Abstract
Endosulfan, a highly toxic insecticide and acaricide, was globally banned under the Stockholm Convention in May 2011 because of its threats to human health and the environment. Despite the ban on its use, endosulfan is still present in the environment, causing poisoning events in domestic animals and wildlife. Toxicologic analysis of poisoned animals conducted by the Istituto Zooprofilattico Sperimentale del Mezzogiorno revealed that in the past 3 y, this pesticide has not disappeared in the regions of interest. More than 650 samples (baits and animal organs) collected from 2013 to 2015 were analyzed through solid-phase extraction followed by gas chromatography–electron capture detection. This methodology allowed us to identify 45 positive samples from the Calabria region of southern Italy (10.2 % of the total samples). Most were samples of gastric content (alpha-endosulfan range: 1.27–3,800 mg/kg) and baits (alpha-endosulfan range: 92–1,497 mg/kg). Lower concentrations of pesticide were found in samples of stomach (alpha-endosulfan: 6.7–118 mg/kg) and liver (alpha-endosulfan: 0.38–14.2 mg/kg).
Keywords
The “persistent organic pollutant” endosulfan is a broad-spectrum organochlorine insecticide first registered for use in 1954 and widely applied to crops, particularly in the developing world. Given its potential to evaporate and travel long distances in the atmosphere, endosulfan has become one of the most widespread pollutants in the world and is found extensively in all ecosystems. This colorless solid has emerged as a highly controversial agrichemical because of its acute toxicity, endocrine effects, and potential for bioaccumulation. It can enter the human body by inhalation, ingestion, and through skin contact because it is liposoluble. Endosulfan has also been observed to have an estrogenic effect in humans.2,11 Clinically, organochlorines cause intense nerve stimulation and hypersensitivity in both humans and animals. An exposed animal becomes hyperesthetic and exhibits tremors and convulsions. Treatment is symptomatic and supportive; the prognosis can be considered good, depending on the exposure dose and the timeliness of decontamination (German Federal Environment Agency–Umweltbundesamt (GFEA-U), Berlin. Endosulfan. Dossier prepared in support of a proposal of endosulfan to be considered as a candidate for inclusion in the Annex I to the protocol to the 1979 Convention on Long-Range Transboundary Air Pollution on Persistent Organic Pollutants (LRTAP Protocol on POPs). February 2007. Available at: https://goo.gl/GQ2RA7).4,10 Although banned in 2006 in more than 50 countries, including the European Union and several Asian and West African nations, endosulfan was still used extensively in many other countries. After many studies indicated its strong toxicity with low-dose exposure, it was banned globally at the Stockholm Convention in May 2011.
As a highly toxic substance, endosulfan is lethal to domestic animals and wildlife, particularly birds, fish, and other aquatic organisms. Insecticides were found to be the primary cause of suspected poisonings reported in Italy and in other European countries (Barbier N. Bilan d’activité du Centre National d’Informations Toxicologiques Vétérinaires pour l’année 2003 [Annual activity report of the National Center for Veterinary Toxicological Information for 2003]. PhD Thesis. Veterinary Sciences. Lyon, France: Université Claude-Bernard-Lyon I, 2005. French).1,3 The use of bait is a serious risk for animals, public health (particularly for children), and the environment, as endosulfan from the bait could enter the food chain or rivers, lakes, and other waterways. In intentional and accidental poisoning episodes involving domestic and wild animals, endosulfan is the most frequently reported organochlorine pesticide.6,7
Between 2013 and 2015, more than 50 baits and 600 suspected poisoned organs were sent to the Istituto Zooprofilattico Sperimentale del Mezzogiorno, as the official control laboratory of the Campania and Calabria regions, for diagnostic and toxicologic analysis. We analyzed tissue samples, such as liver, stomach, and gastric contents, and baits, both received from veterinarians (75%) and from animal owners (25%). Our study describes the identification of 45 endosulfan poisonings through testing of samples collected from the Calabria region.
The analytic methodology used, developed, and validated in our laboratory, combines conventional solid-liquid extraction of the organochlorine pesticide from the sample with gas chromatography–electron capture detection (GC-ECD). A homogenized sample (0.1 g) in a 250-mL flask was treated with 100 mL of diethyl ether for 24 h. The organic phase was then filtered by gravity using a paper filter
a
(90 mm diameter) supported by a glass funnel. Anhydrous sodium sulfate was added and evaporated to dryness using a rotary evaporator at a temperature of ~40°C. The residue was dissolved in 3 mL of
The extract was dried completely under a stream of nitrogen, and the dried residue was dissolved in 1 mL of isooctane. One milliliter of a 1:10 dilution of sample in isooctane was transferred to a 2-mL vial for further GC analysis. Blank samples for the optimization of the method were selected from tissue samples of animals testing negative for endosulfan in a previous analytic investigation. Fortified samples were prepared by spiking with a concentration of 50 µg/L. Prior to the extraction, a blank liver sample (500 mg) with a 100 mg/L standard solution was used as a control. A mixed solution of 100 mg/L of all endosulfan stereoisomers was prepared in isooctane d by using a 10-mL volumetric flask; this solution was stable in an amber flask for 4 m at 4°C and was checked periodically. e In the analytic procedure, 3 dilutions (10, 20, 50 ng/L) of organochlorine pesticide were used to evaluate the concentration of the endosulfan in the specimen. Chromatographic analysis was performed on a commercial GC system f coupled with an ECD and software g for data capture and analysis. The GC system was equipped with a 30 m × 0.25 mm inner diameter × 0.25 µm column, h and the analysis was performed using an injector temperature of 250°C, a detector temperature of 380°C, a flow rate of 3.0 mL/min for the carrier gas helium, 57 mL/min (ECD) for nitrogen, 3.0 mL/min for hydrogen, and 60 mL/min for air. The oven initial temperature was programmed at 100°C, increased to 220°C at 10°C/min, then to 270°C at 4°C/min with a wait of 13.50 min. Endosulfan was detected as a mixture of 2 stereoisomers, with a retention time of 15.39 min for alpha and 16.99 min for beta (Fig. 1). If positive peaks of a pesticide were detected, the results were confirmed by GC-ECD analysis using a different polarity column i (30 m × 0.25 mm inner diameter × 0.25 µm), an oven initial temperature of 100°C, increased to 270°C at 10°C/min, then to 300°C at 5°C/min with a wait of 2 min.
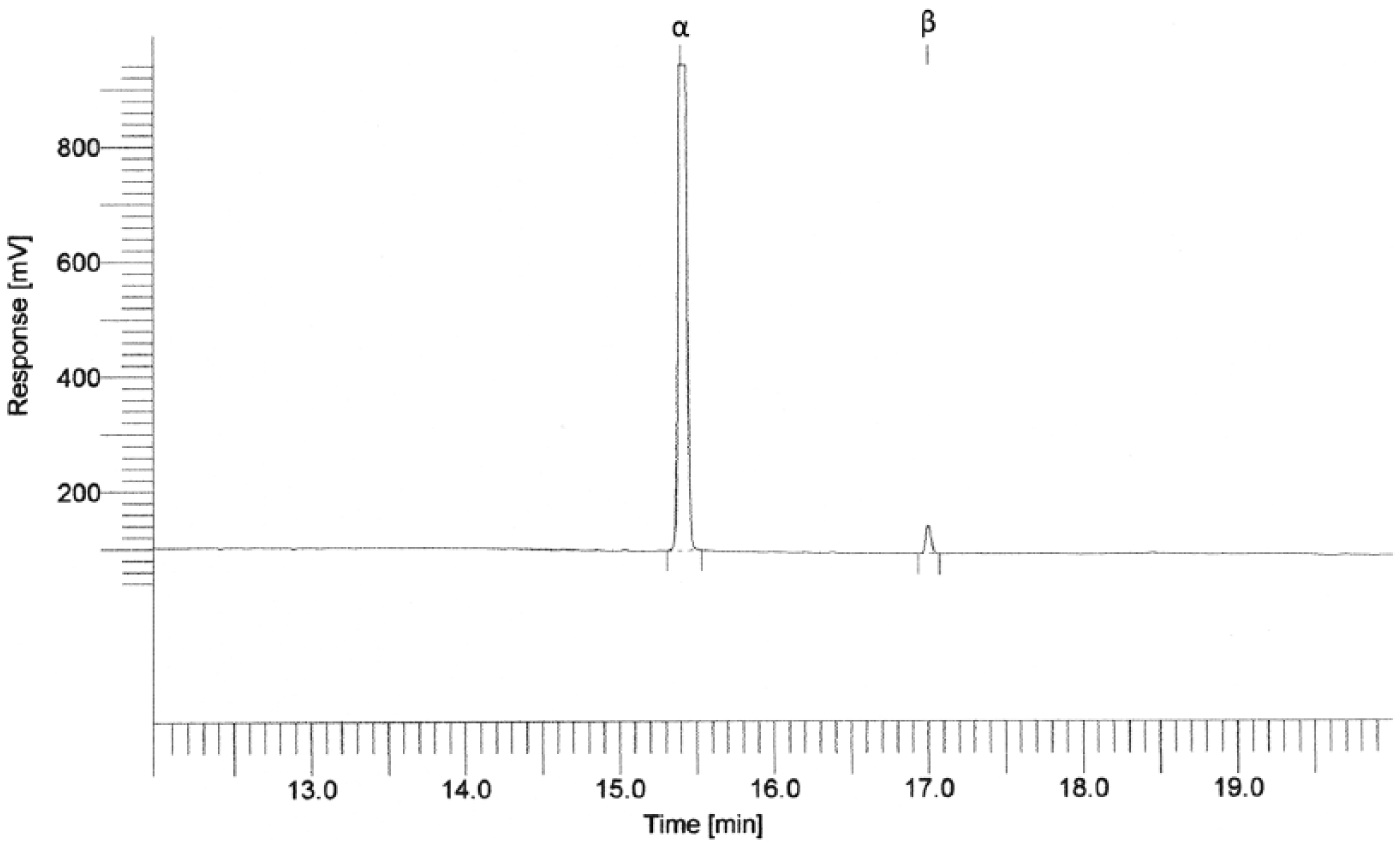
Gas chromatography–electron capture detection of alpha- and beta-endosulfan in a sample of gastric content.
Although the majority of samples tested negative for the presence of the organochlorine pesticide of interest, we confirmed the presence of endosulfan in 45 of 440 samples (10.2%), which had been collected from the Calabria region of southern Italy (Table 1). According to the data recorded from 1996 to 2003 in Italy, 6 dogs were the most commonly poisoned animal (75.5% of calls), followed by cats (6.6%), and foxes (2.2%). The occurrence of fox mortality is indicative of how this substance is also lethal to wildlife. Among the positive samples, 13.3% were poisoned baits; their presence confirms the continued use of endosulfan in intentional poisoning episodes. The majority were samples of gastric content (27; 6%) and stomach (9; 2%). Only 3 were positive samples of liver (6.7%), and the concentration of the pesticide ranged from 0.38 to 14.2 mg/kg for alpha-endosulfan, indicating that liver is not a matrix of choice for the detection of this pesticide.
Endosulfan-positive samples from the Calabria region of Italy with analysis requester, species of poisoned animal, organ, and the amount of alpha- and beta-endosulfan detected.*
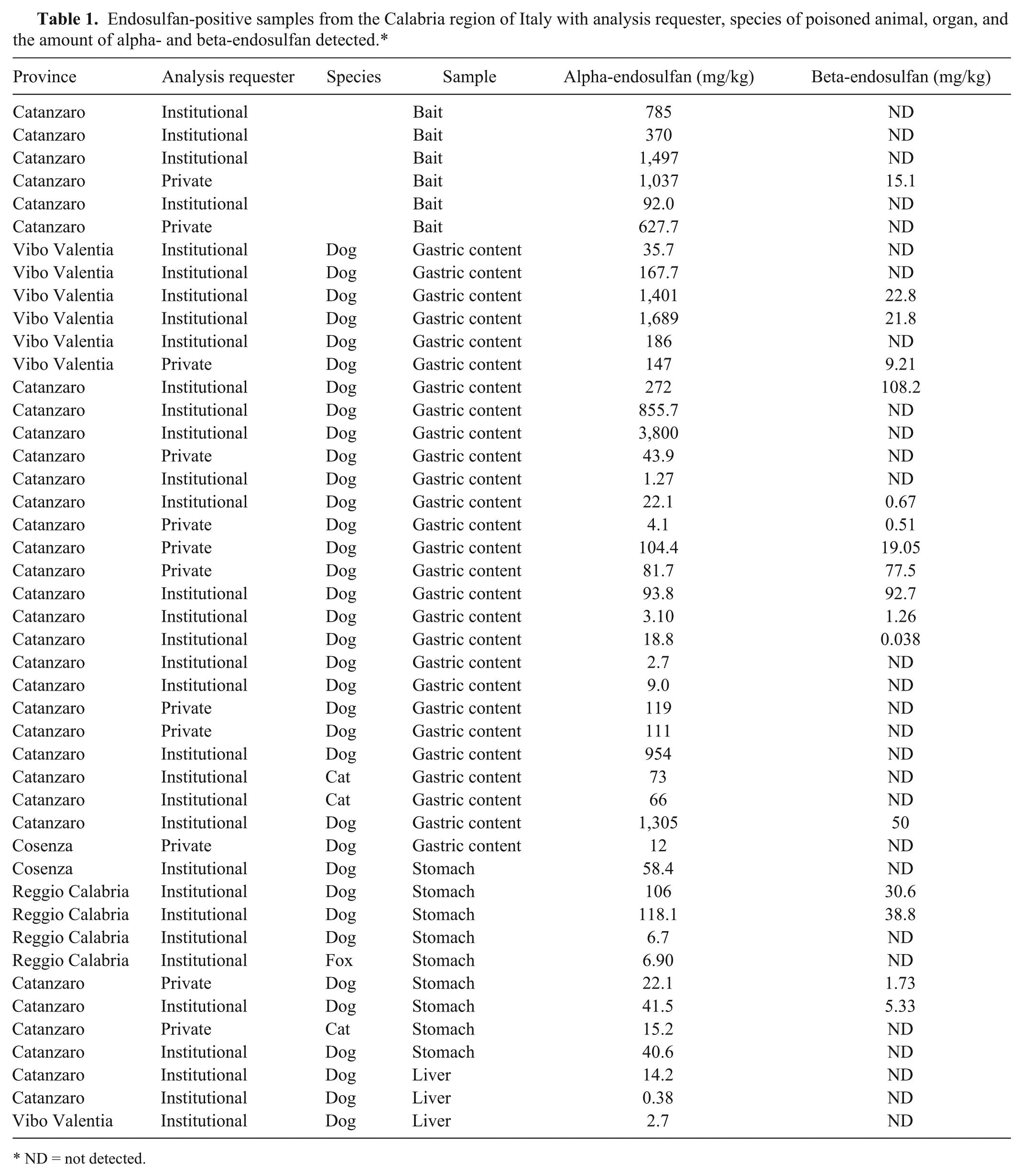
ND = not detected.
In the technical grade form, endosulfan is a mixture of 2 stereoisomers, with an alpha-to-beta ratio of 70:30. 9 Endosulfan is metabolized and excreted from the human and animal body as metabolites such as endosulfan sulfate, alcohol, ether, and lactone. 8 The 2 isomers are also known popularly as endo- and exo- or I and II. We only observed both isomers in 17 samples, and the tissue accumulation of the beta-isomer was always lower than that of the alpha (Table 1). In fact, the presence of beta-endosulfan ranged from 0.038 to 108.2 mg/kg and was identified in 2 samples of gastric content of dogs from Catanzaro. The amounts of alpha-endosulfan ranged from 0.38 mg/kg (dog liver) to 3,800 mg/kg (dog gastric content). Both these dogs were from Catanzaro, which appears to be the area with the greatest number of poisoning episodes.
Samples identified as baits and unidentified material were reported by the finders to have been illegally prepared and laid out in open lands, along paths, or thrown over fences into private gardens. The intent to poison was established for each case according to information provided by the owner or wildlife rehabilitators, and if there was bait or massive amounts of the toxicant in the digestive tract of the animal.
The data analyzed by the Istituto Zooprofilattico Sperimentale del Mezzogiorno confirm that, in the past 3 y, the trend of poisoning continues unabated in the Calabria region. Even though the use of this organochlorine had been gradually restricted and then completely banned, it is still being used for the illegal poisoning of animals. The source of the endosulfan used today is unclear; it could be from old drug stocks of chemists or veterinarians or purchased from online shops that carry agrochemicals containing this pesticide. According to the European Commission Regulation No. 519 of 2012 amending Regulation (EC) No. 850/2004 of the European Parliament and of the Council on persistent organic pollutants as regards Annex I, the placing on the market and the use of articles produced before or on July 10, 2012 containing endosulfan as a constituent of such articles was legal until January 10, 2013. 5
Footnotes
Authors’ contributions
A De Roma and C Rossini contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. C Riverso contributed to acquisition of data and critically revised the manuscript. G Galiero contributed to design of the study and critically revised the manuscript. M Esposito contributed to conception and design of the study and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Whatman no. 40 (90-mm diameter), Anotop 10 LC filter; Whatman International, Maidstone, UK.
b.
EXtrelut NT3 prepacked column with outlet cannulae of 0.60 × 30 mm (1–3 mL sample), Merck, Darmstadt, Germany.
c.
Isolute Florisil prepacked cartridges (1 g for 6 mL of reservoir volume), Biotage, Uppsala, Sweden.
d.
Carlo Erba Reagenti, Milan, Italy.
e.
100 mg/L of alpha and beta endosulfan solutions in isooctane, Dr. Ehrenstorfer Laboratory, Germany.
f.
GC System, Thermo Fisher Scientific, Waltham, MA.
g.
TotalChrome Navigator software, PerkinElmer, Waltham, MA.
h.
Zebron 1710 column (30 m × 0.25 mm ID × 0.25 µm), Phenomenex, Torrance, CA.
i.
Zebron MR-1 column (30 m × 0.25 mm ID × 0.25 µm), Phenomenex, Torrance, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and publication of this article.
