Abstract
Three Florida pumas (Puma concolor coryi) that had spent time in captivity prior to being released in the wild were found exhibiting respiratory signs and reluctance to move. All 3 pumas died shortly after immobilization, despite supportive veterinary care. Significant autopsy findings included necrotizing interstitial pneumonia, with pulmonary edema and hyaline membranes, and suppurative myocarditis. Organisms morphologically consistent with Bartonella henselae were identified in intravascular histiocytes in the lung of one of the pumas on histopathology and confirmed via transmission electron microscopy. B. henselae was detected in fresh lung tissue and confirmed by PCR and sequence analysis (16S-23S spacer region, pap31, and rpoB genes) from one of the affected pumas. In all affected pumas, B. henselae was detected by PCR in formalin-fixed, paraffin-embedded lung tissue, and positively staining organisms were identified in sections of lung by immunohistochemistry for B. henselae. In situ hybridization detected B. henselae DNA in lung tissue from 2 of 3 affected pumas. Our case series suggests that B. henselae can be associated with a fatal disease syndrome in Florida pumas. The cause of susceptibility to fatal disease associated with B. henselae infection in these pumas remains unknown.
Keywords
Florida pumas (Puma concolor coryi) are an intensively managed, endangered subspecies of puma in Florida. Low genetic diversity is a threat to the long-term survival of this population, and inbreeding, prior to a genetic introgression in 1995, resulted in a high incidence of genetic defects including sperm malformations, cryptorchidism, and atrial septal defects. 12
Bartonella are a diverse genus of fastidious, gram-negative, facultative intracellular bacteria that includes several important zoonotic species. The transmission cycles of many bartonellae are unknown or poorly understood, but it is thought that the majority are transmitted by insect vectors. 1 Bartonella henselae is the causative agent of “cat scratch disease,” which usually causes self-limiting lymphadenopathy in humans, but can cause more severe disease manifestations in immunocompromised patients. 3
Domestic cats have long been implicated as the reservoir host of B. henselae, with transmission occurring predominantly via the cat flea, Ctenocephalides felis.1,2 Antibodies to Bartonella spp. are commonly detected in domestic cats worldwide, suggesting that this pathogen is endemic in most domestic cat populations; however, antibody prevalence varies with geographic location. 11 Often assumed to be subclinical, B. henselae infection is being increasingly recognized in association with a variety of clinicopathologic abnormalities including anemia, febrile illness, neurologic dysfunction, endocarditis, and pyogranulomatous myocarditis and diaphragmatic myositis in domestic cats.2,23
A relatively high prevalence of antibodies to Bartonella spp. has been reported in both free-ranging and captive wild felids in North America, with a range of 16–53%, depending on the species.20,24 Such findings suggest that non-domestic felids may also serve as natural reservoirs of Bartonella species, including B. henselae.
Although exposure to Bartonella spp. is common in free-ranging pumas and other wild felids in the United States, cases of clinical disease in wild felids associated with this pathogen are not reported in the literature, to our knowledge. We describe herein a syndrome of necrotizing interstitial pneumonia and suppurative myocarditis in 3 Florida pumas associated with B. henselae.
Case 1 was a 2.5-y-old, male, free-ranging, radio-collared Florida puma that was located following the detection of a mortality signal from the radio collar. This puma had been orphaned at 5-mo-old, raised in captivity in Yulee, FL and released into the wild in southern Florida 8 mo prior to death. Two additional Florida pumas (cases 2 and 3) with similar autopsy findings (described below) were identified in a review of historical causes of death in Florida pumas. 4 Both were 6-y-old males that had been raised together in captivity in the same facility as case 1, fitted with radio collars, and released in southern Florida 4 and 6 wk prior to death, respectively. Based on radiotelemetry data collected prior to death, all cases exhibited decreased movement in the days prior to death. When located, all 3 pumas were found alive and exhibited reluctance to move and some degree of respiratory distress or neurologic abnormalities. All 3 animals were chemically immobilized in the field for transport to a veterinary referral center. One of the pumas died shortly following immobilization, the other 2 died within 24 h of receiving veterinary care.
Autopsy findings common to all 3 affected pumas included diffuse tracheal froth and hemorrhage, thoracic effusion, diffuse dark red-to-purple discoloration of the lungs (Fig. 1), pulmonary edema, and multiple superficial skin lacerations or bite wounds. Other gross findings included poor nutritional condition (2 of 3), lymphadenomegaly (2 of 3), mild gastric or duodenal hemorrhage (2 of 3), bilateral thyroid cysts (2 of 3), hepatomegaly (1 of 3), tan hepatic foci (1 of 3), and mucopurulent nasal discharge (1 of 3). All 3 pumas had at least 1 of the congenital defects that have been previously described in the Florida puma population, including atrial septal defect (case 1), cryptorchidism (case 2), kinked tail (case 2 and 3), and a dorsal hair whorl or “cowlick” (case 2 and 3). Representative tissues were sampled and saved frozen or fixed in 10% neutral-buffered formalin and routinely processed for microscopic slides and stained with hematoxylin and eosin (H&E).
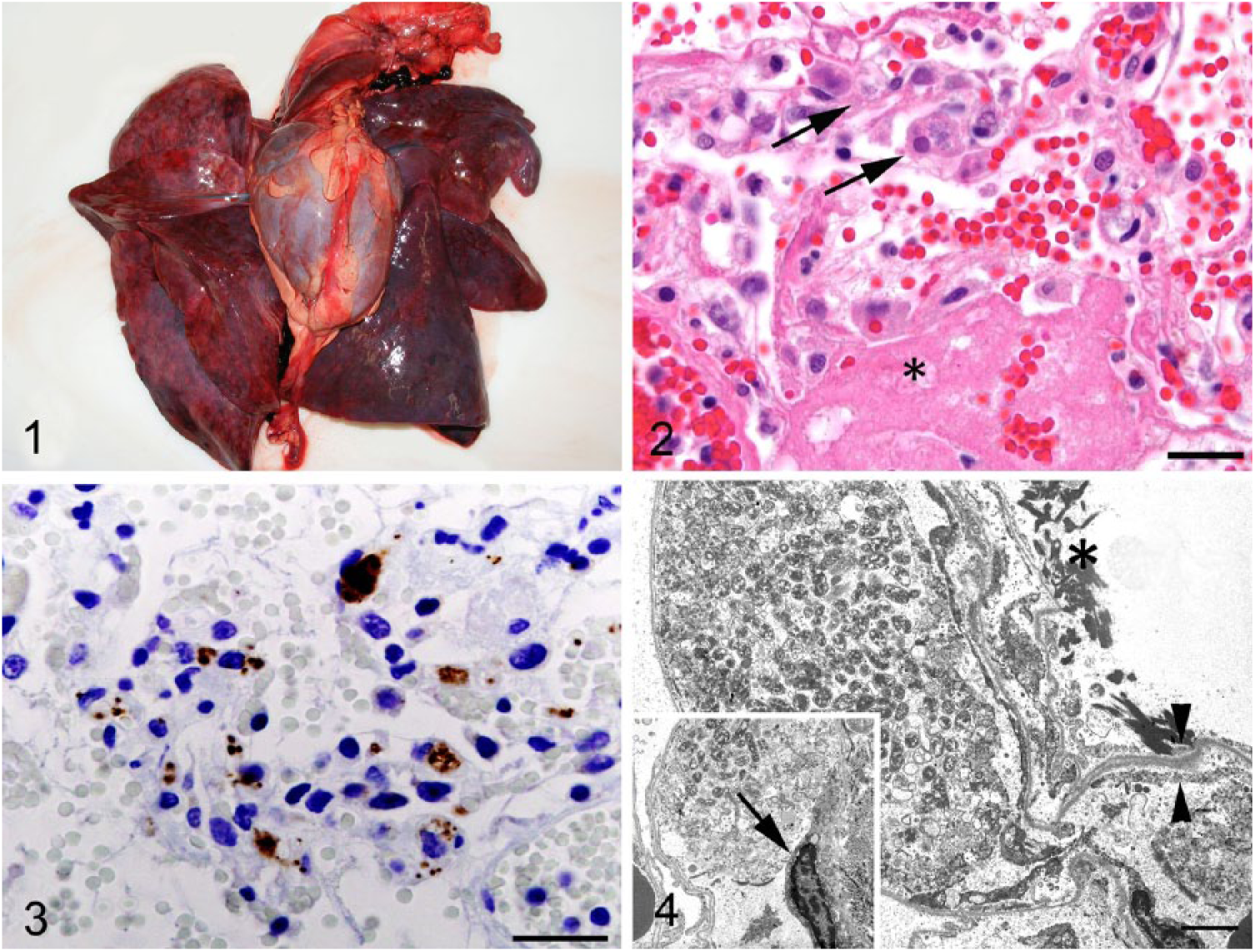
Lungs from an adult male Florida puma (Puma concolor coryi), case 1.
The most striking microscopic lesion common to these 3 pumas was acute, fibrinonecrotizing and hemorrhagic interstitial pneumonia. Alveolar septa were multifocally to diffusely thickened by fibrin, edema, moderate numbers of neutrophils and macrophages, and type II pneumocyte hyperplasia. Alveoli frequently were lined by hyaline membranes and contained pools of hemorrhage, proteinaceous edema fluid, or large aggregates of fibrin (Fig. 2). Perivascular and pleural connective tissues were multifocally expanded by edema or fibrin.
In addition to pulmonary lesions, cases 1–3 had microscopic evidence of myocarditis. Interstitial connective tissues were multifocally to diffusely infiltrated by neutrophils, scattered macrophages, and fewer lymphocytes and plasma cells. Moderate myocardial edema and cardiomyocyte degeneration were variably present. Other microscopic findings in these 3 cases included hepatocellular degeneration or disassociation (3 of 3), cholestasis (2 of 3), biliary and Ito cell hyperplasia (1 of 3), necrotizing vasculitis (1 of 3), mild plasmacytic interstitial nephritis (1 of 3), histiocytic lymphadenitis (1 of 3), pulmonary cholesterol granulomas (1 of 3), and splenic lymphoid depletion (1 of 3).
Only apparent in the lungs of case 1, intravascular macrophages occasionally contained discrete, 3–10 µm diameter, clear, intracytoplasmic vacuoles with numerous 0.5–1 µm, amphophilic, intravacuolar organisms (Figs. 2, 3). Giemsa, Gimenez, and Ziehl–Neelsen staining and CD18 and Mac387 immunohistochemistry (IHC) were performed according to standard procedures on sections of lung from case 1. Intrahistiocytic organisms stained positively with Giemsa and Gimenez, but did not exhibit detectable positive or negative gram-staining and stained faintly light blue with Ziehl–Neelsen acid-fast staining. Bartonella sp. organisms would be expected to exhibit gram-negative staining; the cause of undetectable gram-negative staining in the organisms observed here is unclear. The cells containing the organisms were confirmed to be histiocytes by CD18 and Mac387 IHC. Transmission electron microscopy performed on ultra-thin sections of lung from case 1 revealed numerous intracytoplasmic, pleomorphic, rod-shaped bacteria with trilaminar cell walls, averaging 1.09 µm long by 0.36 µm in diameter within circulating histiocytes, and disruption of adjacent endothelial cells and alveolar epithelial cells (Fig. 4).
Five additional Florida pumas (cases 4–8) and one puma from Kansas (case 9) were selected as case controls based on the absence of microscopic evidence of necrotizing interstitial pneumonia and suppurative myocarditis. Formalin-fixed, paraffin-embedded (FFPE) sections of lung from all 9 pumas were obtained, from which H&E-stained microscope slides were prepared routinely. Ancillary testing for a number of infectious agents and toxins was performed on the cases and case control pumas (Supplementary Table 1).
Conventional polymerase chain reaction (PCR) was performed using primers specific to the B. henselae pap31 heme-binding protein gene and 16S-23S intergenic spacer region, as described previously, 14 on sections of lung from the 2 pumas for which frozen tissues were available (cases 1 and 9). Additionally, conventional PCR was performed using primers specific to B. henselae DNA-directed RNA polymerase subunit beta gene (rpoB), as described previously, 19 on FFPE sections of lung from all cases and case controls. Given the documented cases of contamination from B. henselae–positive FFPE in previous investigations, 22 extreme caution was taken to avoid cross-contamination when creating tissue scrolls from blocks of embedded lung tissue. For each FFPE block, a sterile blade was used to shave the surface to remove any possible previous surface contamination. Following the initial cut, a new sterile blade was used to create a 10–20-µm section of FFPE tissue, which was then stored in a sterile microcentrifuge tube prior to extraction. Nucleic acids were extracted from frozen and FFPE tissues (DNeasy extraction kit, Qiagen, Hilden, Germany). A blank reagent negative control was included in the DNA extractions. All amplicon products from FFPE and fresh tissues were submitted to the Georgia Genomic Laboratory (Athens, GA), for bidirectional sequencing. B. henselae DNA was detected in all 3 Florida pumas with necrotizing interstitial pneumonia and suppurative myocarditis by PCR testing of frozen lung from case 1 and FFPE sections of lung from cases 1–3. Based on genetic sequencing of the 16S-23S intergenic spacer region gene (230 bp) and pap31 gene (525 bp), the isolates detected by conventional PCR on frozen lung from case 1 shared 100% sequence homology with B. henselae. Based on genetic sequencing of the rpoB gene (575 bp), the isolates detected by conventional PCR of FFPE lung tissue from cases 1–3 all shared 100% sequence homology with B. henselae. Of the case control pumas, B. henselae DNA was not identified in the single sample of frozen lung (case 9) or any of the FFPE lung samples tested by PCR.
IHC was performed on 4-µm sections of FFPE lung from all cases and case controls using a 1:100 dilution of B. henselae mouse monoclonal antibody (Biocare Medical, Concord, CA), biotinylated anti-mouse antibody (Vector Laboratories, Burlington, CA), streptavidin conjugated to horseradish peroxidase (Biocare Medical), and diaminobenzidine (Agilent, Santa Clara, CA) as chromogen. Slides were counterstained with Gill II hematoxylin (Leica Camera, Wetzlar, Germany). Immunohistochemical staining for B. henselae on FFPE sections of lung revealed positively staining organisms within pulmonary macrophages in all 3 cases (Fig. 3). Positively staining organisms were not identified in FFPE sections of lung from any of the control pumas evaluated with B. henselae IHC.
In situ hybridization (ISH) of B. henselae DNA was performed on FFPE sections of lung for all cases and 3 of the case controls (cases 4, 6, and 7; Table 1). A Bartonella henselae–specific DNA probe (5′-TGCCCAACCAAATGCTGGCA-3′) was labeled with digoxigenin-ddUTP (DIG; Roche Diagnostics, Mannheim, Germany). Briefly, the DIG-labeled probe was incubated on deparaffinized and protease-digested 4-µm sections of FFPE tissue. Bound DIG-labeled probe was detected with anti-DIG monoclonal antibody conjugated to alkaline phosphatase (Roche Diagnostics) and the chromogen nitro blue tetrazolium chloride (Boehringer Mannheim, Mannheim, Germany). Slides were counterstained with 1% fast green (Poly Scientific, Bay Shore, NY). Definitive positively staining organisms were detected by B. henselae ISH on FFPE sections of lung in cases 1 and 3; results were equivocal for case 2. Positively staining organisms were not identified in FFPE sections of lung from any of the 3 control pumas that were evaluated for B. henselae by ISH.
Results of Bartonella henselae–specific PCR, immunohistochemical (IHC), and in situ hybridization (ISH) testing performed on puma lung tissue.

E = equivocal; FFPE = formalin-fixed, paraffin-embedded; N = negative; NP = test not performed; P = positive.
Our case series provides evidence that B. henselae infection is associated with a unique disease syndrome of necrotizing interstitial pneumonia and suppurative myocarditis that was fatal in 3 Florida pumas. Although a number of studies have demonstrated that both captive and free-ranging pumas and other non-domestic felids have antibodies to B. henselae,20,24 disease associated with Bartonella spp. infection in non-domestic felids has not been described previously, to our knowledge.
B. henselae is most recognized as the causative agent of “cat scratch disease,” a syndrome of regional lymphadenopathy in humans that develops subsequent to cat scratches. 3 Enlarged and reactive lymph nodes were noted in 2 of the 3 affected pumas in our study and have also been described in experimentally infected cats,9,13 but the most striking lesions in these cases involved the lungs and heart. Bartonella spp. infection is increasingly detected in natural cases of endocarditis in humans, domestic dogs, and a single cat.2,7 Pyogranulomatous-to-lymphoplasmacytic myocarditis has been associated with B. henselae infection in experimentally inoculated and naturally infected cats.19,13,24 These findings suggest that B. henselae may have a predilection for cardiac tissues in domestic cats, although the reasons remain unknown. Mild lymphoplasmacytic myocarditis was noted in one of the control pumas. The significance of this finding is unclear.
Although heart lesions in association with Bartonella spp. infections have been reported in domestic cats, there are no previous reports of pulmonary lesions associated with Bartonella spp. infection in any felid species, to our knowledge. There is a single reported case of B. henselae–associated pulmonary lesions in an immunosuppressed woman, 5 and the lung lesions reported in the case were similar microscopically to those observed in the 3 affected pumas. Organisms genetically identical to the strain of B. henselae from the woman’s lung tissue were isolated in the blood of several of this woman’s pet cats, which were presumed to have been the origin of her infection.
It remains unknown why these 3 Florida pumas developed clinical disease associated with B. henselae infections given that exposure in both free-ranging and captive pumas seems common. One potential explanation for Bartonella-associated disease in these pumas is immune compromise, which has been demonstrated to be a significant risk factor in humans and domestic dogs.2,3 Extensive testing on the 3 affected pumas failed to provide evidence of an infectious cause of immunosuppression (such as feline immunodeficiency virus or feline leukemia virus). An underlying congenital immune deficiency in these pumas is considered given the presence of numerous other congenital anomalies in the Florida puma population and in these 3 pumas specifically. However, further investigation would be necessary to evaluate this hypothesis.
Poor genetic diversity had been suspected to play a role in increased susceptibility to infectious agents and atypical disease syndromes in captive cheetahs (Acinonyx jubatus). 16 In a 2004 publication, chronic expression of stress hormones as a result of maladaptation to captivity was hypothesized to cause atypical immune responses in captive cheetahs. 21 All 3 affected pumas in our study spent significant time in captivity (case 1 = 1.3 y; case 2 = 6 y; case 3 = 6 y) prior to their release into the wild, and an association between captivity and fatal B. henselae infection is an intriguing hypothesis. It should be noted that these 3 pumas had been living in the wild for periods of 6 wk to 8 mo prior to succumbing to fatal B. henselae infection, and any causal influence of captivity on their susceptibility to infection cannot be supported by these data alone. Furthermore, numerous pumas have been held for prolonged periods in captivity and survived for years after being released.
Exposure to cat fleas (C. felis) is an important risk factor for B. henselae infection and antibody presence in domestic cats.2,22 Based on previous reports, fleas may be uncommon or under-collected inhabitants of Florida pumas. 8 In contrast to C. felis parasitism, ixodid ticks are common ectoparasites of Florida pumas. 8 Ticks are believed to be a vector of several Bartonella species, and although host–vector–pathogen cycles are poorly understood, there is evidence that tick exposure is a risk factor for Bartonella infection.1,18 Tick-exposed animals that exhibit antibodies to Bartonella species frequently also have exposure to or infection with other potentially pathogenic organisms including Ehrlichia canis, Rickettsia rickettsii, Borrelia burgdorferi, and Babesia spp.1,10,18 Fleas, ticks, and tick-associated bacteria and piroplasms were not detected in any of the 3 affected pumas. However, previous flea and tick exposure or undetected coinfections cannot be excluded as possible contributing factors in the development of fatal B. henselae infection in these animals.
Finally, characteristics of the pathogen warrant consideration as possible contributing factors. Two distinct serotypes and corresponding genotypes of B. henselae have been described, and individual strains of B. henselae exhibit significant genetic heterogeneity and geographic clustering, which suggests clonal evolution of the organism within populations. 15 Certain strains or genotypes of B. henselae may be more pathogenic than others and have been associated with particular disease syndromes.6,17 Although the samples from the 3 affected pumas in our study were not further characterized by multilocus sequence typing or restriction-fragment length polymorphism analysis, virulence of the individual infecting B. henselae strains could have contributed to this unique disease syndrome.
Supplemental Material
DS1_JVDI_10.1177_1040638718789226 – Supplemental material for Necrotizing interstitial pneumonia and suppurative myocarditis associated with Bartonella henselae infection in three Florida pumas
Supplemental material, DS1_JVDI_10.1177_1040638718789226 for Necrotizing interstitial pneumonia and suppurative myocarditis associated with Bartonella henselae infection in three Florida pumas by Elizabeth J. Elsmo, Heather Fenton, Christopher A. Cleveland, Barbara Shock, Mark Cunningham, Elizabeth W. Howerth and Michael J. Yabsley in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the diagnosticians, technicians, and collaborators at Southeastern Cooperative Wildlife Disease Study, past and present, and the numerous staff biologists, veterinarians, and technicians affiliated with state and federal wildlife agencies who submitted the specimens. Special thanks to Deb Carter for her patient technical support with in situ hybridization, Mary Ard for assistance with TEM, Bambi Clemons for providing gross photographs and autopsy assistance, David Onorato for providing genetic analyses, and Dr. A Ayoob (Animal Specialty Hospital, Naples, FL) for assistance with capture and emergency treatment.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was made possible through continued financial support from the member states of the Southeastern Cooperative Wildlife Disease Study provided by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
