Abstract
Pseudogout, also known as calcium pyrophosphate dihydrate (CPPD) deposition disease or chondrocalcinosis, is caused by crystalline deposits of CPPD within the extracellular matrix of articular hyaline cartilage and fibrocartilage, and within articular and periarticular connective tissue. Using a variety of laboratory techniques, we diagnosed pseudogout in the right hindlimb digit V of a 12-y-old Standard Poodle. Histologically, the joint, bone, tendon, and dermis were expanded and effaced by masses of mineralized, rhomboid crystals surrounded by macrophages, multinucleate giant cells, fibrous connective tissue, and chondroid and osseous matrix. Rhomboid crystals exhibiting weak-positive birefringence were identified under polarized light using a first-order red compensator filter. Scanning electron microscopy with energy-dispersive x-ray analysis (SEM-EDXA) revealed that the rhomboid crystals were composed of calcium, phosphorus, and oxygen. Fourier-transform infrared (FTIR) microspectroscopy confirmed the presence of calcium pyrophosphate. In dogs, tophaceous pseudogout, which was the variant of pseudogout in our case, occurs as a single, tumor-like periarticular mass that can be invasive and mimic neoplasia. Having ancillary confirmatory testing (SEM-EDXA and FTIR), particularly in unusual histologic scenarios, such as tophaceous pseudogout in dogs, is desirable for confirming the correct diagnosis, even though it is available only at certain reference centers. The pathogenesis of pseudogout is unknown.
Pseudogout, also known as calcium pyrophosphate dihydrate (CPPD) deposition disease or chondrocalcinosis, is caused by crystalline deposits of CPPD within the extracellular matrix of articular hyaline cartilage and fibrocartilage, and within articular and periarticular connective tissue. Tophaceous pseudogout forms a subset of cases in which there is “massive or tumoral CPPD crystal deposition.” 6 The diagnosis of pseudogout in dogs can be challenging given its rarity and its histologic resemblance to calcinosis circumscripta and sarcoma. Additionally, the literature on pseudogout in dogs can be confusing because the term “chondrocalcinosis,” which is synonymous with pseudo-gout, has been used to describe mineralization of cartilage that is not the result of deposition of CPPD (i.e., is not consistent with pseudogout). 12
We describe herein a classic case of pseudogout in a dog and elaborate on the laboratory techniques that can be used to confirm this diagnosis. Techniques that we used included light microscopy with and without polarizing filters and a first-order red compensator filter, scanning electron microscopy with energy-dispersive x-ray analysis (SEM-EDXA), and Fourier-transform infrared (FTIR) microspectroscopy. Our aim is to familiarize readers with this rare condition and to help pathologists and researchers diagnose pseudogout in dogs.
Digit V of the right hindlimb from a 12-y-old, male Standard Poodle contained a 2.5 × 2.5 × 0.5 cm mass that bulged from the lateral surface of the digit. A preliminary diagnosis of chondrosarcoma was made based on an incisional biopsy of the mass (the incisional biopsy sample was not available for our study). The digit was amputated and submitted whole for examination. Gross findings included white, chalky material associated with the second phalanx and distal interphalangeal joint of digit V (Fig. 1).
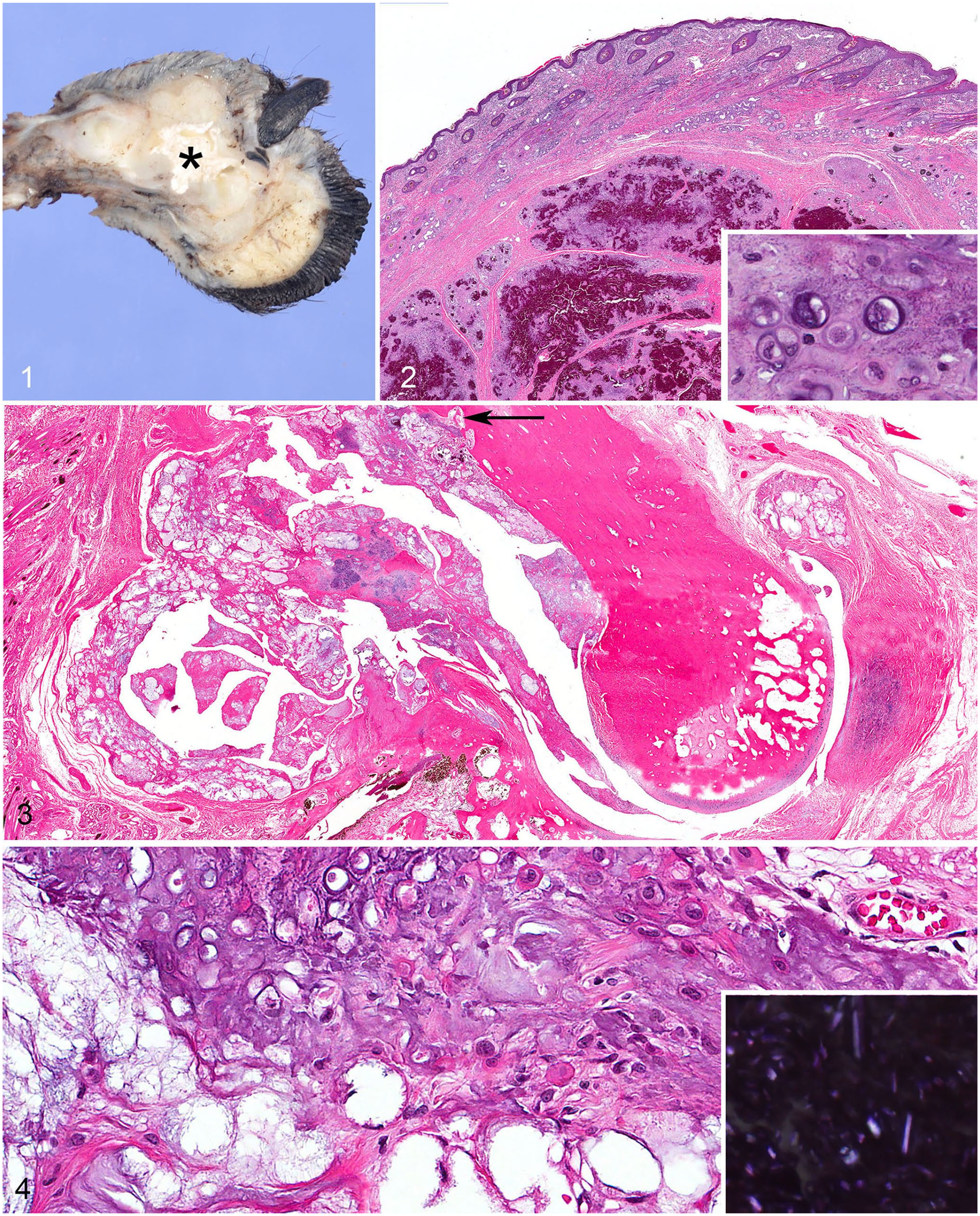
Pseudogout in the digit of a dog.
The digit was fixed for > 24 h in 10% neutral-buffered formalin. A section of soft tissue was removed from the digit prior to demineralizing the rest of the sample (HCl–formic acid in formalin; Poly Scientific) for 5 d. All tissues were processed routinely for light microscopy with paraffin embedding, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). Histologically, a multilobular, moderately cellular, and mildly invasive mass expanded the distal interphalangeal joint, partially encircled and invaded the phalanx, and extended to and expanded the superficial subcutis (Figs. 2, 3). This mass was well-defined and composed of chondroid matrix, epithelioid macrophages, and multinucleate giant cells surrounding pools of mineralized crystals in non-demineralized sections, or artifactual, poorly defined to rhomboid clefts in demineralized sections (Fig. 4). Components of the mass were variably separated by fibroplasia and/or dense fibrous connective tissue with rare osseous metaplasia. Within the chondroid matrix, there was a high density of mature chondrocytes within lacunae. The mass regionally invaded the second phalanx, partially effaced an extensor tendon, and moderately expanded the articular space.
Unstained coverslipped, and H&E-stained, sections examined with 10× and 20× objectives under polarized light microscopy (BX 53 microscope with maximum brightness of illumination [100 W bulb], polarizing filter [U-POT], analyzing filter with a first-order red compensator filter [U-GAN]; Olympus) revealed birefringent rhomboid crystals in both stained and unstained sections (Figs. 4, 5, respectively). Using a first-order red compensator filter (with the orientation indicator parallel to the long axis of the crystals), the crystals appeared blue, indicating weak-positive birefringence consistent with calcium pyrophosphate (Fig. 6). 2

Pseudogout in the digit of a dog.
A tissue section cut from the non-demineralized, formalin-fixed, paraffin-embedded (FFPE) mass was mounted on a polished carbon planchet for scanning electron microscopy with energy-dispersive x-ray analysis (SEM-EDXA; Hitachi S-3400N scanning electron microscope, Hitachi High Technologies; X-Max 80 energy dispersive x-ray spectrometer, Oxford Instruments). Backscatter images and EDXA spectra and maps were obtained at 20 kV and 10 kV accelerating voltages from several areas across the tissue section. SEM backscatter images revealed rhomboid crystals (Fig. 7). EDXA spectra and maps demonstrated the co-location of calcium (Ca), phosphorus (P), and oxygen (O) in the crystals, consistent with a calcium phosphate compound (Fig. 8). Areas of background tissue demonstrated peaks for carbon (C) and O only (Fig. 8). SEM-EDXA also demonstrated Ca-, P-, and O-containing material with a less discrete crystal morphology consistent with other forms of calcium phosphate.
A tissue section from the non-demineralized, FFPE mass was mounted on an aluminum-coated glass slide for infrared (IR) microspectroscopy. IR spectra were obtained (Nicolet 6700 FTIR spectrometer, Continuµm infrared microscope; Thermo Fisher Scientific). Infinity-corrected Reflachromat objectives (15× and 32×) were used to focus the infrared beam on crystals within the tissue section. The reflected IR spectra were collected using a mercury–cadmium–telluride (MCT) detector. Spectra of the specimens were ratioed to background spectra obtained from blank regions on the aluminum-coated slide, and apertures (20–50 nm) were used to direct the IR beam to the areas of interest. Spectra were recorded at 4 per cm spectral resolution, and 128 scans were co-added to reduce noise levels and provide adequate spectral quality. The IR spectra obtained were compared to commercial and internally obtained reference library spectra. The spectra of the rhomboid birefringent crystals were consistent with a mixture of calcium pyrophosphate and tissue protein (Fig. 9).
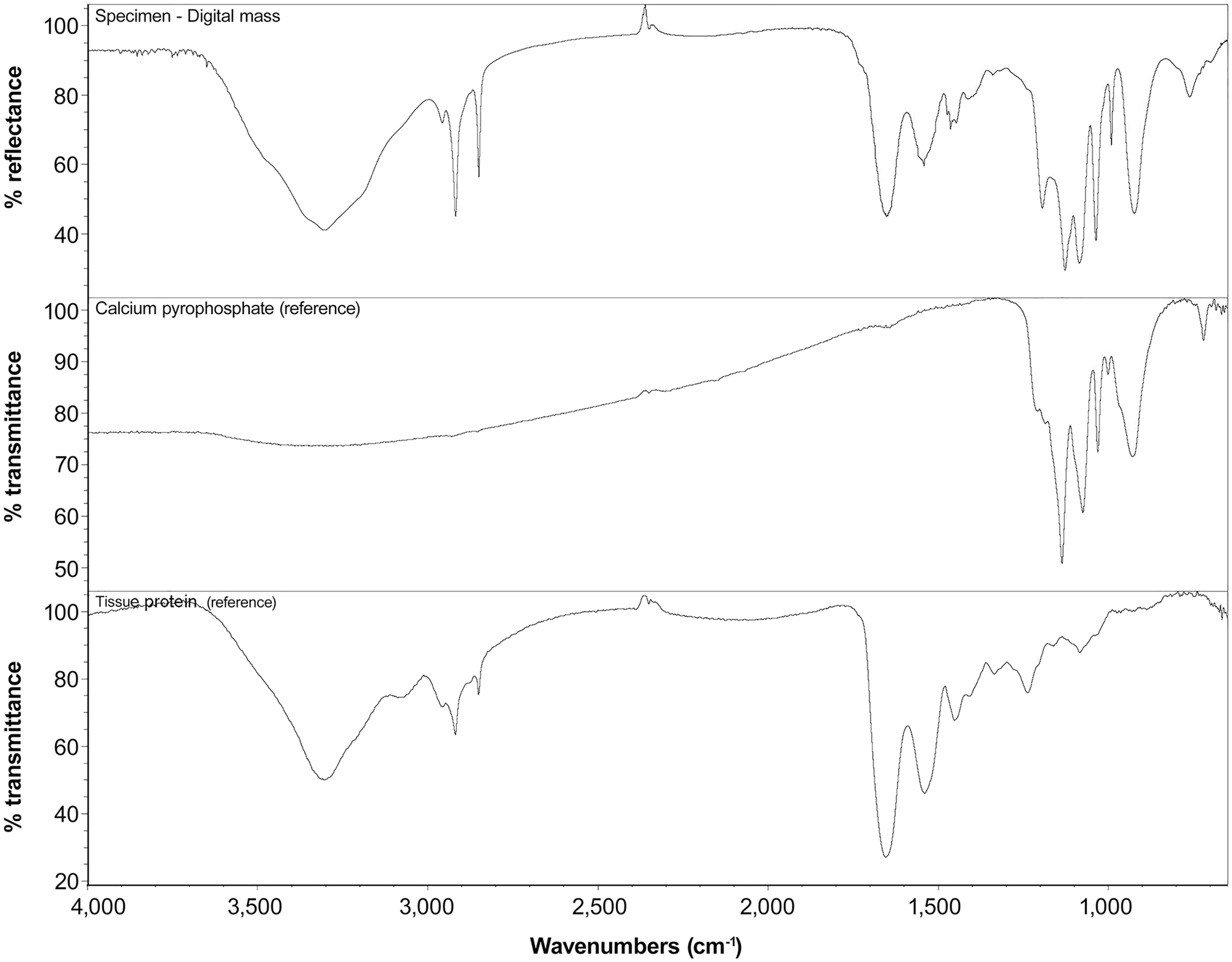
Fourier-transform infrared (FT-IR) microspectroscopy of the digital mass (top graph) is characteristic of a combination of calcium pyrophosphate on a background of tissue protein. Reference library spectra for calcium pyrophosphate (middle graph) and tissue protein (bottom graph) are shown. The combination of peaks from the 2 reference spectra closely mirrors the spectrum of the digital mass. Spectra of both the calcium pyrophosphate reference and the digital mass have similar peaks ranging from 1,300 to 800 wavenumbers, indicating the presence of calcium pyrophosphate within the mass. Spectra of both the tissue protein reference and digital mass have similar peaks ranging from 3,700 to 1,400 wavenumbers, indicating the background of tissue protein in the mass. FTIR.
Ours is a classic case of tophaceous pseudogout in a dog. Although the diagnosis of pseudogout may be straightforward in some cases, the tophaceous form of pseudogout (as reported here) often mimics a neoplasm, requiring additional testing to confirm the diagnosis. Confirmation of the diagnosis of pseudogout can be made several ways, including 1) polarized light microscopy with a first-order red compensator, 2) FTIR, and 3) SEM-EDXA.
A first-order red compensator enhances the optical path differences in a specimen. Using reference points indicated on the compensator and known molecular grains (i.e., direction of linear structure of molecules in the crystal), the color of birefringence will help determine the type of crystal. 5 To our knowledge, pseudogout has not been reported previously in a dog using these filters, perhaps because of limited access to these filters at veterinary pathology laboratories.
SEM-EDXA and FTIR are useful and sometimes necessary in confirming the diagnosis of pseudogout. Although compensated polarized light microscopy is useful in identifying CPPD crystals in optimal conditions, identification of crystals may be hampered by technical issues relating to the strength of polarized light, and is enhanced by examining unstained coverslipped sections rather than H&E-stained sections. 5 In addition, the experience and ability of the microscopist can impact the diagnosis, even in a motivated cohort of reviewers; in a previous study, CPPD crystals were correctly identified in 100% of joint fluid aspirate images by only 1 in 3. 1
SEM-EDXA is a highly sensitive tool that can be used to image and detect elements in tissue because elements have characteristic x-ray scatter patterns. Any detected elements can be mapped and compared to SEM images to determine the elemental composition and distribution within a field in the specimen. 9 SEM-EDXA is only available at some larger pathology centers and in research laboratories. Similar to our case, 2 previous reports of dogs with a presumptive malignant neoplasm or canine pseudogout were later confirmed to be tophaceous pseudogout using SEM-EDXA.4,10
FTIR spectroscopy is a useful ancillary technique to cytology and histology; it provides a “fingerprint” of molecular structure for the components within a sample, such as a FFPE block or cytologic preparation. During FTIR spectroscopy, the amount of IR light that is absorbed and transmitted through a sample produces a measurable spectrum unique to the molecular structure, analogous to the uniqueness of human fingerprints. The main limitations of FTIR in histologic specimens are spatial resolution (ideally analysis is performed on a material 30 × 30 µm or larger), the thickness of the material reducing transmission of IR light through the specimen, and the presence of background materials such as tissue protein. Similar to our case, FTIR was used in a previous report of a dog with a presumptive malignant neoplasm that was later confirmed to be tophaceous pseudogout using this diagnostic technique. 3
The diagnosis of pseudogout can be challenging. Two morphologic differentials for tophaceous pseudogout are sarcoma and calcinosis circumscripta.7,11 The combination of invasive behavior (tophaceous pseudogout frequently invades bone), spindle cells (fibroplasia), and chondroid-to-osseous matrix (hence the name “chondrocalcinosis”) can mimic chondrosarcoma or osteosarcoma. The presence of clear spaces (secondary to washout of crystals) combined with macrophages should assist the pathologist in differentiating chondrosarcoma or osteosarcoma from tophaceous pseudogout.
In non-demineralized sections, tophaceous pseudogout can look similar histologically to calcinosis circumscripta, particularly if a skin biopsy is obtained from around the joint. Both pseudogout and calcinosis circumscripta are characterized by granulomatous inflammation surrounding mineralized material. Evaluation of the mineralized material at higher magnification and/or with polarizing filters reveals that granulomatous inflammation surrounds rhomboid crystals in pseudogout, which is inconsistent with calcinosis circumscripta. 11
The mass in our case of pseudogout was in the distal and interphalangeal joints of digit V of the right pelvic limb. This is similar to a previously reported canine case in which pseudogout formed a mass in the metatarsophalangeal joint of digit V of the right hindlimb. 3 Two other reported cases of canine pseudogout formed masses in the stifle and the carpus.4,10 In humans and animals, most cases of pseudo-gout are idiopathic, as was our case. In humans, pseudogout is less commonly associated with previous joint trauma, multiple metabolic and endocrine disorders, and hereditary conditions such as familial chondrocalcinosis. 8 Having ancillary confirmatory testing (SEM-EDXA and FTIR), particularly in unusual histologic scenarios, such as tophaceous pseudogout in dogs, is desirable for confirming the correct diagnosis, even though it is available only at certain reference centers.
Footnotes
Acknowledgements
We thank John F. Fetsch, formerly of Joint Pathology Center, for reviewing this case. We also thank Clinton Coates of Darnall Medical Library for assistance with obtaining reference material.
Disclaimer
The views expressed in this abstract are those of the authors and do not reflect the official policy of the Department of the Army/Navy/Air Force, Department of Defense, or U.S. Government.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
