Abstract
Rickets can be attributed to nutritional, genetic, hormonal, or toxic disturbances and is classified as a metabolic bone disease. Rickets is most often associated with inappropriate dietary levels of calcium, phosphorus, and/or vitamin D. During a 27-month period (January 2010 through March 2012), the Iowa State University Veterinary Diagnostic Laboratory investigated causes of sudden, unexpected death and lameness in growing pigs throughout the Midwestern United States. Clinical observations from 17 growing pig cases included weakness, lameness, reluctance to move, muscle fasciculations and/or tremors, tetany, and death. Ribs were weak, soft, and bent prior to breaking; rachitic lesions were apparent at costochondral junctions in multiple cases. Acute and/or chronic bone fractures were also noted in multiple bones. Failure of endochondral ossification, expanded physes, infractions, thin trabeculae, and increased osteoclasts were noted microscopically. Decreased bone ash and serum 25(OH)D3, combined with clinical and microscopic evaluation, confirmed a diagnosis of vitamin D–dependent rickets in all cases. In 3 cases, disease was linked to a specific nutrient supplier that ultimately resulted in a voluntary feed recall; however, most cases in the current investigation were not associated with a particular feed company. The present report describes vitamin D–associated rickets and its importance as a potential cause of weakness, lameness, muscle fasciculations, recumbency or sudden unexpected death in swine, and describes appropriate samples and tests for disease diagnosis.
Metabolic bone disease broadly describes disturbances related to bone elongation, mineralization, formation, or remodeling. 28 Rickets, osteomalacia, fibrous osteodystrophy, and osteopenia are various forms of metabolic bone disease. These bone disorders have distinctive features, but can occur simultaneously in the same animals depending on the etiology. 28 Metabolic bone disease in animals is generally associated with dysregulation of parathyroid hormone (PTH) activity or dietary imbalances of phosphorus, calcium, or vitamin D. Mineral deficiency, toxicities, and genetic abnormalities have also been associated with metabolic bone disease conditions. 28 Genetic examples include defective vitamin D metabolism and impaired renal function that interferes with excretion of excess phosphorus.2,6,28
Rickets and osteomalacia have a similar pathogenesis resulting in decreased or defective bone mineralization. Rickets manifests as failure of endochondral ossification in growing bone. Osteomalacia occurs in mature bone when normal remodeling is disrupted. The majority of rickets or osteomalacia cases in production animal medicine occur when feed ingredients are improperly mixed or are in deficient quantities. 28
Swine are sensitive to rickets development because of rapid growth rate and limited exposure to sunlight in modern confinement facilities. 4 Swine also have a tendency to develop fibrous osteodystrophy when faced with metabolic disorders, and this can occur concurrently with rickets as a secondary response.28,29 In the past 2 decades (1990–2010), swine rickets received little attention because historical corn-soy nutritional programs provided adequate calcium and phosphorus, and were supplemented with sufficient vitamin D for normal bone growth and homeostasis. Current diets for market swine are specifically tailored for lean muscle mass growth with less consideration for bone formation. 18 Ingredient alternatives to a traditional corn-soy diet are frequently utilized in swine production because of input costs; the use of distillers’ dried grains with solubles is an example. Therefore, variations in quality or quantity of specific nutrients have the potential to result in subclinical or clinical bone disease.
The current report summarizes 17 different cases of rickets presented to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, Iowa) from growing pigs throughout the Midwestern United States between January 2010 and March 2012. All case submissions were from commercial swine production facilities. At-risk populations ranged from 500–10,000 pigs per case, and the morbidity ranged from 2–40% of the at-risk population.
The first case consisted of 15-week-old mixed-breed finisher pigs located in a 2,000-head, tunnel-ventilated confinement facility in northwestern Iowa in January 2010. Clinical signs reported by the attending veterinarian included an abrupt onset of hind limb weakness and death in approximately 10–20% of the pigs without a prior history of infectious disease. Three clinically affected pigs with hind limb weakness were submitted to the ISU-VDL for a diagnostic investigation. A complete postmortem examination of these pigs revealed mild generalized lymphadenopathy and pliable ribs that easily bowed prior to fracturing. Appropriate tissue samples, including bone growth plates were collected, placed in 10% phosphate buffered formalin for 24 hr, and embedded in paraffin wax. Bone sections were demineralized with a commercial product a for 24 hr according to the manufacturer’s instructions after formalin fixation and prior to paraffin embedding. Tissue sections (4 µm) were stained with hematoxylin and eosin for histopathologic examination. Microscopic lesions were not identified in sections of brain, spinal cord, tonsil, lung, liver, heart, spleen, kidney, intestine, lymph node, pancreas, or skeletal muscle from all submitted pigs. Conspicuous bone growth plate lesions included marked thickening and irregularity of the physis due to hypertrophied chondrocytes forming noncalcified tongues extending into the metaphysis. Large clefts with associated hemorrhage occurring parallel to the physis were occasionally noted at the margin of endochondral ossification. Nonmineralized, retained cartilage cores were common in the primary spongiosa and associated with osteoid accumulation. Numerous osteoclasts bordered the secondary spongiosa, and marrow spaces were frequently replaced by loosely arranged fibrous connective tissue consistent with fibrous osteodystrophy. Cortical bone was also diminished and sometimes replaced by osteoid or fibrous connective tissue. Aerobic and anaerobic cultures of lung, liver, kidney, and brain swabs were negative. Brain swabs were also polymerase chain reaction (PCR) negative for Haemophilus parasuis (ISU-VDL in-house test) and Erysipelothrix rhusiopathiae. 22 Pooled serum samples were PCR negative for Porcine circovirus-2 21 and Porcine reproductive and respiratory syndrome virus b nucleic acid.
The second rib from all pigs was analyzed on a dry weight basis for bone ash according to the ISU-VDL standard operating procedure using the distal portion of the second rib. Total bone ash results for the 3 pigs ranged from 40% to 42%. Normal reference values may range from 45% to 65%.1,9,26,31 Bone ash results from the second rib are comparable to values demonstrated previously in humerus, metatarsal, and vertebrae.1,9,31
Serum analyte analysis, performed at the ISU Veterinary Teaching Hospital, detected abnormal concentrations of calcium, phosphorus, alkaline phosphatase (ALP), and creatine kinase (CK). Specifically, calcium concentrations ranged from 4.6 to 4.8 mg/dl (reference [ref.] interval: 8.0–12.0 mg/dl), phosphorus concentrations ranged from 11.7 to 12.9 mg/dl (ref. interval: 4.5–9.0 mg/dl), and serum ALP concentrations ranged from 410 to 439 IU/l (ref. interval: 25.0–130.0 IU/l). All 3 pigs had serum CK values greater than 2,500 IU/l (ref. interval: 100–2,500 IU/l). Serum 25(OH)D3 levels were measured at Heartland Assays (Ames, IA). 23 All 3 pigs lacked detectable 25(OH)D3 (<2.5 ng/ml). Vitamin D deficiency can lead to hypocalcemia, 27 and acute death with profound hypocalcemia has previously been reported in swine fed diets deficient in vitamin D. 15 Acute, unexpected death and clinical signs were attributed to severe hypocalcemia and marked vitamin D deficiency in this first case. A sporadic feed mixing error was speculated as the cause of hypovitaminosis D; however, feed was not available for testing.
The second definitive case of rickets presented in 11-week-old nursery pigs experiencing tetany, seizure-like muscle fasciculations, and sudden death; over 20% mortality was reported from this site of 8,000 pigs. Ten live pigs with tetany or muscle fasciculations were submitted from this herd to the ISU-VDL in March 2010. Complete necropsy revealed flared costochondral junctions, commonly referred to as rachitic lesions, in 7 of the pigs. All pigs had soft, rubbery ribs that bent approximately 20–30 degrees prior to fracture. Histologic sections of formalin-fixed tissues and demineralized bone were prepared as previously indicated and included thyroid gland, parathyroid gland, and physeal cartilage from the rib, femur, metacarpus, metatarsus, and vertebrae. Microscopic lesions were confined to physeal cartilage and adjacent metaphyseal bone, with ribs being the most consistently and severely affected. Microscopic changes were consistent with failure of endochondral ossification with infractions similar to case 1. Lesions in growth plates of additional bones ranged from normal to severely affected. No significant microscopic changes were apparent in other examined tissues.
Serum samples from the 10 pigs in case 2 were tested for serum 25(OH)D3, calcium, and phosphorus as described above. All pigs lacked detectable 25(OH)D3 (<2.5 ng/ml) and had severe hypocalcemia that ranged from 4.1 to 5.9 mg/dl (ref. interval: 8.0–12.0 mg/dl). Serum phosphorus was within normal limits (4.5–9.0 mg/dl). Total bone ash results for the 10 pigs ranged from 26% to 33%. Case 2 was diagnosed as vitamin D–dependent rickets based on compatible macroscopic and microscopic lesions, abnormal serum vitamin D and calcium levels, and decreased bone ash.
Vitamin D–dependent rickets was diagnosed in 15 additional herd submissions over the subsequent 24 months. Diagnosis in all cases was based on clinical presentation, compatible microscopic bone lesions indicating failure of endochondral ossification, absence of other pathogenic disease processes causing lameness or sudden death, decreased bone ash, and very low serum vitamin D levels. Table 1 summarizes clinical signs of all 17 cases included in the present report. In the summarized cases, growing pigs developed 1 or more of the following clinical signs: lameness, weakness, reluctance to move, dog-sitting, increased humpbacks (kyphosis), bone fractures, seizure-like muscle fasciculations, and tetany (Table 1). Initial clinical signs were often sudden, unexpected death with tremors and/or seizure with no prior history of infectious disease. Submitting veterinarians would communicate that many of these clinical signs were present simultaneously within the herd. Pig age in the case series described herein ranged from 9 to 17 weeks of age or 23–68 kg. Late nursery to early finishing phase of production corresponds to a period of rapid growth for most pigs. Case presentation was more common in late winter and spring; however, sporadic cases were observed during all times of the year (Table 1). An important observation related to these cases made by the ISU-VDL diagnosticians was that barn type was generally a hotel-style nursery or a tunnel-ventilated barn, both of which have minimal sunlight penetration to animal pens based on constructional design.
Summary of vitamin D–dependent rickets cases submitted to the Iowa State University Veterinary Diagnostic Laboratory (Ames, Iowa) from January 2010 through March 2012.
IL = Illinois; IA = Iowa; NE = Nebraska; MN = Minnesota.
Mean age and weight of the pig population at the production facility for each submission.
Serum 25(OH)D3.
Barn designs resulting in lack of sunlight is important because vitamin D3, a fat soluble vitamin, can be synthesized in the skin from 7-dehydrocholesterol following ultraviolet B (UVB) exposure (sunlight). Lack of direct sunlight precludes this process. Vitamin D can also be supplemented in the diet. Following skin synthesis or diet vitamin D absorption, an obligatory two-step hydroxylation process is required for activation of vitamin D. Vitamin D is first hydroxylated in the liver to 25(OH)D, and a second hydroxylation occurs in the kidney forming the metabolically active 1,25(OH)D. 4 Multiple attributes of active vitamin D have been discovered in recent years, 13 but a primary function of vitamin D is to facilitate adequate absorption of calcium, and to a lesser degree phosphorus, from the small intestine. 20 Lack of calcium absorption results in PTH secretion and calcium mobilization from bone. It should be noted that there is still some uncertainty of the role of vitamin D as it relates to bone growth and mineralization; specifically, whether the effects of vitamin D are related to acceptable extracellular concentration of calcium and phosphorus or directed through cellular receptors and stimulation of normal growth plate development. 4
During winter months in northern latitudes above 31°, the angle of the sun prevents penetration of the appropriate wavelength of ultraviolet light (UVB rays). 4 Dietary supplementation of vitamin D3 is crucial to prevent rickets or osteomalacia during this period for all swine. Year-round dietary supplementation of vitamin D is considered a necessity for swine housed in confinement. 29 Relatively few feedstuffs contain vitamin D; natural sources include fish (cod) or fish oils. 4 Plant-based diets contain no vitamin D3 and only minor amounts of vitamin D2.
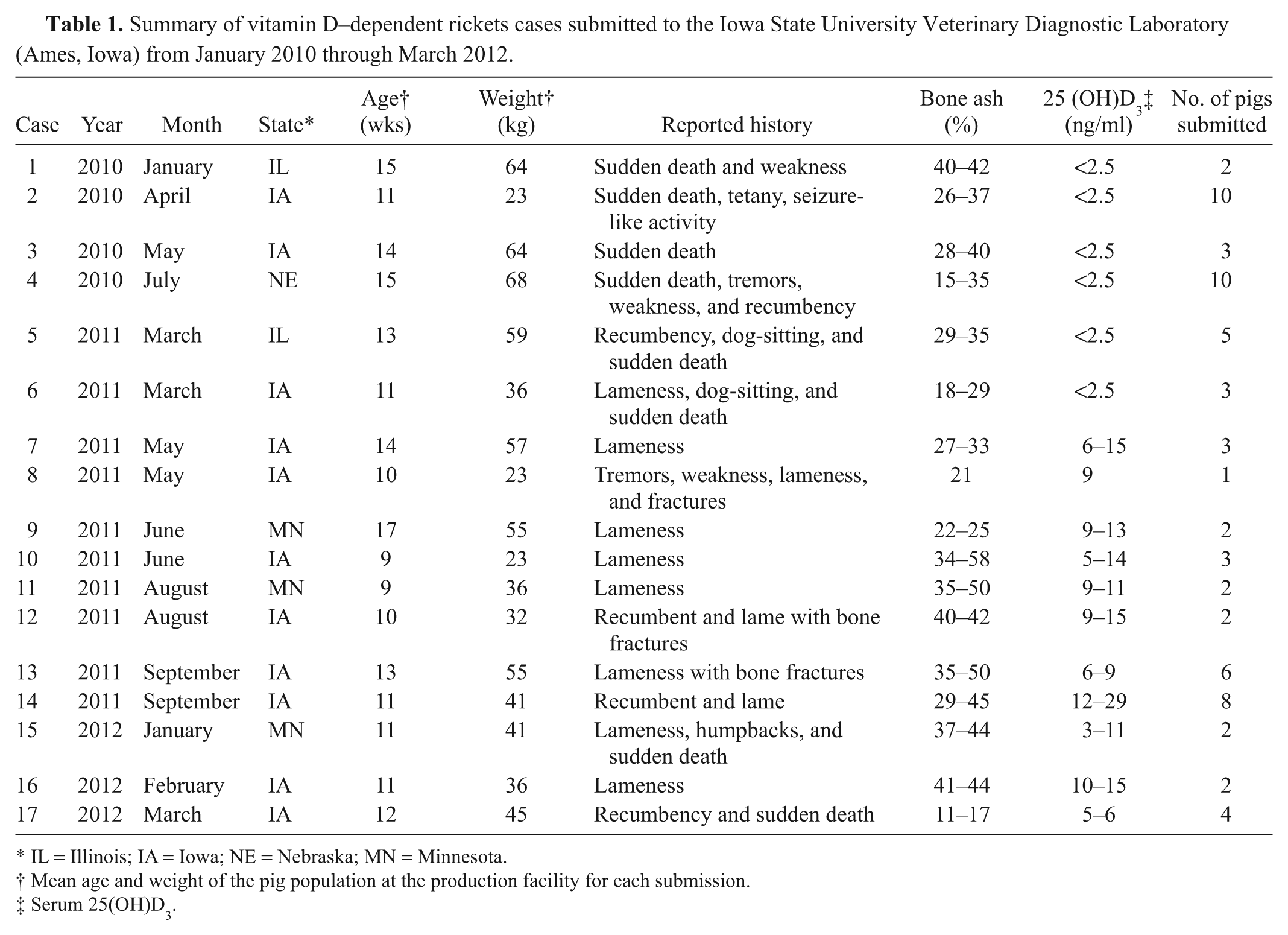
Lameness and bone fractures typically accompany rickets development in swine due to weakened bones (Fig. 1). Walking cautiously, displaying a hunch-backed posture, knuckling-over of forelimbs, and assuming a dog-sitting position are additional clinical presentations that have been previously described in swine. 28 Structural conformation abnormalities including bending, bowing, or diminished length of long bones can also be a feature. 28 Enlarged growth plates can give the appearance of swollen joints, although gross enlargement is not always visible. Lordosis, kyphosis, hirsutism, and decreased feed intake have also been reported in swine suffering from rickets.14,16
Pig; femur and tibia, case 6. There is a chronic diaphyseal fracture with callus formation in the femur (*) at left. Note misalignment of cortical bone. An acute proximal diaphyseal fracture (arrow) with adjacent soft tissue hemorrhage is present in the tibia.
Chronic vitamin D deficiency promotes utilization of body calcium reserves in bone as a compensatory reaction to maintain normal serum calcium. Serum calcium concentrations are finely regulated by the animal and therefore strictly maintained within a narrow range. However, depletion of body reserves can result in acute disruption of calcium homeostasis and hypocalcemia. Clinical signs of acute hypocalcemia in swine include paresis, tetany, muscle fasciculation, and seizures. 15 Hypocalcemia with sudden, unexpected death has been reported in swine fed diets deficient in vitamin D. 15 Hypocalcemia associated with sudden death was a conspicuous but unexpected clinical presentation in a portion of the cases described herein. This observation suggests that vitamin D–dependent rickets in swine has the potential to be associated with lethal hypocalcemia.
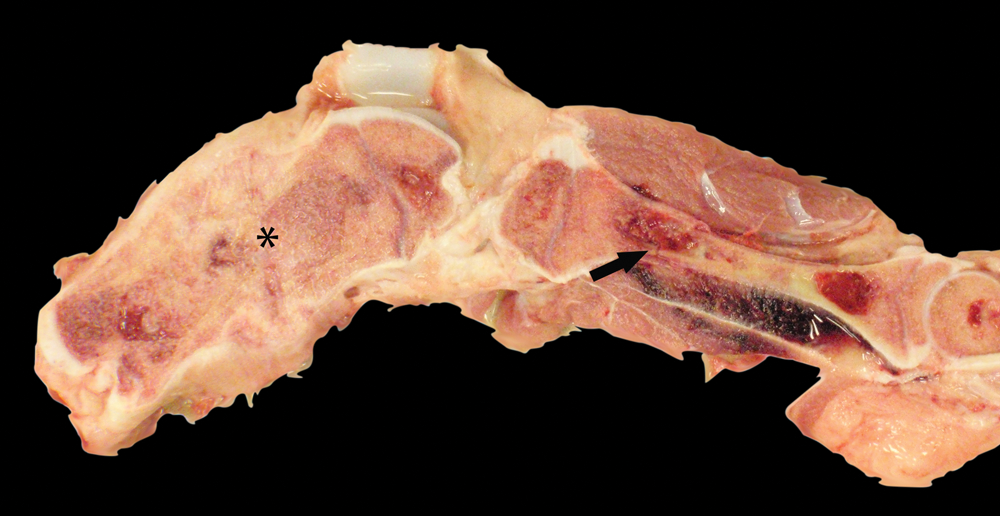
Gross lesions of rickets are highly variable, sometimes inconspicuous, and can take several months to develop, depending upon severity of deficiency. Within a larger population of animals, bone fractures (particularly of ribs) or rachitic rib lesions are frequently apparent. Rachitic lesions are more often visible on caudal ribs because of faster growth compared to cranial ribs (Fig. 2). 28 Widened growth plates can be visualized in multiple locations and are most likely to be associated with sites of rapid bone growth including the distal femur, proximal tibia, proximal humerus, and distal radius. 29 The absence of grossly widened physes does not exclude rickets from the differential list however. In the absence of overt gross lesions, the authors found that assessment of rib-breaking strength was beneficial in determining whether assays directed toward bone hardness needed to be included in the diagnostic workup of the lameness or sudden death presentation. Normal ribs snap distinctly when pressure is applied. Rachitic ribs bend prior to breaking with only a weak audible sound of bone crushing or crinkling.
Pig; rib, case 3. Distal rib is flared at the costochondral junction (rachitic rosary). Physeal cartilage is thickened and irregular (arrows).
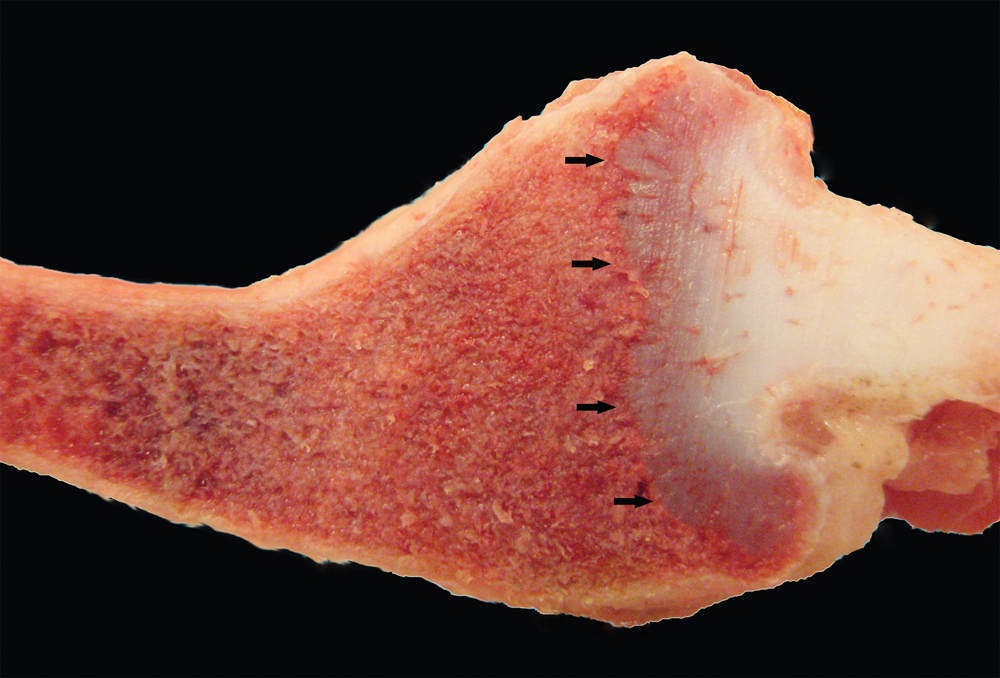
Microscopic lesions associated with rickets are most severe in fast-growing bones in the locations stated above, with the primary change being failure of endochondral ossification. The hypertrophic zone of chondrocytes is thickened and irregular with tongues that extend into the primary spongiosa. Similar lesions can also develop beneath articular cartilage (Fig. 3). 28 Trabeculae of the primary and secondary spongiosa are thinned and sometimes rimmed by unmineralized osteoid. Infractions and hemorrhage can occur due to weakened trabeculae, and fibrous osteodystrophy frequently develops because of decreased serum calcium. 28 Furthermore, large numbers of osteoclasts are commonly observed within Howship’s lacunae (Fig. 3). In the documented cases of the present study, the authors found costochondral junctions to more commonly have compatible microscopic lesions than other locations including the femur, tibia, humerus, radius, metacarpal, metatarsal, and vertebrae. Pituitary glands were not evaluated in the present case series, and parathyroid glands, when evaluated, had no discernible histopathological changes. Not all stated microscopic lesions will be present in every case, but in the authors’ experience, the irregularity of the hypertrophied chondrocyte zone is the most consistent finding.
Bone photomicrographs.
Diagnosis of rickets in swine is not problematic if disease is suspected clinically or following postmortem examination. It is challenging, however, if rickets is unintentionally excluded from a list of differential diagnoses. In many of recorded cases described herein, tremors and/or seizure-like muscle fasciculations and tetany were apparent and often interpreted as central nervous system disease by the attending veterinarian. A presumptive antemortem diagnosis of rickets can be achieved by measuring serum analytes or urine mineral content in conjunction with compatible clinical signs. Vitamin D–dependent rickets or calcium-deficient diets result in secondary nutritional hyperparathyroidism and hypocalcemia if the deficiency is protracted. 27 Serum phosphorus can often be elevated if hemolysis is present or if serum is not separated quickly. Phosphorus can also be elevated in young animals or if there is excessive break-down of bone occurring. 5 Normal to decreased, but not increased, serum phosphorus levels are present in phosphorus-deficient rickets. Serum ALP is elevated following bone reabsorption; however, cautious interpretation is warranted as animals can have elevated serum concentrations of ALP during periods of rapid growth. Elevation in serum CK has also been documented in cases of rickets because of prolonged recumbency or related to hypocalcemic myotonia. 15 Ultimately, in vitamin D–dependent rickets, serum 25(OH)D3 concentrations will be decreased. In phosphorus-dependent rickets, serum 25(OH)D3 will be normal to elevated with normal to decreased PTH levels. 24 Urine calcium-to-phosphorus ratios below 0.05 suggest a calcium or vitamin D deficiency whereas ratios above 1.0 correspond to phosphorus restriction. 8
Extremely low levels of 25(OH)D3 and/or 1,25(OH)D3 vitamin D in serum strengthen a clinical diagnosis of hypovitaminosis D; however, gross or microscopic bone lesions are needed to confirm a diagnosis of vitamin D–dependent rickets. Serum 25(OH)D3 and serum 1,25(OH)D3 are highly correlated in healthy subjects, yet, serum 25(OH)D3 is a more reliable measurement of vitamin D status and calcium absorption ability during deficient states.10,17 Serum levels of 25(OH)D3 are higher and the half-life is longer than serum 1,25(OH)D3. 17 The problem with measuring serum vitamin D in swine is that normal age-based reference intervals for serum 25(OH)D3 and 1,25(OH)D3 have not been well-defined. A 2009 study indicated that serum 25(OH)D3 levels in humans are similar to those in growing pigs. 11 Extrapolation would suggest that pigs can develop rickets if serum 25(OH)D3 is less than 15 ng/ml. 30
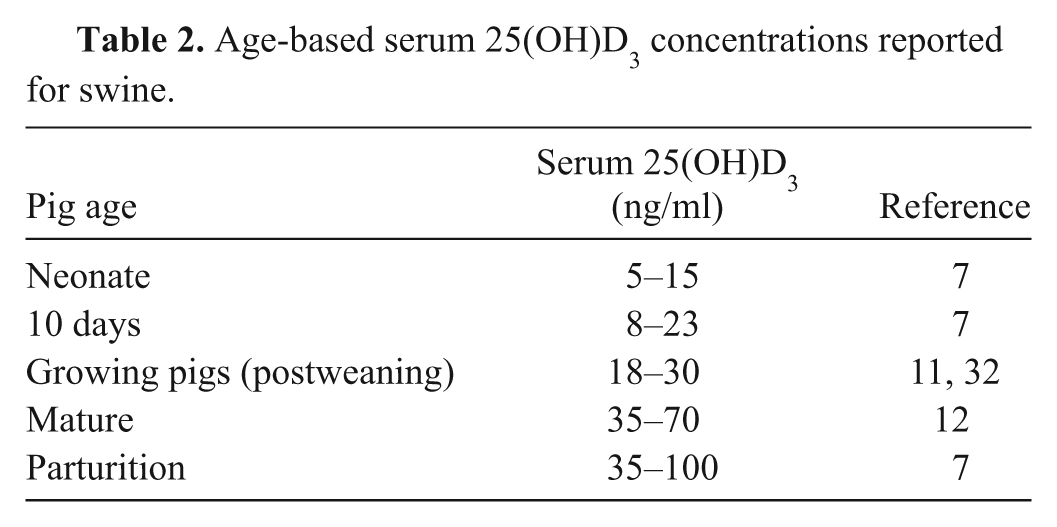
Table 2 is a summary of published data for age-based reference intervals of serum 25(OH)D3 in swine, and the following is a brief explanation for the varying levels based on age or stage of production. During gestation, dam serum vitamin D levels gradually increase leading up to parturition. Levels then begin to fall and return to baseline around 21 days of lactation. 33 Neonatal piglets are born with circulating serum 25(OH)D3; only a small fraction of the dam’s vitamin D crosses the placenta. 7 Piglets receive additional vitamin D via colostrum. Colostral vitamin D levels are approximately 20% of dam serum concentration. Piglet serum vitamin D can increase slightly following absorption from colostrum, but then begins to wane until a fortified diet or sunlight is provided. Apparently, vitamin D is not necessary for milk calcium absorption in suckling pigs, as receptor development has not yet occurred.14,25 By 4 weeks of age, pigs rely on vitamin D for intestinal calcium absorption. 14
Age-based serum 25(OH)D3 concentrations reported for swine.
Approximately 90% of dietary vitamin D is absorbed in the proximal small intestine, and diarrheic animals can have lower vitamin D levels due to malabsorption. 19 Because weaned pigs have marginal serum 25(OH)D3 levels at weaning, postweaning diarrhea may contribute to vitamin D deficiency and the development of metabolic issues.
The most common pathogenic mechanisms for development of rickets are: 1) inadequate dietary supplementation of vitamin D3, 2) inadequate absorption of phosphorus due to low available phosphorus in the diet; often the result of phosphorus bound to phytate and sequestered, or inadequate phytase usage, 3) imbalance of calcium-to-phosphorus ratio in feed (should be roughly 1.2:1), 3 and 4) inadequate dietary calcium, which can contribute to rachitic lesions but is often confounded by accompanying osteoporosis. When rickets or other metabolic bone diseases are demonstrated, dietary factors are implicated as causal irrespective of concurrent infectious disease pressures in the population. Nutritional investigation is warranted in bone disease scenarios, including dietary formulation inspection and/or feed testing. A caveat to feed testing is that feed associated with a clinical problem is seldom the feed that is on-site during an investigation. In one case included in the current series, feed delivered 3 weeks prior to clinical signs was the diet that was found to be deficient in vitamin D. Therefore, routinely saving a feed sample from each delivery is recommended.
Although a portion of the initial cases (cases 1–3; Table 1) were related to a premix production error by a single feed supplier, numerous vitamin D–dependent rickets cases were diagnosed thereafter from multiple locations throughout the Upper Midwestern United States with no common link to a particular feed manufacturer or premix supplier. The authors also highlight the fact that 67 additional and unrelated field cases highly suggestive of vitamin D–dependent rickets were submitted to the ISU-VDL during this timeframe. These other cases had microscopic lesions consistent with failure of endochondral ossification, but were lacking serum or feed samples to confirm inadequate vitamin D levels, and therefore were not included in this case series.
The current report summarizes clinical cases of vitamin D–dependent rickets within growing swine in the United States. The majority of the cases presented in the winter and spring months, perhaps because of shorter sunlight exposure, and some of these cases had confirmed dietary deficiency of vitamin D3. At this time, the authors are uncertain why rickets developed on farms with no demonstrated deficiencies of vitamin D in premix supplements. Investigation of speculative hypotheses has not elucidated root causes or consistent risk factors for the recent increase in frequency of metabolic bone disease diagnoses at the ISU-VDL.
Investigations of lameness or sudden, unexpected deaths in swine should include rickets and other metabolic disease conditions in the differential diagnosis. Assessing bone strength, bone mineralization, serum analyte concentrations, and bone histopathology are essential for definitively diagnosing metabolic bone disease conditions in swine.
Footnotes
a.
DeltaCAL, Delta Medical Inc., Aurora, IL.
b.
TaqMan NA and EU PRRSV, Applied Biosystems, Foster City, CA.
Declaration of conflicting interests
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
