Abstract
Dogs with infectious arthritis may occasionally exhibit positive serum antinuclear antibody (ANA) and rheumatoid factor (RF) titers; however, relevant data are sparse for arthritis secondary to canine leishmaniosis (CanL) caused by Leishmania infantum. We determined the prevalence of positive serum ANA and RF titers in dogs with arthritis secondary to CanL. Blood samples from adult, client-owned dogs with purulent arthritis secondary to CanL, without any comorbidities, were collected for diagnostic purposes. Serum ANA titers were measured by immunoperoxidase test and RF titers by the Rose–Waaler latex test. Twelve of 23 dogs enrolled prospectively in our study had clinical arthritis, and 11 of 23 had subclinical arthritis. Based on LeishVet clinical staging, 7 dogs had clinical stage II disease, 11 had clinical stage III disease, and 5 had stage IV. None of the 23 dogs was seropositive for ANA; 3 of 23 were positive for RF. ANA and/or RF seropositivity, in dogs with CanL-associated arthritis, appears to be weak, if present at all. Based on our results, positive serum ANA and RF titers should not be expected in dogs with arthritis secondary to CanL.
Canine leishmaniosis (CanL) is a multisystemic disease with diverse clinical manifestations, which occasionally include the presence of joint disease (purulent, erosive or non-erosive, mono- or polyarthritis). 15 CanL-associated arthritis is generally thought to be an uncommon clinical entity largely because of its subclinical character, although its reported prevalence ranges widely (4.6–31%).1,15 The condition is thought to develop as a result of an inflammatory response secondary to synovial membrane infection by Leishmania infantum and deposition of immune complexes. 12 Immune-complex deposition and autoantibody formation are widely recognized in the pathogenesis of CanL 9 ; however, the extent to which the latter are associated with arthritis has not been investigated previously.
Antinuclear antibody (ANA) includes a variety of antibodies that react against self-nuclear components. These targeted components are usually histones and ribonucleoproteins in dogs, as opposed to double-stranded DNA in humans. In canine medicine, ANA assays are used primarily in the diagnosis of systemic lupus erythematosus (SLE). However, ANA has also been reported in several immune-mediated and infectious diseases. 20 Rheumatoid factor (RF) is typically an immunoglobulin (Ig)M or occasionally IgA autoantibody that binds to the Fc region of IgG. RF is used in the diagnosis of rheumatoid arthritis in dogs; however, it may occasionally be detected in other forms of autoimmune polyarthritis. 8
ANA and RF assays have been used sporadically in the diagnostic investigation of CanL-associated arthritis, with both positive and negative results reported. 19 However, considering the current lack of clarity about ANA and RF titers in CanL-associated arthritis, other causes of arthritis with positive ANA and/or RF titers (e.g. SLE, rheumatoid arthritis), might be misdiagnosed. 17 Therefore, we investigated the prevalence of positive ANA and RF serum titers in a prospective cohort of dogs with arthritis secondary to CanL in an endemic Mediterranean region. Our hypothesis was that the seropositivity rate would be relatively high, reflecting the proposed immune-mediated pathogenetic component of CanL.
We enrolled prospectively adult client-owned dogs with a diagnosis of arthritis and CanL. All dogs were presented over a 3.5-y period at the Companion Animal Clinic, School of Veterinary Medicine, Aristotle University of Thessaloniki (AUTh), Greece. We included dogs in our study when the following inclusion criteria were met: 1) >1-y-old; 2) no history of medication or vaccination during the preceding 1 mo; 3) clinical and clinicopathologic abnormalities compatible with CanL; 4) anti-leishmanial antibody titer ≥100 in an immunofluorescence assay and direct observation of Leishmania amastigotes in lymph node or bone marrow aspiration smears; 5) evidence of neutrophilic inflammation (neutrophils >10% of all nucleated cells in direct synovial fluid smears) in at least one joint independent of the presence of clinical signs consistent with arthritis; 6) negative serology (Snap 4Dx plus; Idexx) for other vector-borne infections (anaplasmosis, borreliosis, dirofilariosis, and monocytic-granulocytic ehrlichiosis); 7) negative culture of the aspirated joint fluids and urine; 8) absence of radiographic evidence of neoplasia in the joints; and 9) unremarkable thoracic radiographs, abdominal ultrasonography, and echocardiogram.
Synovial fluid was aspirated from the clinically affected joints while dogs were anesthetized with isoflurane after premedication with an acetylpromazine (0.05 mg/kg) and morphine (0.1 mg/kg IM) protocol. A 23-G needle was used for sampling large joints and a 25-G needle for small joints. Synovial fluid samples were submitted subsequently for cytologic examination. Direct smears were prepared from each aspirated synovial fluid and were stained with Giemsa. The total nucleated cell count was calculated as the average of the nucleated cells counted in 20 sequential representative high-power fields (400×). Pleocytosis was defined as >2 cells/400× field. 2 Differential nucleated cell counts were performed based on 200 nucleated cells. Peripheral blood samples were collected via jugular venipuncture for routine diagnostic work, with informed owner’s consent. Samples were collected into 4-mL evacuated serum separator tubes (Eurotubo) that were allowed to clot at room temperature (RT) for 20 min and then centrifuged at 1,500 × g for 10 min to obtain serum. Harvested sera were stored at −80°C for ≤6 mo until further analyzed.
The serum samples were analyzed for ANA using an immunoperoxidase test, as described previously. 6 Briefly, a Hep-2 cell line monolayer slide (MilliporeSigma) was equilibrated in PBS for 30 min. Canine serum samples were subsequently heat inactivated by incubation at 56°C for 30 min and serially diluted in a microtiter tray; similar titrations of control (positive and negative) sera were also made. All samples were tested at a screening dilution of 1:50. The samples were incubated in a humid chamber at RT and were then washed in PBS. Rabbit anti-dog IgG conjugated to horseradish peroxidase (MilliporeSigma) was then added to each well and incubated for 30 min. The slide was then washed, and a diaminobenzidine substrate with peroxide and Mayer hematoxylin was added. All samples were evaluated microscopically at 40× magnification; a titer >40 was considered positive. 6
The presence of RF was assessed using the Rose–Waaler test, which was performed as described previously. 7 Briefly, sheep red blood cells (SRBCs) were sensitized with rabbit anti-SRBC, and a 2% suspension of these cells was added to serial dilutions of patient and control (positive and negative) sera, in a microtiter tray. All samples were tested at a screening dilution of 1:4, and an endpoint titer of 1:64 was used. Unsensitized SRBCs were added to duplicate wells of each dilution as control. After a 60-min incubation at RT, the antibody-coated SRBCs were agglutinated by RF, and the serum titer was determined. 7 RF titers ≥32 were considered positive. All of the procedures carried out in our study were approved by the Ethics Committee of the School of Veterinary Medicine, AUTh.
Data distribution was evaluated using the Shapiro–Wilk test. Statistical analysis was performed using R (https://www.r-project.org/). If data distribution was Gaussian, data were presented as mean ± SD, and if Gaussian, as median and range.
We had included 29 dogs initially in our study, but no overt cytologic evidence of arthritis was noted in 6 of 29, and these dogs were excluded from further analysis. Overall, we included 23 dogs (17 males; 6 females) in our study after applying the defined inclusion criteria. The mean ± SD age of the population was 5.8 ± 2.3 y. The breed distribution of the 23 dogs was as follows: 6 Hellenic Hounds, 4 English Setters, 4 mixed-breed dogs, 3 Doberman Pinschers, 2 Beagles, 1 Brittany Spaniel, 1 German Shorthaired Pointer, 1 Bernese Mountain Dog, and 1 Labrador Retriever. We aspirated 96 joints (3 joints per dog, range: 2–6).
Twelve of 23 dogs had clinical arthritis (i.e. various combinations of lameness, stilted gate, joint pain, or edema on admission) and 11 of 23 dogs had subclinical arthritis (no historical or clinical evidence of joint disease). Five of 23 dogs were diagnosed with monoarthritis after aspirating 3 joints from each dog, and 18 of 23 dogs had polyarthritis. Based on the LeishVet clinical staging for CanL, 18 7 dogs had clinical stage II disease, 11 dogs had clinical stage III disease, and 5 dogs had clinical stage IV disease.
Cytologic evidence of arthritis was seen in 77 of 96 (80%) aspirated joints. Pleocytosis was observed in 85 of 96 (88%) joints (Table 1). L. infantum amastigotes were observed in 14 of 96 aspirated joints (15%; Fig. 1). L. infantum amastigotes were found in at least 1 joint in 5 of 23 dogs. None of the 23 dogs was seropositive for ANA; 3 of 23 were positive for RF (2 with a titer of 64, and 1 with a titer of 32). All of the RF-seropositive dogs had polyarthritis; 2 were subclinical, and 1 was clinical.
Numerical descriptive cytology results of the abnormal synovial fluid samples from 23 dogs with Leishmania-associated arthritis.
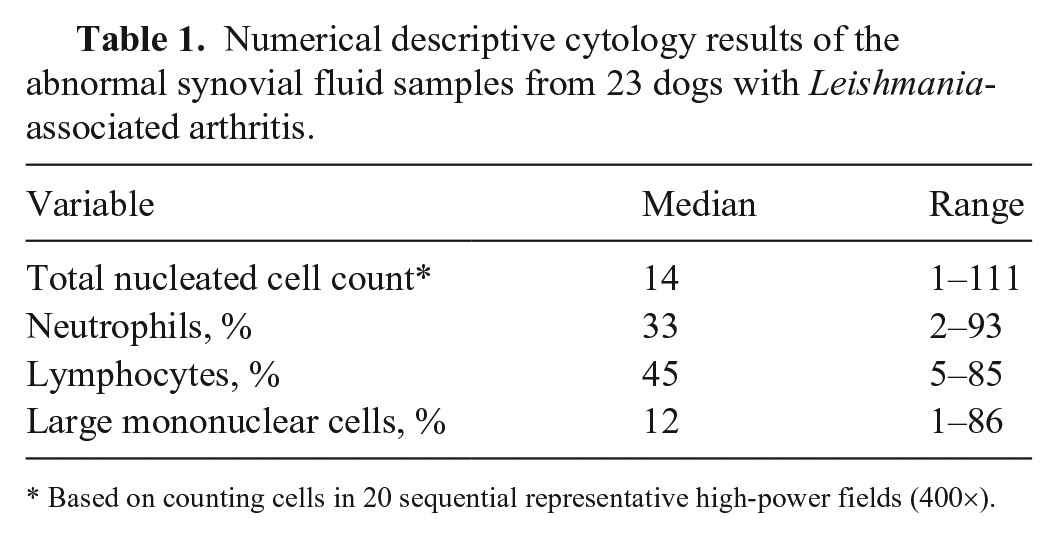
Based on counting cells in 20 sequential representative high-power fields (400×).
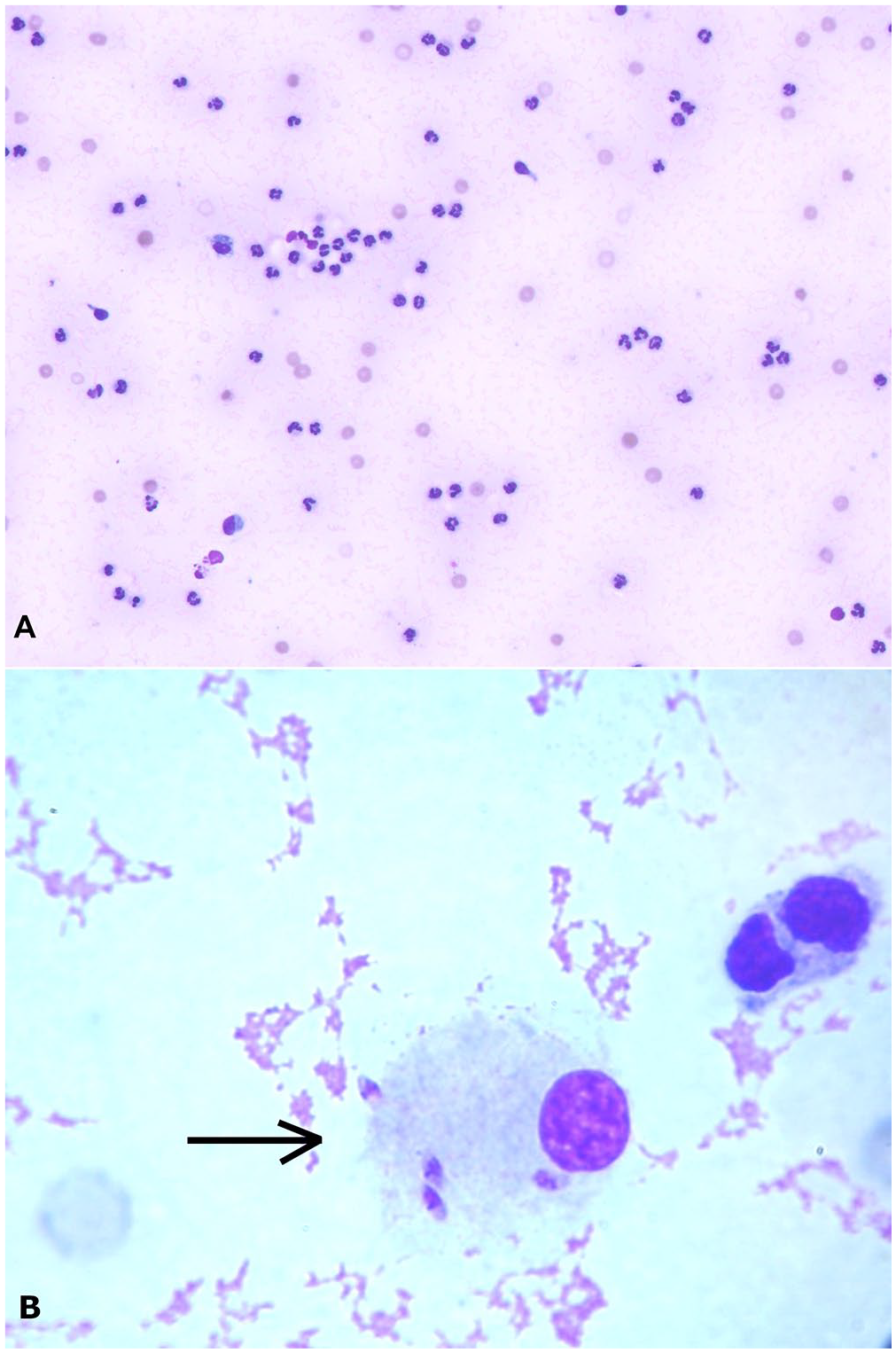
Synovial fluid from a dog with Leishmania-associated arthritis.
We found no ANA-positive dogs in our study; we found 3 RF-positive dogs. Our results corroborate 2 published case reports of negative ANA titers in dogs with CanL-associated arthritis, 6 but are discrepant with case series in endemic areas that reported a 10–47% prevalence of ANAs in CanL.5,13,17 In a published study, the relatively low prevalence (20%) of ANA-positive dogs was attributed to a modification of immunoregulation caused by CanL or other unspecified cause. 17 Indeed, it is known that, as CanL progresses, immune exhaustion occurs, and certain immunity mechanisms, such as L. infantum–specific, interferon gamma–driven Th1 responses, may become compromised. 16
It is unclear if the absence of ANA seropositivity in our study can be attributed solely to the advanced CanL documented in several of the dogs or to other unclarified factors. Given that our results are based on a single ANA measurement, we cannot also predict how the titers would change over time; temporal changes should be considered for future research. The discrepancy between the ANA seropositivity reported by the aforementioned studies and our findings is unlikely to be attributed to the use of an immunoperoxidase assay as opposed to an immunofluorescence assay, given that both assays were found to have comparable performance when the Hep-2 line was used as a substrate for the determination of ANA in humans. 20 However, the discrepancy might be attributed partially to the different specificity of the cell line substrates used. According to one study, the Hep-2 cell line appears to be more specific as a substrate compared to rat liver cell line substrate for the determination of ANA in dogs; specifically, when a titer of 25 was considered as the cutoff value, 20% of healthy dogs were positive for ANA. 11 Therefore, the authors recommended that titers ≥100 should be set as cutoff values when this substrate is used. Considering that 3 previous studies,5,13,14 which reported an ANA seroprevalence of 10%, 15.9%, and 47.2%, respectively, used rat liver cell line as a substrate, it seems reasonable that our seroprevalence was lower. Considering that another study 17 reported a 20% seroprevalence using Hep-2 as a substrate, it can be assumed that ANA seroprevalence in CanL is relatively low. Our findings were in contrast to our initial hypothesis, which was supported by a study on CanL-associated renal disease that also has a strong autoimmunity component; the latter indicated a positive correlation between renal impairment and circulating antibody concentration, implying a pathogenetic effect. 9
The 13% prevalence of positive RF titers, in turn, is in line with the findings of a case report 19 and in contrast with the overall 45% prevalence of RF positivity reported in a group of 14 dogs with CanL and 6 dogs with heartworm disease. 4 However, in the latter study, an ELISA was used to measure RF, and no information was provided about its validation and analytical performance. We used the classic heterospecific Rose–Waaler test, a well-established assay for the detection of RF in the serum of dogs, which was found to perform better than the modified Rose–Waaler test for the measurement of canine RF. 10 However, it is difficult to compare directly the results of the different assays used, because the sensitivity and cutoff points can vary greatly. Moreover, RF has limited applicability for an individual diagnosis given that low-positive titers may be found in a small percentage of healthy dogs or any chronic disease of infectious or noninfectious nature, associated with persistent antigen exposure and immune-complex production. 19 Interestingly, in one study, in which the same method for the measurement of RF was used, 11.7% of 614 sick dogs without arthritis tested positive. 3 Therefore, the prevalence of a positive RF titer in a CanL-associated arthritis case appears to be similar to that of sick animals without arthritis. However, this finding should be confirmed in a large-scale study.
A limitation of our study is that the total nucleated cell count of the synovial fluids was estimated on cytology, but not measured using an automated analyzer. The measurement of ANA and RF titers in paired serum and synovial fluid samples of dogs with CanL-associated arthritis could be considered for future iterations to investigate whether local processes in the synovium could affect the concentration of autoantibodies in the synovial fluid.
Footnotes
Declaration of conflicting interests
Theodora K. Tsouloufi and Ioannis L. Oikonomidis are employees of Idexx Laboratories, which manufactures the Snap 4Dx test. All other authors do not have any conflict of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
