Abstract
Acanthomatous ameloblastoma is a common, locally invasive, nonmetastasizing tumor of the canine oral cavity. The long-term prognosis for canine acanthomatous ameloblastoma is good if complete excision can be achieved, usually by maxillectomy or mandibulectomy. A variant of acanthomatous ameloblastoma with atypical foci was noted in 5 dogs. There was no age, breed, or sex predisposition. Atypical cells were pleomorphic with a high mitotic rate. They were immunohistochemically negative for cytokeratin, vimentin, melan A, PNL2, CD3, Pax5, CD18, chromogranin A, and synaptophysin. Ultrastructurally, the atypical cells had modest amounts of electron-lucent cytoplasm, abundant rough endoplasmic reticulum, zonula adherens junctions, cleaved or irregular nuclei, and occasional cytoplasmic structures consistent with secretory granules or lysosomes. Complete excision was achieved by maxillectomy or mandibulectomy in 3 dogs; the lesion was incompletely excised in 2 dogs. No ancillary therapy was elected in any patient. No local recurrence or distant metastasis was reported in any case. One patient died of heart failure 20 mo following complete excision; all other patients were alive at last follow-up (average follow-up: 18.8 mo, range: 6–30 mo). The histogenesis of the atypical foci is unclear, but atypical foci within acanthomatous ameloblastoma do not appear to be associated with metastasis or with a poor prognosis relative to acanthomatous ameloblastoma with typical histologic morphology.
Introduction
Acanthomatous ameloblastoma is a common tumor arising from rests of odontogenic epithelium in the submucosa and periodontal ligament of the oral cavity. These tumors are locally invasive and destructive, resulting in lysis of alveolar bone and loss of teeth. Because these tumors are so infiltrative, local recurrence is common following incomplete excision, and recommended therapy typically includes maxillectomy or mandibulectomy. 13 If complete excision can be achieved, the prognosis is good, with 98–100% of dogs having no signs of recurrence 1–2 y after complete excision.5,11 The disease-free interval following complete excision has been previously reported as 22.5 mo. 5 Radiation therapy has been advocated in cases in which complete excision cannot be achieved because of the size or location of the tumor, 7 but radiation has been associated with novel oncogenesis at the irradiated site.8,12,14 Importantly, acanthomatous ameloblastoma is not known to metastasize. 15
A variant of acanthomatous ameloblastoma with atypical foci was identified in 5 canine biopsy submissions to the Colorado State University Veterinary Diagnostic Laboratory (CSU-VDL; Fort Collins, CO). The pleomorphic features of cells within atypical foci raised concern for poor clinical outcome in these patients. Herein we report the clinical, histologic, and immunohistochemical features of 5 acanthomatous ameloblastomas with atypical foci. Two cases were additionally evaluated by electron microscopy.
Material and methods
Case identification
The electronic database of the CSU-VDL was queried for canine biopsies diagnosed with acanthomatous ameloblastoma and atypical features. Cases were included in our study if formalin-fixed, paraffin-embedded tissue was available for immunohistochemistry (IHC); the histologic diagnosis was confirmed as acanthomatous ameloblastoma with atypical foci by the authors. Atypical foci were defined as nests or aggregates of large round-to-polygonal cells with modest amounts of cytoplasm and large nuclei with prominent nucleoli (see Results for additional histologic details). Initial biopsies consisted of excisional (patient 2) or incisional specimens (patients 1, 3–5). Additional tissues representing excisional specimens were available for patients 1 and 3. Histopathology submission forms were reviewed to collect signalment information.
Histopathology
Tissue specimens from each case were fixed in 10% neutral buffered formalin for >24 h and then demineralized if necessary in hydrochloric acid solution a for 24–72 h. Trimmed specimens were routinely processed, sectioned at 5 μm, collected on charged slides, and stained with hematoxylin and eosin for evaluation by light microscope. Initial diagnosis of acanthomatous ameloblastoma with atypical foci was made through histologic evaluation of tissue specimens by a Diplomate of the American College of Veterinary Pathologists.
Immunohistochemistry
Select sections from all patients were labeled immunohistochemically with mouse monoclonal antibodies raised against cytokeratin, b vimentin, c melan A, d PNL2, e cluster of differentiation (CD)3, f Pax5, g chromogranin A, h and synaptophysin i following heat-induced epitope retrieval submersion in citrate buffer (pH 6.0) j on an automated staining platform. k Additional sections were labeled with mouse monoclonal anti-canine CD18 l following enzyme pretreatment. m Positive immunohistochemical controls consisted of canine skin (for cytokeratin, vimentin), a canine melanocytic tumor (for melan A, PNL2), normal canine lymph node (for CD3, Pax5, CD18), and canine adrenal gland (for chromogranin A, synaptophysin) incubated with primary antibodies as described. Primary antibodies were replaced with homologous nonimmune sera for negative controls.
Electron microscopy
Tissue blocks from patients 4 and 5 were sent to the University of Georgia (Athens, Georgia) for electron microscopy. Select tissues were deparaffinized and embedded in plastic. Thick sections were prepared and stained with toluidine blue. Areas containing typical odontogenic epithelium and atypical cells were chosen for thin sections, which were prepared with uranyl acetate and lead citrate and examined with a transmission electron microscope.
Case follow-up
Follow-up information was acquired from the primary care veterinarian by phone in each case. Follow-up information included data on local recurrence, distant metastasis, whether additional therapy had been elected (repeat surgery, chemotherapy, or radiation), and ultimate outcome including date of death, if relevant.
Results
Case presentations
Five cases fit the inclusion criteria of the study (Table 1). These 5 cases represented 0.3% of all histologic diagnoses of canine acanthomatous ameloblastomas diagnosed during the 10-y search period. The average age of affected patients was 7 y (range: 4–10 y). There was no sex or breed predisposition. Lesions arose as mass effects of the mandible (2 of 5) or maxilla (3 of 5). In patient 4, the maxillary mass also involved the hard palate with extension into the nasal cavity. Dental radiographs performed in 2 cases (patients 2 and 3) revealed markedly cystic mass effects that displaced adjacent teeth and resulted in osteolysis of the boney arcade, findings suggestive of acanthomatous ameloblastoma.
Signalment of patients with acanthomatous ameloblastoma with atypical foci.*

FS = female spayed; MC = male castrated.
Died of unrelated heart failure.
Histopathology
In each case, the submucosa was expanded by an infiltrative mass of neoplastic odontogenic epithelial cells arranged in thin anastomosing cords and trabeculae supported by variable amounts of periodontal stroma (Fig. 1). Neoplastic odontogenic epithelial cells at the periphery of cords and trabeculae were columnar with palisading morphology and apically placed nuclei, whereas cells located centrally within cords and trabeculae were polygonal with prominent intercellular junctions. The cells had modest amounts of eosinophilic cytoplasm and oval nuclei with stippled chromatin and 0–1 punctate nucleoli. There was mild anisocytosis and anisokaryosis in this population, and mitotic figures were rare (0–1 per 10 high powered fields [hpf], 400×). In many specimens, neoplastic epithelial cells invaded and replaced alveolar bone.
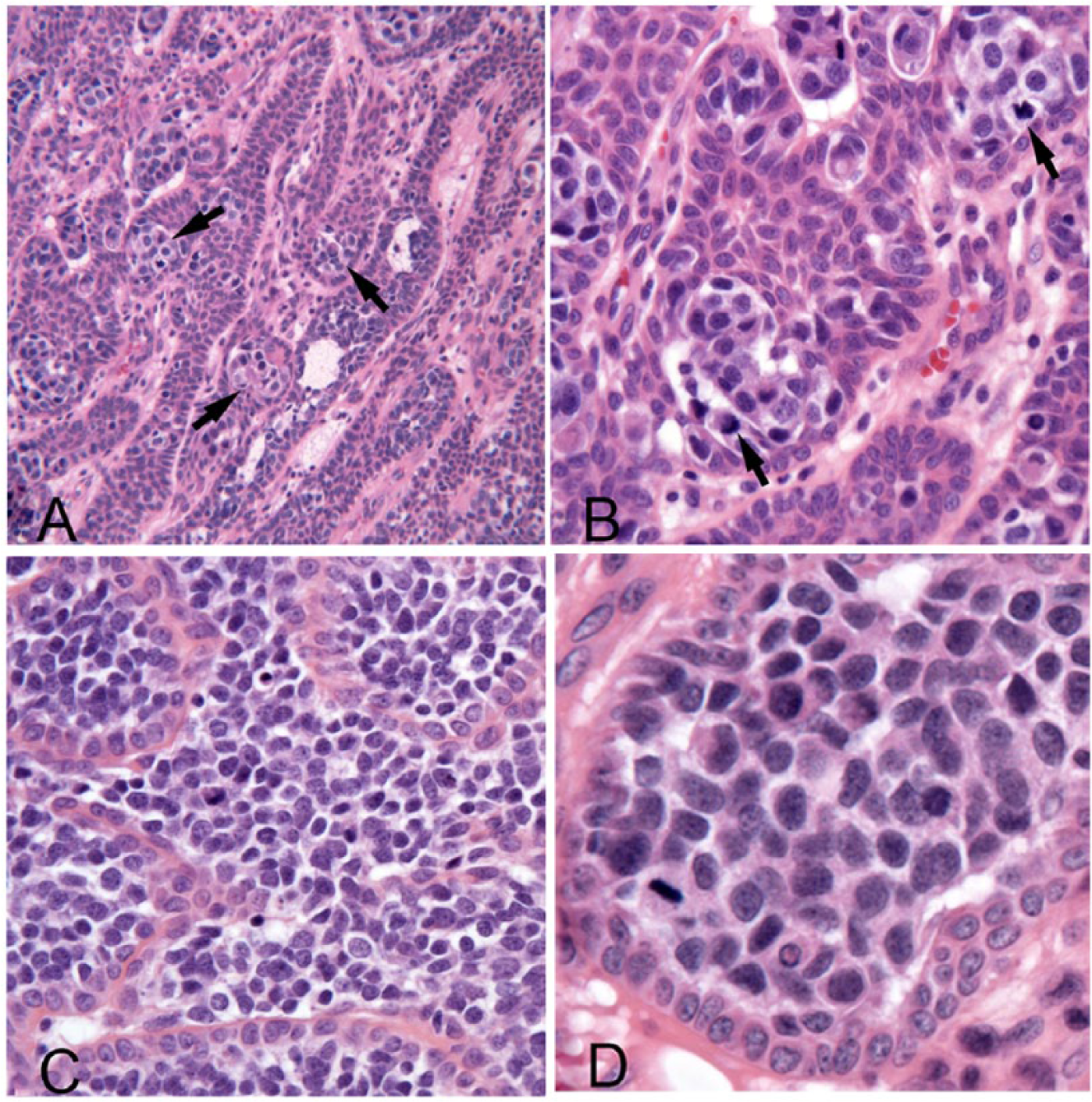
Acanthomatous ameloblastoma with atypical foci in a dog.
Scattered within the typical population of odontogenic epithelial cells were variably sized nests and aggregates of atypical cells. These cells were round-to-polygonal with variably discrete cell borders and sparse-to-modest amounts of finely granular basophilic cytoplasm. Cell nuclei were large (often 30 µm or more) with finely stippled chromatin and occasional prominent nucleoli. There was moderate anisocytosis and anisokaryosis in this population, and mitoses were very common (up to 8 per single hpf, 400×). In some fields, atypical cells predominated and formed sheets that replaced the central regions of cords and trabeculae and extended into the adjacent stroma (Fig. 1C).
Immunohistochemistry
In all 5 cases, typical neoplastic odontogenic epithelium demonstrated modest-to-strong cytoplasmic immunoreactivity for cytokeratin and diffuse negative immunoreactivity for vimentin, whereas cells within atypical foci were negative for both cytokeratin and vimentin (Fig. 2A, 2B). Stromal elements such as fibroblasts and endothelial cells were positive for vimentin (internal positive control). In all cases, the typical odontogenic epithelial cells and atypical cells were diffusely negative for melan A, PNL2, CD18, CD3, Pax5, chromogranin A, and synaptophysin (not shown). Small numbers of CD3- and Pax5-positive mature lymphocytes were occasionally present in the supporting stroma (internal control).
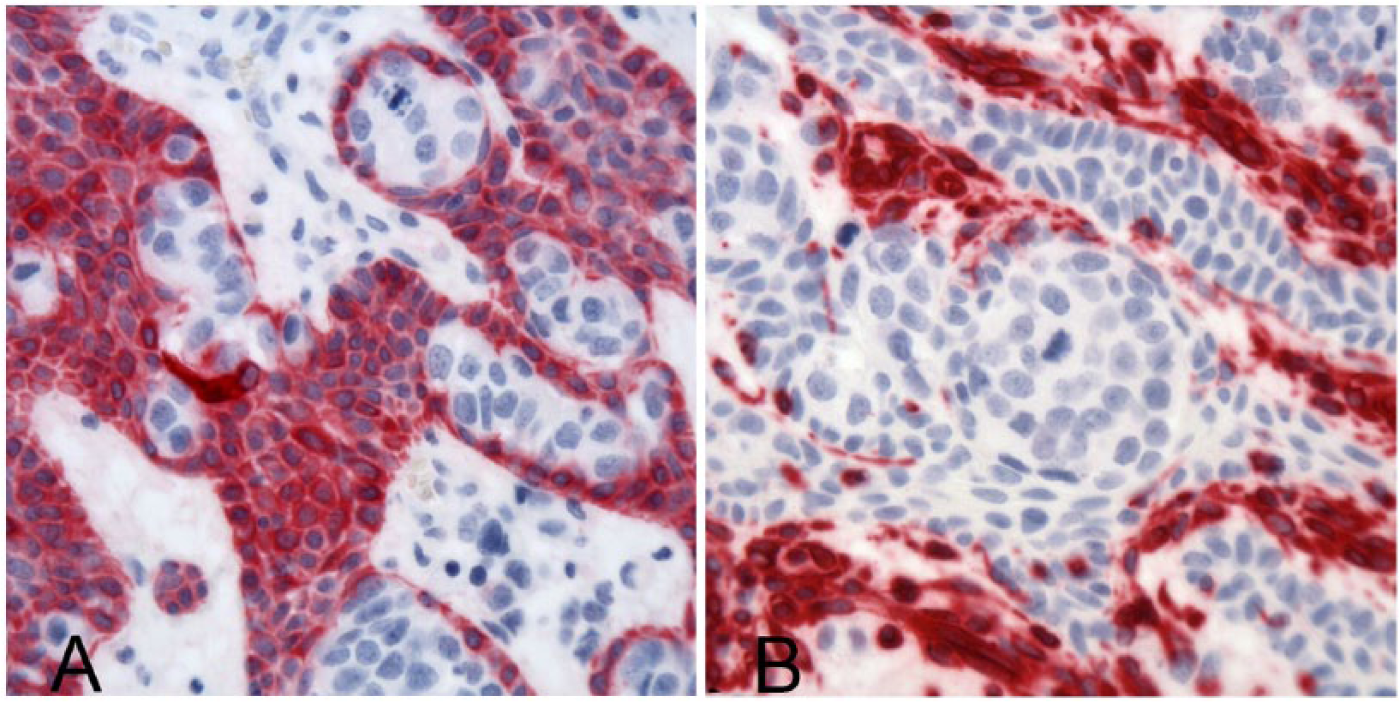
Acanthomatous ameloblastoma with atypical foci in a dog.
Electron microscopy
Typical neoplastic odontogenic epithelial cells (Fig. 3A) were cuboidal or polygonal with discrete cell borders, moderately electron-dense cytoplasm, and many haphazard tonofilaments. These cells had numerous intercellular bridges with desmosomes, and the peripheral palisading cells rested on a basement membrane. Atypical cells were round with indistinct cell borders and more electron-lucent cytoplasm lacking tonofilaments. Nuclei of atypical cells were large and round, irregular, or cleaved with clumped chromatin. Atypical cells had abundant rough endoplasmic reticulum (rER) and junctions most consistent with zonula adherens type (Fig. 3B). Atypical cells contained occasional spherical electron-dense cytoplasmic structures consistent with secretory granules or lysosomes (Fig. 3C).
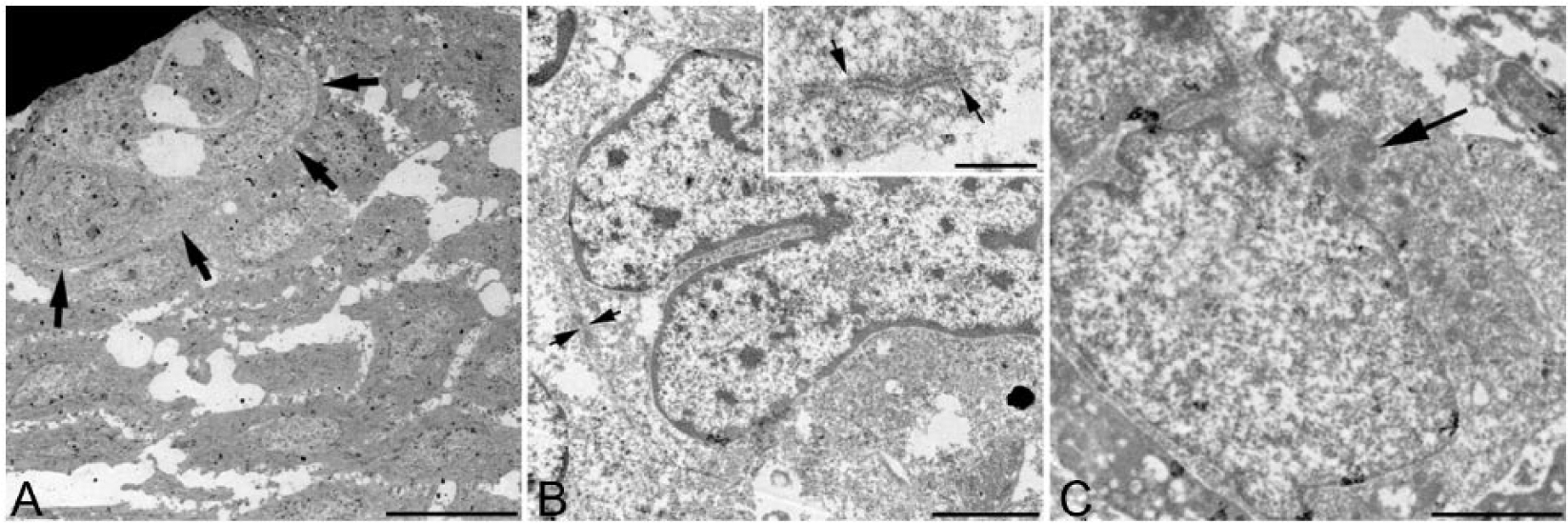
Transmission electron microscopy, uranyl acetate and lead citrate.
Case outcome
Complete excision was achieved in patients 1–3 by mandibulectomy or maxillectomy. Complete excision was not achieved in patient 5 and could not be achieved in patient 4 because of the rostral location of the mass and extension of the neoplastic tissue into the hard palate and nasal cavity. Additional therapy, such as chemotherapy or radiation, was not elected in any of the patients. No patient experienced local recurrence, and metastasis was not reported. Patient 2 died of unrelated heart failure 20 mo following diagnosis and complete excision. All other patients were alive at the time of follow-up with no reported complications or progression of disease. Altogether, the patients had an average follow-up time of 18.8 mo (range: 6–30 mo).
Discussion
Typical canine acanthomatous ameloblastomas are locally invasive tumors of odontogenic epithelium that have a good prognosis following complete excision. Herein we report the features of 5 acanthomatous ameloblastomas with atypical foci. The pleomorphic nature of the atypical cells and increased number of mitoses in this population raised concern for metastasis or poor outcome. However, with the exception of 1 patient who died of unrelated causes 20 mo following complete excision, these patients were alive with no evidence of local recurrence or metastasis documented 6–30 mo after initial diagnosis. Although specific tests to rule out metastasis (e.g., fine-needle aspiration of local lymph nodes, thoracic radiographs, abdominal ultrasound) were not pursued in most patients, and it is possible that metastatic disease may have occurred without detection, the prolonged survival of multiple patients with no relevant clinical signs following surgical excision makes the possibility of metastatic spread very unlikely, especially in cases that were followed for >1 y.
The histogenesis of the atypical cell population arising within acanthomatous ameloblastoma is not clear. Based on histologic morphology, initial considerations included possible collision tumor between a typical acanthomatous ameloblastoma and melanosarcoma, other sarcoma, or round cell tumors. However, the multifocal distribution of atypical foci is not typical for a collision event. The atypical population was immunonegative for vimentin, melan A, and PNL2, making melanocytic origin highly unlikely. The atypical cells were additionally immunonegative for CD18, CD3, and Pax5, suggesting that a histiocytic or lymphocytic histogenesis is unlikely. Although neuroendocrine origin was considered based on the ultrastructural presence of spherical electron-dense cytoplasmic structures (possible secretory granules), the atypical cells were immunonegative for chromogranin A and synaptophysin. Manifestation of epithelial-to-mesenchymal transition was also considered, but this is not supported by the negative immunoreactivity for vimentin.
Unfortunately, little published information is available regarding the ultrastructural features of normal and neoplastic dental and periodontal tissues in dogs. The cases reported herein have ultrastructural features similar to those reported for ameloblastomas in humans. 1 The typical odontogenic cells in these cases rested on a basement membrane and contained tonofilaments, features that are characteristic of ameloblasts in the maturation stage of amelogenesis (i.e., ruffle-ended and smooth-ended ameloblasts), as described in other species. 3 However, other than abundant rER, cytoplasmic granules, and cell junctions, the atypical cells did not have any distinguishing features to indicate origin. Given that these atypical cells are arising within odontogenic epithelium and contain abundant rER and suspected secretory granules, it is considered possible that atypical foci represent ameloblasts in the presecretory or secretory stage of differentiation. 3 However, one would expect such cells to be cytokeratin positive, 4 and these cells were immunonegative for this marker. We also considered that these could be sites of induced odontoblastic differentiation, specifically secretory odontoblasts based on the suspected secretory material, but such cells typically contain thick collagen fibers (von Korff fibers), which were not identified in these cases. 2 Also, odontoblasts are typically immunoreactive for vimentin and certain cytokeratins,4,6 and the atypical cells in our cases were immunonegative for these markers. Odontocytes are also known to have very limited oncogenic potential with no reports of neoplasia or pre-neoplastic changes. 9 Another consideration was that these atypical cells could be analogous to the “clear cells” reported in a number of human odontogenic tumors; however, such cells contain glycogen ultrastructurally and are immunopositive for cytokeratin, thus making this unlikely. 10
Importantly, acanthomatous ameloblastomas with atypical foci do not appear to be associated with a shorter survival time relative to acanthomatous ameloblastomas with typical histologic morphology. Our cases suggest that complete excision alone (by mandibulectomy or maxillectomy) may be curative for canine acanthomatous ameloblastomas that have atypical foci. Indeed, 2 incompletely excised tumors (patients 4 and 5) failed to demonstrate recurrence after 6 and 12 mo, respectively, perhaps suggesting less aggressive oncologic behavior. However, the sample size reported herein is small, and complete excision with follow-up may be warranted for patients until the biologic behavior of this tumor variant is more widely studied and understood.
Footnotes
Acknowledgements
We thank Drs. Jennifer Boyle, Corrine Durand, David Carmichael, and Scott Carpenter for providing follow-up data. Thanks also to Mr. Todd Bass (CSU VDL) for assistance with immunohistochemistry and Mrs. Mary Ard (UGA) for sample handling and processing for ultrastructural analysis.
Authors’ contributions
JL Malmberg, BE Powers, and PA Schaffer contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. EW Howerth contributed to acquisition, analysis, and interpretation of data. JL Malmberg and PA Schaffer drafted the manuscript. EW Howerth, and PA Schaffer critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Surgipath Decalcifier II (3800400), Leica Biosystems, Buffalo Grove, IL.
b.
CK AE1/AE3 (PA0909), Leica Microsystems, Buffalo Grove, IL.
c.
VIM SRL33 (PA033), Leica Microsystems, Buffalo Grove, IL.
d.
Melan A A103 (PA0233), Leica Microsystems, Buffalo Grove, IL.
e.
Pnl2 (N1601), Dako North America, Carpinteria, CA.
f.
CD3 (LN10), Leica Biosystems Newcastle, Newcastle Upon Tyne, United Kingdom.
g.
Pax5 (610863), BD Biosciences, San Diego, CA.
h.
Sp-1 chromogranin A (20085), ImmunoStar, Hudson, WI.
i.
Synaptophysin (PA0299), Leica Microsystems, Buffalo Grove, IL.
j.
Target retrieval solution (pH 6.0), Dako North America, Carpinteria, CA.
k.
Bond-Max, Leica Biosystems Newcastle, Newcastle Upon Tyne, United Kingdom.
l.
CD18 CA16.3C10-IgG1, Dr. Peter Moore, School of Veterinary Medicine, University of California–Davis, Davis, CA.
m.
Enzyme 1, Leica Biosystems, Buffalo Grove, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
