Abstract
A 6-mo-old female Chihuahua was presented with recurrent episodes of hypoglycemia and collapse. Physical examination revealed proportionate dwarfism, retained puppy hair coat, retained deciduous teeth, and open fontanelles. Routine blood tests revealed hypoglycemia, thrombocytosis, hypoproteinemia, and elevated alkaline phosphatase activity. The urinalysis, radiographs, and ultrasonographs were unremarkable. Endocrine testing revealed that insulin-like growth factor 1 was below the detection limit; concentrations of total thyroxine, baseline cortisol, and cortisol stimulated by tetracosactide acetate were within their reference intervals. The pituitary gland showed no organic abnormalities on magnetic resonance imaging. For definitive diagnosis, we conducted the stimulation test for growth hormone (GH) release and diagnosed isolated GH deficiency. Genetic investigation revealed that the present case had 4 point mutations in intronic regions and a 6-bp deletion in exon 5 of GH1. The bioinformatics tool PROVEAN algorithm predicted that the deletion in exon 5 could be deleterious to the function of GH1.
A 6-mo-old female Chihuahua weighing 880 g was referred to the Veterinary Medical Center of the University of Tokyo because of recurrent episodes of hypoglycemia and collapse. The patient collapsed once every ~ 3 d and recovered with 50% dextrose administered orally since 4 mo of age. The dog had no history of exposure to toxins, such as xylitol, ethanol, or salicylate, that could cause hypoglycemia. 6 Physical examination revealed proportionate dwarfism, retained puppy haircoat, retained deciduous teeth (Fig. 1B), and open fontanelles. A complete blood count revealed thrombocytosis (1,027 × 109/L, reference interval [RI]: 143–448 × 109/L). Serum biochemistry analysis indicated hypoglycemia (1.9 mmol/L, RI: 4.2–7.1 mmol/L) and hypoproteinemia (29 g/L, RI: 50–72 g/L). The urinalysis, radiography of the thorax and abdomen, and abdominal ultrasonography were unremarkable. Because juvenile-onset panhypopituitarism was suspected, we conducted endocrine tests. Endocrine testing revealed low insulin-like growth factor 1 (0 nmol/L, RI: 4–95 nmol/L; IDEXX) and normal concentrations of total thyroxine (17.5 nmol/L, RI: 16.7–37.3 nmol/L; Dri-Chem Immuno AU10V; Fujifilm).The adrenocorticotropic hormone stimulation test by tetracosactide acetate (Cortrosyn; Daiichi-Sankyo) revealed normal baseline concentrations of cortisol (54 nmol/L, RI: 27–165 nmol/L; Fujifilm) and post-stimulation cortisol (284 nmol/L).
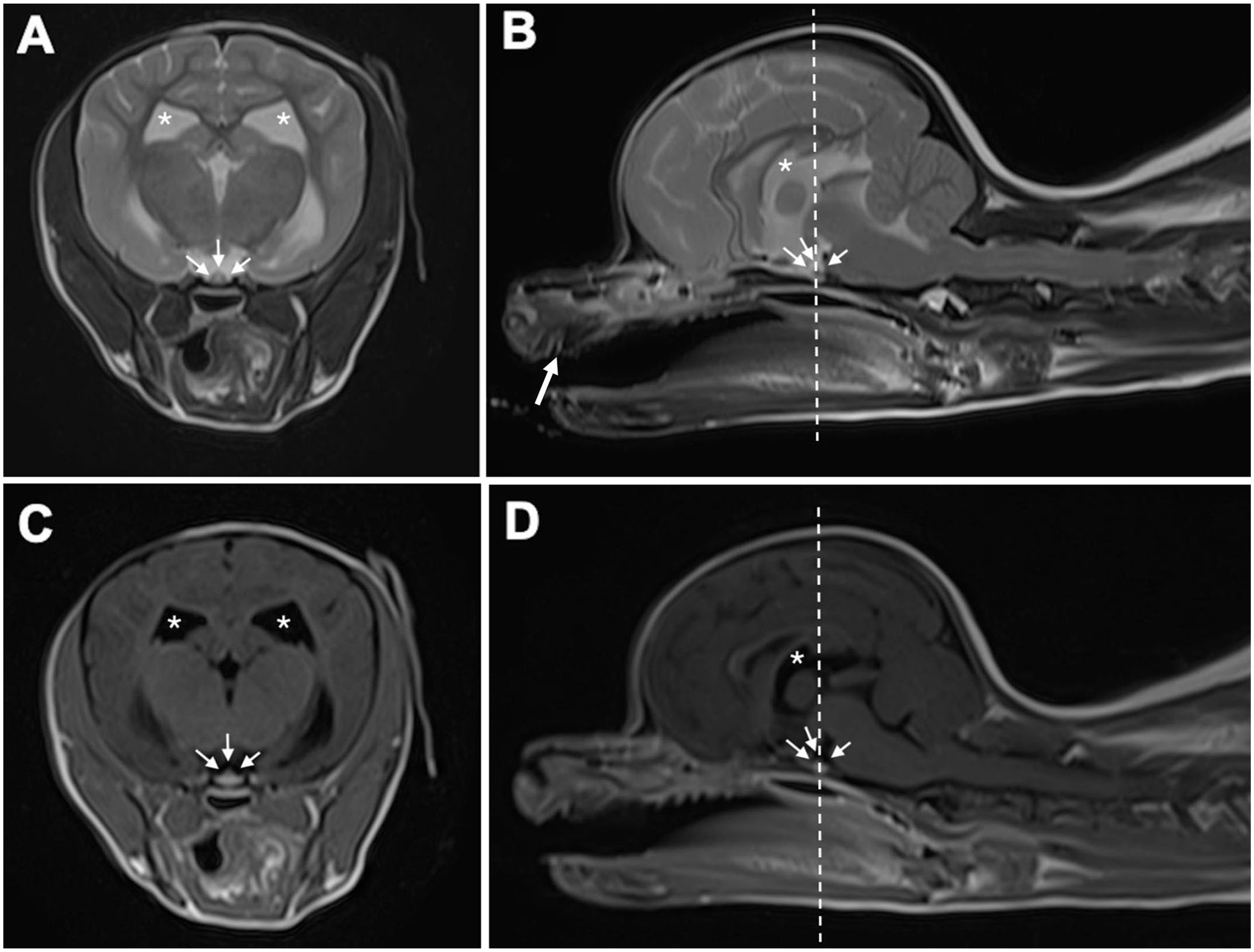
Magnetic resonance imaging of the brain of a dog with growth retardation. T2-weighted plane
For definitive diagnosis, we performed magnetic resonance imaging (MRI) and a growth hormone (GH) stimulation test. MRI of the head revealed no abnormalities in the intracranial regions, including the pituitary gland (Fig. 1). Plasma concentrations of GH were measured using a commercial canine GH ELISA kit (Cloud-Clone) before and 15, 30, and 60 min after an intravenous injection of xylazine (100 μg/kg). 5 The dog did not show the expected elevation in plasma concentrations of GH after stimulation of xylazine, but instead 0.162, 0.154, 0.164, and 0.144 μg/L at 0, 15, 30, and 60 min after xylazine injection, respectively. A previous study reported that baseline GH concentrations in 4 healthy Beagles were 0.83–3.6 μg/L, and 20 min after xylazine stimulation were 5.30–26.6 μg/L. 3 Isolated GH deficiency (IGHD) was hence diagnosed. The dog died suddenly during a hypoglycemic crisis before the initiation of GH replacement therapy using porcine GH. The owner did not consent to an autopsy; thus, no postmortem examination was performed.
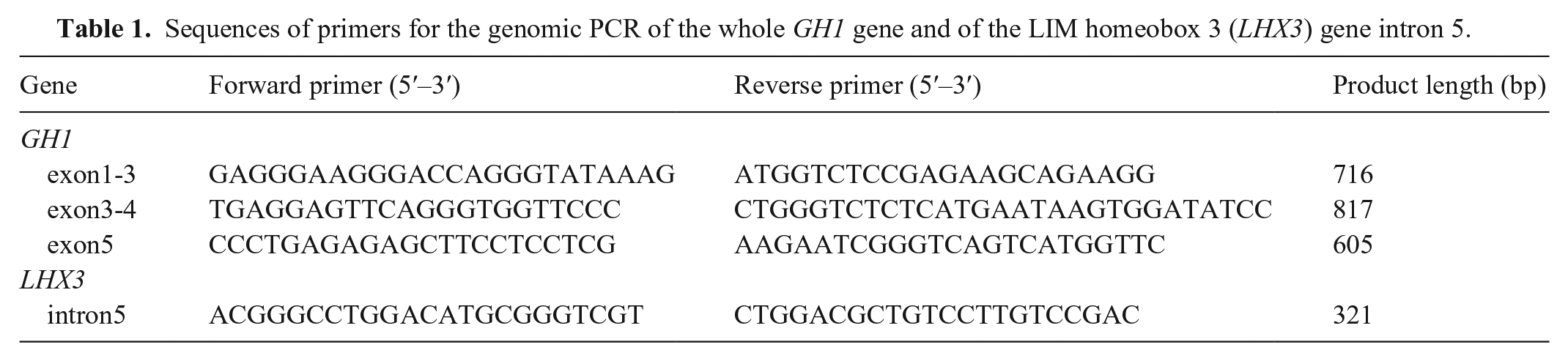
For sequencing analysis, genomic DNA was extracted from EDTA-stabilized peripheral blood samples using a commercial kit (DNeasy blood & tissue kit; Qiagen), following the manufacturer’s protocol. We analyzed the sequences of the whole GH1 gene (accession GCF_000002285.3) and LIM homeobox 3 (LHX3) gene intron 5 (Table 1). PCR was performed (KOD FX Neo polymerase; Toyobo); cycling conditions were 94°C for 2 min, 35 cycles of 98°C for 10 s, 60°C for 30 s, and 68°C for 45 s. PCR products were purified (Wizard SV gel and PCR clean-up system; Promega) and directly sequenced using Sanger sequencing (Genewiz Japan). With sequence alignment, we identified 4 point mutations (c.10+35T>C, c.11–99A>G, c.288+15T>C, and c.289–47_48del) in intronic regions of GH1 (Fig. 2). We found a homozygous in-frame 6-bp deletion (c.573_578del) that resulted in 1 amino acid substitution (K165N) and 2 amino acid deletions (K166del and D167del) in exon 5 of GH1 (Fig. 3). We evaluated the structural destruction of the GH1 protein for these mutations using bioinformatic tools (Protein variation effect analyzer, PROVEAN; http://provean.jcvi.org/seq_submit.php) 4 ; the PROVEAN algorithm predicted that amino acid variations would have a deleterious effect on canine GH protein function with a PROVEAN score of −2.989 for K165N, –12.036 for K166del, and −14.478 for D167del (cutoff = −2.5; scores < –2.5 indicate that amino acid substitution or deletion has a deleterious effect). Additionally, the amino acid sequence comparison between humans and dogs (aligned by ClustalW; https://www.genome.jp/tools-bin/clustalw) revealed that mutation sites K165 and K166 in canine GH corresponded to R167 and K168 in human GH, which have been reported as high-affinity binding sites for GH receptors.1,11 We did not detect a deletion in intron 5 of LHX3 in our case.
Sequences of primers for the genomic PCR of the whole GH1 gene and of the LIM homeobox 3 (LHX3) gene intron 5.
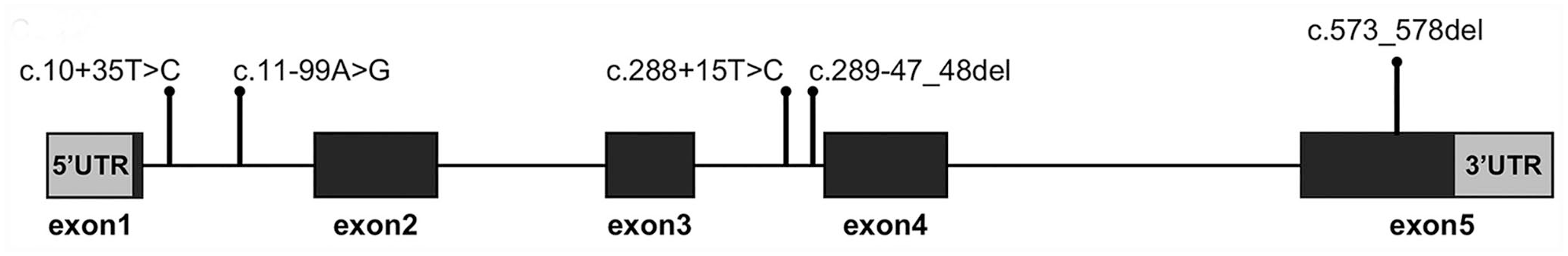
Sequencing analysis of GH1 in a dog with growth retardation. Lollipop plot showing the position and type of mutations in our case. Lollipops represent mutations, and boxes represent exons.
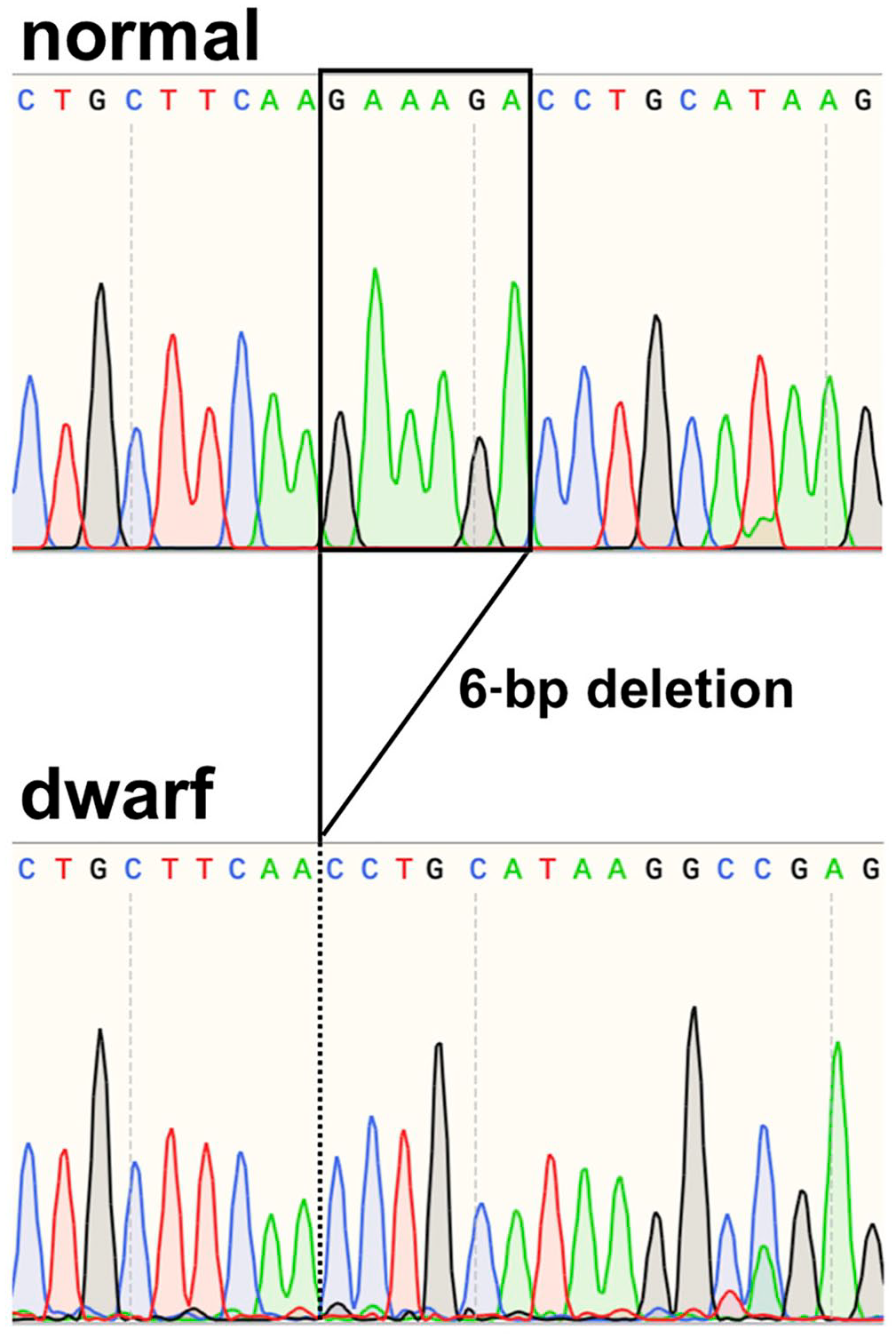
Sanger sequencing of exon 5 of GH1 in a healthy Chihuahua (top row) and our growth hormone–deficient case (bottom row).
To detect canine GH, we used the antibody against the amino acid sequence A51 to F216 of canine GH. Because the immunogen sequence included the mutation site (K165N, K166del, and D167del) and might not recognize mutated GH protein, we may not have measured the actual concentrations of the GH protein. Therefore, we could not conclude that the definitive reason for GH deficiency was a defect in the secretory function of the pituitary gland, a defect in bioactivity of the mutant GH protein, or both.
To our knowledge, isolated GH deficiency with GH1 mutations has not been reported previously in dogs. Congenital GH deficiency, which is relatively common in German Shepherd dogs (GSDs), Saarloos wolfdogs, and Czechoslovakian wolfdogs, has been associated with deletions in intron 5 of LHX3.10,14 GSD patients with GH deficiency have been reported to have combined deficiencies of GH, thyroid-stimulating hormone, and prolactin. 7 Additionally, the presence of pituitary cysts has been reported in GSDs with pituitary dwarfism.7,12 In our case, organic abnormalities were not found in the pituitary gland, and concentrations of serum cortisol and total thyroxine were normal. Furthermore, the deletion in intron 5 of LHX3, which has been reported in certain breeds,10,14 was not found in our case. In human medicine, the involvement of various defects in multiple genes has been reported in the etiology of GH deficiency (Table 2). 2 In veterinary medicine, POU1F1, 9 PROP1, 8 LHX4, 13 and LHX 14 in GSDs, and LHX3 in Saarloos wolfdogs and Czechoslovakian wolfdogs, 10 have only been investigated. In our case, gene sequence analysis revealed that isolated GH deficiency can be caused by mutations in GH1 in the Chihuahua. Our data indicate that the GH1 mutation should be added to the differential diagnosis of canine GH deficiency.
Genes involved in human growth hormone deficiency.

Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by the Grant-in-Aid for Scientific Research from JSPS (KAKENHI) grants 18H02340, 19H00968, and 20K15675.
