Abstract
Bovine trichomoniasis is a concern for the cattle industry. Advances in testing systems have increased the ability to detect the disease in bulls. However, the greatest limitation is proper collection of an adequate sample. The low repeatability observed with most sample collection techniques can cause false-negative results. The aim of our study was to validate a sample collection technique that increases diagnostic sensitivity and is easier and safer to collect than preputial scraping. Commercial bulls (n = 111) of unknown infection status were sampled for detection of Tritrichomonas foetus using 2 different collection methods: 1) preputial scraping with a dry insemination pipette and 2) penile sponging with a 16-ply gauze sponge. Preputial scraping samples were collected by vigorously scraping preputial and penile mucosa using a rigid insemination pipette while applying negative pressure with a syringe. Penile sponge samples were obtained by swabbing the penile and preputial mucosa with a gauze sponge during full extension of the penis. All samples were processed using a commercial medium and submitted under similar conditions for PCR testing. Positive PCR results were detected in 37 of 111 (33%) bulls using the preputial scraping technique; however, 39 of 111 (35%) were positive using the penile sponging technique. The Newton–Raphson algorithm predicted that the sensitivity of the preputial scraping method was 0.919 (95% CI: 0.689–0.983) and the sensitivity of the penile sponging was 0.949 (95% CI: 0.818–0.987). These data indicate that the penile sponging technique is a reliable alternative to the preputial scraping method.
Trichomoniasis, a sexually transmitted protozoal reproductive disease of cattle caused by Tritrichomonas foetus,1,2 has been identified in most western and midwestern U.S. states. 12 Tritrichomonas-infected herds have increased abortions and infertility, extended breeding seasons, and incur significant income loss. 3
The T. foetus protozoa can live in the reproductive tract of the cow and on the surface of the penis of the bull, and most infected bulls will become permanent carriers. 2 Up to 95% of cows will become infected when bred only once by a carrier bull. 9 The organism survives in the vagina and uterus of the cow. After ~50 days, the embryo will die from inflammation within the uterus caused by the protozoa. Cows will eventually clear the infection (2 estrus cycles), develop temporary immunity (2–6 months), and be able to maintain a pregnancy at subsequent breeding. 1 If there is an extended breeding season, cows that develop temporary immunity may be late bred. With shorter breeding seasons, the cow will not have time to develop immunity and will not become pregnant.
Diagnosis of trichomoniasis is either by direct examination of a culture or detection of nucleic acid through PCR testing. However, detecting a T. foetus–infected bull is problematic, and the sensitivity of laboratory tests can be compromised by sample collection. 11 The most common method for sample collection is preputial scraping. 11 To obtain a sample from a bull, the surface of the preputial and penile mucosa is vigorously scraped with a rigid insemination pipette while applying negative pressure with a syringe. Although preputial scraping is the accepted method for collecting samples, it is an imperfect technique. Not only can it be difficult to determine if an adequate sample has been collected, but a previous study 8 reported that right-handed veterinarians were more likely to have culture-positive samples if scraping was conducted from the right side versus the left side, indicating that minor changes in technique can significantly influence test results.
The objective of our study was to validate an alternative sample collection technique that is easier to collect, safer for the bull and the person collecting the sample, and increases diagnostic sensitivity. The protocol was approved by the Iowa State University Institutional Animal Care and Use Committee (8-12-7432-B).
For the project, bulls from high-risk populations in which trichomoniasis was either suspected or known to be a cause of infertility in the herd were sampled. Preputial samples using both methods were either collected on the same day or within 7 days of each other. Private herds that had initially been tested for trichomoniasis by preputial scraping were resampled within 7 days by the sponge method for comparison. In herds where sampling occurred at the same time, preputial scraping was collected first followed by the sponge method to ensure that sponge collection did not preclude the ability to detect trichomonads by preputial scraping. Bulls were restrained in a squeeze chute for sample collection. All samples were placed in a trichomonas sample collection pouch a after collection and sent to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, Iowa) for PCR testing.
For the preputial scraping method, a disposable 53-cm rigid pipette with a 0.3-cm internal diameter was inserted into the prepuce to the fornix. Negative pressure was applied with a 20-mL syringe, and the pipette was moved back and forth in long strokes 5–10 times scraping the preputial and penile mucosa. The sample was then placed in the upper chamber of the sample collection pouch a by flushing the pipette with medium provided in the pouch. a The medium and sample were pushed into the lower chamber, and the pouch was sealed per the manufacturer’s recommendations.
For the sponge method, the penis was extended by electrostimulation with a rectal probe. Once extended, a 16-ply gauze sponge was used to wipe around the glans and down the penile shaft and exposed preputial mucosa 2–3 times (Fig. 1). The sponge was then placed in the upper chamber of the sample collection pouch, a and a disposable applicator stick was used to push the sponge to the bottom of the upper chamber. Air was then removed from the pouch, the pouch sealed, and medium from the lower chamber was expressed into the upper chamber to saturate the sponge. At the ISU-VDL, the medium was manually expressed from the gauze sponge while still in the pouch prior to normal extraction procedures.

Swabbing glans and preputial epithelium with gauze sponge.
Extraction of samples was performed with a commercial RNA/DNA extraction kit b using the manufacturer’s recommendations c for a 115-μL clarified lysate sample with minor modifications. Instead of vortexing the tubes and plates for 3 and 5 min, respectively, these vortex steps were each performed for 30 s. Also, the subsequent centrifugation steps were replaced by simply allowing the tubes and plates to settle for a minimum of 5 min prior to the following steps. Extraction was carried out using a magnetic particle processor d according to the manufacturer’s instructions. e
The PCR assay was performed using a commercial Tritrichomonas detection kit f targeting the internal transcribed spacer region 1 (ITS-1) 5 according to manufacturer’s recommendations. The PCR reactions were run on real-time PCR instrumentsg,h with the following cycling conditions: 95°C for 10 min followed by 40 cycles of 95°C for 15 s and 55°C for 45 s. Data analysis was performed according to the kit insert for runs performed on one instrument. g Instrument default settings were used for analyzing runs conducted on the other instrument. h The PCR kit contains an internal control i to monitor possible inhibition during the PCR reaction. Any sample exhibiting a quantification cycle (Cq) value of ≤40 cycles for the T. foetus target was considered positive. A Cq ≥40 for the T. foetus target and a Cq ≥40 for the control i target is indicative of inhibition of the reaction. In these instances, samples were diluted 1:1 with phosphate buffered saline to dilute out the inhibiting factors, and the extraction and PCR were repeated on the diluted sample.
The sensitivity of each method was calculated with an online application to calculate sensitivity in the absence of a gold standard. j The application j estimates the sensitivity of laboratory tests and generates confidence intervals with a Newton–Raphson algorithm. Model adequacy is evaluated by a goodness-of-fit test. Seven bulls were used in the statistical analysis as reference bulls. Of these 7 animals, 3 bulls, which had previously tested positive by PCR, were purchased by ISU for research purposes and were used as reference bulls in the infected population. Two bulls were positive by both methods, and 1 bull was negative by both methods. The other 4 bulls were from a closed herd with no evidence of infertility and were used as reference bulls from a disease-free population; all tested negative by both methods.
Of the 111 bulls sampled from the high-risk populations, 37 (33%) tested positive by traditional preputial scraping and 39 (35%) tested positive by the penile sponge method (Table 1). Three bulls (2.7%) were negative by scraping and positive by sponge, whereas 1 (0.9%) bull was positive by scraping and negative by sponge. Thirty-six (32%) bulls were positive by both methods, and 71 (64%) were negative by both collection methods.
Real-time PCR results from 111 high-risk bulls and estimated sensitivity of each sample collection method.
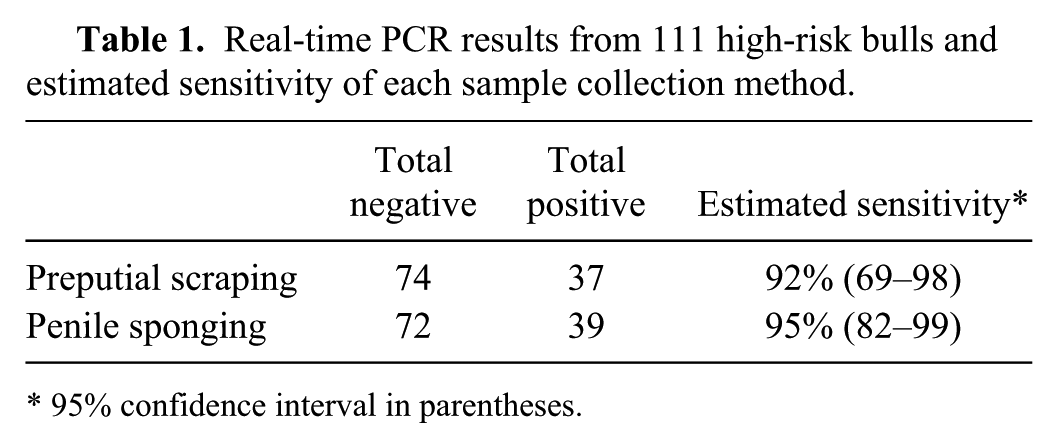
95% confidence interval in parentheses.
Sensitivity of the preputial scraping method was estimated to be 92% (95% confidence interval [CI]: 69–98%), whereas penile sponging was 95% (95% CI: 82–99%). The goodness-of-fit test had a p-value of 0.57 indicating that the model could fit because there was not a statistical difference between the observed and expected results.
The results of our study indicate that penile sponging is an effective method for detecting and diagnosing trichomoniasis in bulls. Both scraping and sponging were comparable in results and were not statistically different. Penile sponging may be an acceptable alternative sampling method for trichomoniasis for diagnostic and regulatory purposes.
Consistent diagnostic accuracy is important for control of reportable diseases. Preputial scraping can be inconsistent. 11 Because of difficulty in collecting an adequate sample, some states have instituted a certification program for veterinarians in order for a submission to be considered an official test (Kansas Department of Agriculture, https://goo.gl/xxkIgY; Wyoming Livestock Board, http://soswy.state.wy.us/Rules/RULES/8063.pdf). One difficulty in collecting an adequate sample is that veterinarians cannot visualize the collection of the specimen, and many use their nondominant hand to control the pipette. The penile sponging technique not only allows veterinarians to visualize the epithelial surface, it also allows for sampling greater surface areas compared with the pipette method. Modified pipettes have been developed to improve sample collection. However, researchers have not reported any improvement of sensitivity with such devices.6,7
Safety for both the veterinarian and patient is important for sampling methodology. Bulls may show discomfort by kicking during preputial scraping. Additionally, veterinarians could potentially cause a preputial laceration or abscess by being too aggressive with a pipette. The sponge technique is less irritating and safer than the pipette method and, typically, when bulls are being electrostimulated, they are less able to react physically.
Another advantage of the sponge method is that it is simple to collect during standard breeding soundness examinations (BSEs). The penile epithelium can be sampled during extension prior to ejaculation. Typically during a BSE, the semen sample is collected first so that there is no chance of blood contamination from the pipette method. This step requires that the pipette sample be collected after the semen has been processed, requiring additional time. One drawback of the sponge method is that it is impossible to collect a sample if the bull will not extend. However, bulls that will not extend can always be sampled with a pipette.
Of the 4 bulls that differed in positive status between the 2 methods, the sponge sample was more likely to detect a positive than the pipette method (3/4 bulls). One bull positive by pipette scraping was negative by the sponge method. This bull was tested with the pipette method 7 days previous to the sponge method. This may have been a potential false-positive result reported on only one particular accession that detected T. foetus in 53% (9/17) of the bulls tested. The Cq value detected in the original positive result using the scraping method was 37.2 with an assay cutoff of 40, the highest Cq of any bull tested in that accession. It is unknown if repeatability of the positive result using the sponge method was affected by the low level of T. foetus in the sample or if the result was actually a result of a lack of specificity.
Handling of sponge samples has an additional minor processing step for testing laboratories in that laboratory personnel need to express the medium out of the sponge prior to pipetting the sample out of the pouch. Before collecting and submitting samples, practitioners need to confirm with the laboratory that they are able to process sponge samples. To date, the sponge sample has not been validated for culture of trichomonas organisms. Although the medium keeps the sponge moist, it may not support growth of the organism. In addition, alternative solutions other than growth medium may be sufficient for testing depending on laboratory protocols. We collected 2 samples from the positive reference bulls with the sponge and then moistened with normal saline prior to laboratory submission, which resulted in similar PCR results to the commercial medium. a
Test validation can be difficult, especially in the absence of a gold standard. Maximum-likelihood methods have been used to estimate sensitivity and specificity parameters when the true disease state is not known. 4 The commercial application used for data analysis uses a Newton–Raphson iterative process to generate the maximum-likelihood estimates and confidence intervals. 10 We have only reported sensitivity in this study, as the specificity depends on the PCR primers utilized by the laboratory. The estimates of sensitivity for both sampling methods used in the study are similar, and the confidence intervals overlap. Additionally, the goodness-of-fit test indicates that the model fits and therefore the sensitivity estimates are valid.
Our study demonstrates that sponge sampling of bulls for trichomonas is a valid method. Regulatory veterinarians should consider allowing sponge samples as an official test for trichomoniasis. The method facilitates easier collection once the penis is extended and is potentially safer for the veterinarian and the bull. Additionally, it may be slightly more sensitive than the pipette method but a larger study needs to be conducted in order to demonstrate statistical differences.
Footnotes
Acknowledgements
We thank the Iowa State University Veterinary Diagnostic Laboratory technicians that helped develop laboratory methods for this procedure. Additionally, we thank the private veterinarians and Iowa State Veterinarians for assisting with access to positive herds.
Authors’ contributions
G Dewell contributed to interpretation of data and drafting the manuscript. G Dewell, P Phillips, and T Dohlman contributed to acquisition of samples. K Harmon and P Gauger contributed to laboratory procedures and supervision. G Dewell, P Phillips, T Dohlman, K Harmon, and P Gauger critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work were appropriately investigated and resolved.
a.
InPouch TF, BioMed Diagnostics Inc., White City, OR.
b.
MagMax pathogen RNA/DNA kit, Thermo Fisher Scientific, Waltham, MA.
d.
Kingfisher96, Thermo Fisher Scientific, Waltham, MA.
e.
MagMax express deep well QRC, Thermo Fisher Scientific, Waltham, MA.
f.
VetMax-Gold Trich detection kit, Thermo Fisher, Scientific, Waltham, MA.
g.
Applied Biosystems 7500 Fast, Thermo Fisher Scientific, Waltham, MA.
h.
SmartCycler, Cepheid Inc., Sunnyvale, CA
i.
Xeno, Thermo Fisher Scientific, Waltham, MA.
Declaration of conflicting interests
The author(s) declared that no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Nolan Hartwig Fund at Iowa State University and a grant from the Iowa Veterinary Medical Association Green Tag Fund (Ankeny, Iowa).
