Abstract
Liposarcomas are rare malignant tumors showing adipocytic differentiation. We report a well-differentiated liposarcoma in a 6-y-old, male neutered cat with a prominent inflammatory component and metastatic spread to the lungs. The patient was initially presented because of fever, lethargy, and a firm subcutaneous inguinal mass. A Tru-cut biopsy of the mass revealed a mixture of well-differentiated adipocytes and lymphoplasmacytic–histiocytic inflammation, interpreted as panniculitis. The mass was surgically excised but recurred 4 mo later. A second excisional biopsy yielded similar histologic findings. A third recurrence of the mass was associated with lung nodules. Histopathology of the recurring and metastatic masses confirmed the diagnosis of well-differentiated inflammatory liposarcoma with pulmonary metastases. The neoplasm had an intense inflammatory component, which obscured the underlying features of liposarcoma and made differentiation from steatitis difficult. This inflammatory variant of a well-differentiated liposarcoma should be considered as a differential in tumorous steatitis-like lesions.
A 6-y-old, male neutered, Domestic Long Hair cat was presented for a 5-d history of inappetence and lethargy. The cat had been regularly vaccinated, fed commercial canned and dry food, and lived indoors with another cat. The physical examination was unremarkable except for a fever of 40.6°C (105°F) and a firm (97 × 89 mm) subcutaneous mass in the inguinal region. Radiographs demonstrated a smoothly marginated, round, soft tissue opacity mass. Ultrasound imaging revealed that the mass contained heterogeneous material with fluid-filled pockets throughout. The cytology specimen of a fine-needle aspirate of the mass was moderately cellular, consisting of a low number of small lymphocytes scattered in a background of basophilic, proteinaceous material. An interpretation of lymphocytic inflammation with a cystic component was made. No neoplastic cells were seen.
No bacterial growth was noted on aerobic and anaerobic cultures of an aspirate of the mass. Hematologic examination revealed an elevated white blood cell count (29.0 × 109/L; reference interval [RI]: 3.4–13.5 × 109/L), with an increase in segmented neutrophils (26.7 × 109/L; RI: 1.5–9.6 × 109/L) and no toxic changes. Slightly elevated glucose (10.1 mmol/L; RI: 4.2–7.4 mmol/L) and globulin (46 g/L; RI: 23–38 g/L) were noted on the chemistry panel. The abnormal findings of the results were most likely the result of an inflammatory process, along with a stress response. Infectious disease–screening results were negative for feline immunodeficiency virus antibody and feline leukemia virus (FeLV) antigen. Feline pancreatic lipase immunoreactivity (fPLI) was mildly elevated at 3.9 µg/L (RI: 0.0–3.5 µg/L).
Tru-cut biopsy samples of the mass consisted of variably cellular samples of adipose tissue with localized areas of fat necrosis. In necrotic areas, there was moderate infiltration by lymphocytes and histiocytes with perivascular lymphoid cuffs. Early fibroplasia was also noted. These features were interpreted as panniculitis with no evidence of neoplasia (Fig. 1A). Fite–Faraco acid-fast stain was used to evaluate for atypical mycobacteria on the histology slides. No acid-fast–stained bacilli were seen. The patient was continued on a commercial balanced diet, and placed on additional vitamin E supplementation and an anti-inflammatory dose of oral prednisolone (0.73 mg/kg/d), which controlled the fever. The mass, however, did not decrease in size. The mass was excised, and histologic examination indicated moderately hypercellular adipose tissue with regions of coagulative necrosis and nodular lymphocytic inflammation; the mass was interpreted at this time as a lipoma with areas of inflammation and necrosis. Incomplete margins were noted.
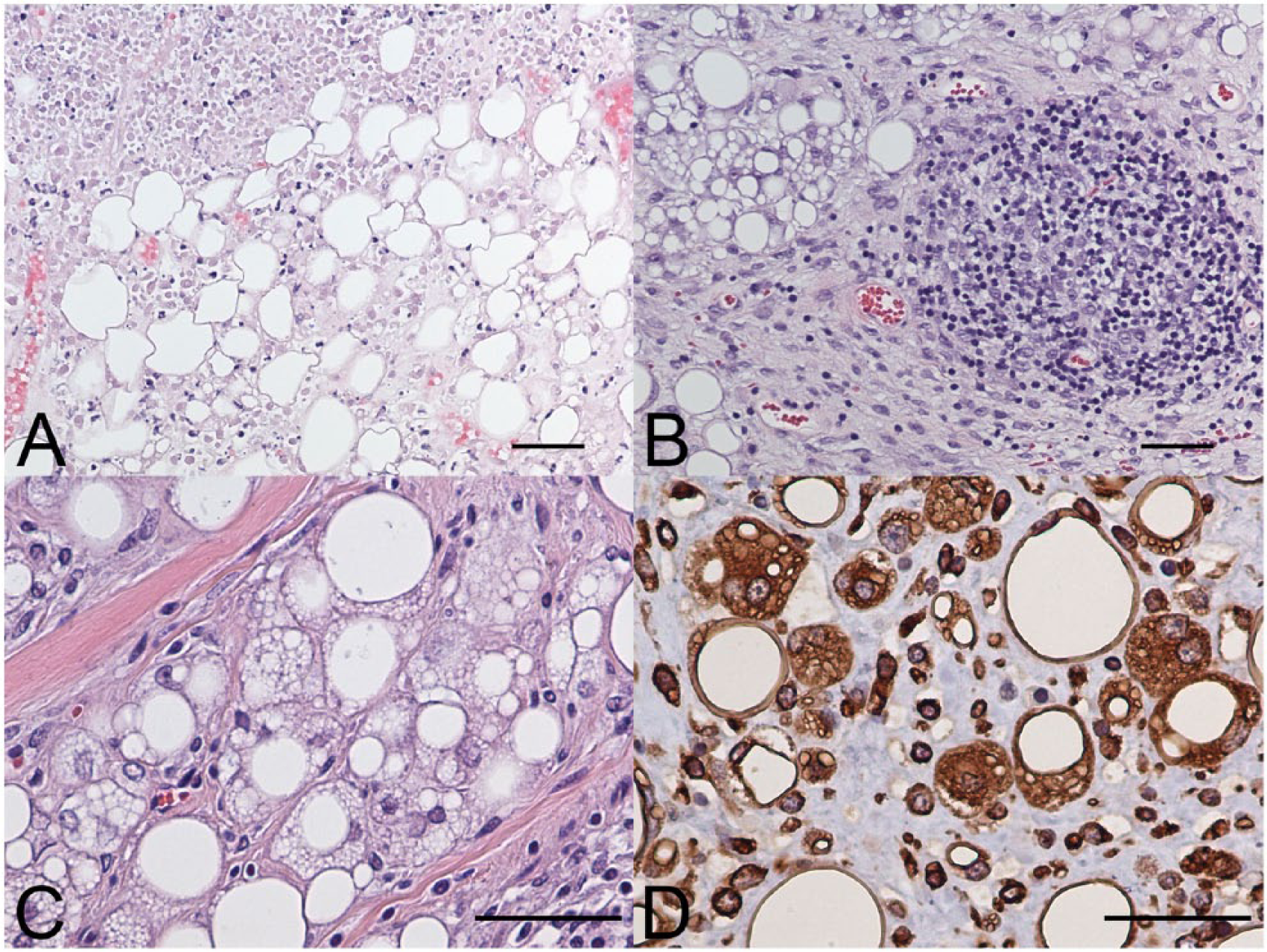
Inflammatory liposarcoma in a cat.
Four months later, a mass recurred in the same area, and the patient was uncomfortable on inguinal palpation, and had an intermittent fever; the mass was again excised. Histopathology of the mass was similar to the initial tissues examined. However, in the second biopsy, there was less necrosis and prominent lymphoplasmacytic and histiocytic inflammation forming discrete lymphohistiocytic aggregates from which the diagnosis of chronic lymphoplasmacytic and granulomatous steatitis was made (Fig. 1B); Fite–Faraco, Ziehl–Neelsen, and Grocott–Gomori methenamine silver stains failed to identify infectious organisms. Continued conversations regarding the unusual clinical progression prompted additional consultation with both veterinary dermatopathologists and a medical pathologist with expertise in soft tissue sarcoma classification. From this collaboration, the diagnosis was revised to a well-differentiated, inflammatory-type liposarcoma based on characteristic nodular lymphoid infiltration, the presence of low numbers of lipoblasts with multiple cytoplasmic vacuoles, as well as low numbers of spindle cells admixed within the more typical adipocytes (undifferentiated lipoblasts). 9
A third recurrence of the mass 7 mo after initial presentation was followed by detection of pulmonary nodules on thoracic computed tomography (CT) scan. The patient did not exhibit signs of illness associated with the regrowth of the primary mass or the pulmonary masses for ~4 mo following the CT. Given the uncertain prognosis and declining quality of life, the patient was euthanized. Autopsy findings corroborated the imaging results and demonstrated 3 masses in the abdominal/inguinal subcutis, and 2 pulmonary masses (Fig. 1C). Histopathology of the recurrent subcutaneous masses as well as the pulmonary masses was similar to previous biopsies, and a diagnosis of well-differentiated inflammatory liposarcoma with pulmonary metastases was confirmed via both hematoxylin and eosin stain and vimentin immunohistochemical reaction (Fig. 1C, 1D). The larger pulmonary mass was morphologically similar to the regrown inguinal mass at the time of necropsy, whereas the smaller mass was less well-differentiated, with minimal lipid vacuolation; both retained an inflammatory component. Declining quality of life was attributed to an unrelated focal encephalopathy noted histologically (mild focal necrosis and gliosis, interpreted as an infarct), rather than clinical effects of the neoplasms.
Diagnosing liposarcoma in this case was difficult because of the intense inflammatory component of the mass, as well as the initial clinical signs in this cat. Two feline cutaneous and/or subcutaneous syndromes characterized by diffuse fat necrosis and inflammation have been described: feline pansteatitis, and systemic lipodystrophy associated with pancreatitis. 6 Other potential etiologies of diffuse fat necrosis and inflammation include infectious agents (bacterial, fungal, viral), physicochemical factors (trauma, pressure, cold stress, foreign body reactions), neoplasia, vasculopathy, and immune-mediated disorders. 6 Pansteatitis is caused by the consumption of high levels of unsaturated fatty acids and/or insufficient intake of vitamin E, leading to inflammation of adipose tissue. Pansteatitis has been reported in cats eating fish-based diets, but diet is not always the underlying cause. Food processing or fat oxidation that inactivate vitamin E can also cause pansteatitis. 6 Common clinical signs of pansteatitis are fever, lethargy, inappetence, and pain on palpation of the skin and subcutaneous nodular masses, which may have a smooth or lumpy consistency. 6 The cat in our case initially had clinical signs similar to those reported in cats with pansteatitis, namely fever, lethargy, and inappetence. Diagnosis of pansteatitis is based on clinical signs, ruling out other causes of fat inflammation, and histologic examination of biopsies. In this case, a commercial cat food was fed, no known trauma was suspected, and infectious causes (including aerobic and anaerobic bacterial infection, and FeLV) were less likely given the negative test results. The fPLI was only mildly elevated, making severe pancreatitis unlikely. The presenting clinical signs, diagnostic work-up, and cytology and biopsy results in our case resulted in a presumptive diagnosis of steatitis of unknown etiology.
An inflammatory variant of liposarcoma has been reported in human medicine, in which the presence of a chronic inflammatory infiltrate can obscure the adipocytic nature of the neoplasm, representing a potential diagnostic pitfall. 9 The adipocytic component could be easily missed or confused with more common forms of fibroplasia associated with inflammatory conditions. 9 The presence of clearly delineated lymphocytic nodules within otherwise unremarkable adipose tissue and without other signs of inflammation should raise suspicion for this unusual liposarcoma variant. Other diagnostic features that suggest liposarcoma include lipoblasts, multinucleation, and variable degrees of intervening fibrous tissue among the adipocyte population, forming a multinodular mass best seen at low magnification. In a small case series in people, tumors recurred in 6 of 8 patients, similar to the recurrence reported in our case. 9 Unlike the feline case we are reporting, the majority of people affected in this human case series were older, with a median age of 62 y, and none had evidence of distant metastasis. 9 In the most recent World Health Organization Classification of Tumours of Soft Tissue and Bone in humans, this entity is 1 of 3 subtypes of “atypical lipomatous tumor/well differentiated liposarcoma,” which are considered locally aggressive with low metastatic potential. 7
Most case reports of liposarcoma in the veterinary literature are from dogs, with isolated reports in cats and other species.3,4,8 In contrast, liposarcoma is the most common soft tissue sarcoma in humans and accounts for ~20% of all mesenchymal malignancies encountered by surgical pathologists. 9 Classification of liposarcomas in animals includes 3 types: well-differentiated, anaplastic–pleomorphic, and myxoid. 8 Although liposarcomas typically develop spontaneously in many species, in cats, liposarcomas are most often found in the subcutis as a manifestation of injection site–associated sarcomas.4,5,10 In addition, there are reports that infection with FeLV may be associated with development of liposarcoma. 4 This patient was FeLV-negative, and the location of the tumor was not coincident with any previous injection sites. Our case deviates from most if not all of the reported cases in cats.3,5,10
The metastatic potential for liposarcomas is considered low in veterinary species, although given the paucity of feline cases, any conclusions on metastatic rate are not well supported. 8 Metastasis may be more likely with injection site–associated liposarcomas4,5; however, metastasis has also been reported in a feline case of a spontaneous liposarcoma. 3 Generally, well-differentiated liposarcoma shows a risk of local recurrence (~30%), but low potential for metastasis. Tumor progression may correlate with increased features of malignancy including changes in morphology such as nuclear atypia, anisocytosis and anisokaryosis, or cellular pleomorphism, and metastatic behavior. 8 With recurrence of this neoplasm, there was loss of differentiation and increased numbers of lipoblasts with variable lipid content. One of the metastatic sites was composed of spindle cells with few-to-no lipid droplets, interpreted as neoplastic lipoblasts.1,8
Recurrence of liposarcoma following excision attempts is common. In one study, 12 of 43 (28%) dogs with liposarcoma had local tumor recurrence after surgical resection. 2 Results of that study also indicated that, in dogs, excision of liposarcomas with wide surgical margins significantly improved survival time, compared with survival times following tumor excision with narrow surgical margins or incisional biopsy. 2 The second time the mass in our case was removed, it was fairly well delineated, but surgical margins were narrow. With early correct histologic diagnosis followed by aggressive resection to obtain wide margins, local recurrence rates can be decreased.
Well-differentiated inflammatory-type liposarcoma should be considered as a differential for tumorous steatitis-like lesions. Distinguishing the 2 entities can be challenging because of intense inflammation, which may obscure the adipocytic cells, and the often subtle differences between normal adipose and well-differentiated neoplastic adipocytes. If neoplasia is suspected based on biologic behavior, complete removal of the mass may improve diagnostic accuracy over incisional biopsy. The true metastatic potential of liposarcomas in cats remains poorly documented, but metastasis is possible. Because of the potential for metastasis of liposarcomas in cats, it is advisable to evaluate feline patients for metastatic disease. Analysis of future cases is important for gaining a better understanding of the behavior of this rare neoplasm.
Footnotes
Acknowledgements
We thank Darya Buehler MD, Department of Pathology and Laboratory Medicine, School of Medicine and Public Health, University of Wisconsin, Madison, WI.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
