterized by hypercontraction bands in myofibers, loss of striations, hypereosinophilia, vacuolation, loss of cytoplasm, and pyknosis (Fig. 3). No significant microscopic abnormalities were observed in any of the other tissues examined.
Samples of lung and liver were inoculated onto Columbia agar and incubated aerobically or anaerobically for 48 h at 37°C. Additionally, samples of liver were processed by PCR for Salmonella spp. 8 No aerobic bacteria were isolated from the liver or lung. Small numbers of Clostridium perfringens (not typed) and Paeniclostridium sordellii were isolated from the small intestine and colon. PCR for Salmonella spp. was negative. Feces were collected from the rectum, and fecal flotation was performed; a small number of coccidian oocysts were observed.
Leaf fragments found in the rumen were submitted to the University of California–Davis Center for Plant Diversity (UCD Herbarium) for identification. Although definitive identification was not possible, N. oleander and several other genera of the Apocynaceae family found in North America were ruled out.
A heavy metal screen, including arsenic, cadmium, copper, iron, lead, manganese, mercury, molybdenum, and zinc, was performed on the liver by inductively coupled plasma–optical emission spectrometry. Rumen contents were analyzed for cyanide (Cyantesmo colorimetric paper; Macherey Nagel). The liver heavy metal screen was unremarkable, and the rumen contents were negative for cyanide.
Rumen contents were also analyzed for oleandrin and peruvoside using a published method for tissue and urine 19 that was modified slightly for rumen content. Briefly, samples were extracted with methylene chloride, solvent exchanged to 50% methanol in water, and analyzed using ultra-high-performance liquid chromatography–triple-stage quadrupole mass spectrometry (UHPLC-MS/MS). The analytical batch included negative control rumen content and control rumen content fortified with 50 ng/g of oleandrin and peruvoside. Peruvoside was detected in rumen content, the chromatogram and spectrum for peruvoside matching that of the fortified sample, with a chromatographic signal ~4 times that of the 50 ng/g fortified rumen content. Oleandrin was not detected in the submitted rumen content but was detected in the fortified rumen content.
Leaf clippings of the plant of concern were originally identified by the UCD Herbarium as Thevetia bicornuta, a species of yellow oleander primarily found in South America. Also known as Cascabela bicornuta, very little information regarding toxicity for either name has been reported in the scientific literature. A full clipping, including leaves, flowers, and fruiting bodies, was later submitted to improve the collection’s representation of the less-common yellow oleander species and was identified as Thevetia peruviana (also called Cascabela thevetia), given differences in the flowers and fruiting bodies.
The extracts from the rumen content and the plant samples were analyzed by UHPLC–high-resolution MS (UHPLC-HRMS) for comparison. Unlike triple-stage quadrupole mass spectrometry, HRMS provides the advantage of non-targeted analysis, potentially enabling the detection of many different cardiac glycosides. A leaf sample was extracted by maceration of 0.21 g of minced leaf in 1 mL of methanol. A 0.25-mL aliquot of the methanol extract was filtered through a 0.22-µm syringe filter into an autosampler vial for analysis. The leaf extract and rumen extract were analyzed (Thermo Q Exactive mass spectrometer, Thermo Fisher; Ultimate 3000 UHPLC system, Dionex). The UHPLC column and mobile-phase conditions were the same as those used for the rumen extract. Electrospray ionization with positive polarity was used. Data were acquired in full scan and parallel reaction monitoring (PRM) modes. The mass resolution was 140,000 (M/ΔM at m/z 200); scan range of m/z 75–1150. For the PRM acquisition, the product ion data from precursor ions at m/z 535.328 (neriifolin) and 549.306 (peruvoside) were acquired. The mass resolution was set at 17,500 (M/ΔM at m/z 200); normalized collision energy was stepped at settings of 15, 25, and 35 V. Prominent product ions for peruvoside included those with nominal masses of m/z 335, 353, and 371. Prominent ions for neriifolin included those with nominal masses of m/z 339, 357, and 371.
Oleandrin was only detected in the control sample (Fig. 4); peruvoside and neriifolin were clearly detected in both the leaf samples (Fig. 5) and rumen extract (Fig. 6). Exact mass data for other known cardiac glycosides were used to review the full-scan, high-resolution data for their presence. None of the other known cardiac glycosides were detectable in either the leaf or the rumen extract.
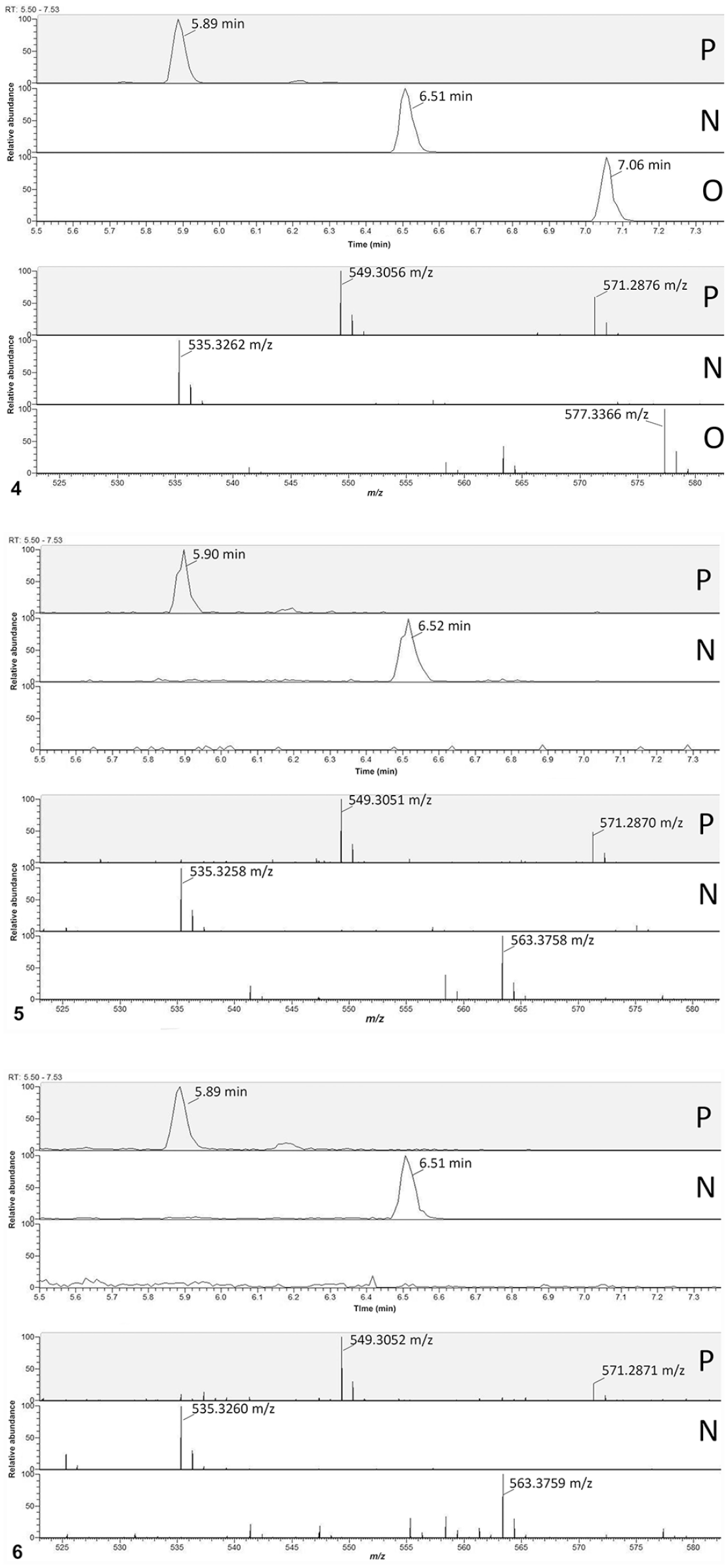
Ultra-high-performance liquid chromatography (UHPLC)–high-resolution mass spectrometry selected ion chro-matograms and full-scan UHPLC–product ion mass spectra for peruvoside, neriifolin, and oleandrin.
The detection of peruvoside and neriifolin in the rumen extract indicates that the goat had been consuming a plant that was likely part of the Thevetia/Cascabela genera. The leaves found in the rumen did not match the T. peruviana clippings or any other known cardiac glycoside–containing plant. Many plant clippings had been offered to the goats that morning and these likely represented other plant species. Peruvoside and neriifolin were detected in the leaf extracts from the T. peruviana clippings, matching the glycosides detected in the rumen contents and confirming the T. peruviana clippings to be the cause of the intoxication in this herd.
There is disagreement in the botany community as to the exact nomenclature for T. peruviana; some consider Cascabela spp. to be part of the Thevetia genus, whereas others consider Cascabela spp. and Thevetia spp. to be separate, yet closely related, genera. Therefore, C. thevetia is also known as T. peruviana. Although some variations exist between Thevetia spp. and Cascabela spp., there are several synapomorphies, including the same number of chromosomes.2,3,20 The Integrated Taxonomic Information System, which is often considered the gold standard for scientific nomenclature, classifies this plant officially as T. peruviana based on review of the most recent literature and leading taxonomic specialists. 12
Thevetia spp. are typically found in tropical America and the West Indies, and are being found more commonly in urban gardens despite being less drought resistant than N. oleander.3,4,7,11 All parts of the plant are toxic, although concentrations of the cardiac glycosides can vary. Ingestion of as few as 1–3 leaves or seeds can result in clinical signs, and fatalities have been reported at these amounts.4,11,14,17 Clinical signs usually occur within a few hours of ingestion and can include gastrointestinal signs (nausea, vomiting, drooling, abdominal pain), cardiac signs (dysrhythmias, atrioventricular block, atrial fibrillation and/or ventricular fibrillation), and neurologic signs (tremors, drowsiness, ataxia, mydriasis, weakness).4,7,10,11
Yellow oleander is a common name used for several different plant species that are becoming more popular in ornamental gardens and is just one example of toxic cultivars quietly invading ornamental plant nurseries. Proper identification of ornamental plants by nurseries and client education about the toxicity of these plants is vitally important to prevent unintentional poisonings in humans, pets, and livestock.11,20 Local botanists will likely need to expand their collections to include these increasingly popular ornamentals being sold to the public. The UCD Herbarium collection contained samples for the majority of the North American cultivars of these species but had very few samples for Central and South American Thevetia spp. and other toxic relatives of N. oleander. Proper identification is quite challenging if botanists do not have representative specimens to compare to, or partial/incomplete samplings from the plant in question, which can delay or prevent a diagnosis. In the absence of adequate plant material for identification, the use of LC-HRMS to identify specific cardiac glycosides can help identify a genus of the plant responsible for intoxication.
Footnotes
Acknowledgements
We thank the tireless and dedicated staff of CAHFS, including the chemists in the toxicology section, the necropsy, histology, and receiving technicians; Vicki Montoya for her help gathering samples and specimens of the plants and facilitating the work up on this case; and Alison Colwell and staff at the UC Davis Center for Plant Diversity for assistance with identification of plants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
