Abstract
Laboratory tests provide essential support to the veterinary practitioner, and their use has grown exponentially. This growth is the result of several factors, such as the eradication of historical diseases, the occurrence of multifactorial diseases, and the obligation to control endemic and epidemic diseases. However, the introduction of novel techniques is counterbalanced by economic constraints, and the establishment of evidence- and consensus-based guidelines is essential to support the pathologist. Therefore, we developed standardized protocols, categorized by species, type of production, age, and syndrome at the Istituto Zooprofilattico Sperimentale delle Venezie (IZSVe), a multicenter institution for animal health and food safety. We have 72 protocols in use for livestock, poultry, and pets, categorized as, for example, “bovine enteric calf”, “rabbit respiratory”, “broiler articular”. Each protocol consists of a panel of tests, divided into ‘mandatory’ and ‘ancillary’, to be selected by the pathologist in order to reach the final diagnosis. After autopsy, the case is categorized into a specific syndrome, subsequently referred to as a syndrome-specific panel of analyses. The activity of the laboratories is monitored through a web-based dynamic reporting system developed using a business intelligence product (QlikView) connected to the laboratory information management system (IZILAB). On a daily basis, reports become available at general, laboratory, and case levels, and are updated as needed. The reporting system highlights epidemiologic variations in the field and allows verification of compliance with the protocols within the organization. The diagnostic protocols are revised annually to increase system efficiency and to address stakeholder requests.
Introduction
Animal health testing has benefited from technologic and scientific advances in laboratory medicine. This progress grew in the early 1990s with the application of molecular biology for infectious diseases, 21 and has contributed to a greater importance of the laboratory from a clinical and epidemiologic perspective in responding adequately to evolving health, social, and production issues. The main factors that have contributed to elevating and making the role of the biomedical laboratory indispensable are: the availability of new technologies; the eradication or reduction of classic diseases; the discovery of new diseases; the decline of diseases diagnosed by their pathognomonic lesions; the occurrence of multifactorial diseases and combined disease expressions that complicate the clinical and pathologic picture; epidemic events in densely populated regions that face increasingly stringent international regulations; the need to contain endemic diseases; and the need to satisfy an increasing demand by farmers and integrated enterprises for usable information.
The expansion of laboratory testing has been made possible by enormous investments in equipment and well-qualified and trained personnel. In recent years, financial resources have been reduced and often have been insufficient for the quantity and quality of healthcare needed by both the public and private sectors. 8 In addition, those same financial constraints have contributed to the affirmation in the veterinary field, already affirmed in human medicine, of the ethics of the organization, for which the person in charge of the laboratory must ask whether the test procedure is appropriate with respect to the goals of the client, while having the economic sustainability of the organization in mind. 2 To this end, appropriateness can be defined as “the extent to which a particular procedure, treatment, test, or service is effective, clearly indicated, not excessive, adequate in quantity, and provided in the inpatient, outpatient, home, or other setting best suited to the patient’s needs.” 13
The principle of appropriateness can be applied through evidence-based practices that integrate the experience of the pathologist with the conscientious, explicit, and judicious use of the best scientific evidence available, through the appropriate use of analytical and statistical methods.6,20 An evidence-based and appropriate approach does not replace the experience and skills of the veterinarian, but organizes, expands, and completes their skillset. By using an evidence-based approach, the diagnostic process is not only more objective and homogeneous, but also supports the choices of the veterinarian and recognizes their fundamental decision-making role in reaching the correct diagnosis.
Help for selecting and using the correct diagnostic approach is now provided within the scientific community through the definition of procedures, protocols, and guidelines.1,18 These tools are considered essential to assist organizations and health professionals to optimize the use of limited resources while improving the quality of outcomes, reducing medical liability, and minimizing the variability in clinical decisions, that is, linked to subjectivity and sometimes lack of knowledge in the definition of diagnostic choices. 14 This information can be analyzed and monitored using information technology (IT) tools. Business intelligence (BI) systems combine data collection, storage, and management with analytical tools to present complex information to planners and decision-makers.15–17 BI provides useful information, delivered at the right time, in the right place, and in the right form to assist decision-makers. BI tools convert data into useful and accessible information and, through user analysis, into knowledge. The user can delve into the information through user-friendly and up-to-date data-driven dashboards, if well designed.
BI systems have come into frequent use in healthcare to better manage organizations and activities.3,5,7,10,11 The impact of BI systems has been enormous and is directed mainly at increasing the level of productivity of healthcare organizations. 22 BI systems are now fully developed and can be applied for epidemiologic and health-monitoring purposes as well.9,19 For the veterinary field, a reporting system for health surveillance has been developed. 12 We illustrate here the BI platform implemented at the Istituto Zooprofilattico Sperimentale delle Venezie (IZSVe) to monitor, manage, and optimize the appropriateness of the diagnostic process in a multicenter pathology service in Italy.
Materials and methods
Development of appropriate panels
IZSVe is a multicenter public health institution, within the Italian National Health Service, whose mission is to conduct prevention, control, and research services in the fields of animal health and food safety for northeast Italy. The IZSVe launched a project in 2013 to standardize the diagnostic process at the autopsy level by defining diagnostic protocols to guide the pathologist in selecting the analyses most appropriate for each case. The project was expected to allow measurement of the quality, efficiency, and cost of the pathology service. To this end, a multidisciplinary team was formed within IZSVe combining skills in data management, business analysis, computer science, epidemiology, laboratory testing, and pathology.
Working groups were defined for each animal species (e.g., bovine, avian, pets) based on the expertise of the veterinarians in charge of the 10 provincial laboratories. Protocols were defined by species, type of production, age, and syndrome through a participatory and shared process, and were built as panels of analyses. Within each panel, certain analyses were defined to be mandatory, which includes analyses considered to be essential or critical to reach the final diagnosis, analyses for those diseases that should be tested according to National and EU Regulation 429/2016 (e.g., avian influenza and African swine fever), 4 and analyses included for epidemiologic purposes. Analyses that were optional within the syndrome panel were defined as ancillary. Ancillary tests can be selected by the pathologist to confirm or exclude the cause of disease suspected on the autopsy floor. Finally, all other analyses available within the IZSVe portfolio, and not included in the panel, were classified as no-panel analyses. After the autopsy, based on the anamnesis and the lesions observed, the pathologist assigned the case to a specific syndrome panel, with the data entered in the results window of the IZSVe laboratory information management system (IZILAB LIMS, Info.C.E.R.; Fig. 1). Because pathologic features could overlap between syndromes, the decision was made to apply the concept of predominant syndrome. The best-fitting syndrome was selected based on lesion severity, accepting the limits dictated by a rigid classification. Furthermore, if the pathologic features did not match any available panels, the pathologist could classify the case as “other/not classifiable.”
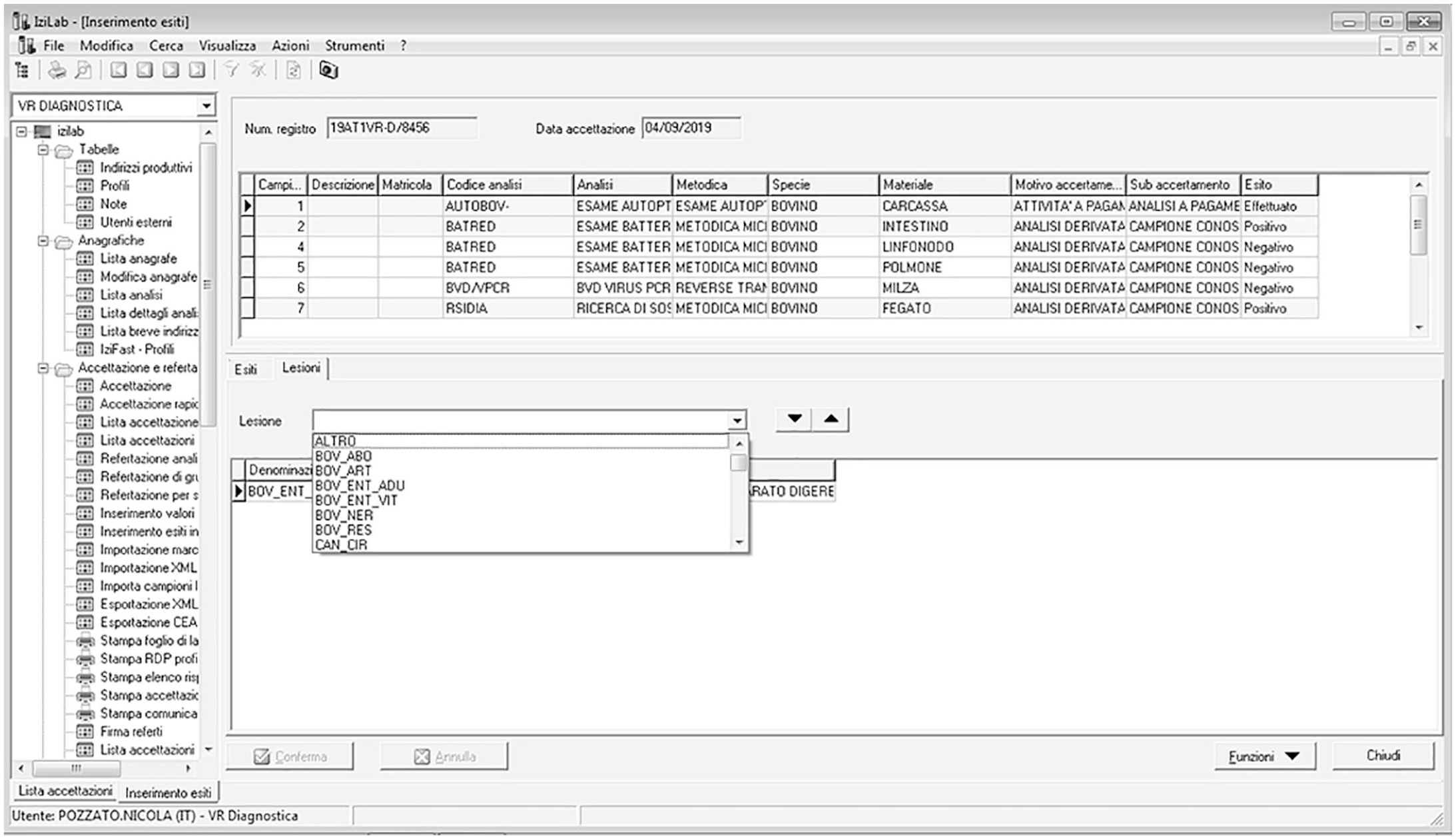
The IZILAB window in which the lesion is registered at autopsy. The codes are defined in Suppl. Table 1.
We defined 72 syndrome panels at IZSVe, classified by species, type of production, age, and disease syndrome or body system (Suppl. Table 1). Examples for cattle are: “bovine enteric calf”, “bovine enteric adult”, “bovine respiratory”; mandatory and ancillary tests filtered for the syndrome are included in a panel dashboard (Fig. 2). Through the examination of baseline aggregated and detailed reports, each pathologist can evaluate the activity at their provincial laboratory relative to the activity of the whole institute.
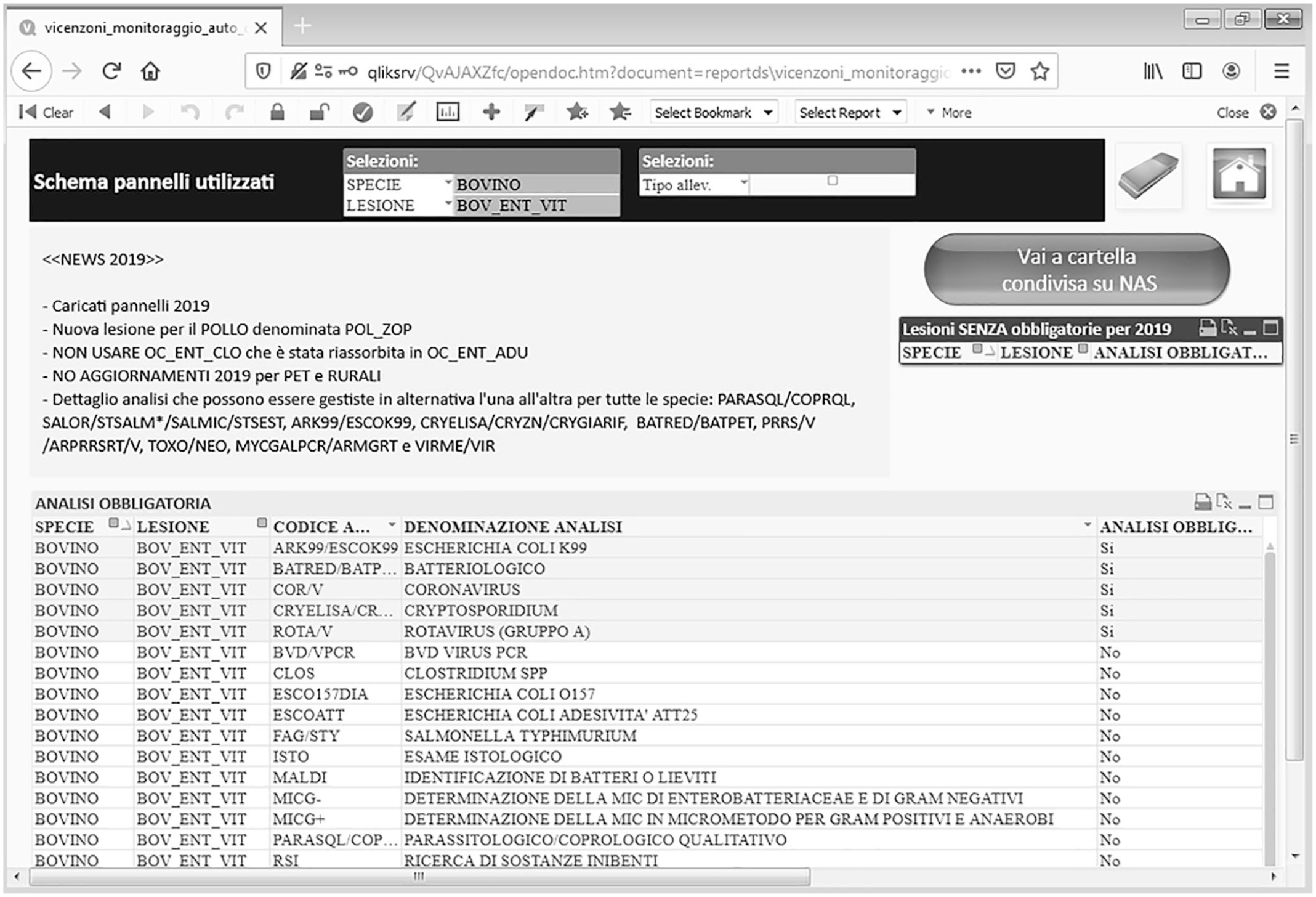
Example of the dashboard representing the list of mandatory (shaded) and ancillary (unshaded) analyses for the “bovine enteric calf” syndrome panel.
Syndrome panels are reviewed in general every year to add or remove analyses and, if necessary, to define new panels according to the evolving epidemiologic situation or the need to account for evolving syndromes. To review syndrome panels, we defined 2 indicators: 1) the ratio between the number of cases that are classified for each species as “other/not classifiable” and all of the cases registered in the year, and 2) the number of no-panel analyses used in the period with respect to the number of ancillary analyses for each syndrome and laboratory. Reviews of syndromes are mandatory if the frequency of these indicators is >10% of baseline.
The dashboard
To monitor each diagnostic protocol, a dashboard with syndrome panels was constructed by IT experts, including a series of analytical and aggregation tables that summarize the activity in each area. This functionality was implemented through a cutting-edge multipurpose BI tool (QlikView; QlikTech International), originally used in IZSVe to analyze the analytical activities of the laboratories and manage administrative aspects. Laboratory information and results are recorded in IZILAB and, utilizing QlikView, these data are processed into a dynamic web-based reporting system (Fig. 3). All tables and charts are interactive, and results are updated in real-time by selecting the appropriate option from the filters or by clicking on a bar in the graphs. Main and secondary dashboards were developed and updated based on current needs, providing access to the information generated by the system. The principal dashboard reports information on the activity and accuracy of the data entered into the IZILAB (Fig. 4). Global filters can display results by year, month, receiving laboratory, species, farm type, and syndrome. Tables can be easily exported with a click into text, Excel, or graphs.
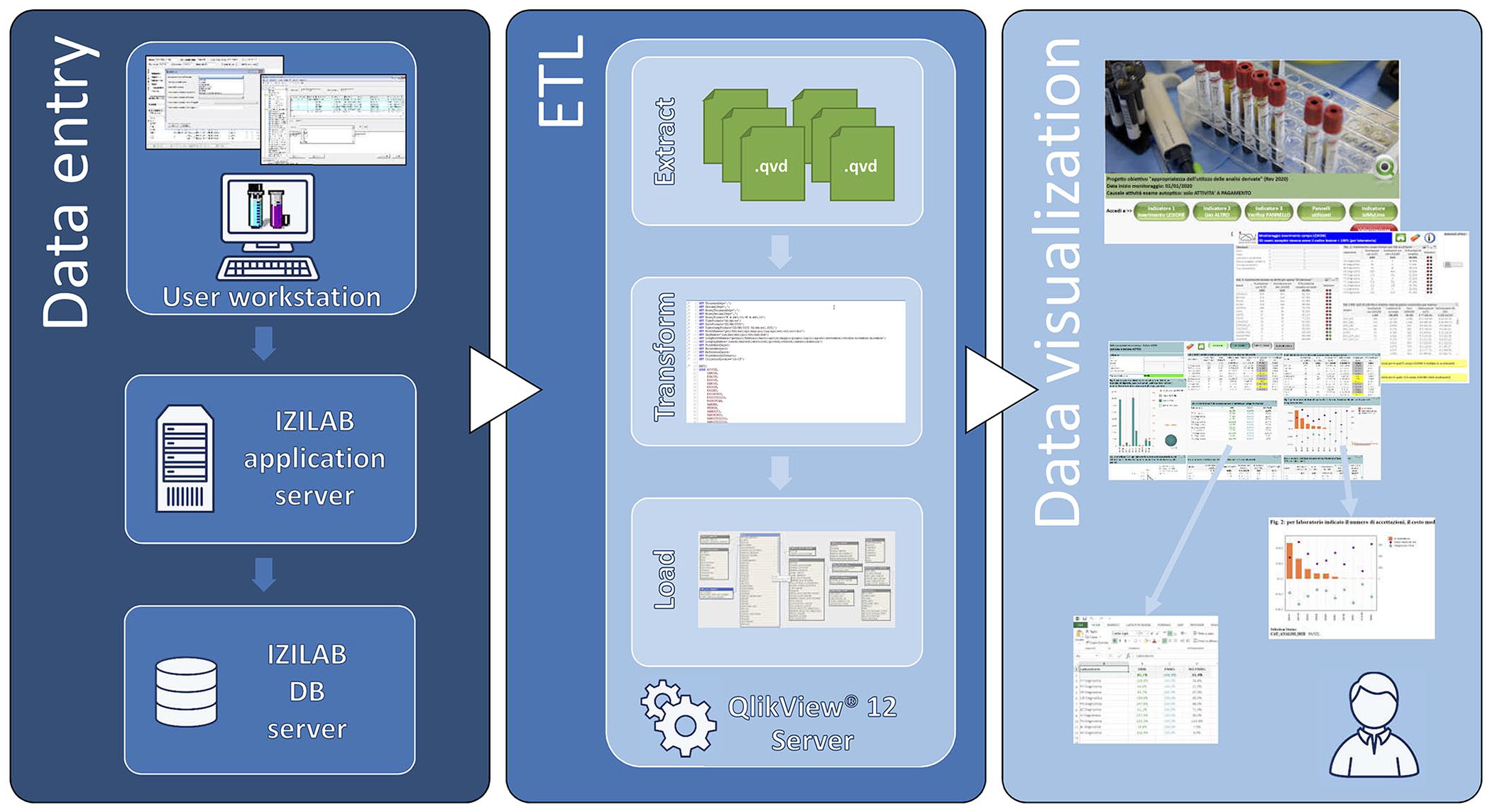
Architecture of the analytical platform and dataflow. Data are entered into the IZILAB LIMS at the user workstation and transferred to the IZILAB DB server through the IZILAB application server. Via the QlikView 12 server, data are selected and extracted, transformed, and then loaded (ETL) into the front-end tool Qlik View. On this level, summary dashboards with drill-down capabilities are generated. Tables and graphs can be exported with a click into text, Excel, or graphics.
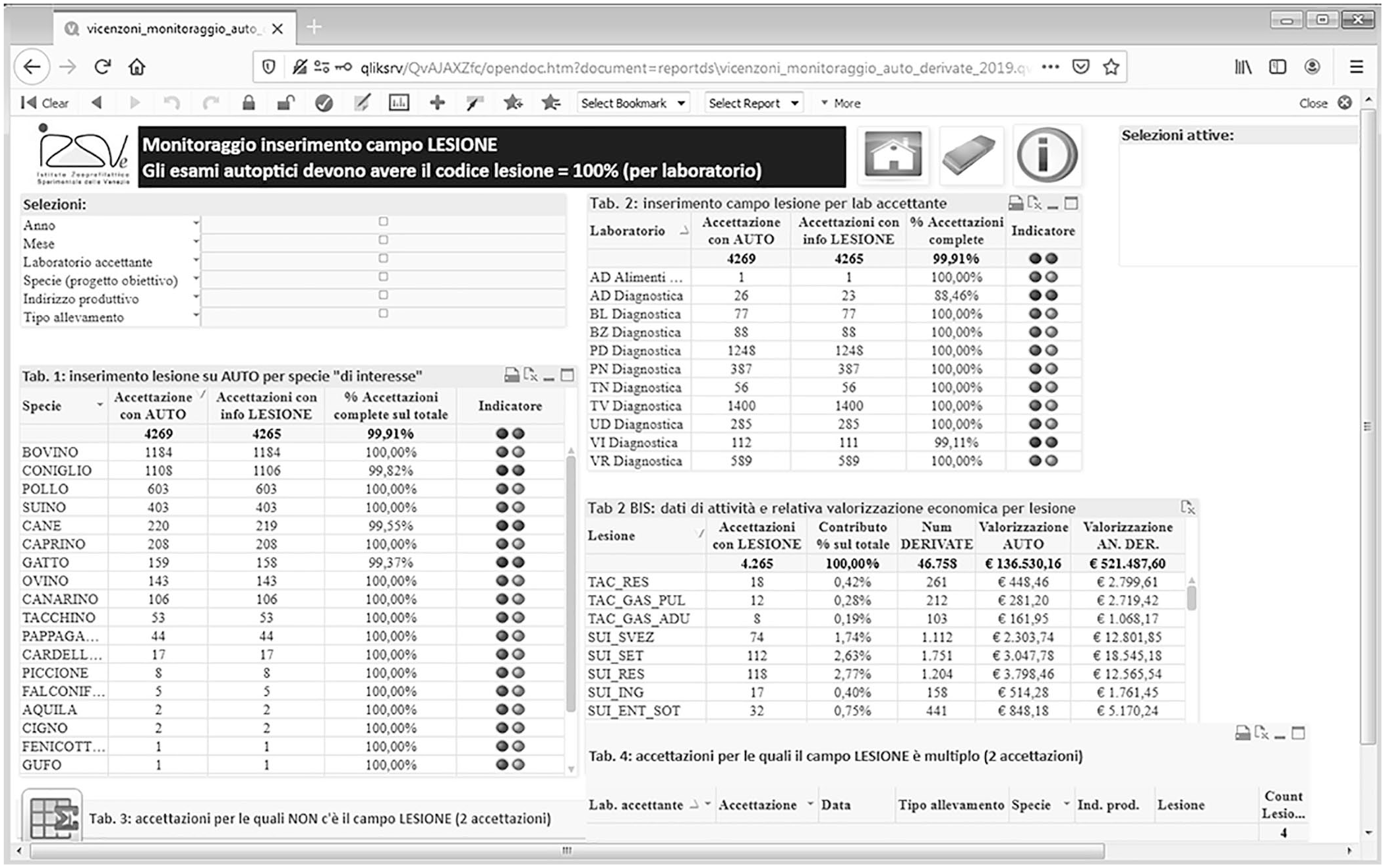
Example of the principal dashboard designed to monitor activity by species, syndrome panel, time period, and laboratory.
The data
The cases described here were submitted for autopsy at the 10 provincial laboratories of the IZSVe in 2017–2019. All animals from intensive, extensive, and small-hold farms as well as pets received for diagnostic purposes were included in the reporting system. Excluded were cases requested by official veterinarians, as suspects of notifiable infectious diseases or forensic cases. Each case could refer to one or multiple animals depending on the number of cadavers received by the laboratory. For poultry, rabbits, pigs, and ornamental birds, each case was usually composed of 5 animals.
Results
In 2017–2019, 11,792 cases were submitted to the 10 provincial laboratories of IZSVe for diagnostic autopsy. Each case included one or more cadavers depending on the species (Table 1) and was supported by clinical anamnesis when available. The most common species examined were cattle, rabbits, and poultry, with 3,203, 2,721, and 2,186 cases, respectively. These 3 species accounted for 68.8% of the cases received. Less frequent were pets and swine. Other species received were primarily ornamental birds.
Distribution of the cases submitted to IZSVe local laboratories for diagnostic autopsy in the period 2017–2019 by species and year.
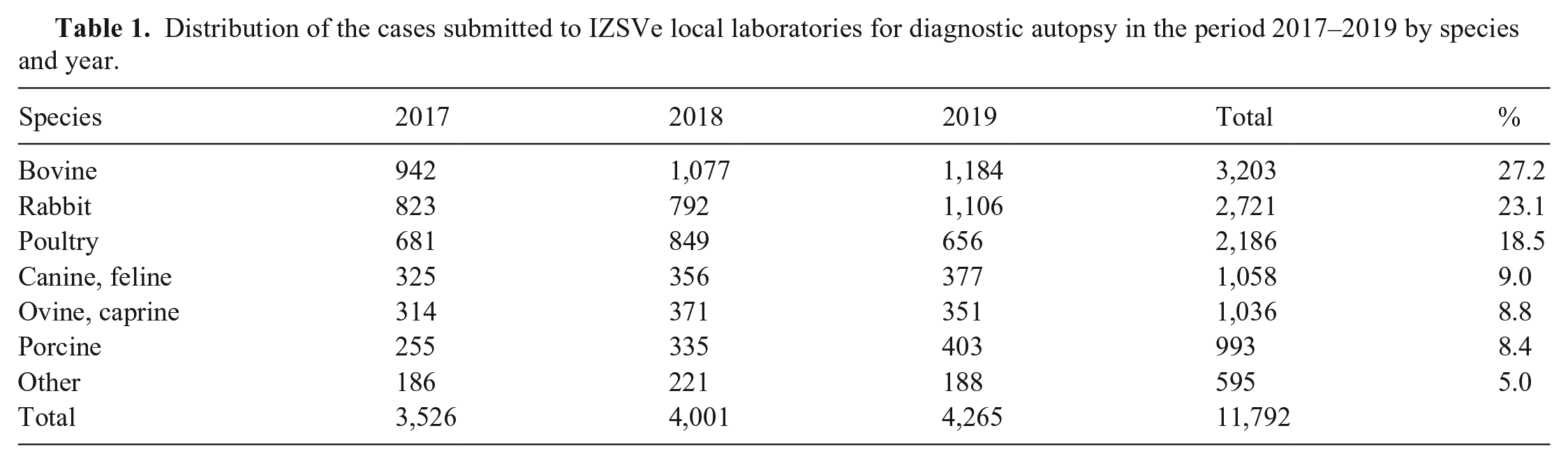
Overall, we noted that the laboratories with a high number of cases for a single species tended to use a lower number of ancillary analyses than laboratories that received small numbers of cases for that species. As an example, among the 993 swine cases examined, the referral laboratory received 527 cases and applied a median number of 6.7 (1–3 quartiles: 4.8–8.1) ancillary analyses. However, the other laboratories applied a median number of 8.3 (1–3 quartiles: 6–11) ancillary analyses. In addition, we found marked differences among the 10 laboratories with respect to the number of ancillary analyses carried out for dogs and cats, irrespective of the number of autopsies carried out. In the 3 y, a median number of 9 (1–3 quartiles: 5–14) ancillary analyses were used, ranging from the extreme of a laboratory with 15 analyses (1–3 quartiles: 8–22) to another with 4 analyses (1–3 quartiles: 2–8.5).
The most common syndrome among all species studied was the enteric syndrome, followed by the respiratory syndrome (Table 2). The prevalence of these 2 syndromes differed widely among species, with enteric versus respiratory cases at a 4:1 ratio in rabbits and small ruminants, 3:1 in companion animals, 2:1 in poultry, 1:1 in cattle, and the extreme opposite in swine with a 1:5 ratio, which could be skewed because of a separate “young” category defined for preweaning piglets. Marked differences in the distribution of the most common syndromes found in cattle, rabbits, and poultry were observed (Fig. 5). In cattle, a substantial change in the proportions among the syndromes was observed, with a 5.8% increase in respiratory cases from 2018 to 2019. In contrast, a decrease of respiratory cases in rabbits was accompanied by an increase of enteric cases in 2018. A progressive drop in respiratory cases was recorded in poultry over the 3 y.
Distribution of the cases submitted to IZSVe local laboratories for autopsy during 2017–2019 by macro category of syndromes and species.
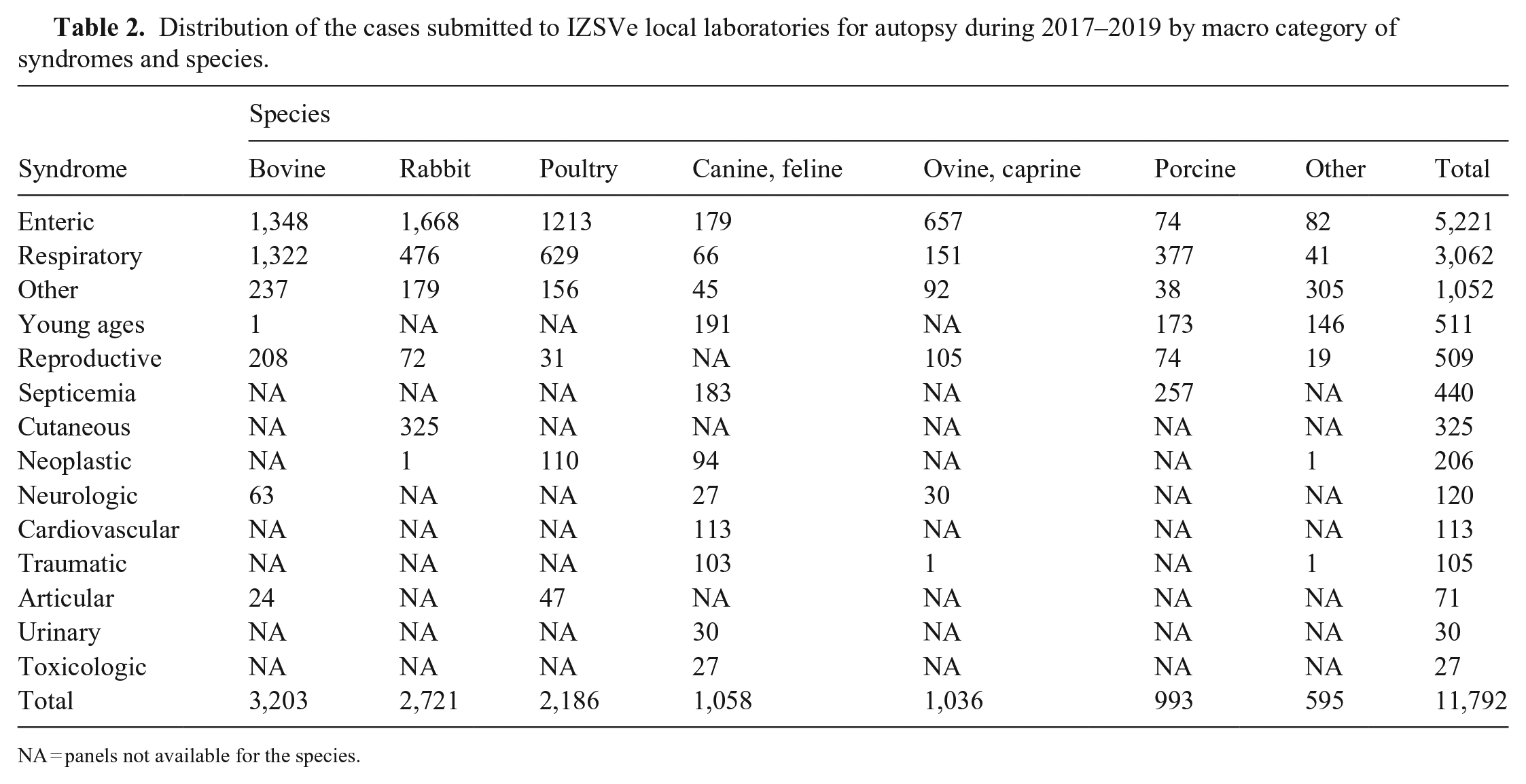
NA = panels not available for the species.
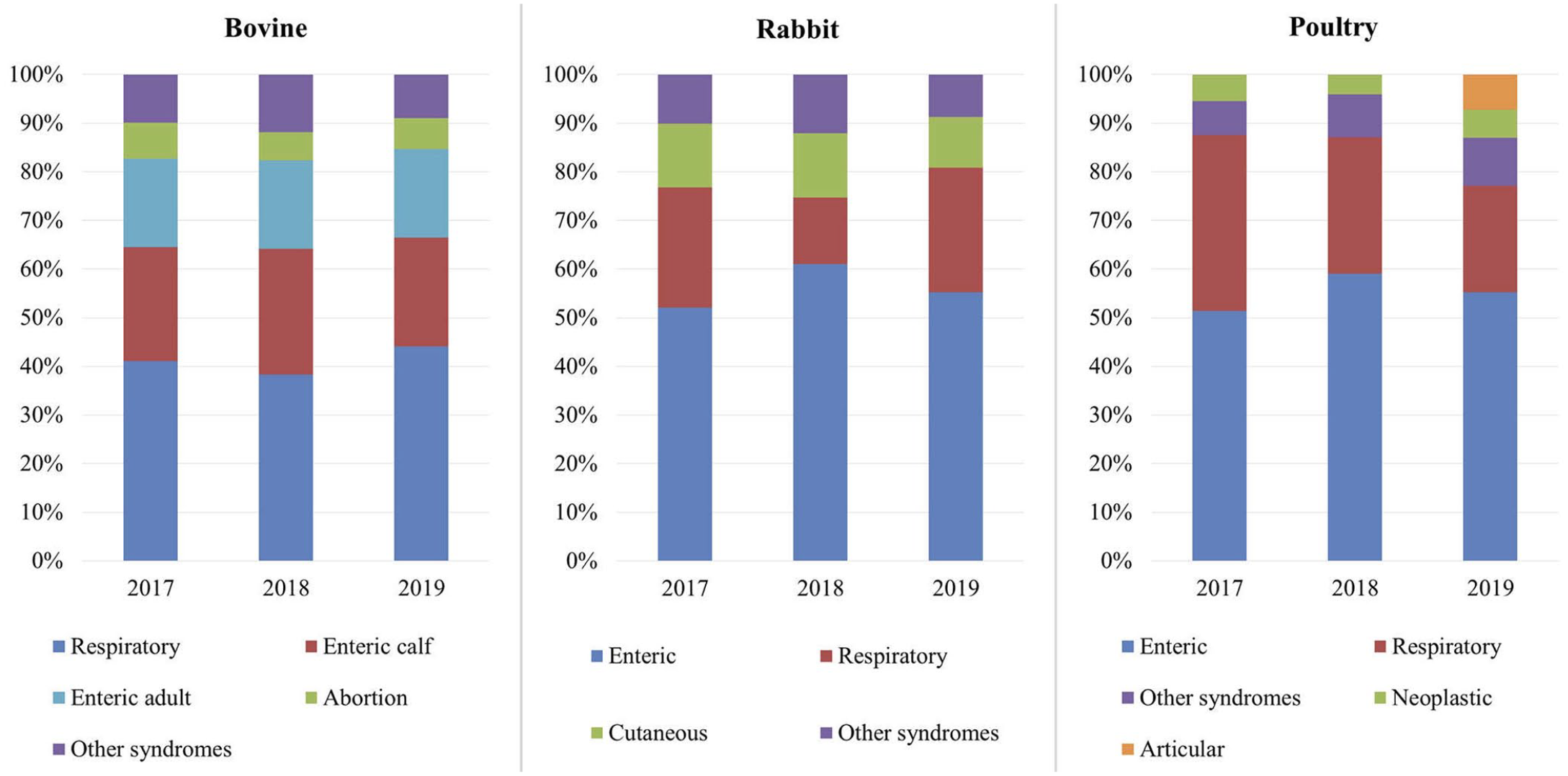
Distribution (%) of the most significant disease syndromes by year in the 3 most common species.
In poultry, a marked increase in the syndrome “other/not classifiable” was observed in 2018. A more detailed analysis of these cases by the avian working group demonstrated articular and skeletal lesions in broilers associated with the isolation of Enterococcus cecorum. Consequently, the avian working group defined a new panel called “articular” and added it in 2019 to the array of panels. This group also recorded a high occurrence of Mycoplasma synoviae infection in the respiratory panel of broiler chickens. These results led to a discussion of possible measures to support our clients in controlling the disease. Hence, the monitoring of 1-d-old chicks by serology to detect possible seroconversion in breeder flocks was advised.
Another interesting outcome of the dashboard in terms of appropriateness was that it highlighted the costs for a positive result for bovine viral diarrhea virus (BVDV) testing. The bovine working group evaluated the detection of BVDV in the bovine panels using 2 tests with different characteristics and costs (direct ELISA and real-time PCR); positive sample results showed 100% concordance between the 2 methods. Based on economics, the group updated all cattle panels that included BVDV testing, advising pathologists to use the direct ELISA only.
To better analyze mandatory, ancillary, and no-panel analyses, the system was implemented in 2019 with secondary dashboards. These include summary tables with the frequency of positive results for analyses with dichotomous results. Contextually, to consider analysis complexity and provide a weighting system, the cost of each analysis has been included as a parameter. Discussion is ongoing on how to structure cost-benefit analysis and performance evaluation of the laboratories that takes into consideration the proportional usefulness of each ancillary analysis in reaching the final diagnosis. Currently, the pathologist inserts the morphologic diagnosis into the IZILAB LIMS as a text file. The absence of unique keywords for the categorization of the final diagnosis does not allow complete integration of the results in the dashboards, hampering the measurement of the appropriateness of the syndrome panels and ancillary analyses applied in each laboratory.
Discussion
Health information technology, with the potential to enhance quality healthcare, is often regarded as an obstacle to effective clinical care. However, our tool, which acquires data from the 10 diagnostic laboratories of the IZSVe, is a multicenter system suitable for taking an analytical approach to animal health problems in the field.
Although aimed at improving the diagnostic capacity of our laboratory network, the dashboards have also facilitated the detection of differences in the application of the diagnostic protocols and thus standardizing the approach within the laboratory network and improving the quality of our service. Over the years, each laboratory has developed species-specific expertise according to the distribution of animal populations in their geographic areas, especially in densely populated areas of industrial farming. For this reason, the most active laboratories in terms of submitted cases have become more specialized in dairy and beef cattle, swine, poultry, or rabbits, and the veterinarians in charge of these laboratories lead each species working group. A higher number of ancillary analyses observed in the less active laboratories may be associated with uncertainty in choosing the most useful and correct analyses to provide a final diagnosis.
Historically, IZSVe and other IZSs were born between the 2 world wars to support farmers in the fight against infectious diseases. In 1970, they become part of the newly built Italian National Health Service, expanding their activities to official testing for food safety. In recent years, the progressive change in human relations with companion animals has led to increased requests for analytical support of small animal clinics, including diagnostic and forensic postmortem examinations. Our monitoring system revealed high variability among laboratories in the number of ancillary tests requested for dogs and cats in the study period, which can be ascribed to a low level of harmonization. An additional explanation might be the different types of requests by clients from urban and rural areas. Moreover, companion animals are often adult-to-old animals, frequently affected by different comorbidities and a complex clinical history, complicating the choice of a single syndrome panel. The activity of the pet working group, in terms of training and sharing information with veterinary clinicians, aims to address and standardize the best diagnostic protocols.
To our knowledge, the only other reporting dashboards in a diagnostic veterinary laboratory network are used in the monitoring program on porcine reproductive and respiratory syndrome in the United States; this group obtained interesting results, applying a different BI software to aggregate, consolidate, and summarize multicenter anonymized data. 23 We can speculate that it would be important in the future to have a comparative evaluation of different platforms.
Our dashboards present information such that prompt interpretation of changes in the epidemiologic situation of farms in the territory is possible, thus allowing improvements in the diagnostic process specifically and in the clinical process more generally. As an example, the poultry working group introduced a specific articular panel as a result of increased detection of Enterococcus spp. infections associated with articular and skeletal lesions. The information provided by the dashboards can also be used to monitor endemic and epidemic diseases of interest and to provide tools for evidence-based decision-making to all stakeholders. As an example, subsequent to the occurrence of M. synoviae infection in broiler chickens, we proposed to our clients the monitoring of 1-d-old chicks by serology to detect possible seroconversion in the breeder flocks. The producer could then better manage new groups in companies that buy 1-d-old chicks from different sources.
Our dashboards have made it possible to detect epidemiologic variations in the field such as the variation of cases ascribed to specific syndromes, or the occurrence of cases attributable to syndromes that require specific analyses, or the introduction of new analyses alongside those already recommended (e.g., the articular syndrome in broilers). In addition, through annual evaluation by species-specific working groups, the diagnostic protocols are reviewed and eventually modified based on accumulated data, which has made the system more efficient (e.g., the BVDV decision). The easy extraction of data and the full report make it possible to discuss and share specific considerations in various sectors with either a wider or a more focused perspective.
To strengthen the diagnostic capacity of the laboratories, we consider it important to include the information related to the final diagnosis. Currently at the IZSVE, the morphologic diagnosis is entered as a text field, which is not always associated with a final comment that takes into account the results of ancillary tests, including histopathology and microbiology. To complete the recording of the diagnostic process, which will allow verification of the effectiveness of the protocols used, it is essential to categorize the possible final diagnoses. We believe that the next step is to define a system to monitor the final diagnosis, but we are conscious that such a fit-for-purpose veterinary classification system for diagnoses is not available. In human medicine, an international disease classification system has been defined by the World Health Organization, the global reference point in public health and the center for identifying health trends and statistics worldwide. This system, called the “International Classification of Diseases” (ICD), was recently updated in its eleventh edition. 24 The ICD contains ~55,000 unique codes for injuries, diseases, and causes of death. It provides a common language that allows healthcare professionals to share health information around the world. The availability of a similar system in veterinary medicine is crucial to categorize lesions and causes of death in various animal species in a standardized way. By following the same methodologic approach, and using, where possible, the categories already defined for human diseases, the reporting system described here could be implemented. It is with this perspective that future developments of BI applications at multicenter institutions and in large geographic areas are to be encouraged.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211003163 – Supplemental material for Business intelligence tools to optimize the appropriateness of the diagnostic process for clinical and epidemiologic purposes in a multicenter veterinary pathology service
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211003163 for Business intelligence tools to optimize the appropriateness of the diagnostic process for clinical and epidemiologic purposes in a multicenter veterinary pathology service by Nicola Pozzato, Laura D’Este, Laura Gagliazzo, Marta Vascellari, Monia Cocchi, Fabrizio Agnoletti, Luca Bano, Antonio Barberio, Debora Dellamaria, Federica Gobbo, Eliana Schiavon, Alexander Tavella, Karin Trevisiol, Laura Viel, Denis Vio, Salvatore Catania and Gaddo Vicenzoni in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Stefano Marangon, former Scientific Director of the IZSVe, for strong support in the development of the project, and all the veterinarians, biologists, biotechnologists, technicians, and administrative personnel from the 10 provincial laboratories and the histopathology, parasitology, toxicology, virology, and laboratory medicine laboratories of the IZSVe for their active collaboration in the project. We also thank Dr. Ivana Pallante for assistance in drafting the manuscript, and Dr. Douwe Bakker (Consultant, Lelystad, NL) for the final review of the manuscript and English editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that this work was supported by the Italian Ministry of Health [grant number IZS VE 17/17 RC] research project: “Avvelenamenti animali: diagnostica tossicologica untargeted ed epidemiologia spaziale per favorire misure di prevenzione e repressione degli atti dolosi.”
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
