Abstract
We analyzed the correlation between minimum inhibitory concentrations (MICs) of antimicrobials used in humans and those used in animals to enable comparison of antimicrobial susceptibility between Escherichia coli isolated from humans and those from animals. We compared the following pairs of MIC data: piperacillin (PIPC) to ampicillin (ABPC), amikacin (AMK) to kanamycin (KM), minocycline (MINO) to oxytetracycline (OTC), and levofloxacin (LVFX) to enrofloxacin (ERFX) using 103 isolates of E. coli from healthy livestock (cattle, pigs, broiler chickens, and layer chickens). Kappa analysis of the agreement for resistance and susceptibility between PIPC and ABPC, AMK and KM, MINO and OTC, and LVFX and ERFX showed almost perfect (κ = 0.81), slight (κ = 0.12), fair (κ = 0.37), and moderate (κ = 0.46) agreement, respectively. Within the antimicrobial pairs, all isolates resistant to the human antimicrobial were also resistant to the veterinary antimicrobial. However, there was less agreement within the pairs for those isolates that were sensitive to the human antimicrobial. The percentage agreement for susceptibility, defined as the percentage of isolates sensitive to both antimicrobials compared with isolates sensitive to both antimicrobials, as well as those sensitive only to the human antimicrobial, was 89.9%, 87.3%, 64.0%, and 89.9% for PIPC and ABPC, AMK and KM, MINO and OTC, and LVFX and ERFX, respectively. Our results suggest that the possibility of missing the resistance for antimicrobials used in human medicine by examining MICs for the equivalent antimicrobials used in veterinary medicine is low.
Antimicrobial resistance affects not only human health but also animal health and food safety. 23 In May 2015, the World Health Organization (WHO) established the Global Action Plan on Antimicrobial Resistance, emphasizing that international multi-sectoral action is needed to combat antimicrobial resistance. 24 The WHO urged all member states to develop relevant national action plans within 2 y. The government of Japan launched the National Action Plan on Antimicrobial Resistance in April 2016, 5 and many other countries around the world have established a national action plan. 25
In Japan, the Ministry of Agriculture, Forestry, and Fisheries conducts antimicrobial resistance monitoring in animal sectors using the Japanese Veterinary Antimicrobial Resistance Monitoring (JVARM) system. 13 The Japan Nosocomial Infections Surveillance (JANIS) program is a national surveillance program organized by the Ministry of Health, Labor, and Welfare (MHLW) of Japan designed to provide basic information on the incidence and prevalence of nosocomial infections and antimicrobial-resistant bacteria in Japanese medical settings. 12 However, some of the antimicrobials used for antimicrobial susceptibility testing differ between the JANIS and JVARM schemes. For example, piperacillin, amikacin, minocycline, and levofloxacin are only used in JANIS. 12 Although integrated surveillance programs are carried out in North America and Europe,1,21 it remains difficult to estimate the resistance against human antimicrobials given that the antimicrobials used may not necessarily be common between humans and animals.
Among the third-generation cephalosporins, ceftiofur is used in the JVARM system, whereas cefotaxime is used in the JANIS program. We have confirmed the good relationship between the minimum inhibitory concentration (MIC) of ceftiofur (used in veterinary medicine) and cefotaxime (used in human medicine). 6 However, the correlations between some classes of antimicrobials used in the veterinary and human sectors are not clear. We, therefore, assessed the validity of results of antimicrobial susceptibility tests conducted with different antimicrobials in the same class used in human and veterinary medicine to evaluate the effect on human medicine as a result of antimicrobial resistance among the bacteria from animals.
We selected 103 Escherichia coli isolates with a broad distribution of MIC values for the relevant antimicrobials. The isolates were from fecal samples of healthy animals (22 from cattle, 20 from pigs, 30 from broiler chickens, and 31 from layer chickens) in Japan from July to September in 2009 under the JVARM system. These isolates were isolated from animals (1 isolate/animal/farm) at 84 farms except for 19 farms; on 19 farms, we obtained 2 E. coli isolates with different resistance patterns from a single animal.
We performed antimicrobial susceptibility testing according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI). We tested 4 antimicrobial agents that are approved for use in animals in Japan, namely, ampicillin (ABPC), kanamycin (KM), oxytetracycline (OTC), and enrofloxacin (ERFX). 14 The MICs of these 4 agents were determined by an agar dilution method. 3 We also tested piperacillin (PIPC), amikacin (AMK), minocycline (MINO), and levofloxacin (LVFX), which are used in the JANIS program. 12 The MICs of these 4 agents were determined by a broth microdilution method. 4 Breakpoints established by the CLSI were used for ABPC, ERFX, KM, PIPC, AMK, MINO, and LVFX.2,4 The breakpoint for OTC was from the JVARM system. 15 E. coli ATCC 25922 was used as a quality control strain.2 –4 Analysis of scattergrams that correlate the MICs of antimicrobials used in the JANIS program to those used in the JVARM system was performed using the percentage agreement for resistance and susceptibility. In addition, the Cohen kappa coefficient (κ) was used to determine the agreement between the results determined in the pairs of human and veterinary antimicrobials. 11
We found good correlation between susceptibilities of LVFX and ERFX, and PIPC and ABPC (Fig. 1, Table 1). Fluoroquinolones inhibit the action of DNA gyrase and topoisomerase IV, thereby preventing DNA replication. 7 The major mechanisms that lead to fluoroquinolone resistance involve mutations in the quinolone resistance–determining regions (QRDRs) of DNA gyrase and topoisomerase IV. 7 The increased number of mutations in the QRDRs of DNA gyrase and topoisomerase is correlated with the level of quinolone resistance in E. coli 22 ; the relationship between the number of mutations and the elevation of MIC is regarded as clear. 17 The MICs of various fluoroquinolones are well correlated with each other.
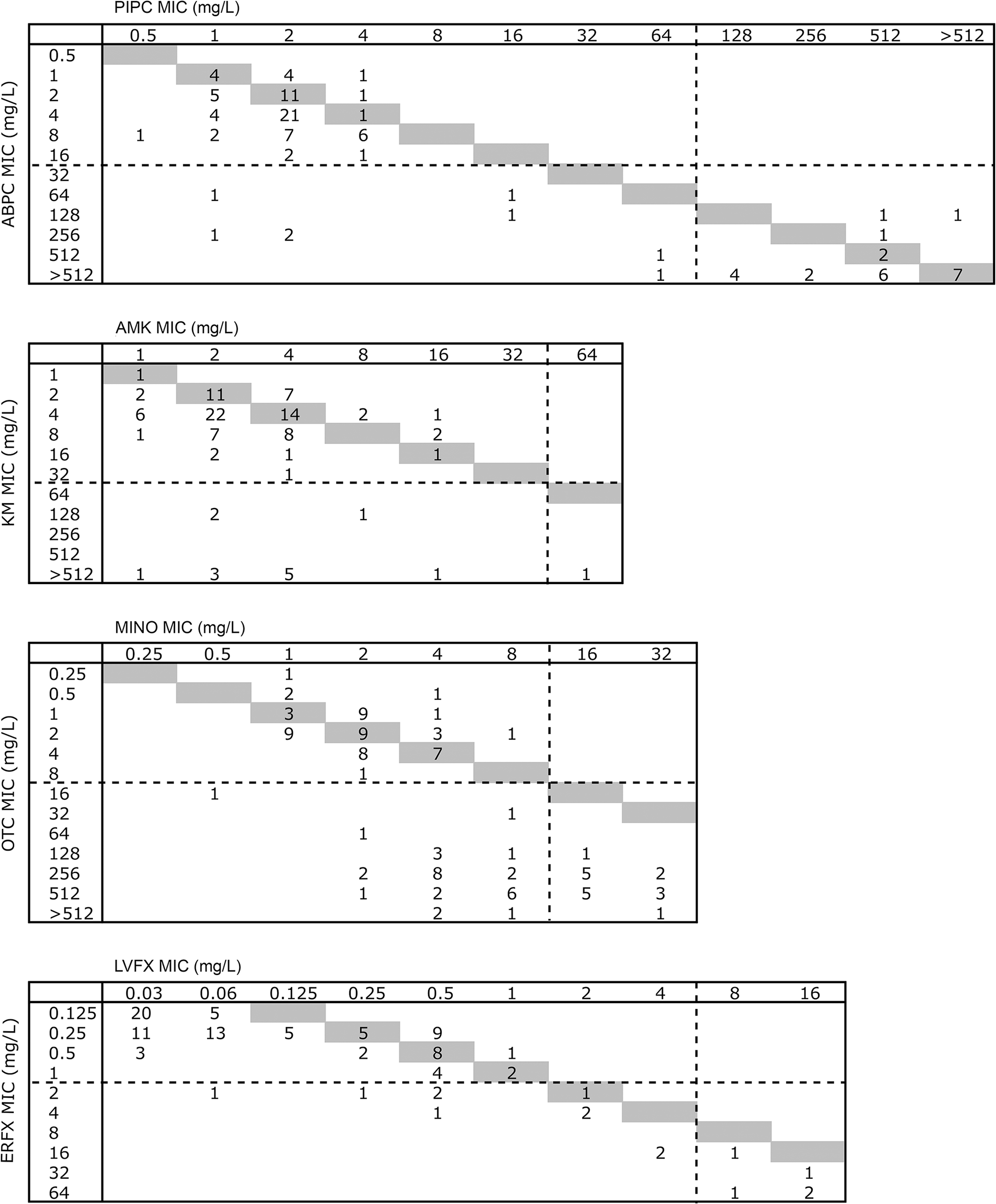
Scattergrams showing the minimum inhibitory concentration (MIC) of piperacillin (PIPC) compared to ampicillin (ABPC), amikacin (AMK) compared to kanamycin (KM), minocycline (MINO) compared to oxytetracycline (OTC), and levofloxacin (LVFX) compared to enrofloxacin (ERFX). Numbers within the scattergrams indicate the number of Escherichia coli isolates. Vertical dotted lines represent CLSI breakpoints for PIPC, AMK, MINO, and LVFX. 4 Horizontal dotted lines indicate the respective CLSI breakpoints for ABPC, KM, and ERFX 2 and the JVARM breakpoint for OTC. 15
Classification results, percentage agreement for resistance, percentage agreement for susceptibility, and kappa statistics for each pair of antimicrobial agents examined in this study.
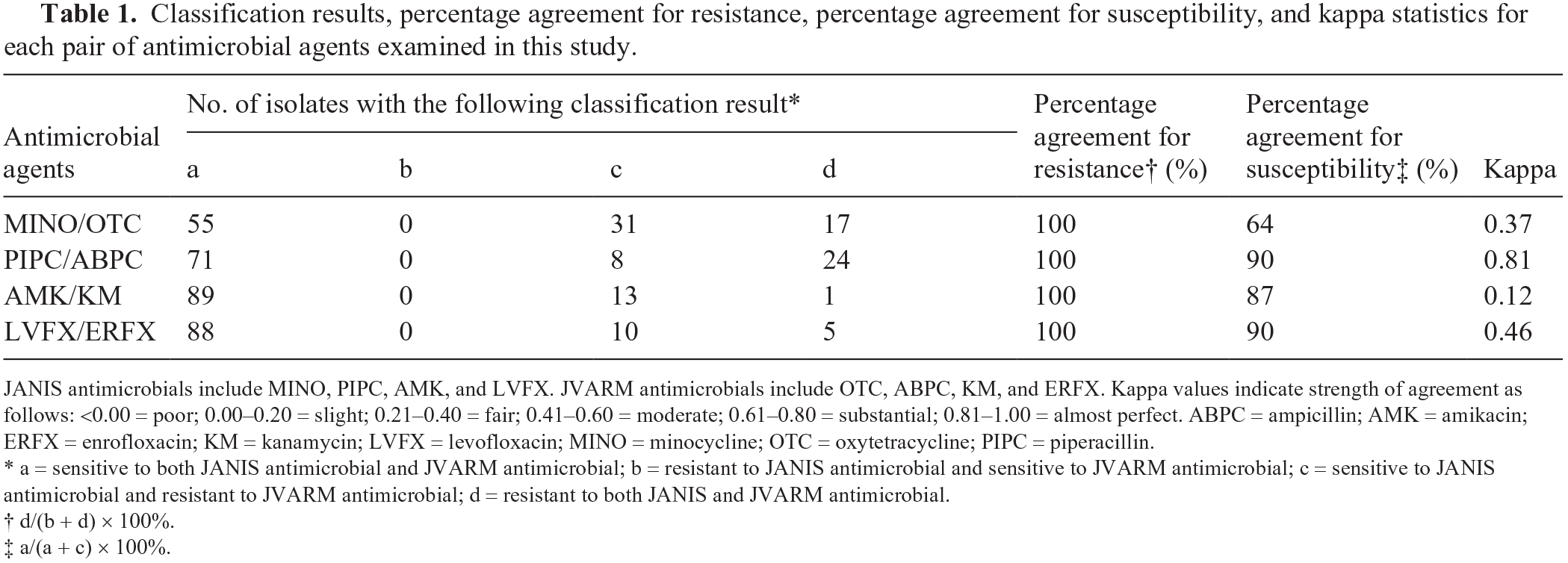
JANIS antimicrobials include MINO, PIPC, AMK, and LVFX. JVARM antimicrobials include OTC, ABPC, KM, and ERFX. Kappa values indicate strength of agreement as follows: <0.00 = poor; 0.00–0.20 = slight; 0.21–0.40 = fair; 0.41–0.60 = moderate; 0.61–0.80 = substantial; 0.81–1.00 = almost perfect. ABPC = ampicillin; AMK = amikacin; ERFX = enrofloxacin; KM = kanamycin; LVFX = levofloxacin; MINO = minocycline; OTC = oxytetracycline; PIPC = piperacillin.
a = sensitive to both JANIS antimicrobial and JVARM antimicrobial; b = resistant to JANIS antimicrobial and sensitive to JVARM antimicrobial; c = sensitive to JANIS antimicrobial and resistant to JVARM antimicrobial; d = resistant to both JANIS and JVARM antimicrobial.
d/(b + d) × 100%.
a/(a + c) × 100%.
PIPC is a broad-spectrum β-lactam antibiotic of the ureidopenicillin class 19 and is an ABPC derivative formed by conversion of the α-amino group to a ureido group. 16 A previous study showed that ABPC is an accurate predictor of the in vitro activity of PIPC against Enterococcus faecalis. 9 The almost perfect agreement (κ = 0.805), the percentage agreement for resistance (100%), and the percentage agreement for susceptibility (89.9%) that we found showed that E. coli susceptibility to PIPC could be predicted by susceptibility to ABPC.
In our results, the percentage agreement for susceptibility to OTC or MINO was low (64%), and the kappa statistics indicated only a slight correlation between the resistance to AMK and KM (κ = 0.117; Table 1). Therefore, we looked for antimicrobial resistance genes to explain these findings. Detection of the following aminoglycoside-modifying enzyme genes and tetracycline-resistance genes was performed by PCR: aac(3)-IIa, aac(6′)-Ib, ant(3′′)-Ia, aph(3′)-IIa, tet(A), tet(B), strA, and strB. The primer sets and PCR conditions have been described previously.8,26
The low specificity of OTC to MINO was a result of the high frequency of MINO-susceptible and OTC-resistant isolates (31 of 103). The results of resistance gene detection showed that most of the MINO-susceptible and OTC-resistant isolates contained the tet(A) gene (77.4%; Table 2). A previous study suggested that E. coli isolates harboring the tet(A) gene showed tetracycline resistance, whereas those harboring the tet(B) gene generally showed both tetracycline and MINO resistance. 20 Most of the MINO- and OTC-resistant isolates in our study contained the tet(B) gene (88.2%), in agreement with the results of the previous study. 20 The tet(A) and tet(B) genes encode tetracycline efflux pumps and predominate in E. coli from humans and animals.10,20
Tetracycline resistance genes found in 31 Escherichia coli isolates expressing various tetracycline susceptibilities.

MINO = minocycline; OTC = oxytetracycline.
We observed that the degree of deviation between the MICs of AMK and KM was large in AMK-susceptible and KM-resistant isolates, and that there was no clear relationship between aminoglycoside resistance genes and MICs of AMK (Table 3). The disagreement between MICs of AMK and KM may be caused by the difference in aminoglycoside resistance genes possessed by each isolate. It has been reported that aac(6′)-Ib and aacA4 are responsible for AMK resistance. 18 Although 3 isolates in our study possessed aac(6′)-Ib, 2 of 3 isolates did not exhibit AMK resistance. This may be the result of variable expression levels of the gene in these isolates.
Relationship between minimum inhibitory concentration (MIC) to amikacin (AMK) and kanamycin (KM) and aminoglycoside resistance genes.
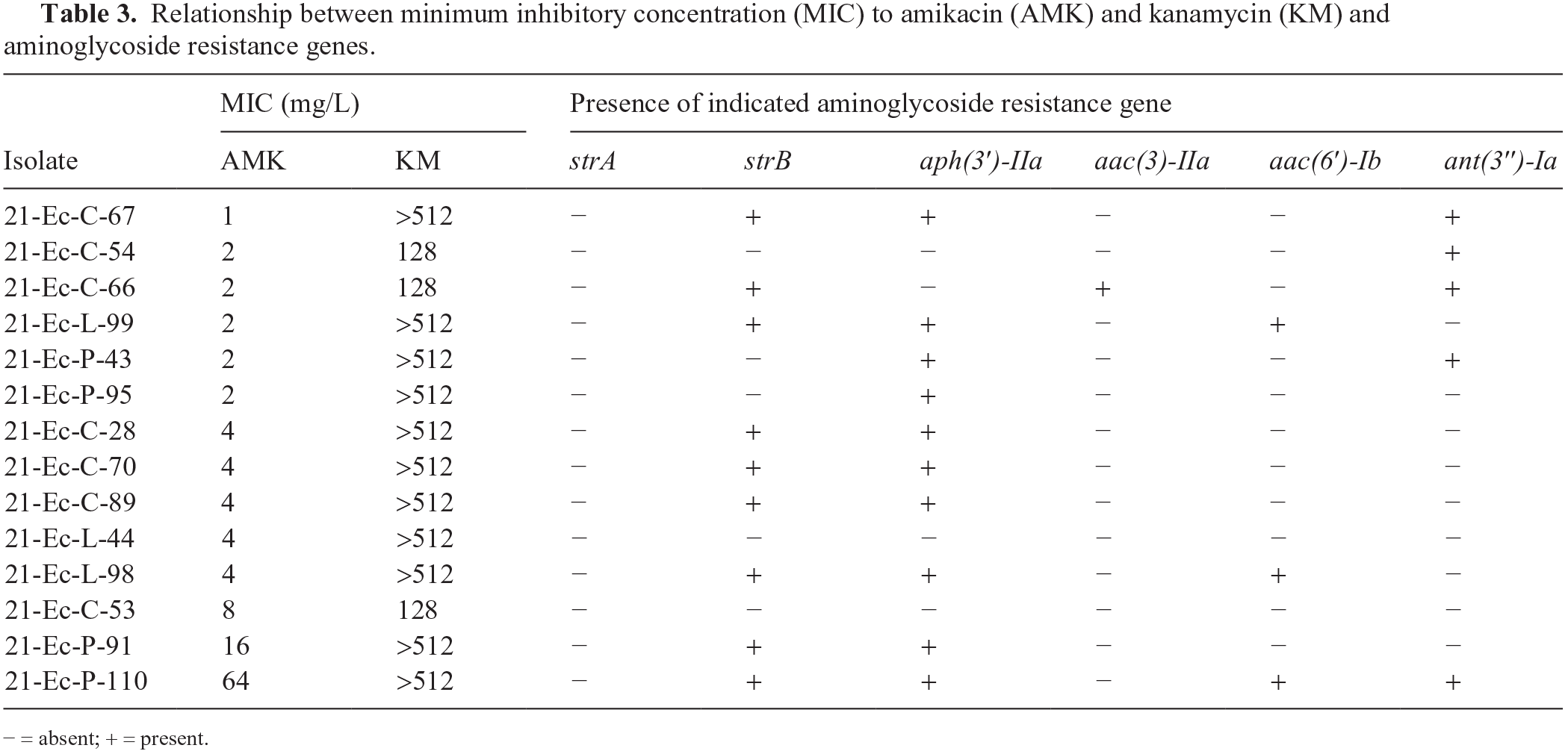
− = absent; + = present.
We found the MICs of antimicrobials used in animals to generally be higher than those of antimicrobials used in humans. Our results suggest that many isolates showing resistance to antimicrobials used in animals remain susceptible to those used in humans. In all classes of antimicrobials examined in our study, the agreement of resistance was 100% (Table 1), suggesting that, for example, an isolate that is resistant to MINO is always resistant to OTC.
We have demonstrated that the E. coli isolates resistant to antimicrobials used in humans are also resistant to antimicrobials belonging to the same class used in animals. These results suggest that if the resistance to antimicrobials used in humans is evaluated by the MICs of antimicrobials used in animals, the possibility that a resistant isolate would be regarded as sensitive is low.
Footnotes
Acknowledgements
We thank the staff of the Livestock Hygiene Service Centers across Japan for providing us with E. coli isolates.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported in part by a grant-in-aid from the Japanese Ministry of Health, Labor and Welfare (H27-Shokuhin-Ippan-008).
