Abstract
We evaluated serologic responses of cattle, bison, elk, and swine representing negative control, early vaccination (4–8 wk), late vaccination (21–28 wk) or booster vaccination, early after-experimental challenge (2–4 wk), and late after-experimental challenge (8–21 wk), in a brucellosis fluorescence polarization assay (FPA; n = 10 sera per species per treatment) using negative control sera from cattle, bison, elk, and swine (n = 5 per species). Sera from cattle shedding Brucella abortus strain RB51 in milk were also evaluated against the 20 negative control sera. The species of negative control sera used in the FPA could increase (p < 0.05) delta millipolarization (mP; delta mP = sample mP − negative control mP) results. In general, the species of negative control sera did not alter the interpretation of FPA results in control, vaccinated, or infected animals. Even after repeated RB51 vaccinations in bison, cattle, or elk, or in cattle shedding RB51 in milk, serologic results from the FPA remained negative. Species differences in FPA results were noted; elk developed robust humoral responses very quickly after infection that resulted in strong positive FPA results. In cattle and bison, humoral responses appeared to develop over a longer period of time, and greater delta mP values were detected at later times after infection. Sensitivity of the FPA for detecting infected animals was greatest for elk in early challenge samples and bison in late challenge samples. Our data suggest that species of origin of negative control sera does not influence interpretation of the FPA in natural hosts of Brucella abortus.
Introduction
Brucellosis in domestic livestock and wildlife is caused by infection with various Brucella spp., which are intracellular bacteria that are associated primarily with fetal losses or infertility in natural hosts. 9 Although animals are the natural hosts, the agent can be transmitted to humans, in which clinical signs and symptoms can be more severe and chronic than in animals.4,10 Addressing the disease in its natural host is the most economical and efficient approach for preventing brucellosis in humans. 16 To control brucellosis in domestic livestock, regulatory programs that have been in place in many countries for decades rely primarily on the serologic detection of Brucella-infected animals.
Although antibody responses are not correlated with protection against brucellosis, detection of these responses is an efficient means of identifying animals that have been exposed to Brucella infection. Virulent B. melitensis, B. abortus, and B. suis field strains express the O side chain (perosamine residue) on the lipopolysaccharide (LPS) and are considered to be “smooth” strains. In comparison, some vaccine strains (e.g., B. abortus strain RB51) do not express the O side chain on the LPS and are considered to have a “rough” phenotype. Because the O side chain is immunodominant, most brucellosis surveillance tests are based on the detection of antibodies against this antigen. 8
Although several brucellosis serologic tests are available, the fluorescence polarization assay (FPA; DOO Biotehnika) is commonly used for screening and confirmatory testing in the United States. The test is based on the detection of rotational differences that occur when antibodies bind to a small, soluble, fluorochrome-labeled antigen in solution. 7 Small molecules rotate randomly at a rapid rate resulting in rapid depolarization of light whereas larger complex molecules rotate more slowly and cause a reduced rate of depolarization of light. When serologic responses of cattle were compared at a reference laboratory, the FPA was slightly more sensitive than other tests, including complement fixation, serum agglutination, rose bengal, and competitive ELISA, and had comparable specificity. 13 Other studies have suggested that the FPA has similar or slightly lower specificity for detecting swine brucellosis compared to other conventional brucellosis serologic tests.14,18
Although the FPA is widely used for the detection of brucellosis in cattle, it is also used for screening sera from other species such as bison, elk, and swine. However, performance of the FPA in these species has not been well characterized. In addition, questions have been raised regarding the possible influence of negative bovine control sera (included with the test kit) on results for other species. We were also interested in evaluating whether multiple B. abortus strain RB51 (RB51) vaccinations, or persistent infections with RB51, could cause suspect or positive results in the FPA. We designed our study to determine the influence of negative control sera from different species on test performance, characterize species differences in FPA results, and evaluate FPA results after vaccination or infection with rough Brucella strains in cattle, bison, elk, and swine.
Materials and methods
Brucella abortus sera
Sera from bison, elk, and cattle (n = 50 samples per species) were randomly selected from serum banks at the National Animal Disease Center (NADC) from published and unpublished studies (n = 16 studies) for the following treatment groups (n = 10 samples per treatment per species): (1) negative control; (2) 4–8 wk after vaccination with the rough RB51 vaccine (early vaccination); (3) 21–28 wk after RB51 vaccination, or 4–8 wk after booster vaccination (late vaccination); (4) 2–4 wk after experimental challenge with virulent B. abortus strain 2308 during pregnancy (early challenge); and (5) 8–21 wk after experimental challenge with strain 2308 during pregnancy. All animals in treatments 4 and 5 were confirmed as culture positive at autopsy. Treatment group 6 included 8 serum samples from 3 adult Jersey cows that were continuously shedding B. abortus strain RB51 in milk, confirmed by culture isolation.
Brucella suis sera
Swine sera (n = 40) were selected randomly from frozen serum banks at the NADC for 4 treatment groups from previous studies (n = 10 samples per treatment): (1) negative control; (2) 4–8 wk after vaccination with the rough B. suis strain 353-1 vaccine (early vaccination); (3) 12–18 wk after B. suis strain 353-1 vaccination (late vaccination); and (4) 5–6 wk after experimental challenge with virulent B. suis strain 3B (early challenge).
ELISA
Humoral responses to RB51 in all sera from bison, elk, and cattle for treatments 1, 2, and 3 were determined by an ELISA described previously using methanol-killed RB51 bacteria.1,6 In a similar manner, antibody responses of bison, elk, and cattle to strain 2308 in treatments 1, 4, and 5 were determined by an ELISA using methanol-killed strain 2308 bacteria. The peroxidase-labeled secondary antibody for bison and cattle sera was a rabbit anti-bovine IgG (H+L) used at a 1:35,000 dilution (Jackson Immunoresearch). The secondary antibody for elk sera was a peroxidase-labeled, rabbit anti-goat IgG (H+L) used at a 1:5,000 dilution (Jackson Immunoresearch). Humoral responses of swine were determined using an ELISA using methanol-killed B. suis strain 353-1 (vaccination) or B. suis strain 3B (experimental infection). The secondary antibody for swine was a peroxidase-conjugated, rabbit anti-swine IgG (whole molecule) used at a 1:2,000 dilution (MilliporeSigma).
Negative control sera for the FPA
Background negative control sera were selected from serum banks at the NADC from cattle, bison, elk, and swine (n = 5 per species) that were known to be either from brucellosis-free herds or had been demonstrated to be seronegative on brucellosis serologic tests. All background sera had millipolarization (mP) values < 95 on the FPA, consistent with negative serologic responses for brucellosis, and had been stored at −70°C in serum banks. Sera from 4 cattle from 3 herds in Iowa, Minnesota, and Missouri were included as background samples. The negative control sample included with the test kit was the fifth cattle serum tested. Five negative control bison sera from captive bison herds across multiple states in the western United States were obtained. Similarly, sera from 5 elk originally free ranging in Wyoming and a captive herd in Minnesota were selected. Background sera for swine included 4 domestic swine from 2 herds in Iowa and 1 feral swine from South Carolina. The single positive bovine control serum used in each assay was included with the FPA kit.
FPA evaluation
Assays were run on a portable, single-well, fluorescence polarization instrument (Sentry 201 FP; Ellie). Data were recorded in a Microsoft Excel spreadsheet in which mP values were calculated by the FPA reader based on differences in measurements of the sample with and without fluorescent-labeled conjugate (tracer). Delta mP was calculated by subtracting the mean of 3 measurements of the tested background sera from the mP value of the control, post-vaccination, or post-challenge samples. Delta mP values < 10 were classified as negative; 10–20 as suspect, and > 20 as positive.
Each of the 20 background sera (n = 5 for each species) was used as the negative control in the evaluation of all B. abortus and B. suis sera from all species selected for evaluation (n = 240). All B. abortus or B. suis samples from a single species were tested against a background serum, and before initiating testing with another background serum.
Field data comparing responses of cattle, bison, and elk
For comparison of delta mP results from field samples, we obtained data from a state diagnostic laboratory (Montana Department of Livestock, Montana Veterinary Diagnostic Laboratory, Bozeman, MT) in which fresh sera were run using the FPA on a multi-detection microplate reader (Synergy 2; BioTek Instruments) using the single negative bovine control sample included in the FPA kit. Data included assays of 427 cattle sera, 179 bison sera, and 100 elk sera. We compared the mean delta mP of the 3 species.
Statistical analysis
Optical densities (ODs) in the ELISA were compared by species using a general linear model procedure (SAS Institute). FPA data (mP and delta mP) were compared by species of background sera using an analysis of variance model. For analysis of FPA data obtained under field conditions, repetition was included as a variable in the model. Chi-square analysis was used to compare differences in test interpretation (positive, suspect, or negative) to determine if background sera influenced overall test results. In the chi-square analysis, we compared positives alone; negative and suspect results combined; and positives and suspect responses combined compared to negative results. Treatment means within a species for OD, mP, and delta mP were separated by the use of a least significant difference procedure (p ≤ 0.05).
Results
ELISA
Comparing ELISA results for Brucella strains that do not express the lipopolysaccharide O side-chain (rough), sera from cattle, elk, and swine had mean ODs in early vaccination that were greater (p < 0.05) than the control sera but did not differ (p > 0.05) from sera at later times after vaccination, or after booster vaccination (Table 1). Sera from bison differed (p < 0.05) across all 3 sets of samples, with the highest ODs in the late vaccination response, followed by early vaccination, and negative control samples.
Humoral responses of cattle, bison, elk, and swine after vaccination or experimental infection as measured by optical density in an ELISA using methanol-killed rough (B. abortus strain RB51 or B. suis strain 353-1) or smooth (B. abortus strain 2308 or B. suis strain 3B) strains as antigens.
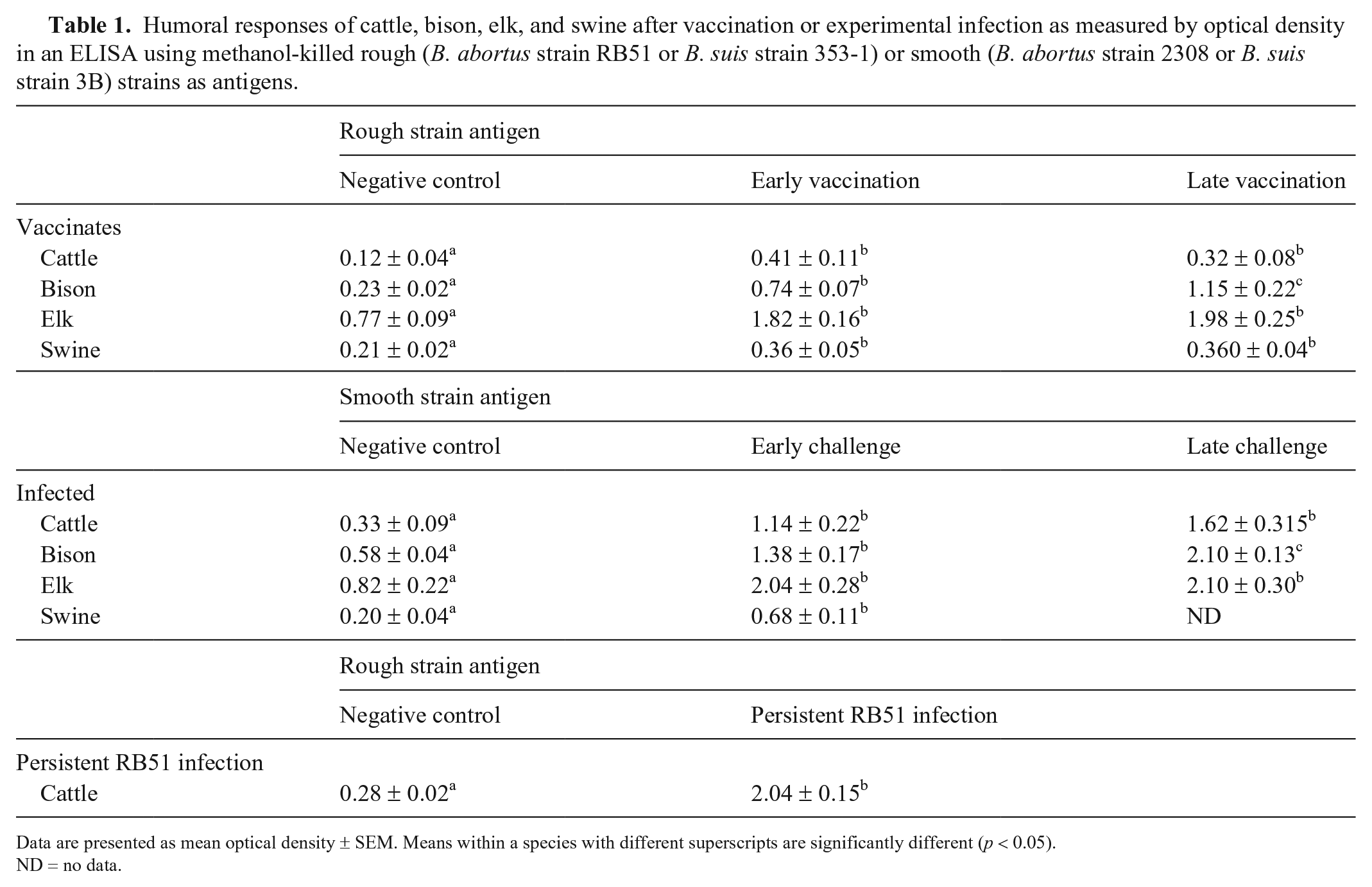
Data are presented as mean optical density ± SEM. Means within a species with different superscripts are significantly different (p < 0.05).
ND = no data.
Comparing responses to virulent Brucella strains (smooth) after experimental infection, elk and cattle ODs in early challenge were greater (p < 0.05) than ODs of negative control sera but did not differ (p > 0.05) from results for sera obtained in late challenge. Similarly to the trend noted after vaccination, sera from bison differed (p < 0.05) across all 3 sets of samples, with late challenge sera having the greatest OD, followed by early challenge, and then negative control samples. Sera obtained from swine in early challenge had greater (p < 0.05) OD than negative control sera.
Sera from cattle shedding RB51 persistently in milk had greater (p < 0.05) ODs than control samples.
FPA
Swine background sera had a lower mean mP (p < 0.05) than background sera from bison, cattle, and elk (Table 2). The lower mean mP in swine sera was a result of the feral swine sample having the lowest mP readings for any background serum. Mean mP of negative control sera from bison, cattle, and elk did not differ (p > 0.05).
Mean mP results of background sera from cattle, bison, elk, and swine in the brucellosis fluorescence polarization assay.

Means with different superscripts are significantly different (p < 0.05).
Comparing FPA data from control, vaccinated, or infected sera within a species, no significant effects (p > 0.46) were noted by species of background sera on mean mP results. However, when delta mP of control, vaccinated, or infected sera were compared within a species, the source of negative background sera did have statistical importance for bison (p = 0.026) and swine (p < 0.0026), but not cattle (p = 0.82) and elk (p = 0.169).
Mean delta mP values, but not mean mP results, in control, early vaccination, and late vaccination treatment groups, were influenced (p < 0.001) by species of background sera when data were analyzed across all species. Species, as the source of the serum being tested, was highly significant (p < 0.001) in the statistical model, indicating species differences in FPA responses in these treatment groups. In control and early vaccination treatment groups, mean delta mP for cattle, when compared against all background sera, was greater (p < 0.01) than comparable responses of bison, elk, and swine. Mean delta mP of all 3 species in the control and early vaccination treatments (range: 0.77 to −2.63) remained well below the cutoff value of 10 delta mP used for interpretation of samples as suspect. In the late vaccination treatment, mean delta mP for swine, when compared across all background sera, was greater (p < 0.01) than mean results of samples from other species (bison, cattle, and elk). However, as noted for results in control and early vaccination treatments, the mean delta mP for all species in the late vaccination treatment (0.09 to −1.86) remained well below the suspect cutoff value.
Delta mP and mP responses of sera obtained from cattle, bison, elk, and swine in the early challenge treatment were not influenced (p = 0.38) by the species of background sera used. However, mean delta mP values did differ (p < 0.001) by species for sera obtained at the time of early challenge (Fig. 1). Sera from elk had greater (p < 0.05) mean mP and mean delta mP (105.8) results when analyzed across background sera and compared to results for sera from the other 3 species. Mean delta mP for cattle, bison, and swine in this treatment did not differ (p > 0.05) and were 36.9, 25.2, and 15.1, respectively.
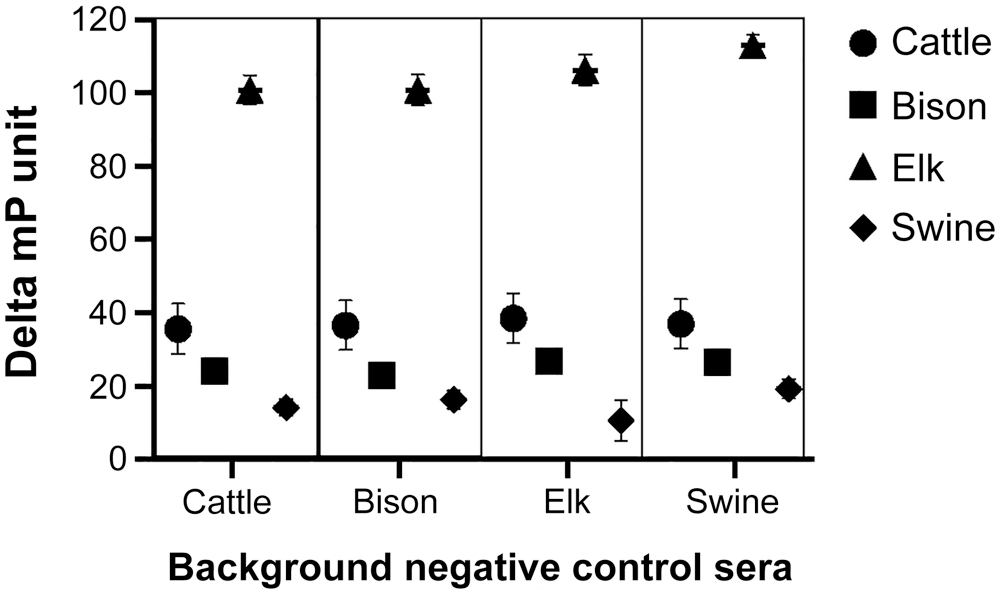
Serologic results for cattle, bison, elk, and swine in the brucellosis fluorescence polarization assay at 2–4 wk after experimental infection with 107 CFU of B. abortus strain 2308 (cattle, bison, and elk) or B. suis strain 3B (swine) when evaluated using each species of background negative control sera (cattle, bison, elk, or swine). Data are expressed as mean delta millipolarization unit (delta mP = [sample mP − background mP]) ± SEM.
Delta mP responses in the late challenge treatment (Fig. 2) were not influenced by the source of background sera (p = 0.92). Mean delta mP in this treatment did differ by the species from which the treatment sera samples were obtained (p < 0.001). In late challenge sera, bison had greater (p < 0.05) delta mP results (108.9) compared to results of sera from elk and cattle (91.4 and 86.5, respectively). No samples were evaluated for swine in the late experimental challenge treatment.
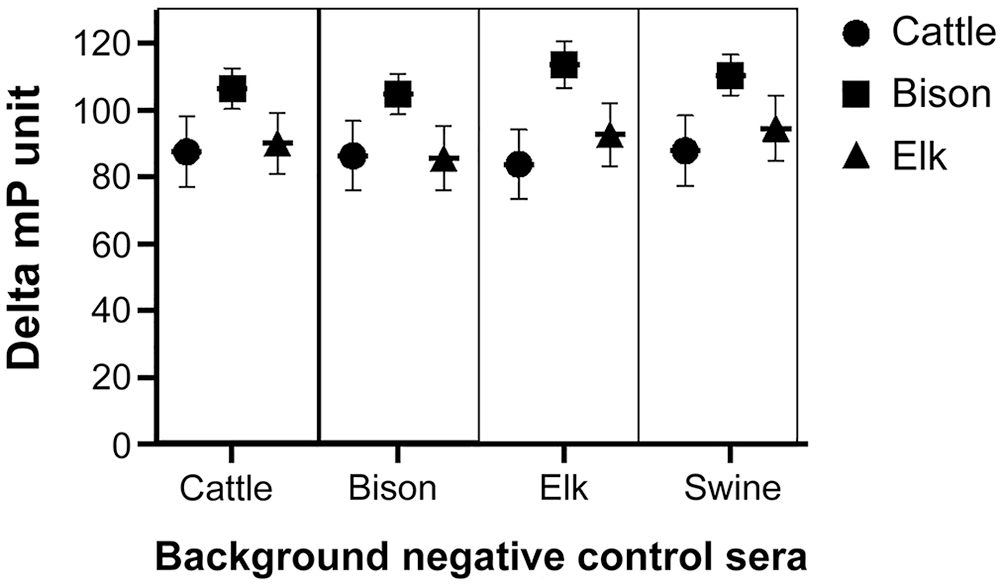
Serologic results for cattle, bison, and elk in the brucellosis fluorescence polarization assay at 8–21 wk after experimental infection with 107 CFU of B. abortus strain 2308 (cattle, bison, and elk) when evaluated using each species of background negative control sera (cattle, bison, elk or swine bkgd). Data are expressed as mean delta millipolarization unit (delta mP = [sample mP − background mP]) ± SEM.
Delta mP values for sera obtained from cattle persistently shedding RB51 in milk were not influenced (p > 0.05) by the species of background sera. Means obtained using each background serum remained < 10 delta mP for interpretation as a suspect (range: 0.62 to −5.5).
Interpretations of FPA results
The use of cattle, bison, or elk background sera did not influence whether samples in any treatment were interpreted as positive (with or without suspects) or negative. However, the feral swine background serum had the lowest mean mP of all background sera, resulting in a trend for increased delta mP in all sera tested against this background serum. This resulted in the analysis demonstrating that using swine serum as a background resulted in greater (p < 0.05) numbers of samples in control, early vaccination, late vaccination, and RB51 milk shedder treatment groups being classified as positive or suspect when compared to the use of bison, cattle, or elk sera as background control sera in the FPA. If the feral swine serum was not included in the analysis, the statistical effect on test interpretation was eliminated. The effects of the feral swine background serum on interpretation was not significant for early or late challenge sera from cattle, bison, elk, or swine. This suggests that the increase in delta mP, caused by the lower mP for the feral swine background serum, was relatively small when compared to the change in mP associated with positive responses in Brucella-infected animals.
Although the samples analyzed were limited, it was also of interest to note differences in sensitivity of the FPA between species. Given that all samples in early and late challenge were from culture-positive animals, and background sera did not influence test interpretation at these times, a rough estimate of sensitivity was calculated. At early times after experimental infection (5–6 wk in swine and 2–4 wk in the other species), the FPA had the greatest average sensitivity in detecting elk as suspect or positive (99.5%), followed by bison (73.5%), cattle (59.5%), and swine (50%). At 8–21 wk after experimental challenge, the FPA tended to be more sensitive for detecting infected bison (98.5%), followed by elk (61.0%) and cattle (62.5%). The possibility cannot be excluded that the rough sensitivity estimates were influenced by the magnitude of the delta mP values in each species, and the limited number of samples evaluated in each species after infection.
Species differences in FPA results for field samples
Comparing data from negative sera from cattle, bison, and elk obtained in a diagnostic laboratory setting using a Synergy 2 instrument and performed per the manufacturer’s recommendations, sera from cattle (0.18 ± 0.15) had the smallest mean delta mP. Bison sera demonstrated a greater (p < 0.01) mean delta mP (4.98 ± 0.24) compared to cattle samples, and elk sera (7.55 ± 0.41) had the greatest (p < 0.01) mean delta mP value when run against the bovine negative control sera included in the test kit.
Discussion
Our results demonstrate that there are species differences in sera properties in the FPA and that species of sera used as negative controls in the assay can influence results, but in general, do not influence test interpretation. Our data suggest that Brucella-negative sera from cattle tend to have higher mean mP values than sera from other species. This observation was supported by field data demonstrating species differences in delta mP values when sera were evaluated in a diagnostic laboratory setting. Although our data are limited, the results suggest that swine may have lower baseline values on the FPA. Species differences causing lower mP values for sera could potentially lead to small elevations in delta mP values for bison, elk, and swine when a negative control bovine serum (included in the test kit) is used in the assay. Although our data suggest that delta mP values rise very quickly after infection, the impact of the cattle background serum on test interpretation with bison, elk, and swine samples could cause some samples to be interpreted as suspect. Although this would be problematic for regulatory officials, our data suggest that a retest of bison and elk in 4–8 wk should be beneficial in detecting acute infections because FPA results should be significantly higher by this time.
Compared to bison, cattle, and swine, our data also emphasize the robust humoral response by elk that occurs early after infection with a virulent strain of Brucella appropriate to that host. Antibody responses by elk appear to plateau relatively early after infection given that our FPA data obtained later in infection tended to have lower mean values regardless of background sera used. In comparison, our data suggest that bison may develop stronger responses as indicated by the FPA results later in infection. In contrast to early in infection, data from our study suggest that bison samples from late infection have FPA results that are equal to or greater than responses of comparable samples obtained from infected elk.
Consistent with previous reports that RB51 does not induce seropositive responses after vaccination, 17 our data reaffirm that RB51 vaccination (bison, cattle, and elk), or when using a B. suis rough vaccine (swine), does not cause positive FPA results even after booster vaccination. In addition, cattle persistently infected with RB51, as evidenced by shedding in milk, remain seronegative even though RB51 ELISA results are significantly higher than peak titers after initial vaccination. It has been reported that RB51 expresses low levels of a B. melitensis–like O side chain on its LPS, suggesting that sufficient exposure to RB51 might induce positive results on brucellosis serologic tests. 2 Atypical results on brucellosis serologic tests associated with RB51 vaccination have been reported 5 and questions have been raised whether booster vaccination under field conditions could impact serologic responses. Our data suggest that these concerns are not valid for serologic responses evaluated using the FPA.
Similar to other reports that serologic tests have relatively low specificity and sensitivity for detection of brucellosis in swine,3,11,12 our data also suggest that the FPA is less sensitive in swine compared to other species. Recognizing the limitation in the number of samples that we evaluated, there was a nonsignificant trend for the assay to interpret more samples from infected swine as suspect or positive when swine serum was used as background. However, use of this approach does not appear to be appropriate given that sensitivity of the FPA was not dramatically improved, and specificity was reduced as more negative samples were interpreted as suspect or positive. Our data suggest that humoral responses of swine after infection with B. suis demonstrate significant differences compared to FPA results for cattle, bison, and elk infected with B. abortus. Our results and data from other reports3,15 emphasize the need for improved brucellosis serologic tests for swine.
Our study indicates that the species of negative control sera can influence FPA measurements, but the species does not appear to significantly alter the interpretation of sera as positive or negative. The commercial FPA is widely used for detecting brucellosis in natural hosts (cattle and wildlife), and our data suggest that continued use of the assay per the manufacturer’s directions in cattle, bison, and elk is acceptable and will not significantly alter the detection of seropositive animals.
Footnotes
Acknowledgements
We thank Darl Pringle and Lilia Walther for their technical assistance. Names are necessary to report factually on available data; however, the USDA neither guarantees nor warrants the standard of the product, and the use of the name by USDA implies no approval of the product to the exclusion of others that may also be suitable.
Declaration of conflicting interests
Miladin Kostovic is the owner of Ellie LLC, a commercial source for FPA instruments and the commercial FPA kit. The other authors (Olsen, Crawford, Fuentes, Boggiatto) have no potential conflicts of interest with respect to the research, authorship, and or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
