Abstract
Fungi in the genus Aspergillus are some of the most common fungal pathogens in veterinary species, primarily affecting the respiratory tract. In both human and veterinary cases, calcium oxalate crystals have been documented in sites of Aspergillus infection. Cases in multiple species (16 birds, 15 horses, 5 dogs, 1 ox, and 1 dolphin) were identified that had either positive cultures for Aspergillus sp., or had conidiophores present that could be identified as belonging to the genus Aspergillus. Histologic slides were examined to confirm the presence of oxalate crystals and how often they were identified on the original report. Calcium oxalate deposition was detected in 14 of 38 cases examined, including A. fumigatus, A. versicolor, A. niger, and unspecified Aspergillus sp. infections. Calcium oxalate crystals were identified in 11 of 16 avian cases, as well as in 1 of 1 bovine, 1 of 15 equine, and 1 of 5 canine cases. Crystals were described in only 3 of the 14 original pathology reports of these cases, indicating that identification and reporting of crystals in histologic specimens could be improved. All the tissues with crystals were respiratory tissues with air interfaces, including nasal sinus, trachea, syrinx, lung, and air sac. In cases with crystals identified on H&E-stained sections, crystals were frequently not present or were fewer in number in tissue sections stained with Gomori methenamine silver and periodic acid–Schiff. Routine polarization of slides of fungal infections, especially in the respiratory tract, should be considered to check for calcium oxalate crystals.
Introduction
Aspergillus is a genus of ubiquitous saprophytic fungi that are opportunistic pathogens. Aspergillus fumigatus is the most common cause of aspergillosis in mammalian and avian species, although infections with various other Aspergillus spp., including A. terreus, A. flavus, A. nidulans, and A. niger, have been reported.2,17,36,37 Feline Aspergillus infections are primarily sinonasal or sino-orbital, 17 and canine infections are primarily sinonasal, bronchopulmonary, or disseminated.35,37 The most frequent manifestations of aspergillosis in ruminants are abortion, placentitis, pneumonia, and gastroenteritis, whereas equine aspergillosis mainly manifests as guttural pouch mycosis and pneumonia. 35 In birds, aspergillosis predominantly affects the respiratory tract, and a number of factors, including environmental conditions, stress, and altered immune status, can predispose birds to infection. 2 In marine mammals, aspergillosis has been reported to cause lesions in numerous organs, including lung, skin, brain, alimentary tract, lymph nodes, bone marrow, and middle and inner ear; disseminated infections have also been reported.1,8,9,29,32 Aspergillus may also be angioinvasive, allowing dissemination throughout the body. 18
In the asexual development phase of Aspergillus, colonies form aerial hyphal branches. Some of these branches can develop into conidiophores, 19 which produce the asexual spores, or conidia. Conidiophore development and formation of conidia require exposure to air, and both exposure to oxygen as well as sensing of the phase change (solid to air) likely regulate conidiogenesis. 4 Therefore, conidiophore formation and conidiogenesis in vivo are generally observed at an air interface, which is most often in the respiratory tract.
Aspergillus niger and other Aspergillus species synthesize oxalic acid as a product of the tricarboxylic acid cycle. 34 Precipitation of calcium oxalate crystals occurs at a physiologic pH, as oxalate complexes with calcium in the tissue and blood. 28 Oxalic acid and oxalate crystals are thought to cause damage to tissues,20,26,34 including pulmonary blood vessels, with resultant pulmonary hemorrhage occasionally reported in human patients.20,34 Calcium oxalate is believed to cause tissue injury via iron-dependent generation of free radicals. 13
In the veterinary literature, oxalate crystal formation has been reported in an alpaca and a great-horned owl with respiratory A. niger infections.25,40 Oxalate crystals were also identified in pneumonia caused by Metarhizium anisopliae var. anisopliae in an American alligator (Alligator mississippiensis), 16 and in 2 American alligators with pulmonary infections with Beauveria bassiana. 12
Only 2 cases of oxalate crystals in Aspergillus infections have been reported in veterinary medicine, in contrast to a much larger number of cases and a retrospective study in humans. Our study objectives were to determine the incidence and location of oxalate crystals in Aspergillus infections in animals, to determine if crystals were identified in the pathology reports for these cases, and to determine which species of Aspergillus were associated with oxalate crystals.
Materials and methods
A retrospective analysis of postmortem examinations and surgical pathology cases that had been presented to the University of Florida (UF) Anatomic Pathology Service was performed. A case-insensitive text search was performed for the truncated term “aspergill” on cases with computerized records that were searchable (postmortem reports from January 1, 1995–February 28, 2013, surgical pathology reports from January 1, 2006–February 28, 2013). There were no restrictions placed on species, sex, age, or the tissues involved. Reports were read to determine if fungal infection was identified histologically. Only cases in which an Aspergillus species was cultured, or conidiophores were noted histologically and genus identification was considered possible, were included in this study.
Hematoxylin and eosin (H&E)–stained slides of tissues with fungal infections were viewed microscopically under normal and polarized light for evidence of crystals consistent with calcium oxalate. All of the sections, including both normal and abnormal regions of the tissues, were examined under polarized light, with normal tissue acting as an internal control. Calcium oxalate crystals are needle-like, colorless to pale yellow, strongly birefringent, and frequently have a wheat-sheaf or rosette arrangement 11 (Fig. 1). Cases were all examined by one pathologist, who was blinded to the report when determining whether crystals were present. After examination of slides, the corresponding pathology reports were read to determine if oxalate crystals were identified in the initial report. Case 1 had multiple biopsy specimens and a postmortem examination, and the initial biopsy report that diagnosed the fungal infection was used to determine whether crystals were identified on the report.
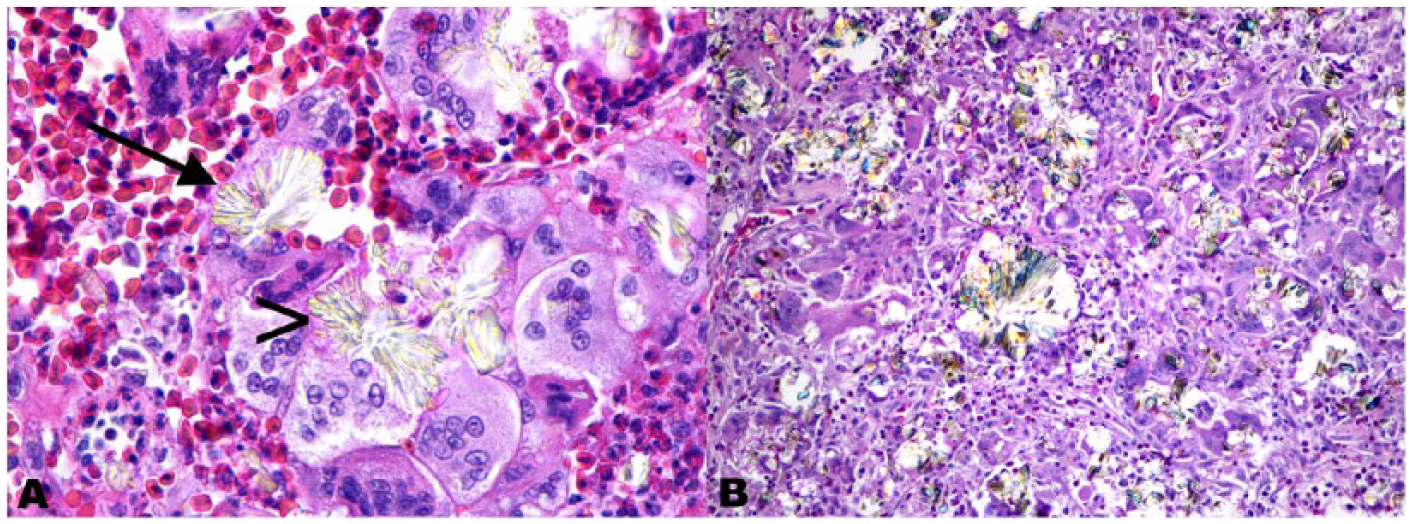
Granulomatous tracheal mass from a blue and gold macaw (case 1).
Aspergillus sp. conidiophores were identified by the conidial heads, which are composed of the vesicle, phialides, conidia, and, in some species, metulae 30 (Fig. 2). The vesicle is a terminal enlargement of the conidiophore. 22 Phialides, the flask-shaped cells that produce the conidia, may be connected directly to the vesicle (uniseriate) or can be connected to the vesicle via supporting cells called metulae (biseriate). 30 Round conidia in chains are produced by the phialides. 30 Species identification was attempted using conidiophore morphology, including seriation, arrangement of the phialides, and pigmentation.22,38 Sections stained with Gomori methenamine silver (GMS) and periodic acid–Schiff (PAS) were available for some cases, and were also examined for calcium oxalate crystals. When available, histologic sections of kidney, with or without fungal infection, were examined under polarized light for the presence of calcium oxalate crystals.
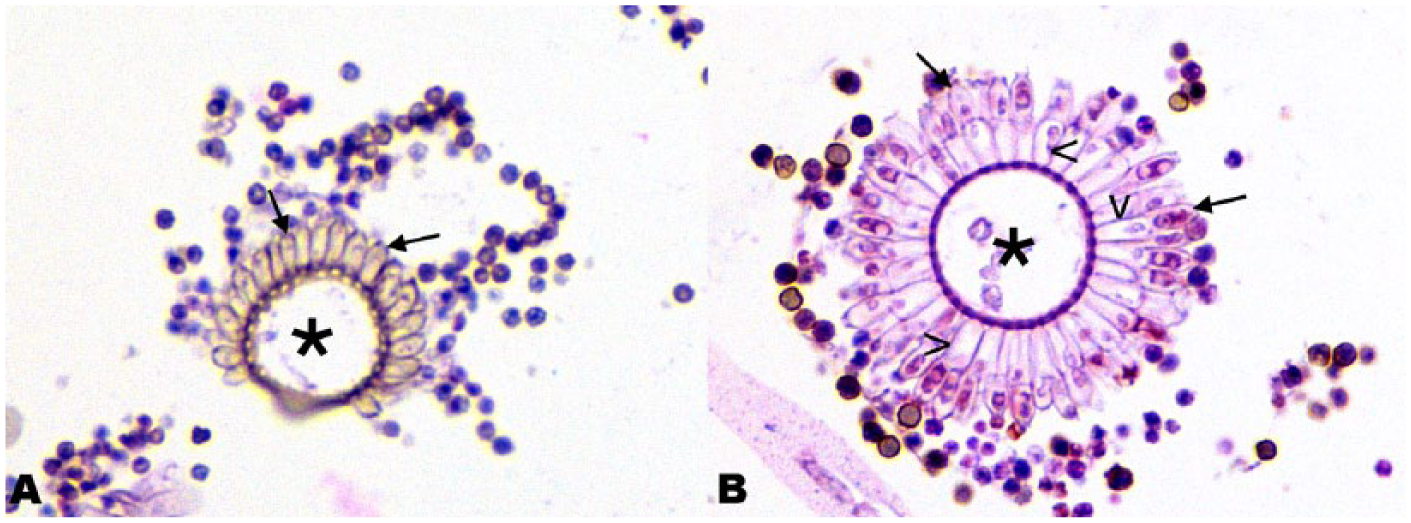
Conidiophores in the lung of a Holstein cow (case 17). H&E-stained section.
Results
Via a computer text search, 259 reports were identified that contained the truncated term “aspergill.” Elimination of cases that did not have fungal elements identified in the report yielded 74 reports. Restricting the case set to those in which an Aspergillus species was cultured from affected tissue, and/or conidiophores consistent with Aspergillus sp. were identified histologically, reduced the set to 38 reports. Five of these reports were from 1 animal with 4 separate biopsy specimens and a postmortem examination, so the total number of cases from the search was 34. An additional biopsy report from this animal from 2004 (not included in the computer text search) was identified from the subsequent biopsy reports; this sample was also included in the study. Four additional culture-positive cases were identified from postmortem examinations from 1979 to 1982 from a search of paper records, for a total of 38 cases. Eight cases (cases 19–22 and 35–38) were biopsy specimens, 1 case (case 23) was a submission of formalin-fixed tissues from a postmortem examination, and 28 cases were postmortem examinations performed by the UF Anatomic Pathology Service. Animal species affected included 16 birds, 15 horses, 5 dogs, 1 ox, and 1 dolphin. In 34 of 38 cases, fungal infection was identified in the respiratory tract, although some cases also had additional tissues affected (Table 1).
Evaluation of aspergillosis cases for the presence of calcium oxalate crystals.
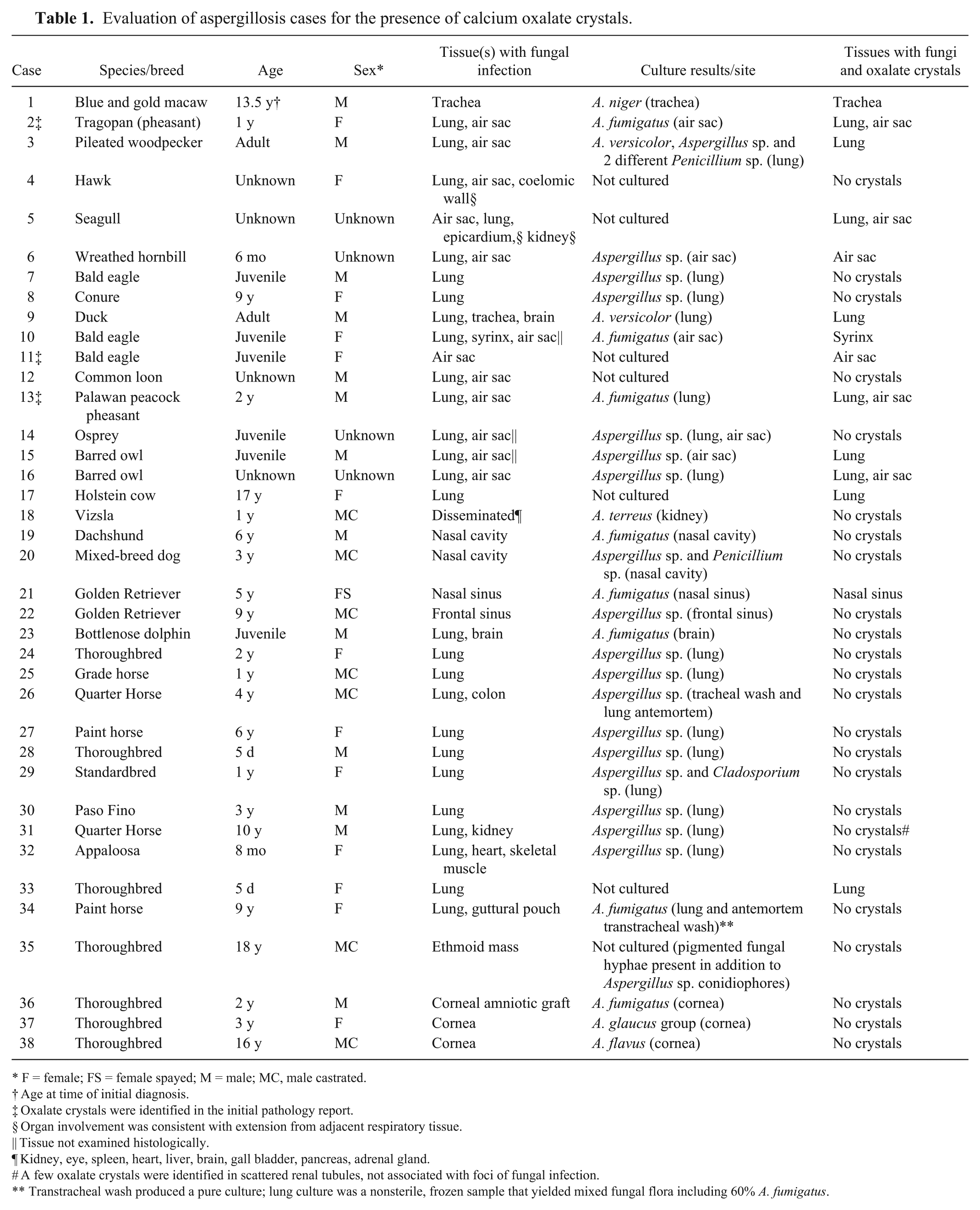
F = female; FS = female spayed; M = male; MC, male castrated.
Age at time of initial diagnosis.
Oxalate crystals were identified in the initial pathology report.
Organ involvement was consistent with extension from adjacent respiratory tissue.
Tissue not examined histologically.
Kidney, eye, spleen, heart, liver, brain, gall bladder, pancreas, adrenal gland.
A few oxalate crystals were identified in scattered renal tubules, not associated with foci of fungal infection.
Transtracheal wash produced a pure culture; lung culture was a nonsterile, frozen sample that yielded mixed fungal flora including 60% A. fumigatus.
Of the 38 cases, 31 were confirmed as Aspergillus via culture. Seventeen of the culture results were not identified to the species level (reported as Aspergillus sp.), 8 were A. fumigatus, 1 was A. versicolor, and 1 was A. versicolor and an additional Aspergillus sp. that was not further identified; A. niger, A. glaucus group, A. terreus, and A. flavus were identified in 1 case each. The remaining 7 cases were identified as Aspergillus species based on microscopic conidial morphology.
Fourteen of 38 cases were found to have various degrees of calcium oxalate crystal deposition. The crystals were identified in the initial pathology report in 3 of these 14 cases; all 3 of these were birds.
Crystals were identified in 4 cases that were not cultured (histologically identified as Aspergillus species by conidial morphology) and in 10 cases with cultures performed, including 3 reported as Aspergillus sp. (not identified to species), 4 A. fumigatus, 1 A. versicolor, 1 A. versicolor together with Aspergillus sp., and 1 A. niger. The frequency of calcium oxalate crystals in each fungal species were as follows: 3 of 17 reported as Aspergillus sp., 4 of 8 reported as A. fumigatus, 1 of 1 reported as A. versicolor, 1 of 1 reported as A. versicolor and Aspergillus sp., 1 of 1 reported as A. niger, and 4 of 6 that were histologically identified as Aspergillus spp. Additional genera of fungi were occasionally cultured together with the Aspergillus. In case 3, 2 different Penicillium spp. were also cultured along with A. versicolor and an Aspergillus sp. that was not further identified. In case 29, Cladosporium sp. was also cultured together with Aspergillus sp.
Calcium oxalate crystals were identified in 11 of 16 avian cases, as well as in 1 of 1 bovine, 1 of 15 equine, and 1 of 5 canine cases. Crystals were identified in the trachea or syrinx (3 of 14 cases), lung (9 of 14 cases), air sac (6 of 14 cases), and nasal sinus (1 of 14 cases). In 4 cases, there were crystals in both the lung and air sac. Crystals were only noted in respiratory tissues with air interfaces (Fig. 3), including air sac, lung, trachea, and nasal sinus, and only in areas of the tissue that contained fungal elements. Regions of normal tissue in affected organs did not contain oxalate crystals. In 9 of 14 cases with crystals, fungal infection was noted in multiple respiratory tissues (cases 2, 3, 5, 6, 9, 10, 13, 15, 16). All cases with crystals in multiple respiratory tissues were birds (9 of 11 avian cases with crystals). In these cases, crystals were noted in all affected respiratory tissues in only 4 of those 9 cases. In 1 of the remaining 5 cases (case 15), the lung and air sac were both affected, but sections of air sac were not examined histologically. Crystal deposition was also not uniform within an individual organ, with some having numerous crystals throughout, and others having crystals in only a small portion of the lesions. In some cases with multiple affected air sacs, only some of the air sacs contained crystals.
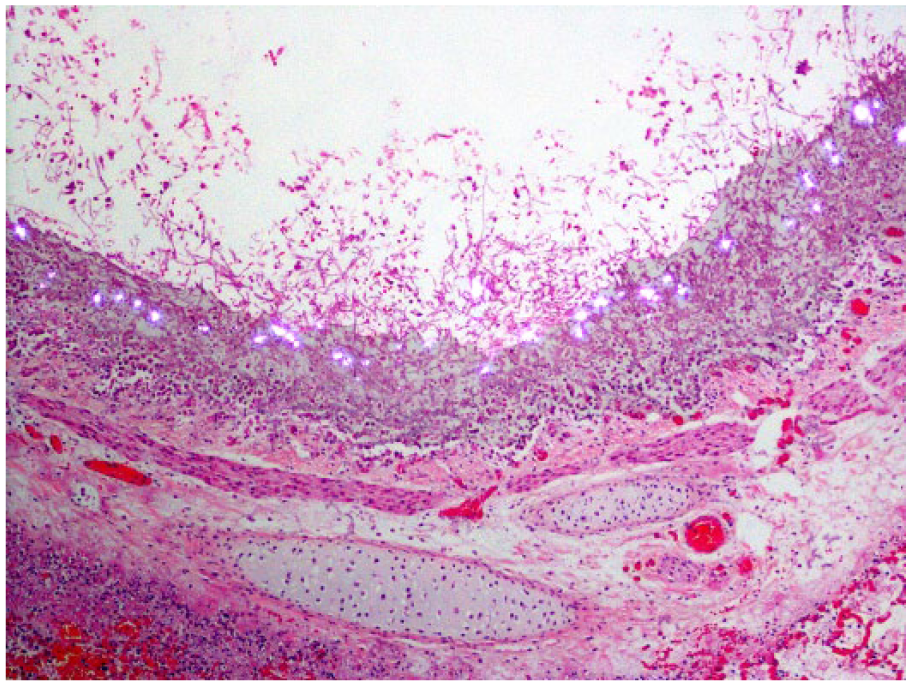
Necrotizing bronchitis caused by Aspergillus sp., in the lung of a Thoroughbred horse (case 33), with extensive fungal colonization of the bronchial wall. Calcium oxalate crystals are present at the air interface. H&E, polarized light.
GMS-stained sections were examined in 10 of the 14 cases with crystals (cases 1, 2, 5, 6, 9–11, 13, 17, 33), as well as in 8 of the 24 cases without crystals (cases 7, 8, 18, 23, 34–36, 38). PAS stains were available on 6 of the 14 cases with crystals (cases 5, 6, 9, 11, 17, 33) and 1 of the 24 cases without crystals (case 8). All cases with PAS stains also had a GMS stain performed. Crystals were not identified in the majority of the GMS- or PAS-stained sections where crystals were identified on H&E. Only cases 1 and 5 had crystals present on these sections. In case 1, a GMS stain was done on the tracheal sections from the postmortem examination, and fewer crystals were identified than were present in the H&E sections. These crystals were embedded in fibrous connective tissue deep in the section. In case 5, GMS staining was done on the lung and air sac, and PAS staining was done on the air sac only. The air sac in both the GMS- and PAS-stained sections had crystals that were embedded within the inflammation deep in the section, but the more numerous superficial crystals on the H&E-stained section were absent (Fig. 4). The GMS-stained section of lung did not contain crystals, although they were present on the H&E-stained section.
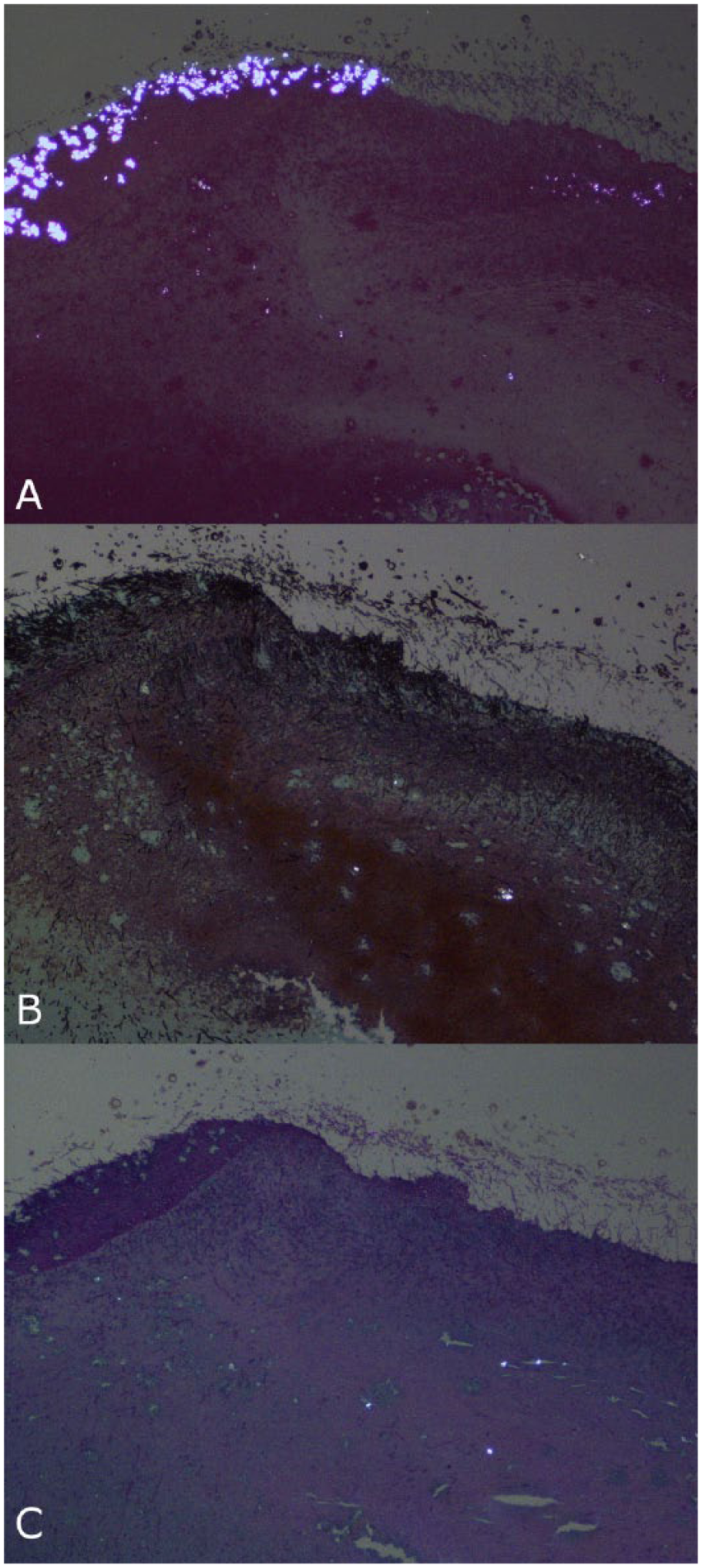
Fungal airsacculitis in a seagull (case 5).
Kidneys were examined if sections were available (12 of 14 cases with crystals, 10 of 24 cases without crystals). Rare to few crystals consistent with calcium oxalate were identified in renal tubules in 3 cases (cases 18, 31, 33), 1 dog and 2 horses. In cases 18 and 33, these were limited to 2 crystals in a single tubule. Only case 33 had oxalate crystals in the tissue with fungal infection (lung).
Serial samples were present for one case (case 1), a male blue and gold macaw, whose initial diagnosis of tracheal aspergillosis was made at 13.5 y of age. This bird lived an additional 32 mo after the initial diagnosis. Crystals were present but not identified in the report from the initial biopsy specimen. Four additional biopsy specimens were taken 24–32 mo after the initial diagnosis, with a few fungal hyphae on the second biopsy specimen and none in any subsequent samples. The second through fifth biopsy specimens all contained oxalate crystals that were identified in the reports. At postmortem examination (32 mo after the initial diagnosis), there was extensive tracheal stenosis, with a large amount of fibrosis and granulomatous inflammation associated with crystals. Fungi were not identified via special stains or culture of the tracheal lesion from the postmortem examination.
Discussion
Of the specimens that were positive for calcium oxalate crystals (14 of 38 cases), crystals were described in only 3 of 14 of the original reports. Although only a few scattered case reports of oxalate-producing Aspergillus infections exist in the veterinary literature, the true prevalence of oxalate crystals in Aspergillus infections appears to be much higher. When the crystals were sparse and/or colorless, they were difficult to detect without polarization. Polarization of all cases with fungal infection could provide further insight into the actual occurrence of calcium oxalate crystals.
The 14 cases with crystals were read by 11 different pathologists; 2 pathologists read the 3 cases with crystals identified in the report, and 9 pathologists read the 11 cases where crystals were not identified in the report. Although it is possible that crystals were seen but not recorded in the report, this seems unlikely as it is not routine for most pathologists to polarize slides on fungal cases, and the reports typically had fairly detailed histologic descriptions.
Oxalate crystals were only noted in the respiratory tract, in tissues with an air interface. This may occur because fungi that synthesize oxalic acid do so as a product of the tricarboxylic acid cycle, 34 which requires oxygen. Human cases also predominantly involve the lung, with occasional cases involving the nasal cavity, 27 sinuses, 27 trachea, 15 and nasopharynx. 10 Individual human cases of A. niger infections with oxalate crystals have been reported outside of the respiratory tract, including otomycosis with crystals in the tympanic membrane, 21 and 2 cases of infection in a gangrenous foot.24,33 Although these lesions were outside the respiratory tract, the presence of conidiophores in all 3 of these cases21,24,33 suggested that there was an air interface at the infection site. One human autopsy case with disseminated aspergillosis had oxalate crystals in lesions in both the lung and hilar lymph node. 27 Crystals in the hilar lymph node could speculatively have drained from the lung rather than being produced within the lymph node.
One retrospective study of human autopsy cases with Aspergillus infection identified crystals in 11 of 68 cases. 27 Our incidence was higher; however, it is possible that the number of Aspergillus cases in the human autopsy study was incorrect, as only 10 of the cases (including 4 with crystals) were identified by culture and/or conidial morphology. The remaining 58 cases were diagnosed only by hyphal morphology. 27 As the hyphal morphology of Aspergillus is similar to other fungal species, some of the cases may have been misclassified as Aspergillus, altering the incidence.
In the human literature, oxalate crystals have been associated with significant tissue damage,20,26,34 including pulmonary hemorrhage.20,34 However, no studies have definitively determined whether oxalate crystals are a negative prognostic indicator. The prognosis of aspergillosis in humans largely depends on the site of infection and comorbidities. 23 As 12 of 14 of our specimens with oxalate crystals were from postmortem examinations, it was not possible to determine if oxalate crystals factor into prognosis. In the one case with serial samples (case 1), crystals were more numerous than fungi after the initial biopsy. The second biopsy submission, taken 2 y after the initial biopsy, was composed primarily of crystals with heterophilic and granulomatous inflammation and fibrosis (Fig. 1), with relatively few fungal hyphae. Inflammation and fibrosis associated with the oxalate crystals persisted even when fungi could not be identified by special stains or culture. It is possible that these crystals remain in the tissue and incite an inflammatory response even after treatment clears the fungal infection. This animal survived for 32 mo after the initial diagnosis, and crystals were present in the initial biopsy. However, it is difficult to draw conclusions from a single case. Although fungal invasion of vessels was observed in some cases, sometimes with various degrees of hemorrhage in the surrounding tissue, large amounts of hemorrhage associated with vessel damage were not identified in any tissues with oxalate crystals.
During the course of our study, we discovered that it was very difficult to definitively identify Aspergillus species based on conidiophore morphology, given the high degree of phenotypic overlap. Some infections had conidial morphologies that could be consistent with more than 1 species. In case 17, there were uniseriate and biseriate conidiophores (Fig. 2). Additionally, the orientation of the conidiophores in sections, such as sections that only included the conidial head (Fig. 2B), did not always allow for the determination of the arrangement of the phialides. Although we were confident that these were Aspergillus species based on conidial morphology, species identification was not possible. As hyphal morphologies overlap between Aspergillus and multiple other species of fungus, only cases with positive cultures or cases with conidiophores identified histologically were considered in this study. Fungal culture should be considered a much more accurate means of identification.
In human aspergillosis cases, calcium oxalate crystals are usually associated with A. niger infection, and less commonly with A. fumigatus or A. flavus.10,11,28 In our cases, A. fumigatus was the most common species associated with oxalate crystals (4 of 14 cases), which may also reflect that it is the most common Aspergillus species identified in veterinary cases of aspergillosis. Only one of our cases (case 1) had A. niger cultured, and crystals were identified in this case. Other fungi, including A. versicolor and A. fumigatus, as well as multiple cases with Aspergillus sp. that were not further classified, also had calcium oxalate crystals. In the human literature, there is one report of pulmonary zygomycosis, in which Rhizopus sp. was cultured, that also had calcium oxalate crystals. 31 In veterinary cases, a few other fungi, including M. anisopliae var. anisopliae and B. bassiana, have been associated with oxalate crystals in American alligators, therefore culture, molecular testing, and/or morphology must be used to definitively determine the etiology.12,16 Only Aspergillus spp. have been associated with calcium oxalate crystals in mammals in reported veterinary cases.
Several cases had mixed fungal infection. In cases 3, 20, and 29, either Penicillium sp. or Cladosporium sp. were also identified in culture. In case 34, an antemortem transtracheal wash produced a pure culture of A. fumigatus, whereas a postmortem lung culture of a nonsterile, frozen sample yielded mixed fungal flora including 60% A. fumigatus. Case 35 had pigmented fungal hyphae, in addition to hyphae and conidiophores consistent with Aspergillus sp. Of these 5 cases of mixed infection, only case 3 had crystals. Although Penicillium sp. are capable of producing oxalic acid6,7 and oxalate crystals, 14 only a single case of Penicillium sp. infection in birds has been reported—P. cyclopium infection in the beak of a macaw. 3 Conidiophores were not present histologically to differentiate between Aspergillus and Penicillium. The Penicillium sp., a common air-borne mold, was likely a contaminant in this case, and oxalate crystals were likely related to the Aspergillus infection, as this is the most common fungus in avian respiratory infections. However, infection with Penicillium sp. cannot be definitively ruled out as a cause of oxalate crystal formation.
Calcium oxalate crystals were most commonly identified in avian species (11 of 14 cases with oxalate crystals). One possible reason for this high prevalence is the relatively high rate of respiratory aspergillosis in birds relative to mammals, as crystals in this study were only identified in tissues with an air interface.
Fungal elements from Aspergillus spp. are often readily visible on H&E-stained sections, so special stains are often not necessary for the diagnosis. Slides stained with GMS and PAS were also available for examination in some of the cases. It was noted that birefringent crystals present in tissue stained with H&E were only rarely identified in the tissue sections stained with GMS or PAS. In the 2 cases in which crystals were identified, they were fewer than on the H&E-stained sections, and they were embedded within the tissue. It is presumed that the staining process must in some way dissolve the crystals; the acid used in these stains could be a possible solvent.
In humans, significant numbers of renal tubular oxalate crystals have been identified in patients with oxalate-producing Aspergillus infections,27,34,39 and are believed to be related to hyperoxaluria secondary to excessive oxalosis in the respiratory tract. 39 In our case series, renal crystals were identified in only 3 cases, 1 with and 2 without oxalate crystals in the tissues with fungal infection. The paucity of crystals in the kidney, as well as their presence in both cases with and without oxalate crystals within fungal infections, suggest that renal crystals were more likely endogenous rather than related to fungal infection. However, renal oxalosis appears to be a relatively rare occurrence in human cases, and could potentially occur in veterinary cases. There is 1 case report of a white-tailed deer with A. fumigatus infection in the lung and brain with oxalate crystals in the renal tubules. 41 Oxalate crystals were not identified in the lung, but the report did not mention examination of lung sections under polarized light, and the authors did not link the 2 disease processes. Oxalate crystals may be observed in the kidneys in multiple other disease processes, including ingestion of oxalate-containing plants and ingestion of moldy feed with oxalates produced by Aspergillus spp., 5 therefore the oxalate crystals in the kidneys of the deer could have been unrelated to the pulmonary infection.
Footnotes
Acknowledgements
We thank Dr. Richard Montali for planting the idea for this study.
Authors’ contributions
CL Payne contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. MJ Dark contributed to acquisition and interpretation of data, and critically revised the manuscript. JA Conway contributed to design of the study; contributed to acquisition of data; and critically revised the manuscript. LL Farina contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
