Abstract
Porcine circovirus 3 (PCV3) is widespread in pigs worldwide. Diverse clinical signs and lesions have been associated with PCV3, but the role of PCV3 as a cause of disease in swine remains unclear. We investigated the association of PCV3 with clinical signs and histologic lesions in 730 diagnostic swine cases between February 2016 and January 2018. The cases contained 2,177 samples submitted from 474 sites located in 21 states in the United States. PCR assay results were positive for PCV3 for 577 of 2,177 (27%) samples, 255 of 730 (35%) cases, 181 of 474 (38%) sites, and 17 of 21 (81%) states. We detected PCV3 in 19 of 28 specimen types and in pigs of all ages and clinical presentations, including healthy pigs, with the highest detection rate in adult pigs. PCV3 detection was not associated with respiratory, gastrointestinal, or CNS signs, weight loss, or sudden death. Of 58 types of histologic lesions evaluated, PCV3 detection was associated with myocarditis, cardiac vasculitis, and interstitial pneumonia in growing pigs. A high PCV3 detection rate was observed in aborted fetuses.
In the summer of 2015, porcine circovirus 3 (PCV3; Circoviridae, Circovirus) was identified by next-generation sequencing (NGS) while investigating poor reproductive performance and high sow mortality in a North Carolina swine herd. 22 In that study, PCV3 was also detected in samples of pigs from Mexico and Canada. 22 During the same time period, PCV3 was identified in 3 cases with unexplained cardiac and multi-organ inflammation from 3 U.S. states (Minnesota, Missouri, South Dakota) through NGS, and PCV3 mRNA was confirmed in tissues by in situ hybridization (ISH). 23 Subsequently, PCV3-positive samples were found by PCR in Argentina, Brazil, China, Colombia, Denmark, Germany, Hungary, India, Italy, Japan, Malaysia, Poland, Russia, Serbia, South Korea, Spain, Sweden, Thailand, the United Kingdom, and Vietnam, proving the presence of PCV3 throughout swine populations worldwide.3,5,7–9,11,12,15,18,19,21,30,32,34,36,38,40,43,44 Furthermore, PCV3 was detected in archived historical samples from as early as 1993 in Sweden, 43 1996 in Spain and China,17,35 and 2002 in the United Kingdom. 5 Overall, the accumulated data indicate that PCV3 is widespread and has been circulating in swine populations for decades.
The role of PCV3 as a cause of disease in swine is still under investigation. Diverse clinical signs have been reported in pigs infected with PCV3, mainly including respiratory,15,16,23,24,27,33,45 enteric,24,27,45 reproductive,2,3,8,16,18,22,28,32,39 integumentary,2,22 and neurologic signs.2,4 However, many of these reports were descriptive studies, and did not provide evidence for association between clinical signs and the presence of PCV3. Some studies did not fully rule out other pathogens as the cause of the reported clinical signs,3,15,16 some lacked healthy pigs as a control group, 32 or did not include postmortem examinations of the pigs.4,14,18 Furthermore, several studies have detected PCV3 in pigs without clinical signs.9,14
A few studies have provided more insightful information on the potential role of PCV3. The first PCV3 clinical reports found cutaneous and renal lesions in sows with anorexia, abortions, and decreased conception rate, and PCV3 was detected within the renal lesions by immunohistochemistry. 22 In another study, PCV3 was detected by NGS and ISH in 3 cases, with clinical signs including anorexia, weight loss, swollen joints, respiratory disease, rectal prolapse, and neurologic signs in pigs from 19-d to 10-wk-old. 23 These pigs had lesions of nonsuppurative myocarditis and/or cardiac arteriolitis, mild perivascular lymphocytic encephalitis and meningitis in the cerebellum, interstitial pneumonia, and acute bronchitis. Another study detected PCV3 nucleic acid in the lesions by ISH in fetuses with myocarditis, weak-born piglets with encephalitis and myocarditis, pigs with porcine dermatitis and nephropathy syndrome (PDNS), and pigs with systemic periarteritis. 2
Four experimental infections of nursery pigs with PCV3 have been published to date.13,20,37,41 In the first experiment, a PCV3 DNA infectious clone was inoculated into 4- and 8-wk-old specific pathogen–free pigs. The 4-wk-old pigs developed anorexia, coughing, sneezing, and diarrhea within 8 d post-infection (dpi) following hyperpnea, lethargy, skin erythema and papules, and convulsions within 15 dpi. 13 Microscopic lesions included bronchointerstitial pneumonia, lymphoid depletion in mesenteric lymph node and spleen, interstitial nephritis, and necrotic and eosinophilic myocarditis. 13 The 8-wk-old pigs developed only skin lesions during a 28-d observation period. 13 Two other experimental infections were conducted with PCV3-positive tissue homogenates or a cell culture–propagated PCV3 virus in 5-wk-old or 6-wk-old caesarean-derived, colostrum-deprived piglets, respectively.20,37 Although all animals were clinically unremarkable, histologic lesions, including multisystemic inflammation and perivasculitis, were observed in both studies.20,37 Finally, in a 2021 study, nine 3-wk-old cesarean-derived, colostrum-deprived pigs inoculated with isolate PCV3-China/DB-1/2017 did not develop obvious clinical signs or macroscopic lesions. 41
Therefore, diverse clinical signs and lesions have been associated with PCV3. A few in-depth studies have described potential associations with lesions, but a limited number of pigs and cases were examined. Our objectives were 1) to describe PCV3 detection frequencies in veterinary diagnostic samples; and 2) to investigate the presence of PCV3 and its association with clinical signs and histologic lesions in a large database of clinical samples from pigs in the United States.
Materials and methods
Data collection and frequency of PCV3
Data were obtained from the University of Minnesota Veterinary Diagnostic Laboratory (MN-VDL; St Paul, MN, USA) laboratory information management system (LIMS). Diagnostic sample submissions between February 2016 and January 2018 were considered. A submission was defined as a single sample or a group of samples from one farm site sent to the laboratory on a particular day. We selected submissions originating in the United States that had at least one PCV3 PCR test performed (n = 730). The data from the MN-VDL LIMS contained the date, site, farm location, specimen type, and PCR test results for PCV3, porcine circovirus 2 (PCV2), and porcine reproductive and respiratory syndrome virus (PRRSV; Arteriviridae, Betaarterivirus suid). All test procedures had been performed following routine standard operating protocols at the MN-VDL. For positive PCR results, the cycle threshold (Ct) values were used to estimate the viral load.
The PCV3-positive submission rate was calculated as the number of submissions with ≥1 PCV3-positive test divided by the total number of submissions with PCV3 PCR performed (n = 730). Within the 730 submissions, 2,177 samples were tested with a PCV3 PCR assay. Samples could represent an individual pig, a small pool of 2–5 pigs, or a larger group of pigs (from aggregate specimens such as processing fluids and oral fluids). The PCV3-positive sample rate was defined as the number of PCV3-positive samples over the total number of samples with PCV3 PCR performed (n = 2,177). The PCV3-positive site rate was calculated as the number of sites with ≥1 PCV3-positive case over all sites tested (n = 474). The PCV3-positive state rate was the number of states with ≥1 PCV3-positive submission over the total number of states with submissions (n = 21).
Association of PCV3 with clinical signs, histologic lesions, and age
For the evaluation of PCV3-associated clinical signs, only results from submissions with available tissues were selected, which included results from 1,323 pigs. These submissions were cases of clinical disease, and all were tested for PCV3 regardless of the age, clinical signs, or clinical diagnosis reported by submitting veterinarians. Information on clinical signs was retrieved directly from the MN-VDL LIMS, and/or from the diagnostic reports and submission forms. Submissions were classified into 8 categories based on clinical signs: respiratory, gastrointestinal disease, weight loss, lameness, sudden death, CNS disease, fetal death, and other.
Results from submissions that included pathology reports were used for histologic lesion and age analysis (n = 1,239 pigs). Samples were classified into 6 swine age groups based on North American swine production systems (fetus, piglet, nursery, finisher, adult, unknown). The ages included in each age group were: fetus (aborted fetuses and mummified and stillborn pigs), piglet (<19-d-old), nursery (19–70-d-old), finisher (71–180-d-old), adult (>180-d-old), and unknown (age not specified). The PCV3 positivity rates were calculated for each age group. In addition, for tissue samples with PCR test results available for PCV3, PCV2, and PRRSV, the positivity rate for each virus and co-detection rates were analyzed for each age group.
Histologic lesion information was mined from pathology reports, and then categorized based on organs and lesion types. For each lesion, each pig was classified into 1 of 3 outcomes: lesion present, lesion absent, or tissue not available.
Description of fetal death cases
Fetal death cases, with a PCV3 PCR test performed, were investigated between February 2016 and January 2018 at the MN-VDL. Results for PRRSV, PCV2, and porcine parvovirus 1 and 2 (PPV1, PPV2; Parvoviridae, Ungulate protoparvovirus 1 and 2) were also collected for further analysis given that they can cause abortions and fetal mummification. The association between PCV3 detection and fetal age (estimated by the fetal crown-rump [CR] length by measuring from crown of the head to anus) was investigated.
Statistical analysis
Descriptive statistics of samples and frequency of PCV3 were performed using Excel v.16.60 (Microsoft), and the geographic distribution of the PCV3 positive state rate was drawn using R v.4.1.0, the usmap and ggplot2 packages (https://www.r-project.org/).
The Fisher exact test was used to investigate the association between the presence of PCV3 and clinical signs, histologic lesions, or the detection of PRRSV and PCV2 using Epi Info v.7.1.3.0 (https://www.cdc.gov/epiinfo/index.html). The odds ratio (95% CI) and p values were reported. The Wilcoxon nonparametric rank-sum test was used to investigate the association between clinical signs or histologic lesions, and PCV3 load was estimated by Ct values using Epitools (http://epitools.ausvet.com.au). For this analysis, only PCV3-positive results were included. Estimated Ct value difference and p values were reported.
Results
PCV3 frequency in states and specimens
Between February 2016 and January 2018, 730 swine submissions, containing 2,177 samples, were retrieved from the MN-VDL LIMS. The cases originated from 474 sites in 21 U.S. states (Fig. 1). PCV3 was identified in samples from 17 states (81%), indicating that PCV3 was widespread in U.S. swine farms. Positive samples were not found in 4 states; however, very limited numbers of samples were available from those states. Most samples were from Minnesota (n = 1,224, 56%; Table 1). Twenty-seven percent of the samples (577 of 2,177) were PCV3 positive; the PCV3-positive submission rate was 35% (255 of 730). The PCV3-positive site rate was 38% (181 of 474).
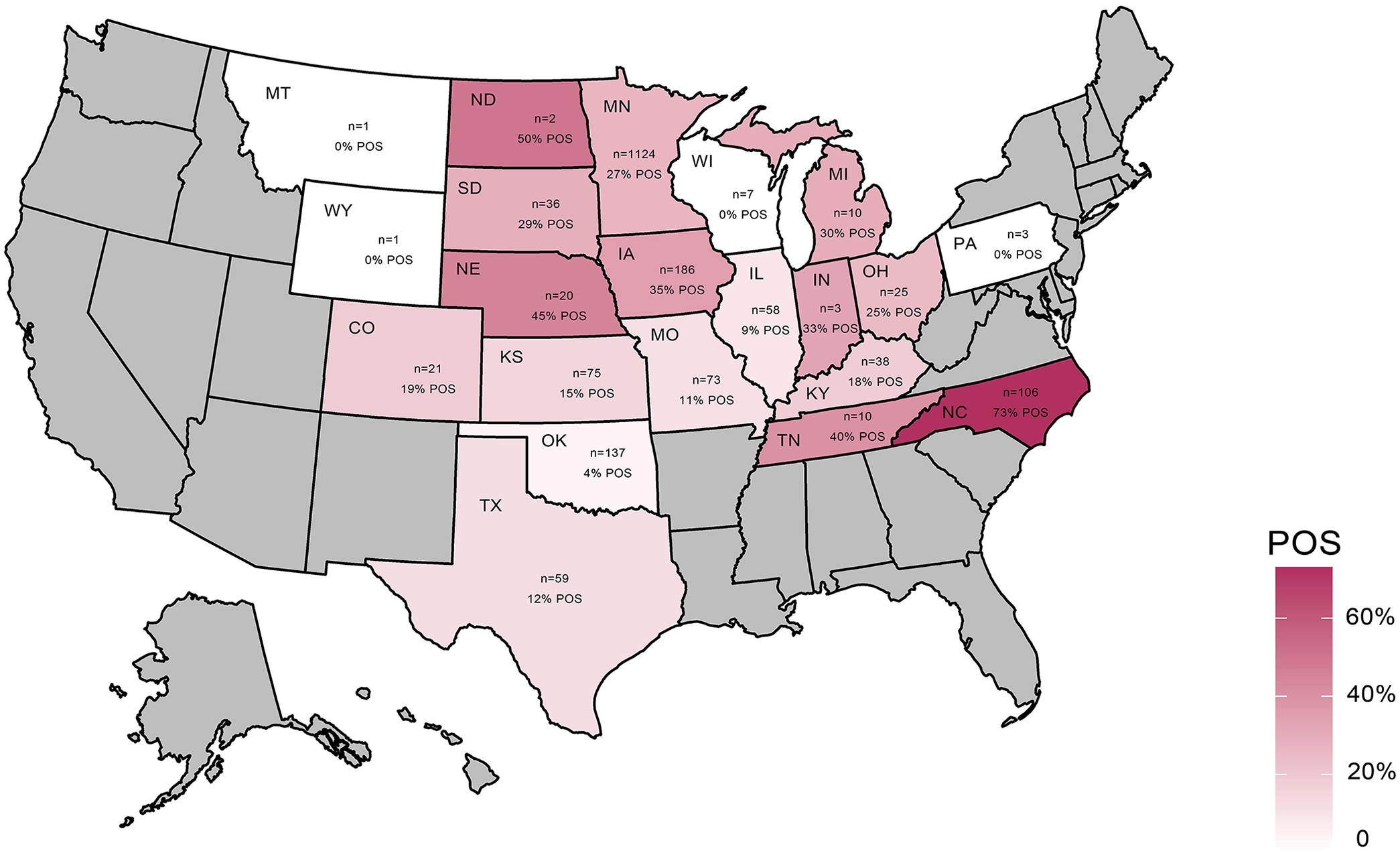
Geographic distribution of porcine circovirus 3 (PCV3) from clinical samples submitted from U.S. swine farms. The number of samples per state is illustrated with states in maroon gradient background. States with no samples submitted are in gray. PCV3-positive sample rate and the number of samples per state are displayed within each state. POS = positive.
Frequency by U.S. state of origin of porcine circovirus 3 (PCV3)-positive PCR results in swine samples, by site, case, and sample.
Of 28 different specimen types tested for PCV3, 19 specimen types had ≥1 sample positive for PCV3. Tissue homogenate (a mixture of fresh tissues including lung, heart, kidney, spleen, and lymph nodes) was the most common specimen followed by serum, processing fluid, and oral fluid. PCV3 was detected in the following specimens: tissue homogenate, serum, oral fluid, processing fluid, lung, blood, heart, testis, bulbourethral gland, epididymis, prostate gland, vesicular gland, intestine, feces, lymph node, spinal cord, bone marrow, and skeletal muscle (Table 2). Oral fluids, processing fluids, and serum samples were among the specimens with the highest positive rates.
Frequency of porcine circovirus 3 (PCV3)-positive PCR results in swine samples, by specimen type (n = 2,177).

The specimen types are ranked by the total number of samples tested. Samples of unclear specimen type are not presented.
PCV3 frequency in pigs with various clinical signs
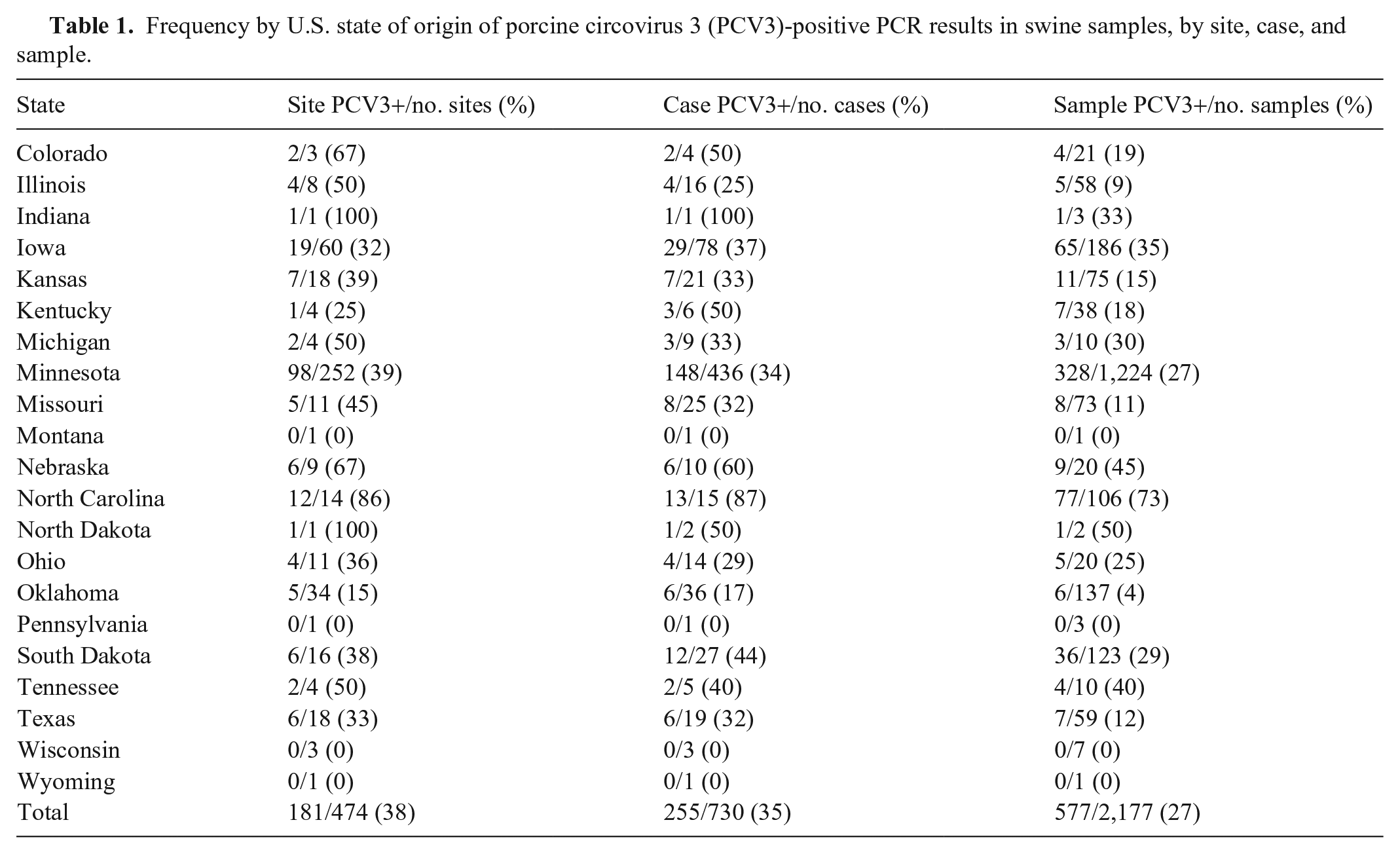
Clinical sign information was available for analysis from 1,323 pigs from 730 submissions. The positivity rate of PCV3 in pigs with various clinical signs ranged from 13% in pigs with CNS signs to 28% in pigs with lameness or weight loss (Table 3). Significant associations were lacking between clinical signs and the presence of PCV3. The median Ct values ranged from 31 in samples from pigs with respiratory disease to 38 in samples from pigs with CNS signs. Significant differences (p < 0.05) in Ct values were observed for pigs with CNS signs and weight loss; these pigs had higher PCV3 Ct values than pigs without those clinical signs.
Frequency of porcine circovirus 3 (PCV3)-positive PCR results in swine samples, by clinical sign.
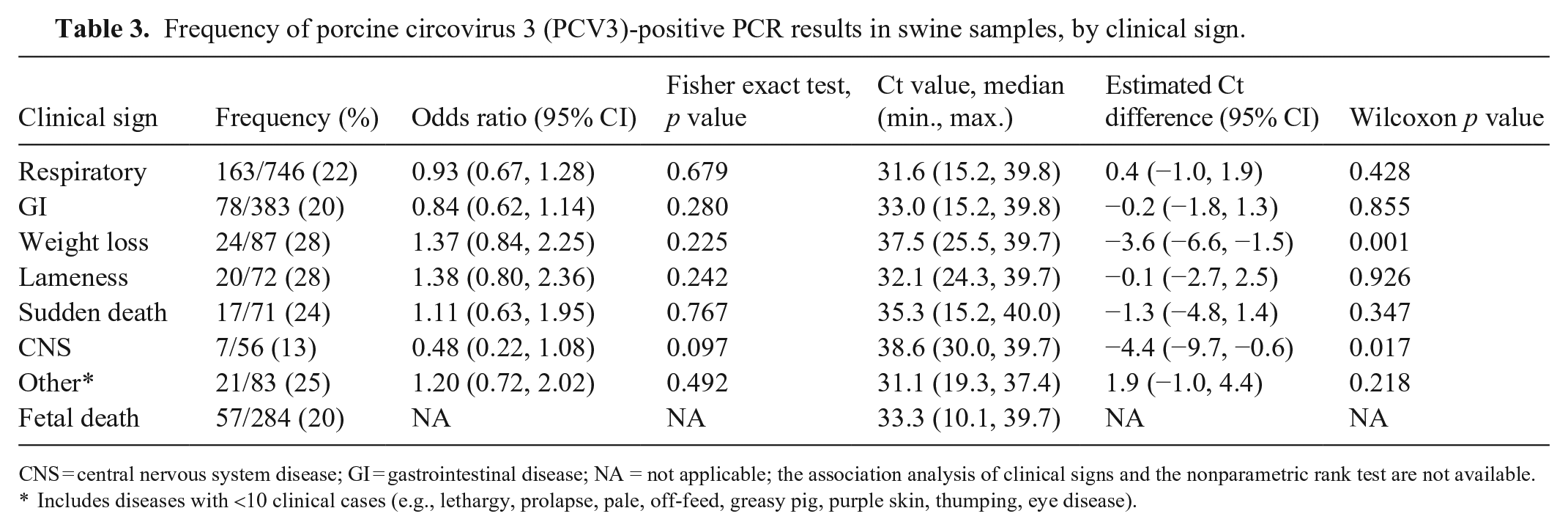
CNS = central nervous system disease; GI = gastrointestinal disease; NA = not applicable; the association analysis of clinical signs and the nonparametric rank test are not available.
Includes diseases with <10 clinical cases (e.g., lethargy, prolapse, pale, off-feed, greasy pig, purple skin, thumping, eye disease).
PCV3 age distribution and PCV2+PRRSV co-detection
PCV3 was detected in pigs of all ages. Of 1,239 pigs with histopathology reports, the PCV3 positivity rates among fetuses (210), piglets (158), nursery pigs (476), and finishing pigs (289) was 15–21% (Figs. 2, 3; Suppl. Table 1). The PCV3 positivity rate in adults (77) was 58%, which was the highest rate among all age groups (Figs. 2, 3). The pigs in the unknown age group (n = 29) had a PCV3 positivity rate of 38%.
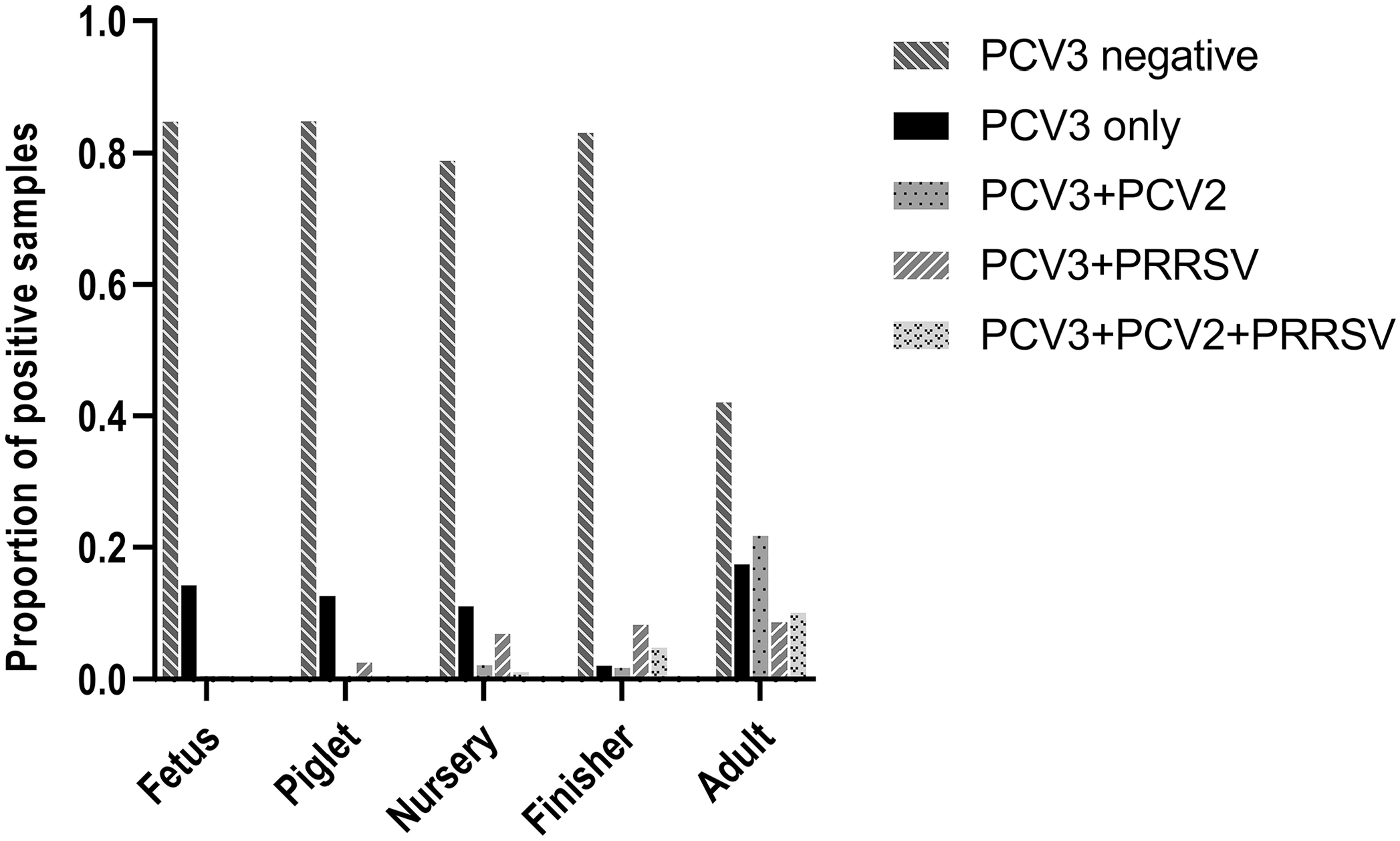
Porcine circovirus 3 (PCV3) detection, alone or in combination with porcine circovirus 2 (PCV2) and/or porcine reproductive and respiratory syndrome virus (PRRSV), in different age groups.
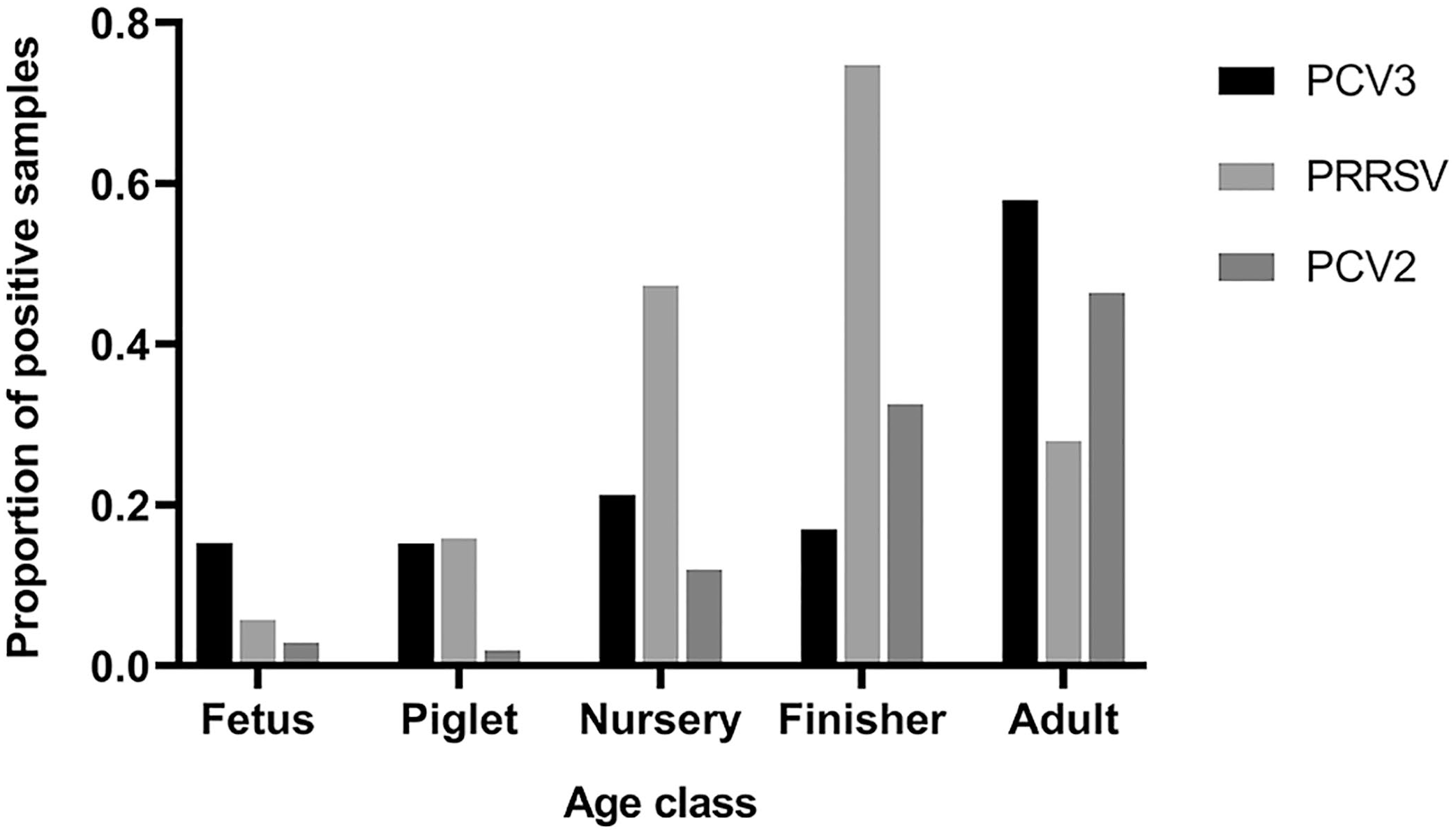
Detection of porcine circovirus 3 (PCV3), porcine circovirus 2 (PCV2), and porcine reproductive and respiratory syndrome virus (PRRSV), alone or in combination(s), in different age groups.
The total PCV3+PRRSV, PCV3+PCV2, and PCV2+PRRSV co-detection rates were 9%, 5%, and 9%, respectively (Table 4). PCV3 detection was significantly associated with PCV2 detection (p < 0.001), and PRRSV detection was significantly associated with PCV2 detection. However, associations were lacking when co-detections were analyzed at the different age-group levels.
Frequency of porcine circovirus 3 (PCV3), porcine circovirus 2 (PCV2), and/or porcine reproductive and respiratory syndrome virus (PRRSV) co-detection in swine tissue samples, by age group.
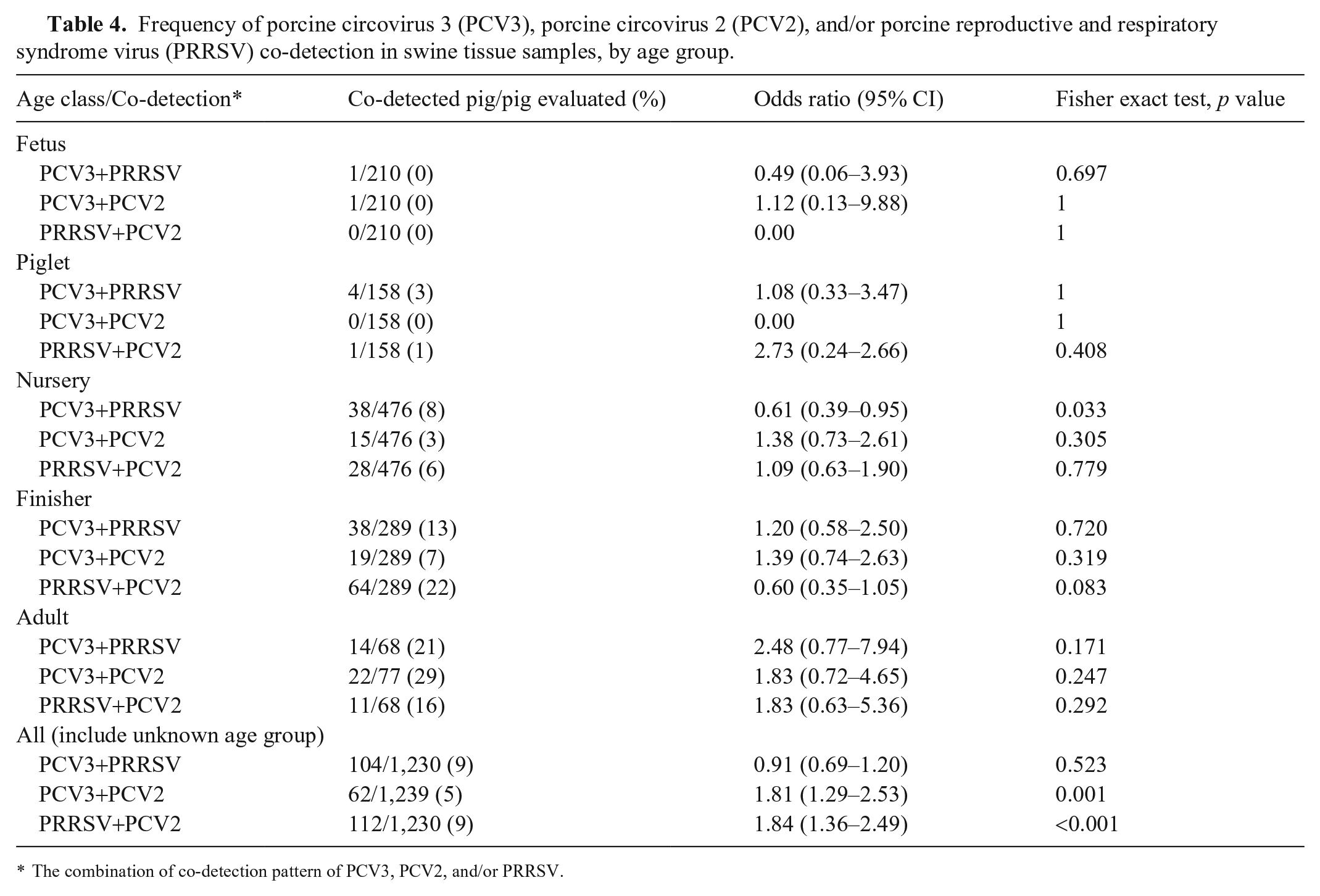
The combination of co-detection pattern of PCV3, PCV2, and/or PRRSV.
PCV3 frequency in pigs with various histologic lesions
Histologic descriptions were available for 1,239 pigs across all age groups. After removing fetuses (210), records for 1,029 pigs were available to analyze the frequency of PCV3 in pigs with various lesions. We identified 58 different histologic lesions (Suppl. Table 2). PCV3 frequency in pigs with various lesions ranged from 0% in pigs with ear necrosis or epidermitis to 63% in pigs with cardiac vasculitis or perivasculitis. In PCV3-positive pigs, the median PCV3 Ct value ranged from 22 in pigs with cardiac vasculitis or perivasculitis to 38 in pigs with purulent arthritis (Suppl. Table 2).
PRRSV and PCV2 can cause some of the same lesions as PCV3 (Suppl. Table 2). Therefore, to avoid the potential confounding effect of these viral infections, an additional analysis was performed including only the records from pigs that were negative for PRRSV and PCV2 (n = 425; Suppl. Table 3). Using that subset of data, PCV3 detection significantly increased the odds of pigs having myocarditis, cardiac vasculitis or perivasculitis, or interstitial pneumonia (Table 5). Ct values were significantly lower in pigs with myocarditis, cardiac vasculitis or perivasculitis, interstitial pneumonia, or hepatitis.
Frequency of porcine circovirus 3 (PCV3)-positive PCR results in tissue homogenates from swine with selected histologic lesions.
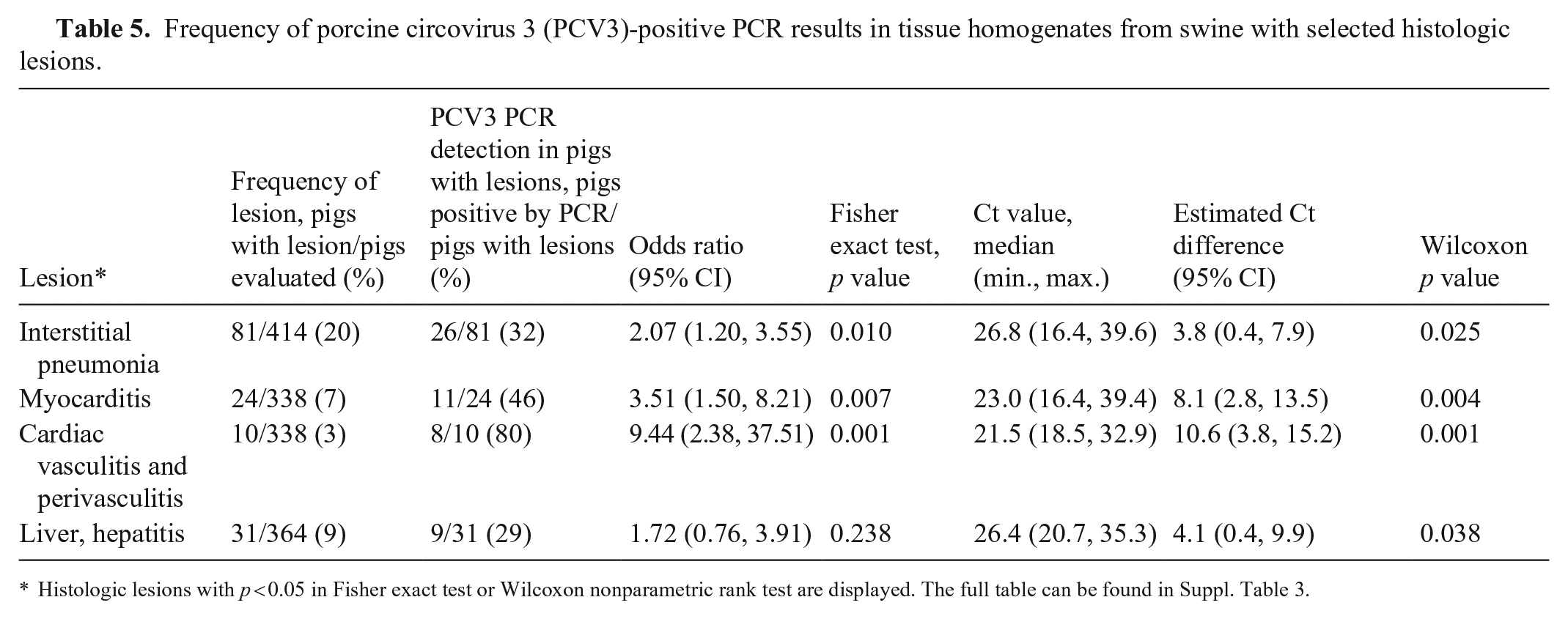
Histologic lesions with p < 0.05 in Fisher exact test or Wilcoxon nonparametric rank test are displayed. The full table can be found in Suppl. Table 3.
Description of fetal death cases
We retrieved 64 fetal death submissions, including 283 fetuses, from the MN-VDL LIMS (Table 6). Most fetuses (199 of 283; 70%) were negative for all of the tested viruses. Seventy-nine fetuses had single detections: PCV3 (58), PRRSV (10), PCV2 (5), PPV1 (5), or PPV2 (1). In addition, 2 fetuses had co-detection of PCV3+PPV1, 1 fetus had PCV3+PCV2, 1 fetus had PCV3+PRRSV, and 1 fetus had PRRSV+PPV1. No fetus had >2 agents co-detected.
Frequency of detection of porcine circovirus 3 (PCV3), porcine reproductive and respiratory syndrome virus (PRRSV), porcine circovirus 2 (PCV2), and porcine parvovirus 1 and 2 (PPV1, PPV2) in swine fetal tissues (n = 64 cases; n = 283 samples).

The CR length was available for 172 fetuses and was 1.5–33 cm with a median of 20 cm. Specifically for PCV3-positive fetuses, the CR length was 10–33 cm with a median of 20 cm (Suppl. Fig. 1). There were no significant differences in the C-R measurements between PCV3-positive and -negative fetuses.
Histologic descriptions were available for 184 of 283 fetuses assessed. Tissues, including heart (184), liver (181), lung (179), kidney (177), and brain (172), were evaluated; 125 of 184 (68%) fetuses lacked lesions. The most common lesion in fetuses was epicarditis (51 of 184). Other lesions, such as bronchopneumonia, pleuritis, myocarditis, endocarditis, and hepatitis, were found in <7 fetuses. Of all fetuses with histologic analysis, 44 fetuses had single viral detections: PCV3 (26), PRRSV (8), PCV2 (5), PPV1 (4), and PPV2 (1). Of PCV3 single detected fetuses, 7 fetuses had epicarditis, 1 fetus had epicarditis and bronchopneumonia, and 1 fetus had cardiac vasculitis and perivasculitis. Of PRRSV single detected fetuses, 4 had epicarditis, and 2 had epicarditis and endocarditis. Of PCV2 single detected fetuses, 3 had epicarditis. Of PPV1 single detected fetuses, 1 fetus had epicarditis and 1 fetus had myocarditis. No lesions were observed in the fetus with single PPV2 detection. Significant association was lacking between epicarditis and PCV3 detection (p = 0.824) or with the PCV3 Ct value (p = 0.203).
Discussion
PCV3 is commonly found in swine populations worldwide. We found that as many as 27% of clinical samples from the United States were positive for PCV3. In addition, PCV3 was found in various specimens, from pigs of all ages, and from many states. It is no surprise that PCV3 was detected in pigs with a variety of clinical signs, as well as in healthy pigs. However, establishing an association between the virus and any particular clinical signs or lesions remains a challenge. Only a few studies had provided insights into these associations by investigating a few specific field cases in detail.2,22,23 However, most of the studies focused on a few affected, PCV3-positive pigs, and lacked a comparison group. In contrast, we took advantage here of a large, well-characterized database of routine clinical cases processed in a systematic manner in a diagnostic laboratory setting. During the period of study, all of the tissue cases received at the MN-VDL that contained systemic tissues (lung, heart, kidney, spleen, lymph node) were tested for PCV3 regardless of the submitting veterinarian’s request, therefore reducing the selection bias. However, the results from our study are still biased to some extent given that most of the samples came from sick populations.
Significant associations were lacking between specific clinical signs and the presence of PCV3. The power of our analysis is probably limited because: 1) PCV3 is a common virus that can cause subclinical infections, and 2) various pathogens can cause the same clinical signs. When the viral loads of the positive samples were analyzed, significant associations were observed between certain clinical signs (weight loss, CNS signs) and a smaller viral load. These associations are most likely affected by confounders such as the age of the pig.
Multiple studies have reported that PCV3 could be detected in a variety of samples, including mummified and stillborn fetal tissues, fetal thoracic fluid, serum, heart, lung, brain, lymph node, liver, kidney, spleen, tonsil, oral fluid, pleural effusion, peritoneal cavity, intestine, colostrum, umbilical cords, and salivary gland.2,4,5,10,14,19,22,42,44,46 In our study, PCV3 was detected in 19 different specimen types. Oral fluids, processing fluids, and serum samples were among the specimens with the highest positive rates. It is important to note that, unlike tissue samples, these sample types do not represent all of the samples received at the MN-VDL, but a subset of cases in which the PCV3 PCR test was specifically requested by the veterinarian. Therefore, oral fluids, processing fluids, and serum samples are more likely to come from farms in which PCV3 was suspected. Nevertheless, our study supports the use of serum, oral fluids, and processing fluids as good specimens for PCV3 surveillance. However, the interpretation of a positive sample without history, clinical signs, and full diagnostic workup is difficult because pigs can be PCV3 positive without expressing any clinical signs.
The frequency of PCV3 detection increased with age, which could result from more exposure to PCV3 and/or decreasing maternal antibodies over time as pigs grow older. The PCV2 infection rate was also lower in fetus, piglets, and nursery pigs, which could be explained by mass vaccination programs for PCV2 in U.S. swine farms and delayed circulation of the virus. We found an apparent association between the presence of PCV2 and PCV3 in tissues; however, the association was not observed when the data were analyzed at each age group. Therefore, age probably acts as a confounder given that both viruses were more common in older pigs. In contrast, PRRSV was detected more commonly in samples from nursery and finishing pigs, which could be explained by the high frequency of this virus in pigs in this age in the United States and by the common use of attenuated vaccines that yield positive results in the PCR test.
As many as 58 histologic lesions were described in the cases in our study. Several positive and negative associations were found between the detection of PCV3 and some of these lesions. However, some of the associations found could be confounded by the common presence of 2 other viruses, PRRSV and PCV2, which are known to cause lesions such as interstitial pneumonia and myocarditis.25,26,31 Fortunately, our database included PCR results for PRRSV and PCV2 for most of the pigs in the study, and we were able to remove from the analysis pigs that tested positive for these viruses. After removing PRRSV- and PCV2-positive pigs, significant associations with PCV3 were observed for myocarditis, cardiac vasculitis, and interstitial pneumonia. This finding supports studies of myocarditis and cardiac vasculitis associated with detection of PCV3 in tissues by ISH.2,23 Our results also suggest a potential role of PCV3 in interstitial pneumonia. Interstitial pneumonia has been described in PCV3-positive pigs in other case reports6,23,29 and in one experimental infection trial. 13 It is important to note that the PCV3 PCR test in our study was performed for each pig on a homogenate of multiple tissues, not on individual tissues. Therefore, the PCV3-positive status can only be determined at the pig level, not at the tissue level. Further studies looking at associations between the positivity of the tissue and the lesions in that particular tissue would be necessary to evaluate the strength of these associations.
A study has suggested an association between PCV3 and encephalitis in weak-born neonatal piglets, 2 which was not evident in our study. However, the small sample size available for our analysis (brain was only available in a small subset of cases) may have prevented the detection of that association. In contrast, we did not observe associations with granulomatous lymphadenitis, characterized by moderate cortical lymphoid depletion, and with PDNS as reported in previous studies,2,22 even though the availability of kidney and lymphoid tissues was extensive.
Diagnosing the cause of fetal death in clinical cases can be frustrating because viruses may not infect all of the fetuses in a litter, and the infectious agent may be undetectable at the time of abortion or farrowing. In fact, several diagnostic submissions involving multiple litters are often needed before a cause can be detected. 1 For that reason, detecting PCV3 in as many as 22% of fetuses in our study was unexpected given that this infection rate was much higher than that of well-known causes of fetal death such as PRRSV, PCV2, or PPV1. Although the detection of PCV3 in fetal tissues does not necessarily imply that it was the cause of death, it does suggest that PCV3 can cross the placental barrier easily and replicate in fetal tissues, in some cases producing high viral loads. The lack of correlation of PCV3 detection with lesions is not unexpected given that histologic lesions are not very common in porcine fetuses. Overall, our results suggest that PCV3 could cause fetal death, as has been proposed by others.2,22,32
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221099538 – Supplemental material for Frequency of porcine circovirus 3 detection and histologic lesions in clinical samples from swine in the United States
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221099538 for Frequency of porcine circovirus 3 detection and histologic lesions in clinical samples from swine in the United States by Zhen Yang, Douglas G. Marthaler and Albert Rovira in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Michelle Leonard and Mary Thurn for their assistance managing the laboratory information management system, and Kimberly VanderWaal for her help with statistical analysis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was funded by the Swine Health Information Center grant 18-201, Summer Scholar Programs of College of Veterinary Medicine at the University of Minnesota, US-China Joint DVM scholarship program, and the Chinese Scholarship Council.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
