Abstract
Pasteurella multocida is the causative agent of fowl cholera, an economically important disease of commercial and backyard poultry. Turkeys are particularly susceptible to fowl cholera; both backyard and commercial turkeys can succumb to disease. On April 10, 2018, a dead 9-mo-old male Bronze turkey was submitted to the California Animal Health and Food Safety Laboratory System (CAHFS)–Turlock branch for postmortem examination. History included previous housing and fighting with another male turkey, after which separation by a fence was instituted. Fighting continued, and depression and anorexia of 2 d duration was followed by acute collapse and death. At autopsy, blood clots markedly expanded the pericardium, and a tear was visible in the left ventricular free wall. Vegetative aortic valvular lesions were observed. Microscopically, infarcts were observed in kidney, liver, heart, spleen, and pancreas, with large numbers of gram-negative bacterial colonies present in most organs. P. multocida was isolated from multiple organs, and identified as serotype 2,5 and fingerprint 1604. Septic embolization from the vegetative valvular aortic lesions likely led to infarcts in multiple organs, including the left ventricular free wall, which ultimately caused weakening of the ventricular wall, ventricular rupture, and exsanguination into the pericardial space. Rupture of the left ventricular free wall has not been previously documented in turkeys with P. multocida infection, to our knowledge, and demonstrates an atypical presentation of fowl cholera in this backyard turkey.
Fowl cholera is caused by the gram-negative non-motile bacterium Pasteurella multocida. P. multocida subspecies multocida is the main cause of fowl cholera, although occasional outbreaks are caused by subspecies septica or gallicida. Fowl cholera occurs in most avian species and is contagious; turkeys are very susceptible, with infections frequently resulting in high mortality in a flock. 7 Fowl cholera is frequently seen as an acute septicemic disease with high mortality and gross anatomic lesions resulting from vascular disturbances. Other domesticated farm animals are carriers and can shed P. multocida; strains vary in virulence for avian species. 10 Once infection occurs, birds may succumb in the acute septicemic phase, may survive the acute phase and succumb in the days following, may become chronically infected, or may ultimately recover. 7
P. multocida can enter the host through the mucous membranes of the upper respiratory tract, conjunctiva, and cutaneous wounds. 7 Stressors, such as changes in environment, nutrition, and injury, can affect the course of the disease. Chronically infected carrier and convalescent birds are a major source of infection because carrier birds shed the bacteria for life. 7 Chronic conditions can occur in which localized infections predominate.7,9,14 P. multocida can survive in the environment for up to 2 wk and in dried blood for up to 8 d. 7 If separated from organic material, P. multocida is destroyed easily by common disinfectants, sunlight, drying, and at temperatures >56°C. 7
Several typing methods exist for differentiation of P. multocida strains. Restriction enzyme analysis (REA) is used frequently to determine genotype, and agar gel immunodiffusion (AGID) is used to determine serotype. P. multocida isolates can be differentiated into 5 capsular serogroups (A, B, D–F), and 16 somatic serotypes (1–16). 7 Serotypes are known to predominate within geographic areas, with serotypes 1,3 and 3,4 commonly isolated from California poultry and wildlife.4,15,18 We describe herein a case of fowl cholera in a 9-mo-old backyard turkey (Meleagris gallopavo) with infarcts in parenchymatous organs, minimal lung lesions, vegetative valvular endocarditis, and acute rupture of the left ventricular free wall.
A 9-mo-old male Bronze turkey died on April 7, 2018, and was presented to the California Animal Health and Food Safety Laboratory System (CAHFS)–Turlock branch, on April 10 for postmortem examination. The turkey had been rescued 1 mo previously, and resided at an animal sanctuary with one other adult male turkey. The 2 turkeys were housed together in an outdoor paddock with access to shelter and perches. They were separated into neighboring paddocks after aggressive behavior caused injury to the submitted turkey’s head, snood, and feet; however, fighting continued through the fence. Two days before death, the subject was depressed and anorexic, and received 15 mg of meloxicam (Mobic, Cipla, Miami, FL) orally as recommended by the veterinarian. The submitter reported continued depression and anorexia, with acute collapse and death. Many species of domesticated animals reside at the sanctuary, and the subject had close contact with other chickens, ducks, geese, and turkeys. Information on diet and management was not available.
The turkey was moderately autolyzed and in good body condition based on skeletal muscle evaluation, with scabs present along the beak margins and snood. The pericardium was severely distended with clotted blood and contained a few foci of pericardial emphysema (Fig. 1A). Upon incision of the pericardium, a 1-cm long tear in the free wall of the left ventricle was noted midway between the base and apex of the heart. Clotted blood adhered to the torn myocardial surface and extended into the left ventricular chamber. The edges of the rupture appeared pale in contrast to the surrounding myocardium (Fig. 1B). Upon opening the left atrium, tan, firm, vegetative lesions were observed thickening the aortic semilunar valves (Fig. 1C). The liver was moderately enlarged and congested (Fig. 1A). The kidneys were dark green (postmortem artifact), with 4–8 mm diameter, multifocal depressed gray areas. Clotted blood was present in the abdominal air sacs and renal capsule. The lungs were dark green (postmortem artifact), with no exudate observed on the serosal surface or cut section. The crop was distended by gas and contained minimal feed; the gizzard contained a mixture of pelleted feed and grass.
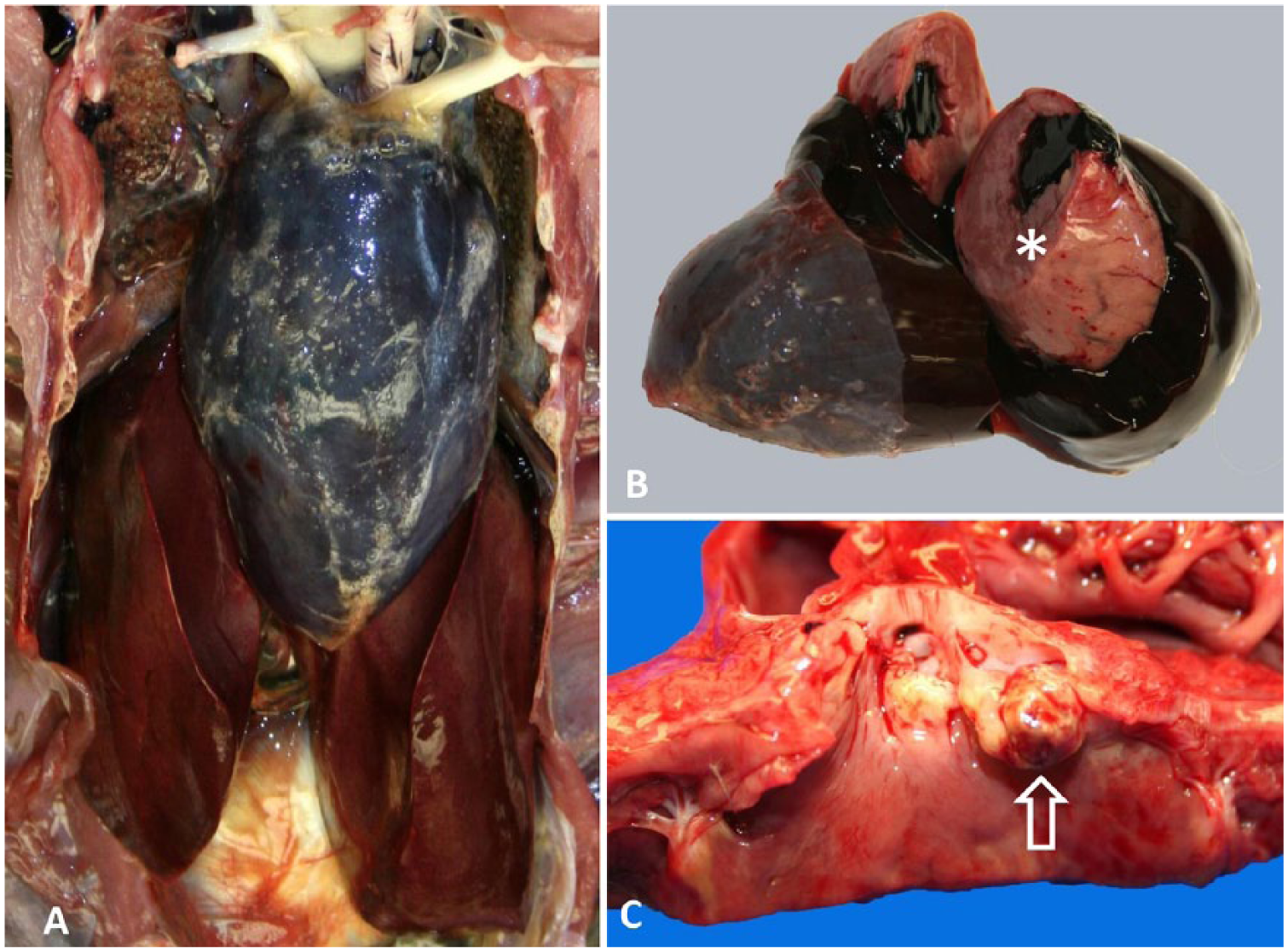
Macroscopic lesions in a 9-mo-old backyard turkey with fowl cholera and acute cardiac rupture.
Mild multifocal nongranulocytic perivascular cuffing and meningitis were present in brain sections. Avian paramyxovirus 1 immunohistochemistry on brain sections was negative. The kidney contained areas of ischemic necrosis bordered by mixed inflammatory cell infiltrates. Large numbers of gram-negative bacterial colonies were visible in renal parenchyma and thrombosed blood vessels. Glomerular arterioles were thrombosed, and tubules were infiltrated by granulocytes (Fig. 2A). The lung contained emboli of gram-negative bacteria associated with vascular thrombosis.
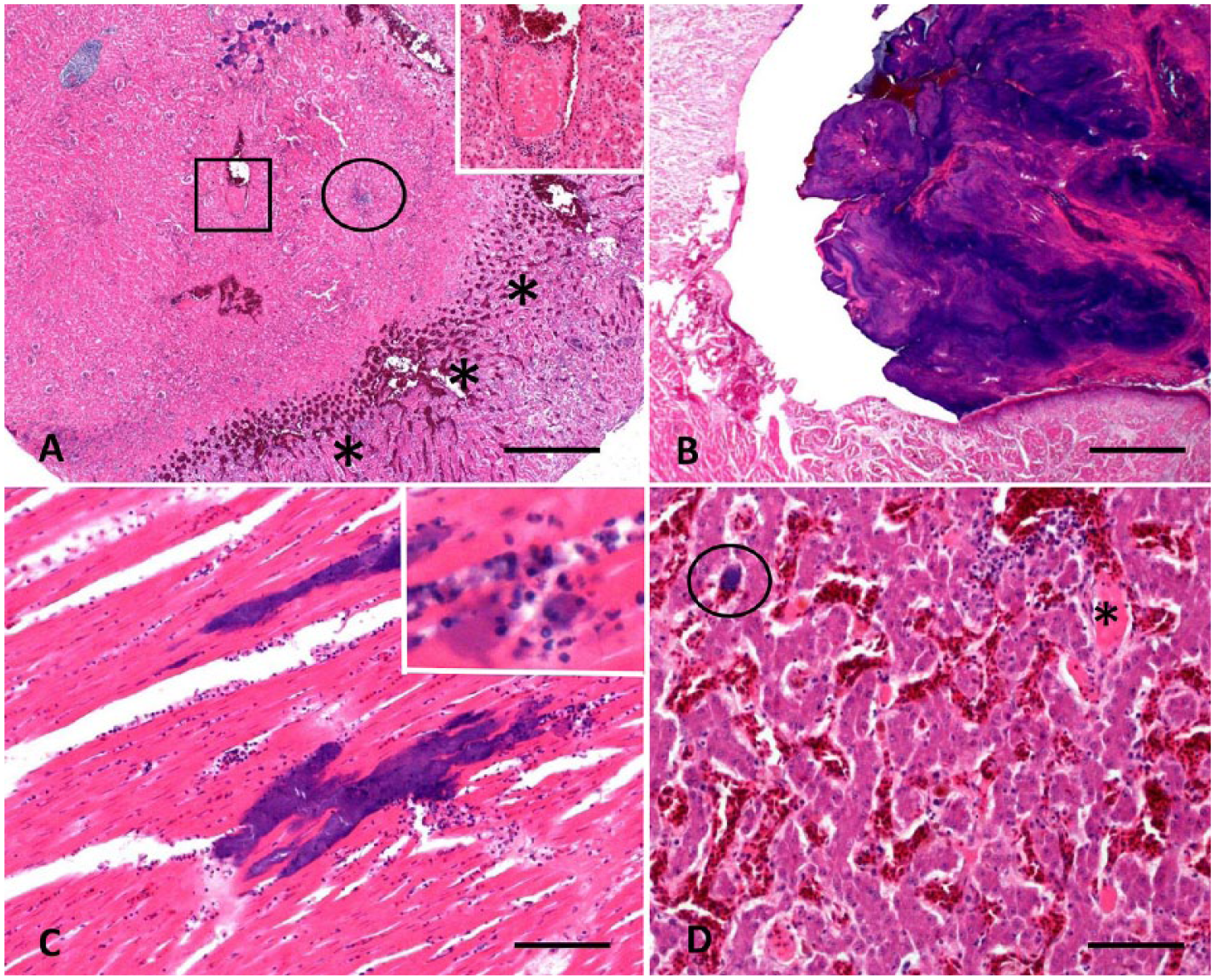
Histologic lesions in a 9-mo-old backyard turkey with fowl cholera and acute cardiac rupture.
The aortic valve was expanded by a polypoid mass composed of alternating layers of gram-negative bacterial colonies and fibrin, admixed with degenerate heterophils. The valve was moderately edematous with mixed inflammatory cell infiltration and fibrin; mineralization was present along the mass edge (Fig. 2B). The left ventricular free wall contained scattered foci of acute necrosis throughout the myocardium and bordering the rupture, characterized by fragmented and swollen cardiomyocytes with reduced striation. Abundant gram-negative bacterial colonies were observed in the myocardium and within thrombosed vessels (Fig. 2C).
Foci of necrotic and vacuolated hepatocytes were observed in the liver, with fibrin thrombi in sinusoids and vessels. Abundant gram-negative bacterial colonies were observed in the hepatic parenchyma and sinusoids (Fig. 2D). The spleen had fibrinoid necrosis of arteriolar walls, mineralization, moderate lymphocyte depletion, and moderate numbers of gram-negative bacterial colonies in the parenchyma. No bacterial colonies were observed in the pancreas; however, fibrinoid necrosis and thrombosis of blood vessels were observed. The gross anatomic lesions observed and histologic findings led to a presumptive diagnosis of bacterial septicemia with multiple infarcts as well as rupture of the left ventricular wall. Bacteriologic examination followed.
Lung, liver, kidney, heart, and aortic valves were cultured on 5% sheep blood agar and MacConkey agar at 37°C and 7% CO2 for 48 h. After 48 h, bacterial colonies compatible with P. multocida were confirmed by phenotypic and biochemical characteristics. 3 The P. multocida isolate was sent to the National Veterinary Services Laboratories (NVSL; Ames, IA) for AGID testing and DNA fingerprinting by REA. AGID was performed according to the World Organization for Animal Health (OIE) protocols (Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2017, http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.03.09_FOWL_CHOLERA.pdf). REA was performed as described previously. 21 Serotype 2,5 and fingerprint 1604 were identified.
According to the CAHFS protocol for avian influenza surveillance, an oropharyngeal swab was collected; the sample tested negative for the presence of influenza A virus by reverse-transcription real-time PCR (RT-rtPCR; National Animal Health Laboratory Network protocol, CAHFS, Davis laboratory). Given a history of heavy metal toxicosis in previous cases submitted from this premises, the liver was analyzed for lead, manganese, cadmium, iron, copper, molybdenum, zinc, mercury, and arsenic concentrations by inductively coupled plasma–optical emission spectrometry. 6 Results were within normal limits. The final diagnoses were acute pasteurellosis and left ventricular rupture with cardiac tamponade.
Fowl cholera infection in turkeys can cause both acute and chronic disease. Multiple factors are involved in determining the course of disease, including the virulence of the particular strain of P. multocida, immune status, and environmental factors.2,4,8,11 Infection typically occurs in turkeys 3 wk of age and older as a result of environmental exposure from many potential sources.2,7 The virulence of a particular strain of P. multocida varies greatly depending upon several factors, including the presence of a capsule, endotoxin production, outer membrane proteins, heat shock proteins, and antibody cleaving enzymes.4,8,11,13,19
Various stressors to the bird such as a change in environment, change in feed, and injury can influence the course of disease. The production of corticosterone in stressed avian species is a known immunosuppressant, causing suppression of both humoral and cellular immune responses. 5 The turkey in our report had been moved to the animal sanctuary with another male turkey 2 wk prior to death, and the change in environment, change in feed, stress, and injuries caused by fighting were likely important factors causing immunosuppression. Our turkey had exposure to other poultry, waterfowl, swine, wild birds, and rodents while at the sanctuary, which may have increased the exposure of our turkey to P. multocida. Farm animals are known carriers of P. multocida; however, most species are avirulent for poultry. 7 Swine are a known exception, with several studies demonstrating virulence for poultry of P. multocida isolates from swine. 10 Wild birds and rats may also carry P. multocida and serve as a potential source of infection. 7
Acute infections are characterized by septicemia, hyperemia, congestion, hemorrhage, and necrosis of abdominal organs. 7 Infected turkeys frequently suffer from acute infections and have pneumonia and lung consolidation. 9 When a virulent strain is encountered, septicemia can occur in as few as 6–12 h post-infection. 14 Chronic infections are more common in chickens, and are characterized by granulocytic lesions typically involving cranial bones, respiratory tract, hock joints, foot pads, coelomic cavity, and oviduct. 7
Our turkey had gross anatomic lesions indicative of acute septicemia, with an enlarged liver and pale foci in the myocardium and kidneys. Microscopically, infarcts were observed in myocardium, liver, kidney, spleen, and pancreas. The extent of bacterial colony proliferation observed was likely impacted by postmortem proliferation, given that the carcass was held for 3 d prior to submission. The immediate cause of death was rupture of the left ventricular free wall and exsanguination into the pericardial space with cardiac tamponade. Septic embolization from the vegetative aortic valvular lesions led to ischemic necrosis, a weakened myocardium, and ultimately myocardial rupture. Although cases of left ventricular free wall rupture in avian species have not been documented previously, to our knowledge, multiple cases in the human medical literature describe rupture of the left and right ventricular free walls, as well as ventricular septal rupture, as complications of myocardial infarction.20,22
Turkeys can be afflicted by several cardiovascular diseases including spontaneous aortic rupture; however, such diseases are more common in commercial rather than backyard turkeys given the differences in management and environment. Spontaneous aortic rupture occurs typically in 12–16-wk-old heavy and fast-growing male turkeys. High blood pressure, genetics, arteriosclerosis, diets including sweet peas (Lathyrus spp.), intensive management, and low levels of copper are possible etiologies. 16 Aortic or coronary artery rupture was ruled out in the backyard turkey in our report given the absence of lesions in the heart and vessel walls. The clotted blood in the renal capsule and abdominal air sacs in our turkey may have been the result of traumatic injury from fighting prior to death, congested vessels rupturing postmortem because of weakening of the vascular walls from infarcts, or passive congestion causing leakage of blood into the coelomic cavity.
Although serotype 2,5 was identified by AGID, AGID serotyping has issues with cross-reactions, reproducibility, and reliability.1,7 Serotyping and genotyping results are not correlated, further highlighting the limitations of this test. 17 Serotypes 1,3 and 3,4 are commonly identified in California P. multocida isolates, with strain recurrence on a premises frequently occurring in outbreak situations.12,17 Fingerprint 1604 was identified by REA, which offers higher specificity than serotyping.1,7 NVSL currently offers only genotyping by REA and serotyping by AGID for P. multocida strain differentiation. 1
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
