Abstract
Two adult mixed-breed ewes were presented with a 2-wk history of upper respiratory disease. Both animals were depressed, with bilateral serosanguineous nasal discharge and harsh bronchovesicular sounds accompanied by crackles and wheezes on auscultation. One animal was recumbent and was euthanized at presentation. The other animal with similar signs, as well as exophthalmos, was euthanized because of a mass in the nasal passages. On autopsy, severe pyogranulomatous and necrotizing ethmoidal rhinitis with focal pyogranulomatous pneumonia was diagnosed in both animals. An intralesional fungal organism was identified in the nares and lungs of both animals. The organism could not be isolated via fungal culture but was identified as Trichosporon sp. by a PCR assay. Trichosporon spp. are rarely associated with disease in veterinary medicine. This ubiquitous fungus might cause disease following trauma to the nasal passages or secondary to immunocompromise.
Trichosporon spp. are ubiquitous fungi, often isolated from water, soil, and feces.5,6 These organisms have been isolated from the skin, airways, gastrointestinal tract, and reproductive tract of normal humans and animals.6,15 Of the 12 current species, less than half have clinical relevance.3,6,13 Trichosporon spp. have been reported to affect humans and non-human primates, and of these reports, most are mild and dermatologic. 6 There are far fewer reports of Trichosporon affecting other species.6,13 We report here an infection of clinical significance in sheep, which, to our knowledge, has not been reported previously. We retrieved no cases of nasal trichosporonosis in a search of several academic resources, including Google, PubMed, and CAB Direct, using the search terms “nasal rhinitis, fungal rhinitis, ovine rhinitis”, suggesting that this condition has not been reported previously in sheep.
Two white mixed-breed ewes, 2- and 3-y-old, were presented to the Mississippi State University College of Veterinary Medicine (Starkville, MS, USA) for examination following reports of respiratory difficulty in the flock and nasal discharge of 2-wk duration. One of the 2 animals was also presented for elective enucleation of the proptosed right eye. The condition of both animals had been steadily declining for 2 wk before presentation. Both animals were born on the farm where they resided, in a flock of 30 sheep. Both ewes had received injectable ivermectin (200 µg/kg, SC) for suspected Oestrus ovis infection and oxytetracycline (9 mg/kg, SC) for bacterial bronchopneumonia. An unnamed ointment had been applied to the proptosed eye. At the time of presentation, both ewes were dull and quiet, had bilateral serosanguineous nasal discharge, and intermittent dyspnea. Harsh lung sounds were accompanied by crackles and wheezes, most noticeably affecting the right cranioventral lung lobe. Ewe 1, a 2-y-old in adequate body condition, was recumbent with marked nasal discharge. Given the severity of disease and poor prognosis, she was euthanized and autopsied. Ewe 2, a thin 3-y-old weighing 28.1 kg, had a proptosed right eye; skull radiographs revealed a large, space-occupying lesion in the nasal cavity that extended into the right orbit. She was euthanized given the severity of disease and was autopsied.
On gross examination of ewe 1, the turbinates of the ethmoid region and caudal nasal cavity, particularly on the right side, were distorted, and tissues were firm to slightly fibrous (Fig. 1). A 2-cm, bulging, tan-to-pink, spherical mass in the central aspect of the left caudal lung lobe contained several 2–5-mm firm tan nodules. No other gross abnormalities were observed.
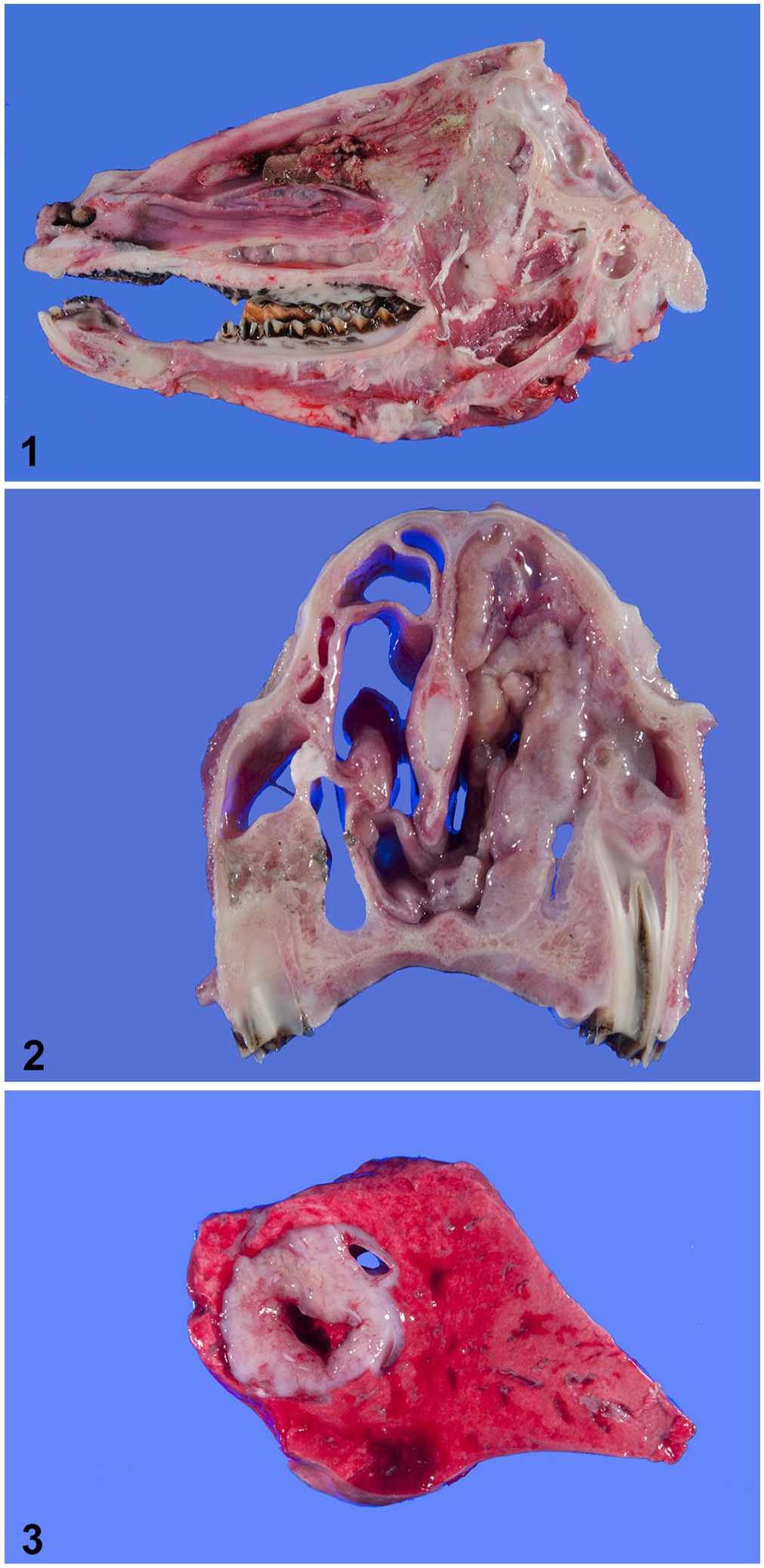
Nasal trichosporonosis in 2 mixed-breed ewes.
The gross examination of ewe 2 verified proptosis of the right eye. The cornea was diffusely thick and ulcerated; a 5-mm rupture was present at the limbus. The vitreous humor was hemorrhagic. The orbit was partially filled by a firm, tan, finely cobblestoned mass that emanated from the ethmoid region and extended rostrally into the nasal cavity and caudally into the olfactory bulbs of the brain. The mass filled the right nasal cavity and infiltrated the concha rostrally to the level of the premolars (Fig. 2). The left caudal lung lobe also had a 2-cm, well-demarcated, pale-tan nodule that bulged when cut, and had a 1-cm area of central necrosis (Fig. 3). No other gross abnormalities were observed.
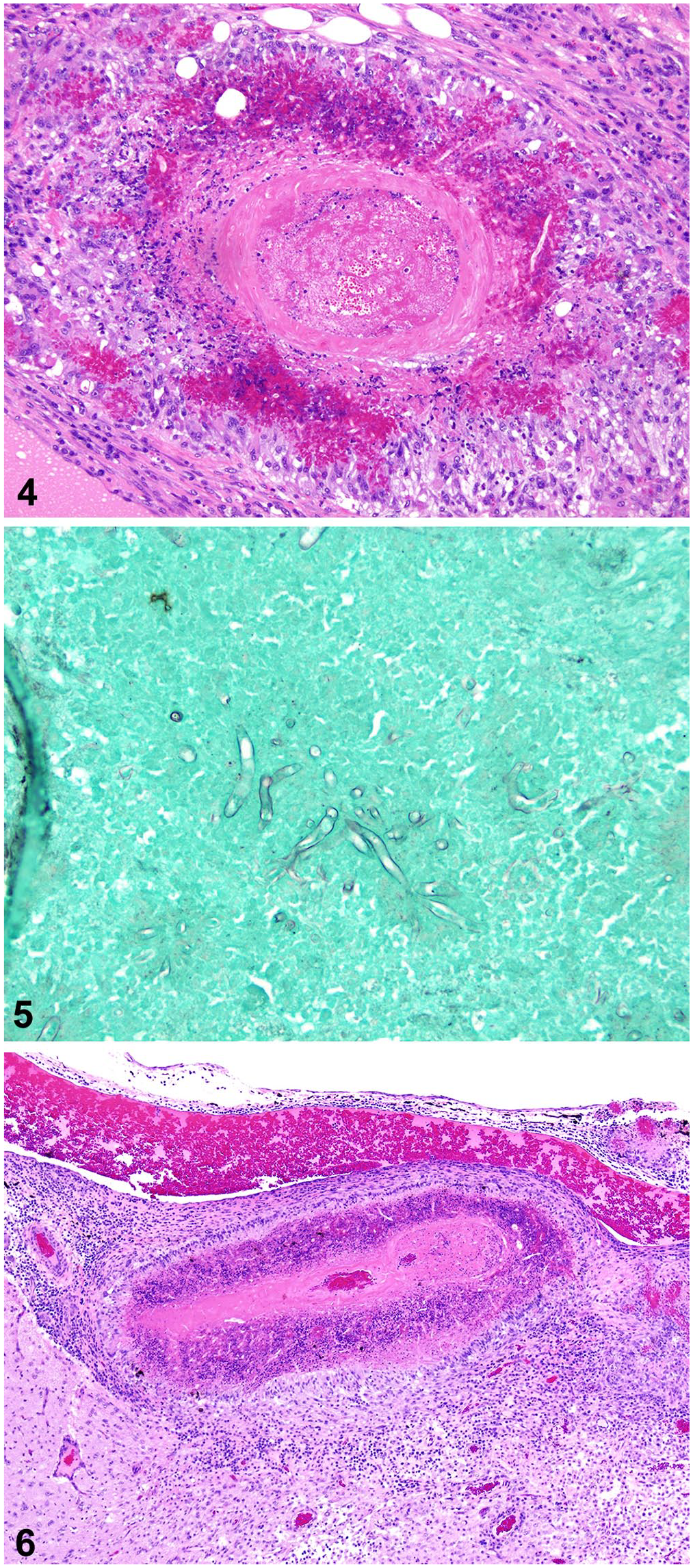
Histologic examination of the nasal tissues of both ewes revealed severe inflammation in association with hemorrhage, thrombosis, and coagulative necrosis. Large numbers of neutrophils and epithelioid macrophages, with fewer multinucleate giant cells, lymphocytes, and plasmacytes, surrounded Splendore–Hoeppli material (Fig. 4). Fungal hyphae within the material were 10-µm wide, with parallel walls, no appreciable septa, and dichotomous branching (Fig. 5).
Nasal trichosporonosis in 2 mixed-breed ewes.
Histologically, the cerebral meninges of ewe 1 were expanded many times by fungal periarteritis and transmural thrombotic arteritis (Fig. 6). Meningitis extended into Virchow–Robin spaces and the encephalon multifocally. Many layers of necrotic macrophages surrounded collections of radiating, magenta, club-like, 30–80-µm wide formations that surrounded poorly preserved fungal hyphae, consistent with Splendore–Hoeppli material; epithelioid macrophages and multinucleate giant cells delimited this exudate (Fig. 6). Macrophages and gitter cells infiltrated the neuropil in association with areas of gliosis, neuronal necrosis, edema, and liquefaction; such areas comprised < 5% of the brain sections examined. The pulmonary nodule was composed of similar inflammatory infiltrates and was associated with bronchi. The inflamed bronchial epithelium was often ulcerated or hyperplastic. Surrounding alveoli were filled with edema fluid and fibrin, with many large foamy macrophages, neutrophils, and Splendore–Hoeppli material.
Our diagnoses were severe pyogranulomatous and necrotizing ethmoidal rhinitis, with intralesional fungi and subsequent sinusitis, osteomyelitis, and meningoencephalitis, plus focal fungal pyogranulomatous pneumonia.
Gross differential diagnoses in our cases included enzootic nasal tumor based on the mass effect seen on radiographs, as well as the clinical signs of progressive nasal discharge, loss of condition, dyspnea, and the proptosis of the right eye in ewe 2. 12 Both ewes were also of the typical age for this disease presentation. 12 Jaagsiekte sheep retrovirus (Betaretrovirus) was also considered as the cause of the lung lesions given that this agent causes clinical signs similar to those caused by the ovine enzootic nasal tumor virus (Betaretrovirus), and would result in the lung lesions noted. 12 Bacterial infection with Salmonella enterica subsp. diarizonae has also been reported to cause proliferative rhinitis similar to the lesions described. 16 Additional infectious causes of rhinitis, such as herpesvirus and Pasteurella multocida, could also be considered, although the lesions that they cause are less consistent with the findings in our cases. 13 Microscopic findings and identification of the intralesional Trichosporon spp. confirmed via PCR and Sanger sequencing from submitted tissue (Washington Animal Disease Diagnostic Laboratory, Washington State University, Pullman, WA, USA) confirmed fungal infection as the primary etiology.
Most of the few reports in the veterinary literature of significant disease caused by Trichosporon refer to dogs and cats. Trichosporonosis has been reported most commonly as a cause of otitis externa and dermatitis in dogs,2,9 with one report of meningoencephalitis in a dog secondary to Trichosporon (Apiotrichum) montevideense 4 and a second report of systemic T. (Apiotrichum) loubieri infection in a cat. 13 In cats with cutaneous fungal infections caused by other fungal species, lesions are thought to be secondary to systemic infection introduced through the nares.11,13 In the case of the cat with the systemic T. loubieri infection, it is plausible that respiratory disease preceded the other signs. 12 However, there have been select reports of Trichosporon spp. solely affecting the upper airways. T. loubieri has been reported to cause rhinitis in cats, without accompanying systemic infection.13,14 The isolation of T. (Cutaneotrichosporon) jirovecii with Rhodotorula mucilaginosa from the laryngotracheal area via bronchoscopy was reported from a dog with laryngitis; the dog had received steroids and was being treated for hypothyroidism. This was the first report of a respiratory tract infection caused by these fungal organisms. 3
Trichosporonosis in livestock species has been described only rarely. T. beigelii has been reported as a cause of mastitis in dairy cattle,7,8,10 and Trichosporon spp. have been isolated from the gastrointestinal flora of horses. 15 Little else is known of this organism in livestock. Consequently, infection in the 2 sheep described here is most likely opportunistic.
There was no history of dietary deficiencies or underlying disease in this flock, or therapy with immunosuppressive medication such as steroids. Exposure to noxious substances in the air that can occur commonly in a farm setting, such as ammonia or machinery exhaust, may have predisposed the flock to infection as a result of injury to the nasal mucosa. Although a full flock assessment was not completed, it is possible that other sheep in the flock were affected, given that all animals were subjected to the same management and environment. Based on our findings, fungal disease should be considered in the differential diagnosis of rhinitis in sheep. Including fungal disease on a differential list will help direct diagnosis, treatment, and prognosis, although attempted treatment of trichosporonosis has proven unsuccessful because of species-related microbial resistance within the Trichosporon genus. 1 Furthermore, in food animal species, treatment options are limited in animals for human consumption, and the cost of therapy can be a limiting factor. Additional testing, such as advanced imaging, transtracheal washing, or bronchoalveolar lavage, may be pursued antemortem in such cases given that this information can further direct producer decision-making.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
