Abstract
Histochemical techniques used in examination of muscle biopsies typically require frozen sections. Given that most of the specimens submitted to a veterinary laboratory for diagnosis are formalin-fixed, the choice of staining methods is limited. We aimed to further advance the diagnostic capabilities of pathologists presented with formalin-fixed muscle samples and to describe the differences in immunohistopathologic findings between neurogenic and myogenic muscle disorders. Based on hematoxylin and eosin staining, we defined in dogs the histologic lesions in 4 neurogenic disorders (degenerative myelopathy and polyneuropathy) and 2 myogenic disorders (dystrophin-deficient muscular dystrophy). In cats, we defined the lesions in 2 neurogenic disorders (lymphoma of nerve roots and spinal cords) and 1 myogenic disorder (laminin α2–deficient muscular dystrophy). Immunohistochemistry for slow and fast myosins revealed angular and group atrophy of type 1 and type 2 fibers in dogs and cats, and fiber type grouping in dogs. These immunohistopathologic findings were specific to neurogenic muscle disorders. Immunohistochemistry for nestin and myogenin revealed nestin-positive fibers and myogenin-positive nuclei in dogs and cats. They were not specific, but these fibers in myogenic disorders can be interpreted as regenerating fibers. The immunohistochemical method described herein appears to be useful for discriminating neurogenic and myogenic disorders in formalin-fixed, paraffin-embedded muscle tissue of dogs and cats.
Introduction
Muscle disorders may be roughly categorized into neurogenic disorders and myogenic disorders (myopathies). Neurogenic muscle disorders are characterized histopathologically by angular atrophy, small- or large-group atrophy, and pyknotic nuclear clumps. 12 Histopathologic findings of myopathies vary, but include variation in myofiber diameter, and necrosis and regeneration of myofibers. 12 Such myopathic findings are also occasionally observed in neurogenic muscle disorders and vice versa. Hence, discriminating histologically between neurogenic and myogenic muscle disorders is challenging. 29
Histochemical methods such as ATPase staining are widely used for myofiber typing.4,5,12,14 In normal muscle, and most myopathies, there is a mosaic or random pattern of type 1 and type 2 fibers. In neurogenic muscle disorders, the groups of atrophic fibers are of both types, and grouping of fibers of the same type (fiber type grouping) is indicative of re-innervation following denervation.12,13 Acid or alkaline phosphatase staining is also used for the detection of necrotic or regenerating fibers. Acid phosphatase highlights the presence of macrophages in necrotic fibers, and alkaline phosphatase reaction is positive in some regenerating fibers.9,12,14,27 These stains are also useful for discriminating between neurogenic and myogenic disorders. However, such histochemical techniques typically require frozen sections. Because most of the specimens submitted to a veterinary laboratory for examination in Japan are formalin-fixed, the choice of staining methods is limited.
In human muscle pathology, immunohistochemical techniques using antibodies to myosin are commonly used instead of ATPase staining.2,25 Muscle fiber types are related to their corresponding myosin heavy chain isoform contents.1,34,35,37 Immunohistochemistry can be used to define macrophages in necrotic fibers using CD68 or Iba-1 antibody.11,13 Immunohistochemical techniques using antibodies to nestin and myogenin are used instead of alkaline phosphatase staining, and nestin-positive fibers with myogenin-positive nuclei are interpreted as regenerating fibers.33,38,43 During the development and regeneration of muscle fibers, nestin is expressed in myotubes (regenerating fibers).3,7,23,28,39 Myogenin is expressed in myonuclei during the differentiation of myoblasts to myotubes.15,21,42,44 In people, most of these procedures are performed on frozen sections of muscle samples, but many of these immunohistochemical methods can also be applied to formalin-fixed, paraffin-embedded muscle samples.
Several defective proteins associated with the sarcolemma are responsible for various forms of muscular dystrophy. Dystrophin is a muscle cytoskeletal protein, and mutations in dystrophin cause Duchenne or Becker muscular dystrophy. 13 Laminin α2 is localized to the basal lamina of muscle fibers, and an absence of laminin α2 causes merosin-deficient congenital muscular dystrophy. 13 Abnormalities of these proteins can usually be detected by immunohistochemistry on frozen section, but it is also established in people that immunohistochemical techniques on paraffin sections can reveal abnormalities and support the definitive diagnosis.16,31
We investigated the application of immunohistochemical methods using formalin-fixed, paraffin-embedded tissues for canine and feline muscle disorders. We focused on immunohistochemical differences between neurogenic and myogenic muscle disorders.
Materials and methods
Samples of canine and feline skeletal muscles and nerve tissues (spinal cord, nerve root, and peripheral nerve) were collected from 4 autopsy and 5 biopsy cases in our laboratory. All cases were histologically diagnosed as muscle disorders (Table 1).
Case information and muscles examined from dogs and cats with muscle disorders.*
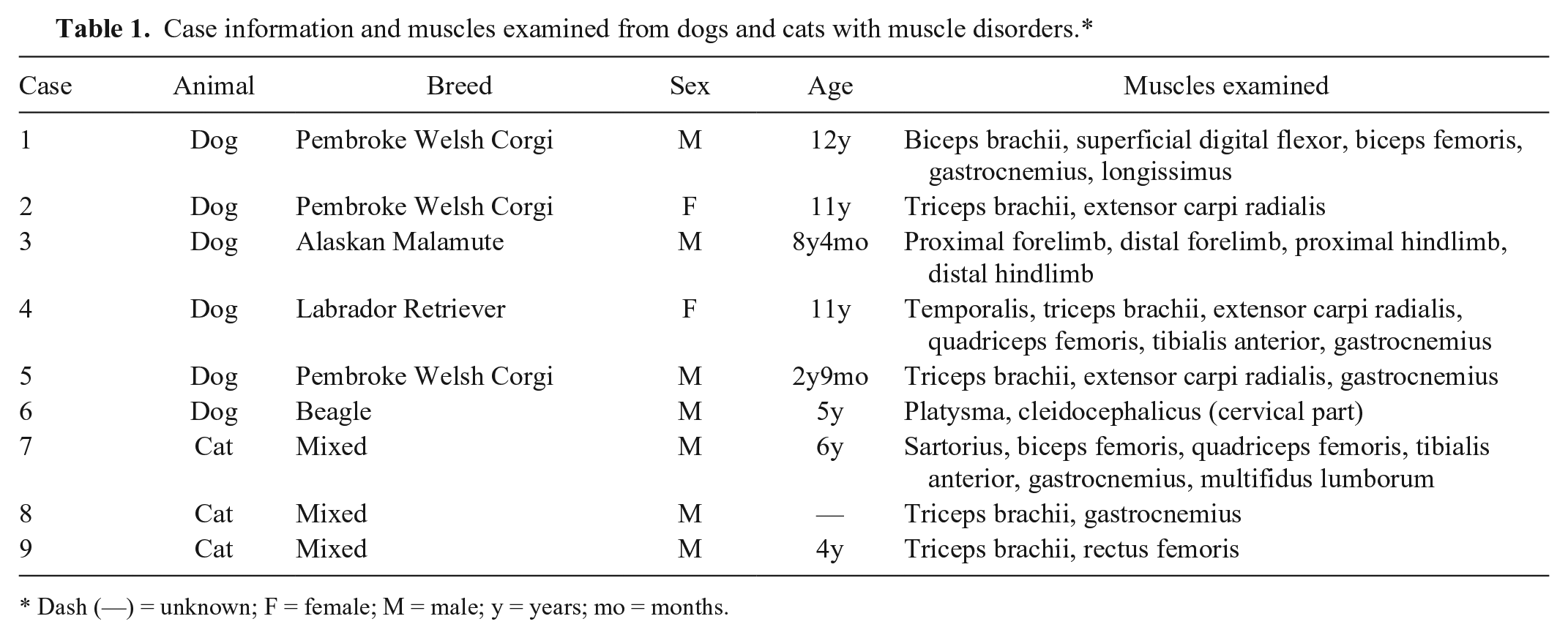
Dash (—) = unknown; F = female; M = male; y = years; mo = months.
Tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, and embedded in paraffin wax. Paraffin tissue sections (2 μm thick) were stained with hematoxylin and eosin. Nerve tissues were also stained with luxol fast blue and immunostained for neurofilament to evaluate demyelination or axonal degeneration. All cases were tentatively divided into 2 groups based on histologic examinations: neurogenic and myogenic disorders.
A commercial anti-canine fast myosin antibody (Sigma-Aldrich, Tokyo, Japan) was used. A rabbit was immunized with an antigen peptide of the canine fast myosin (amino acids 1929–1937; UniProt ID Q076A7; [H] CXSREVHTKVI [OH], X = 6-aminocarponic acid).
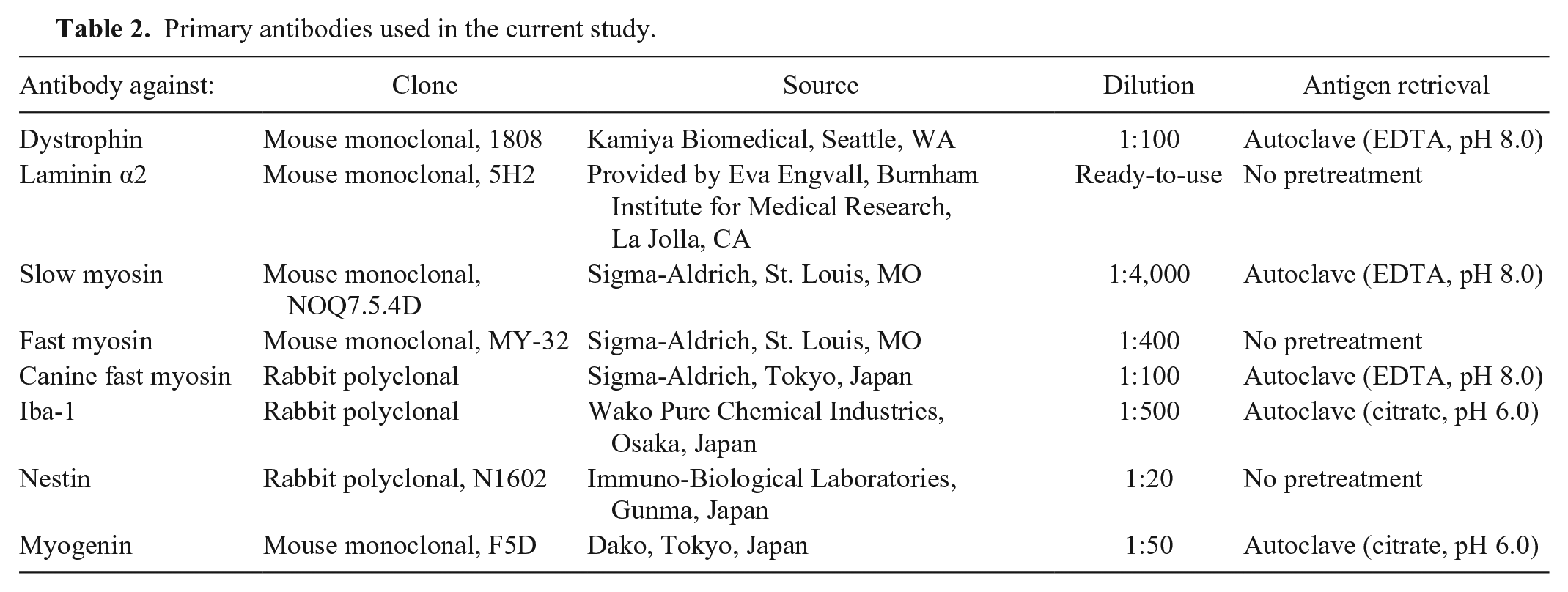
Consecutive sections were reacted with each primary antibody after antigen retrieval (Table 2). In order to inactivate endogenous peroxidase, deparaffinized sections were first immersed in 1% hydrogen peroxide in methanol for 4 min. After washing with Tris-buffered saline (TBS), the sections were incubated with 5% skimmed milk in TBS at 37°C for 30 min in order to avoid nonspecific reactions. After being incubated with the primary antibody at 4°C overnight, the sections were incubated with a horseradish peroxidase–labeled polymer at 37°C for 40 min, and reacted with 0.05% 3,3’-diaminobenzidine plus 0.03% hydrogen peroxide in Tris–hydrochloric acid buffer. Finally, the slides were counterstained with hematoxylin. Negative controls were obtained by omitting the primary antibodies.
Primary antibodies used in the current study.
Formalin-fixed sections (8 μm thick) from cases 5 and 6 (dogs) were immunostained for dystrophin and laminin α2 to confirm the diagnosis. Frozen samples of these cases could not be obtained. Formalin-fixed and frozen muscle tissue sections (8 μm thick) from case 9 (cat) were immunostained for dystrophin and laminin α2. Autopsy and biopsy cases within normal limits were used as normal controls. The anti–laminin α2 antibody that we used in this study did not exhibit immunoreactivity against canine formalin-fixed and frozen tissues and feline formalin-fixed tissues.
Results
Pathologic features of canine cases
In cases 1–4, significant pathologic changes were observed in nerve tissues (Table 3). Demyelination was observed in all of these cases. Cases 1 and 2 were diagnosed as degenerative myelopathy (DM) based on the detection of a SOD1 gene mutation and histologic lesions in nerve tissues. In addition, axonal loss was detected in both spinal nerve roots and spinal cords of case 1. Cases 3 and 4 were diagnosed as polyneuropathy; case 4 was the peripheral type. In cases 5 and 6, no significant lesions were observed in nerve tissues.
Histopathologic findings and immunohistochemical results of neurogenic and myogenic disorders in dogs and cats.*
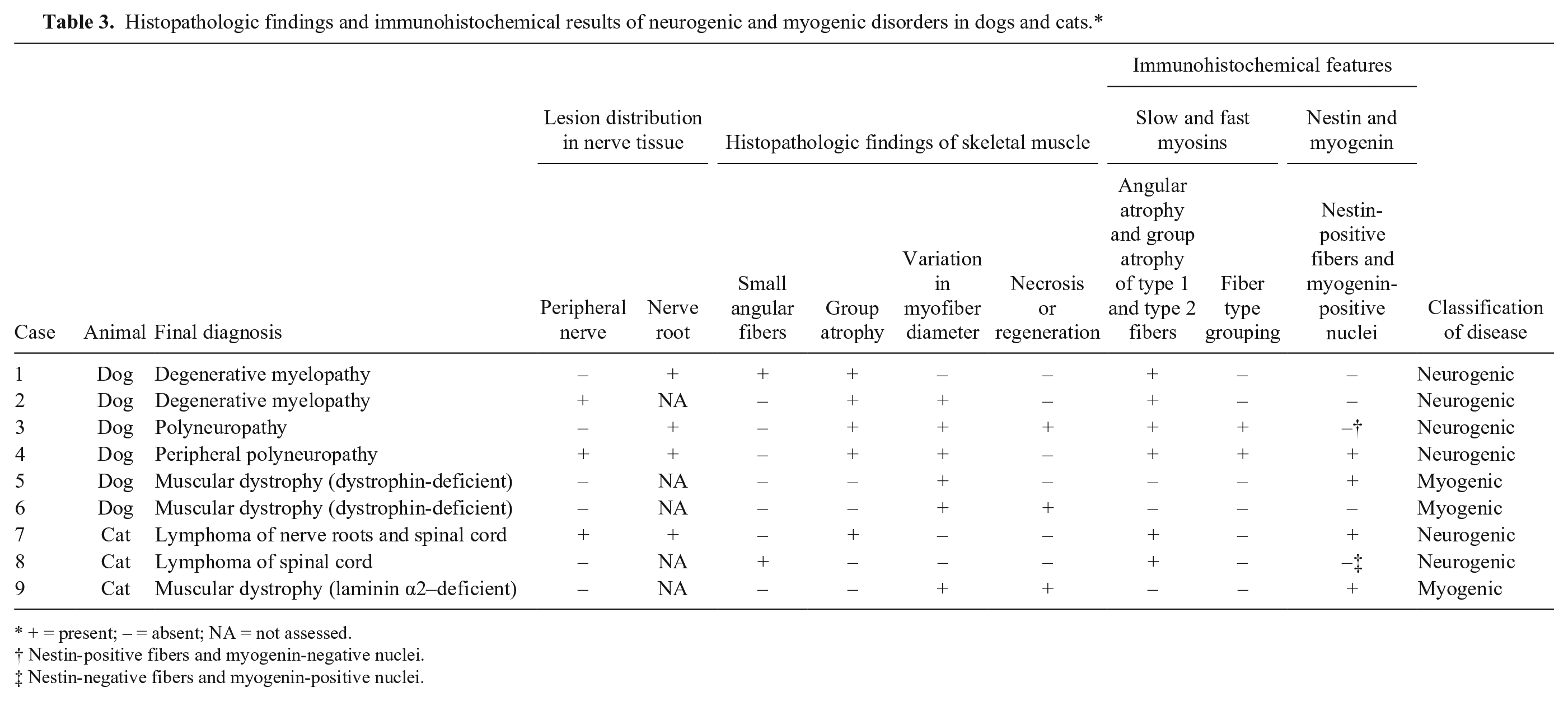
+ = present; – = absent; NA = not assessed.
Nestin-positive fibers and myogenin-negative nuclei.
Nestin-negative fibers and myogenin-positive nuclei.
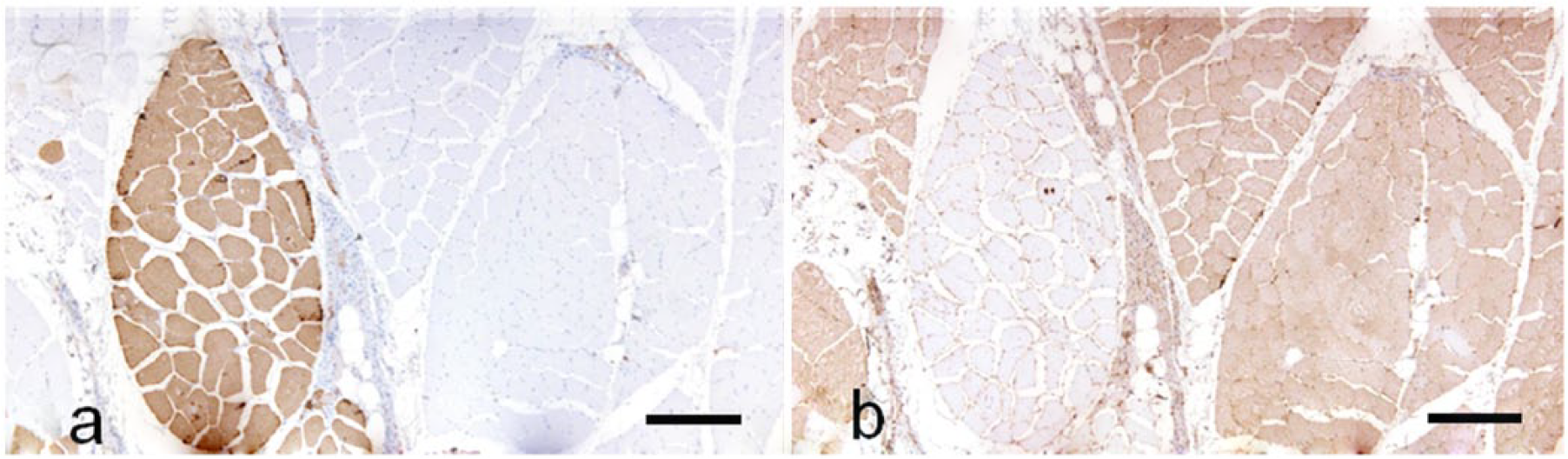
Formalin-fixed muscle sections taken from normal canine controls showed strong immunolabeling of dystrophin at the sarcolemma (Fig. 1a). In cases 5 and 6, there was no immunolabeling of dystrophin at the sarcolemma in formalin-fixed sections (Fig. 1b) and thus these cases were diagnosed as dystrophin-deficient muscular dystrophy.
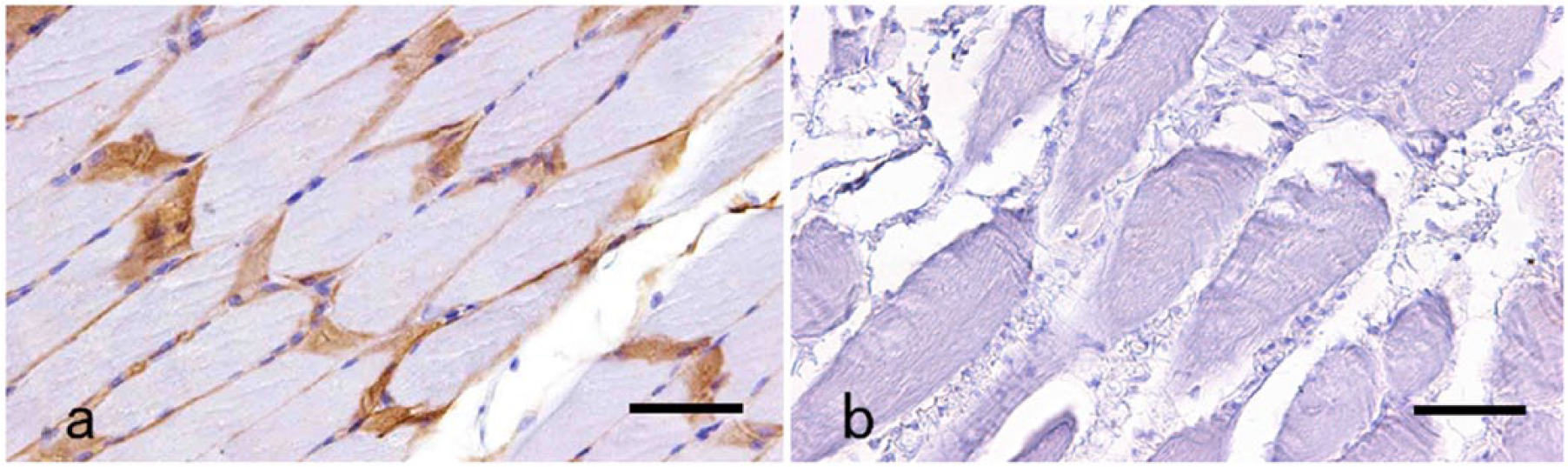
Immunolabeling of dystrophin in muscles of a normal dog and a dog with muscular dystrophy (case 5).
Histopathology and immunohistochemistry of canine muscle tissues
Major histopathologic findings included small angular fibers, small- or large-group atrophy, variation in myofiber diameter, and the necrosis and regeneration of myofibers (Table 3). Small angular fibers and/or small- or large-group atrophy were observed in cases 1–4 (Fig. 2a, 2b). In these cases, immunohistochemistry for slow myosin and fast myosin revealed angular and group atrophy involving both type 1 and type 2 fibers and occasional hybrid fibers immunopositive for both slow and fast myosins (Fig. 2c, 2d). Obvious fiber type grouping was also observed in cases 3 and 4 (Fig. 3a, 3b). In case 4, a large number of nestin-positive fibers were observed in the area of large-group atrophy (Fig. 4a). Some of the nestin-positive fibers had small nuclei with obscure nucleoli. These myonuclei and pyknotic nuclear clumps were immunopositive for myogenin (Fig. 4b). In case 3, although all nuclei were negative for myogenin, many nestin-positive fibers were observed in the lesion of large-group atrophy. In other cases, myofibers and myonuclei were immunonegative for nestin or myogenin.
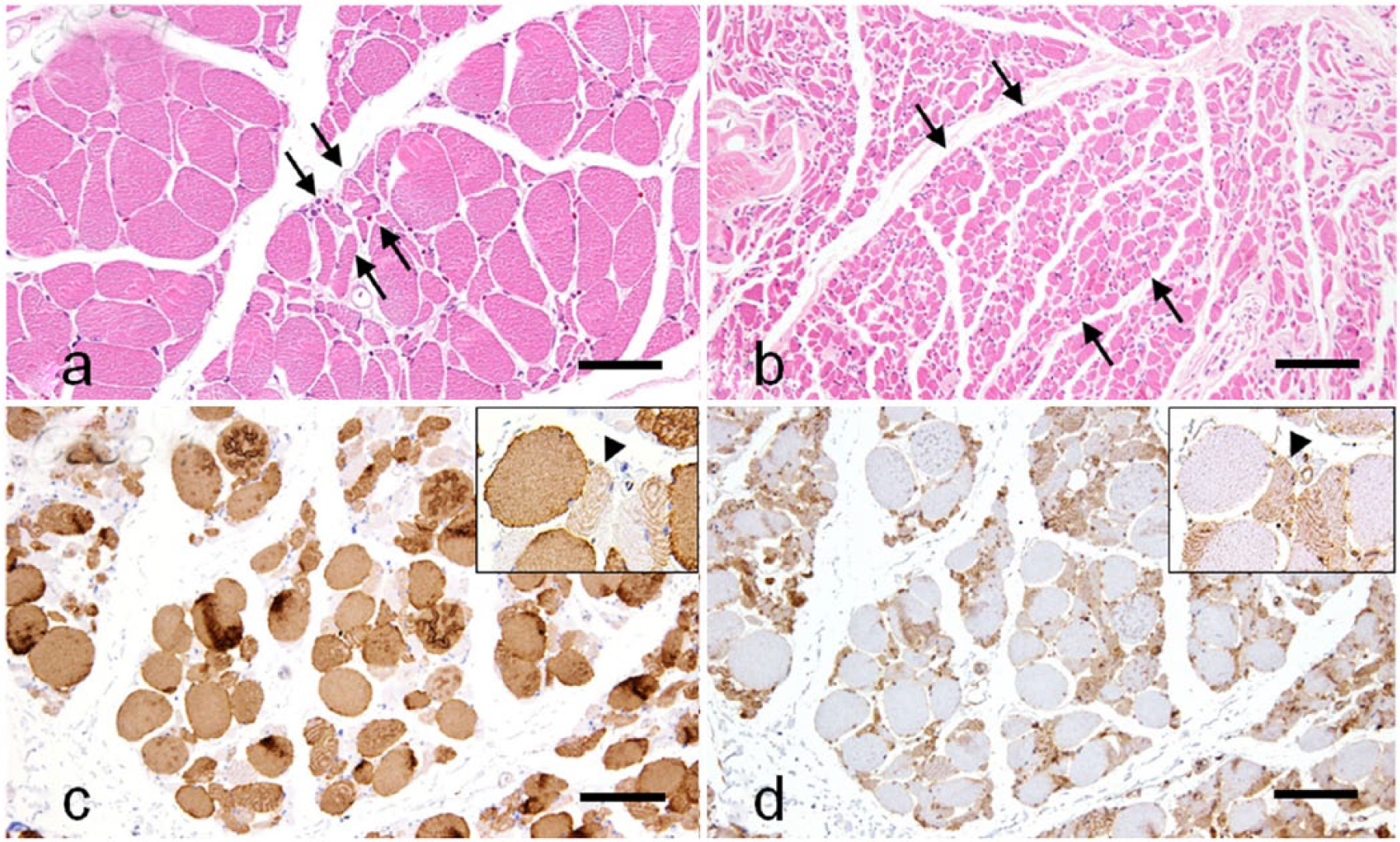
Neurogenic changes in a dog (case 1) with degenerative myelopathy.
Neurogenic changes in a dog (case 3) with polyneuropathy.
Neurogenic changes in the gastrocnemius muscle of a dog (case 4) with peripheral polyneuropathy.
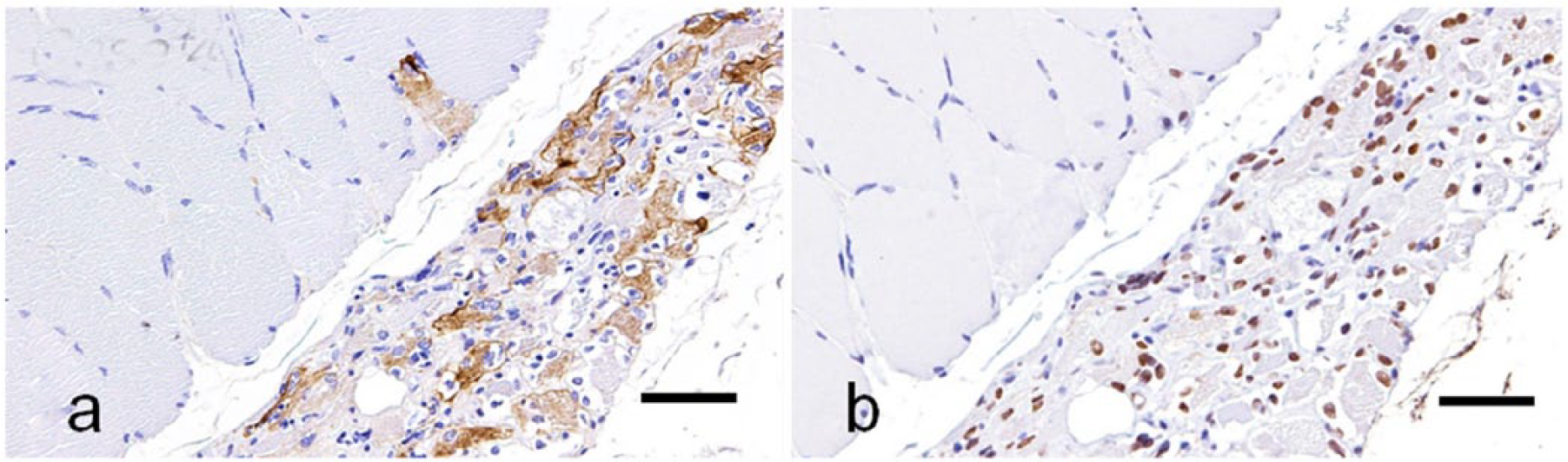
In cases 5 and 6, variation in myofiber diameter and the random pattern of muscle fiber types was observed, and no lesion of fiber type grouping was observed. In case 5, many regenerating fibers were immunopositive for nestin, and their nuclei were immunopositive for myogenin. Some regenerating fibers were included in hybrid types that were immunopositive for both slow and fast myosins. In case 6, macrophages and myoblasts were observed within necrotic lesions. Macrophages were immunopositive for Iba-1, but their nuclei were immunonegative for myogenin. Myoblasts were immunonegative for Iba-1, but their nuclei were immunopositive for myogenin. Based on these results, cases 1–4 were diagnosed as neurogenic muscle disorders. Cases 5 and 6 were diagnosed as myopathies (Table 3).
Pathologic features of feline cases
In cases 7 and 8, significant pathologic changes were observed in nerve tissues (Table 3). Marked infiltration of neoplastic lymphocytes in the spinal cords and resulting demyelination and axonal loss in the spinal nerve roots and peripheral nerves were observed in cases 7 and 8. These cases were diagnosed as lymphoma. In case 9, no significant lesions were observed in nerve tissues.
Formalin-fixed muscle sections taken from normal feline controls showed strong immunolabeling of dystrophin at the sarcolemma. In case 9, there is no immunolabeling of laminin α2 at the sarcolemma on frozen sections, and thus case 9 was diagnosed as laminin α2–deficient muscular dystrophy.
Histopathology and immunohistochemistry of feline muscle tissues
Major histopathologic findings included small angular fibers, small- or large-group atrophy, variation in myofiber diameter, and the necrosis and regeneration of myofibers (Table 3). Small angular fibers and/or small- or large-group atrophy were observed in cases 7 and 8. In these cases, immunohistochemistry for slow myosin and fast myosin revealed angular and group atrophy involving both type 1 and type 2 fibers and occasional hybrid fibers immunopositive for both slow and fast myosins. Fiber type grouping was not observed. The loss of myofibers and pyknotic nuclear clumps was often observed in case 7. Nestin-positive fibers were detected in the area of large-group atrophy. Myonuclei and pyknotic nuclear clumps were positive for myogenin. In case 8, small angular fibers were immunonegative for nestin but the peripheral nuclei were immunopositive for myogenin.
Variation in myofiber diameter was observed in case 9 (Fig. 5a). Immunohistochemistry for slow and fast myosins revealed a random pattern of atrophy and hypertrophy of type 1 and type 2 fibers and occasional hybrid fibers (Fig. 5b, 5c). Many regenerating fibers (Fig. 6a) were immunopositive for nestin (Fig. 6b), and their nuclei were immunopositive for myogenin (Fig. 6c). Some regenerating fibers were included in hybrid types that were immunopositive for both slow and fast myosins (Fig. 6d, 6e). Based on these findings, cases 7 and 8 were diagnosed as neurogenic muscle disorders. Case 9 was diagnosed as myopathy (Table 3).
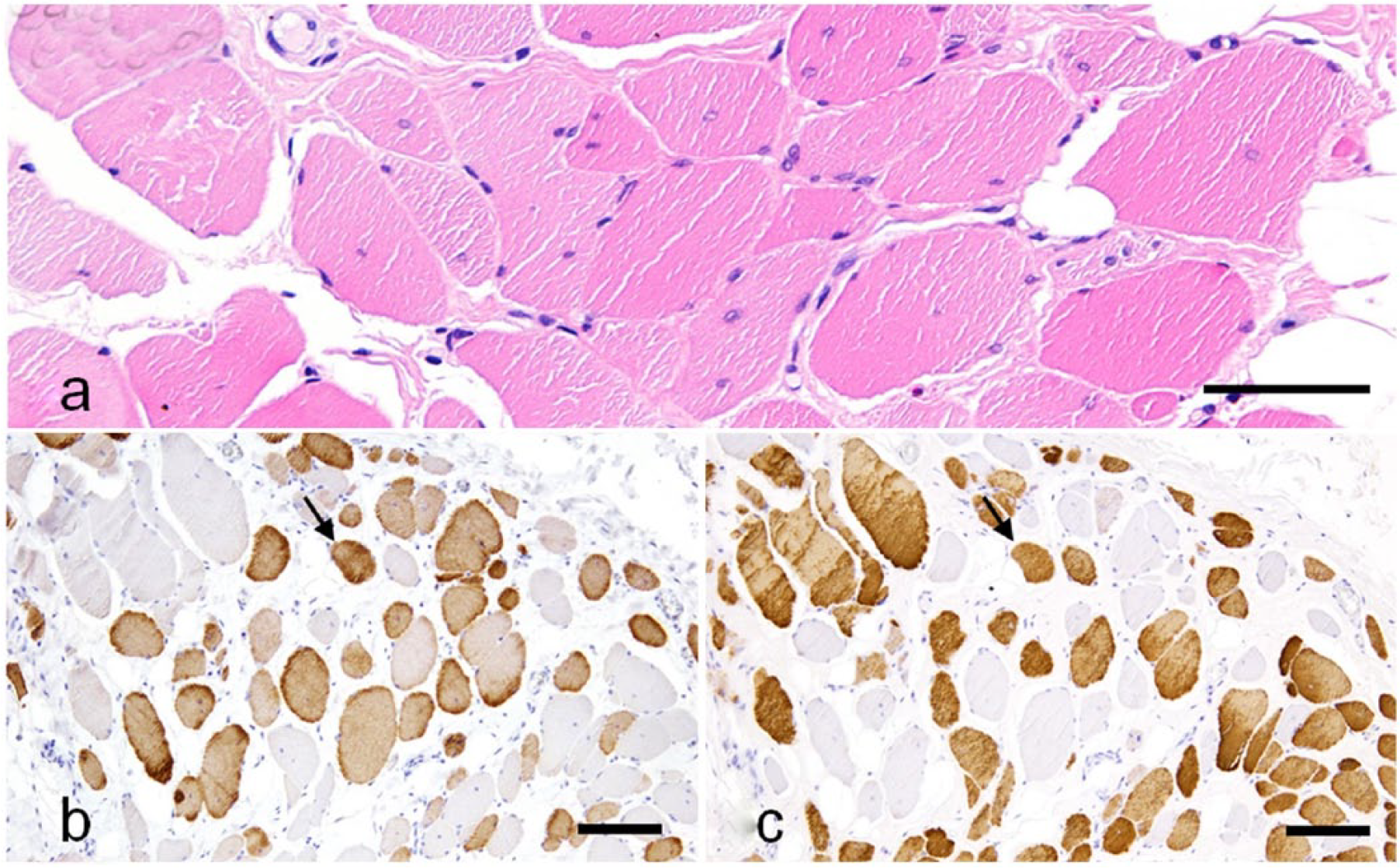
Myogenic changes in the triceps brachii of a cat (case 9) with muscular dystrophy.
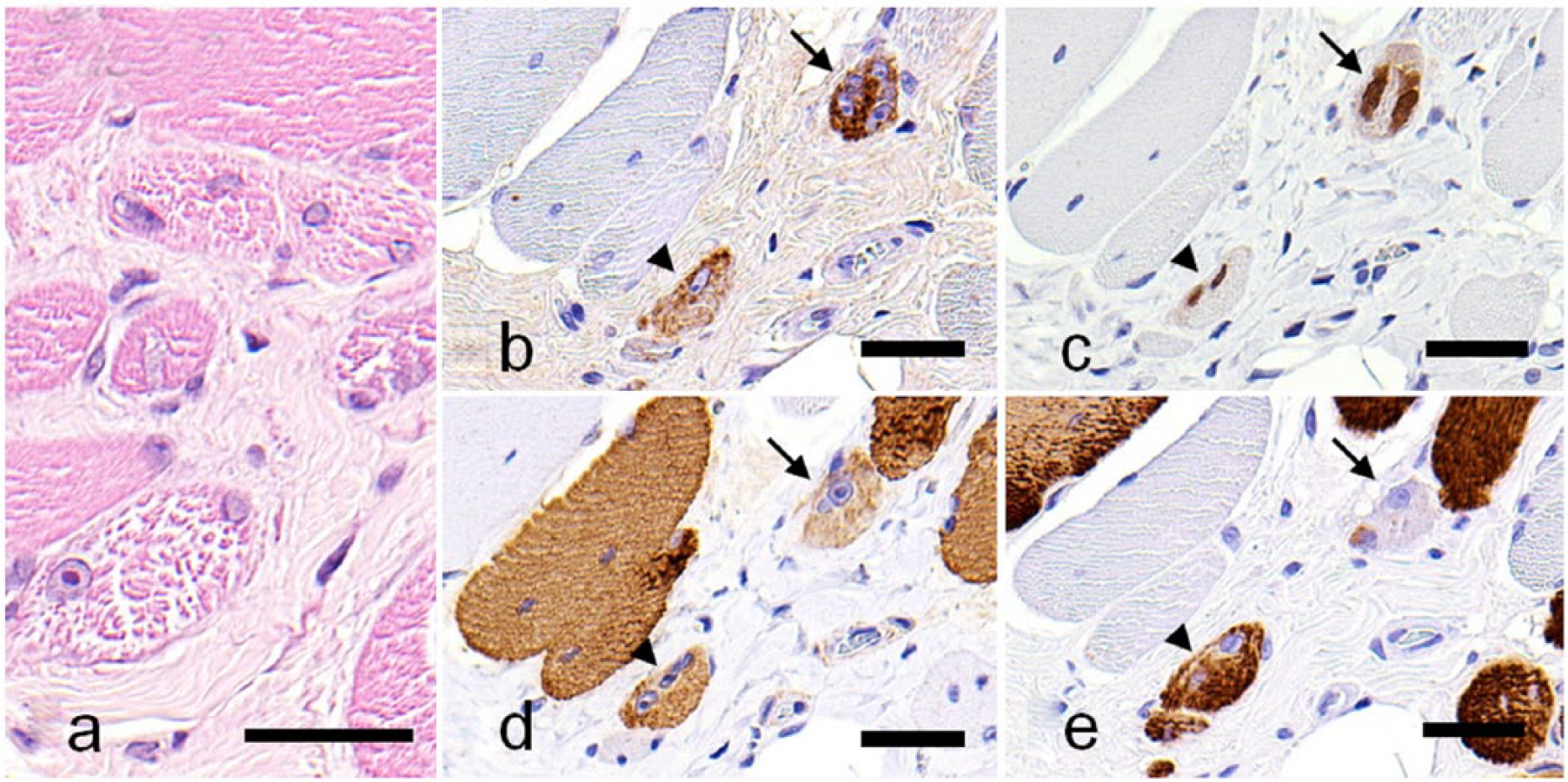
Myogenic changes in the triceps brachii of a cat (case 9) with muscular dystrophy.
Discussion
DM has been identified in dogs including Pembroke Welsh Corgis,8,18 and peripheral neuropathy is a feature of late-stage DM. 30 Polyneuropathy has been described in Alaskan Malamutes.6,26 In canine cases diagnosed as neurogenic muscle disorders, we detected the axonal loss only in case 1. Although demyelination alone does not result in denervation, axonal degeneration is thought to have existed in nerve tissue samples not examined in other cases.
Immunohistochemical analysis of muscle biopsy is a powerful method for the identification of a protein deficiency and the diagnosis of muscular dystrophy in animals, but it usually requires frozen sections.22,24,32,40,41 Our results suggest that the commercial dystrophin antibody can provide reliable staining in canine sections. However, this conclusion should still be considered preliminary because frozen muscle sections of our dystrophin-deficient muscular dystrophy cases could not be obtained. The anti–laminin α2 antibody did not exhibit immunoreactivity against canine and feline formalin-fixed tissues and thus it is not part of the study.
The present immunohistochemical method using paraffin-embedded sections and antibodies to slow and fast myosins has the ability to detect the normal mosaic or random pattern of type 1 and type 2 and angular and group atrophy involving both type 1 and type 2 fibers, and fiber type grouping of muscle fibers in dogs and cats instead of ATPase histochemical staining. Because commercial antibodies to human fast myosin did not react with the canine counterpart, we developed a new antibody against canine fast myosin.
An essential feature of denervation is angular and group atrophy involving both type 1 and type 2 fibers. Fiber type grouping is considered pathognomonic of denervated muscle,12,13 although it can be observed in some myopathies. 17 Angular and group atrophy of type 1 and type 2 fibers were definitive for the diagnosis of neurogenic muscle disorders in canine and feline cases. Fiber type grouping was also pathognomonic in canine cases, although it was not obvious in all cases. No feline case in our study showed this lesion, which may have been the result of disease staging. The immunohistochemical method used in our study is expected to reveal fiber type grouping in cats. Hybrid fibers (type IIC) are immature, regenerating, or remodeling (i.e., conversion of the fiber type during re-innervation) fibers.12,13 The hybrid fibers observed in case 1, which was classified as a neurogenic muscle disorder, may be in the remodeling process, suggesting re-innervation occurring in myofibers in addition to the lesion of group atrophy. The hybrid fibers observed in case 9, which was classified as myopathy, may be degenerating or regenerating.
Iba-1 antibody was useful to identify macrophages in canine formalin-fixed skeletal muscle samples. Other antibodies, such as CD markers that recognize canine and feline macrophages, might also be useful. 10 The expression of nestin or myogenin in myoblasts and regenerating fibers has been reported in human neuromuscular diseases.33,38,43 Similar expression patterns for the markers in myoblasts and regenerating fibers were observed in dogs and cats in our study. Nestin-positive myofibers with myogenin-positive nuclei were scattered throughout fascicles in canine and feline cases of myopathies. These were interpreted as regenerating fibers. On the other hand, the diameter of nestin-positive fibers with myogenin-positive nuclei was small, and these fibers were only observed in the lesion of large-group atrophy in neurogenic disorders. Pyknotic nuclear clumps were immunopositive for myogenin in cases 4 and 7 with neurogenic muscle disorders. Although the expression of nestin and myogenin is known to be up-regulated in denervated fibers,19,20,36,39 we were unable to clarify whether the expression of nestin and myogenin resulted from regeneration or denervation in neurogenic disorders.
Footnotes
Acknowledgements
We thank Dr. S Okuno for providing samples of canine and feline skeletal muscles and nerve tissues.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
