Abstract
The measurement of N-terminal pro–B-type natriuretic peptide (NT-proBNP), a biomarker for heart stress detectable in blood, has been shown to have clinical utility in cats with heart disease. A second-generation feline enzyme-linked immunosorbent assay (Cardiopet® proBNP, IDEXX Laboratories Inc., Westbrook, Maine) was developed to measure NT-proBNP in routine feline plasma or serum samples with improved analyte stability. Results of the analytical validation for the second-generation assay are presented. Analytic sensitivity was 10 pmol/l. Accuracy of 103.5% was determined via serial dilutions of 6 plasma samples. Coefficients of variation for intra-assay, interassay, and total precision were in the ranges of 1.6–6.3%, 4.3−8.8%, and 10.1–15.1%, respectively. Repeatability across 2 lots for both serum and plasma had an average coefficient of determination (r2) of 0.99 and slope of 1.11. Stability of the analyte was found to be high. In serum samples held at 4°C for 24–72 hr, the mean percent recovery from time zero was ≥99%. In serum samples held at 25°C for 24 hr, the mean percent recovery from time zero was 91.9%, and for 48 hr, 85.6%. A method comparison of the first- and second-generation assays with a clinically characterized population of cats revealed no difference in the tests’ ability to differentiate levels of NT-proBNP between normal cats and cats with occult cardiomyopathy (P < 0.001). Results from our study validate that the second-generation feline Cardiopet proBNP assay can measure NT-proBNP in routine feline plasma and serum samples with accuracy and precision.
Introduction
In cats presenting with respiratory distress, it is challenging to differentiate congestive heart failure from primary respiratory disease as a cause of observed clinical signs. Studies5,7 have demonstrated that the measurement of circulating N-terminal pro–B-type natriuretic peptide (NT-proBNP) enhances the ability to distinguish between congestive heart failure and primary respiratory disease in presenting animals. Measurement of NT-proBNP has also shown utility in differentiating between normal cats and cats with occult cardiomyopathy (OCM).8,12 For both applications, accurate and precise measurement of NT-proBNP is critical to making an accurate diagnosis.
NT-proBNP is the inactive cleavage product of the prohormone B-type natriuretic peptide (BNP), which is stored in cardiomyocytes. In response to stretch and stress on the cardiac muscle, BNP is released and cleaved into a biologically active hormone (C-terminal BNP) and the inactive NT-proBNP. 10 NT-proBNP is released in proportion to the amount of stretch and stress on the myocardium, and the concentration is often increased in cats with heart disease relative to cats without.8,12
The first-generation feline NT-proBNP immunoassay
a
provided linear quantitative measurement of NT-proBNP in ethylenediamine tetra-acetic acid (EDTA) plasma samples in a range of 24–1,500 pmol/l, with intra- and interassay precision (%CV) of
To address the sample handling limitations, a second-generation immunoassay for the quantitative measurement of feline NT-proBNP was developed. The analytical validation of the second-generation immunoassay is presented in this publication. The linear range, accuracy, and precision of the assay is preserved or improved upon relative to the first-generation test. In addition, the second-generation assay supports routine sample submission of EDTA plasma or serum without the use of specialized protease inhibitor tubes.
Materials and methods
The second-generation feline NT-proBNP assay is a sandwich enzyme-linked immunosorbent assay (ELISA) containing unique anti-feline NT-proBNP capture and detection antibodies, targeting epitopes directed to the N-terminal portion of NT-proBNP. Antibodies directed to this portion of NT-proBNP were found to facilitate analyte detection without specialized protease inhibitor tubes. The capture antibody is immobilized on 96-well microtiter plates, b and the detection antibody is conjugated to horseradish peroxidase c (HRP). A set of 6 calibrators, containing increasing levels of synthetic NT-proBNP in a protein containing buffer base, are included in each test run and are used to create a calibration curve. Three controls, containing low, medium, and high levels of synthetic NT-proBNP in a protein containing buffer matrix, are also included in each test run. In addition, the test kit contains a sample dilution buffer, wash concentrate, 3,3′,5,5′-tetramethylbenzidine d (TMB) substrate, and a stop solution (1 M sulfuric acid).
To perform the assay calibrators, controls and feline samples are diluted 1:4 into the sample dilution buffer. A portion (75 µl) of the sample mixture is added to the microtiter wells, and the plates are incubated at 25°C for 1 hr. Plates are washed 5 times with a wash solution in an automatic plate washer, then 75 µl of detection antibody-HRP conjugate is added to each well and the plates are incubated at 25°C for 1 hr. Plates are washed as above, followed by the addition of 100 µl of TMB substrate to each well; plates are then incubated at 25°C for 15 min. The color development is stopped by adding 50 µl of the provided stop solution. Optical density (OD) values are collected spectrophotometrically at 450 nm. A calibration curve is constructed using a commercial spreadsheet program, e and the concentration of NT-proBNP from each control or patient sample is calculated from this calibration curve.
Sample collection
Feline serum and plasma samples for all studies except the method comparison were obtained from IDEXX Reference Laboratories and stored frozen (–20°C) until use. Samples were randomly collected from the population of cats submitted to the Reference Laboratories for NT-proBNP testing. Sex, age, and breed information for samples collected from the Reference Laboratories were not provided. Samples for the method comparison study were prospectively recruited asymptomatic “at-risk” cats, with “at-risk” factors defined as the presence of a heart murmur, gallop, arrhythmia, and/or breed predisposition. Breeds represented include Abyssinian (n = 1), Bengal (n = 2), Cornish Rex (n = 2), Domestic Long Hair (n = 4), Domestic Medium Hair (n = 3), Domestic Short Hair (n = 40), Maine Coon (n = 4), Persian (n = 3), Ragdoll (n = 1), Sphynx (n = 4), and 2 were unrecorded. Forty-three cats were male (mean age: 7.5 years), 22 cats were female (mean age: 6.2 years), and the age and sex of 1 cat was not recorded.
Analytical validation
Methods used for analytical validation testing were based on industry standard guidance documents published by the United States Food and Drug Administration (FDA, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM). Guidance for Industry Bioanalytical Method Validation. 2001. Available from: http://www.fda.gov/downloads/Drugs/Guidances/ucm070107.pdf), the Clinical and Laboratory Standards Institute,1–4 and internal best practices. Statistical analysis was conducted with commercial software.f,g
Analytic sensitivity of the assay was determined by measuring the limit of blank (LoB) and limit of detection (LoD). Sixty replicates of a negative sample (porcine serum) and 60 replicates of a low positive sample (synthetic NT-proBNP spiked into porcine serum) were tested on the second-generation Cardiopet® test. a The LoB was determined by calculating the dose at 1.645 times the standard deviation (SD) of the 60 OD values. The LoD was calculated using the equation LoD = LoB + cβ (SD of sample readback), where cβ = 1.645/(1 – 1/(4 × f)) and f = degrees of freedom.
To determine accuracy and dilution linearity of the assay, 2-fold serial dilutions of 6 high concentration (>1,500 pmol/l) feline EDTA plasma samples (4 native and 2 pools spiked with synthetic NT-proBNP) were generated using low concentration feline EDTA plasma pools (28 and 29 pmol/l). Each sample was tested in duplicate on the second-generation Cardiopet assay and the dose calculated. As an analytical method for determining NT-proBNP dose has not been defined, the value of each sample was determined from the linear portion of the curve and a dilution factor was used to calculate the remaining predicted doses. Generally, the 1:4 dilution was used to define the assigned value. Ratios of the mean observed dose divided by the expected dose were calculated for each sample. Precision was measured by testing 4 feline serum sample pools (69, 124, 319, and 871 pmol/l) and 2 control samples (47 and 236 pmol/l). The concentrations of the sample pools and control samples were determined by testing each on 3 independent assay lots and averaging the average measured dose. Each sample was tested as 3 sets of duplicate wells (independent sample dilutions) on 2 plates per run with 3 independent assay lots. Two runs per day were conducted over 3 days by 3 operators. A total of 324 tests per sample were conducted. The testing was designed to determine intra-assay and interassay precision. For each sample, the mean, SD, and coefficient of variation (CV) was calculated.
Assay repeatability was measured by testing 317 matched serum and EDTA plasma feline samples on 2 independent lots of the assay. Deming regression correlation was obtained for both sample types.
To determine if the presence of commonly occurring sample components would interfere with the performance of the feline NT-proBNP assay, the impact of elevated levels of hemoglobin, lipids, and bilirubin were evaluated. Each component was spiked into a low NT-proBNP (64 pmol/l) and high NT-proBNP (521 pmol/l) feline serum pool at varying concentrations. A commercial system h was used to measure hemoglobin in each neat sample following the manufacturer’s instructions. Replicates of 2 were tested, with the mean concentration used to define levels. For the low-dose serum pool, hemoglobin i levels ranged from 30 to 555 mg/dl and, for the high-dose serum pool, levels ranged from 30 to 560 mg/dl. Lipid levels were measured by diluting samples 10-fold with phosphate buffered saline. An ultraviolet–visible spectroscopy spectrophotometer j was blanked with a phosphate buffered saline sample at an OD of 660 nm. The OD at 660 nm of 2 replicates of each diluted sample was then measured. Measured OD units were multiplied by the dilution factor of 10, and mean values were calculated. For the low-dose serum pool, lipid k levels ranged from values of 0.27 to 6.67 OD at 660 nm and, for the high-dose serum pool, lipid levels ranged from values of 0.25 to 6.44 OD at 660 nm. The total bilirubin concentration for each sample was determined using a commercial instrument l following the manufacturer’s instructions. Each sample was run neat in replicates of 2, and the mean concentration was calculated. For the low-dose serum pool, bilirubin m levels ranged from 0.28 to 17.35 mg/dl and, for the high dose serum pool, levels ranged from 0.23 to 17.68 mg/dl. For all components, the low level represents the base level measured in the unspiked feline serum pool. For each sample, 4 sets of duplicate wells (independent sample dilutions) were tested on multiple plates of a single lot of the assay. NT-proBNP doses were measured and compared to the unspiked feline serum pools via 1-way analysis of variance with an alpha value of 0.05.
Stability of NT-proBNP
Integrity of NT-proBNP as measured by assay performance was evaluated with a population of 92 feline serum samples stored without specialized protease inhibitor tubes. Samples spanned the range of the assay from 24 to 1,500 pmol/l (n = 29, <100 pmol/l; n = 15, 101–270 pmol/l; n = 48, >270 pmol/l). Samples were baseline tested on the assay (t = 0) then held at either 4°C (24, 48, 72, and 168 hr [7 days]) or 25°C (24 and 48 hr). A subset of the samples (n = 83) was held at 25°C for 72 hr. Samples were tested after storage, and the percent dose recovery of baseline was calculated. Paired t-test was used to compare the dose of NT-proBNP at 4°C and 25°C for each time point. In a second experiment, matched EDTA plasma and serum samples from 11 cats were stored at either 4°C or 25°C for 24, 48, and 72 hr. NT-proBNP dose was measured at each time and temperature and the mean dose percent recovery calculated as above. Paired t-test was used to compare mean dose recovery between EDTA plasma and serum.
Method comparison
Agreement between the first- and second-generation feline NT-proBNP immunoassays was examined using a clinically characterized population of 66 feline plasma samples, tested on both assays. A Bland–Altman analysis was performed using the mean values of the first-generation and second-generation assays, and the bias between methods was determined. A receiver-operator curve (ROC) analysis was performed to assess the performance of the first- and second-generation feline NT-proBNP assays at differentiating normal cats from those with OCM. In addition, median and interquartile range (IQR) NT-proBNP dose values were calculated for normal cats and cats with OCM, and a comparison between the groups was made via the Mann–Whitney test.
Results
Analytical validation
Analytical sensitivity of the assay was determined by measuring LoB and LoD. Figure 1 summarizes the results of the testing with a histogram. LoB was calculated to be 7 pmol/l (range: 0–9 pmol/l), and LoD was 10 pmol/l (range: 12–22 pmol/l).
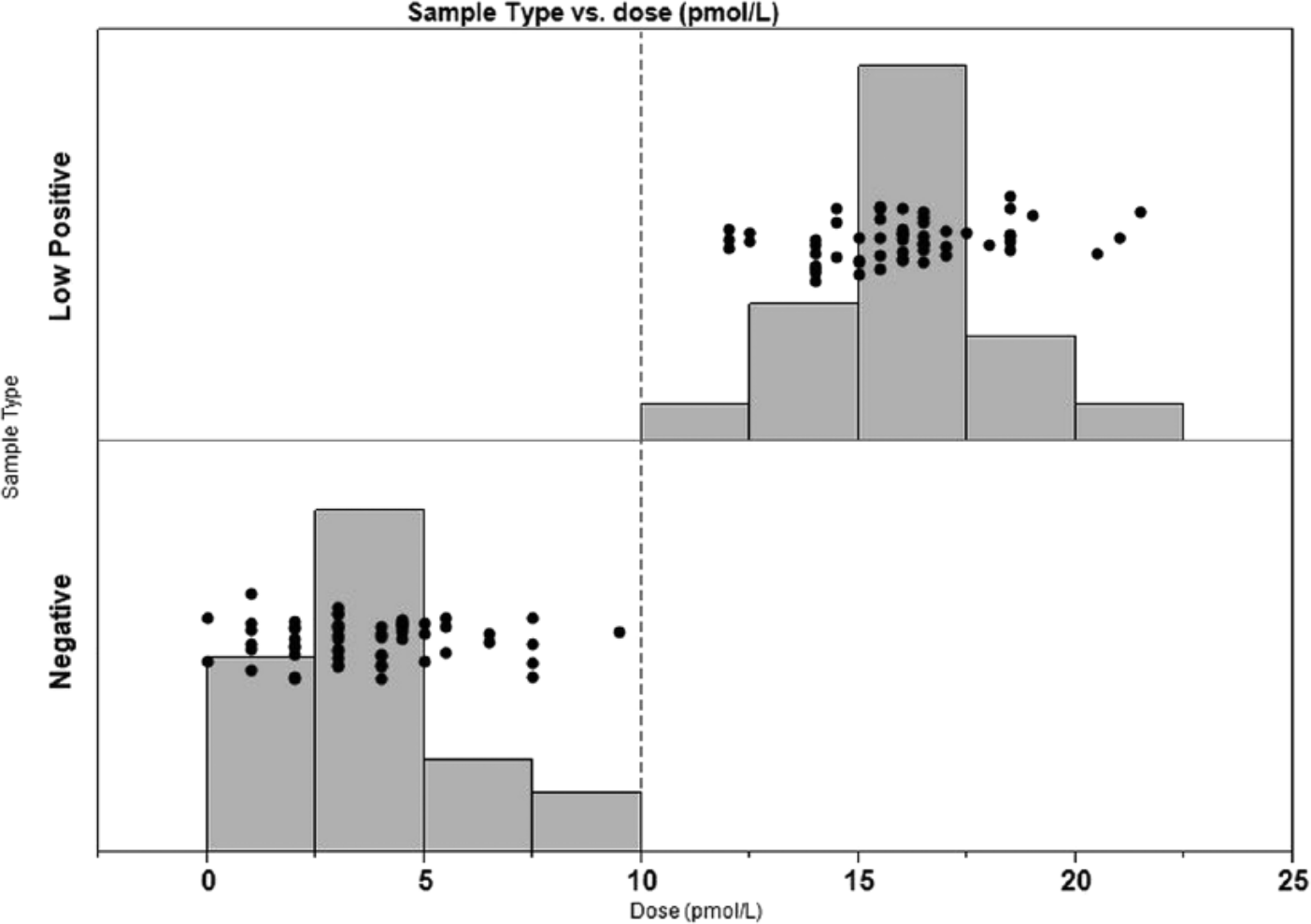
Histograms of 60 replicates of a low positive (top graph) and a negative (bottom graph) sample. N-terminal pro–B-type natriuretic peptide (NT-proBNP) dose in pmol/l is defined on the X-axis. Individual replicates are noted with a dot (●). The vertical dashed line indicates the limit of detection (LoD; 10 pmol/l) of the assay.
Accuracy and dilution linearity of the assay, defined by calculating a ratio of the mean observed dose divided by the expected dose for all samples tested, was found to be 103.5% ± 11.5%. The measured dose compared to the predicted dose for all samples, with a 45° identity line indicated, is graphed in Figure 2. Slopes of the linear regression lines for each sample ranged from 0.824 to 1.24 (data not shown).
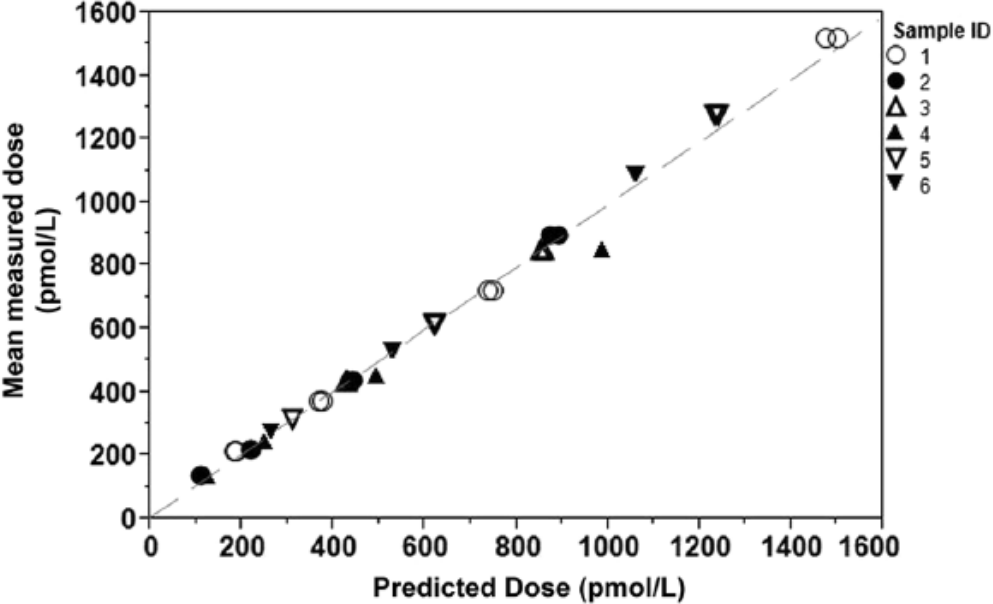
Twofold serial dilution of 6 independent samples with mean measured N-terminal pro–B-type natriuretic peptide (NT-proBNP) dose (pmol/l) plotted against predicted NT-proBNP dose (pmol/l). Dashed line indicates a 45° identity line. The calculated ratio of the mean observed dose divided by the expected dose across all samples is 103.5% ± 11.5%.
Intra-assay and interassay precision was measured for 4 feline serum sample pools (69, 124, 319, and 871 pmol/l) and 2 control samples (47 and 236 pmol/l). The intra-assay %CV ranged from 1.6 to 6.3%, the interassay %CV from 4.3 to 8.8%, and the total %CV from 10.1 to 15.1% (Table 1).
Summary of precision testing for 4 native sample pools (69, 124, 319, and 871 pmol/l) and 2 control samples (47 and 236 pmol/l).*
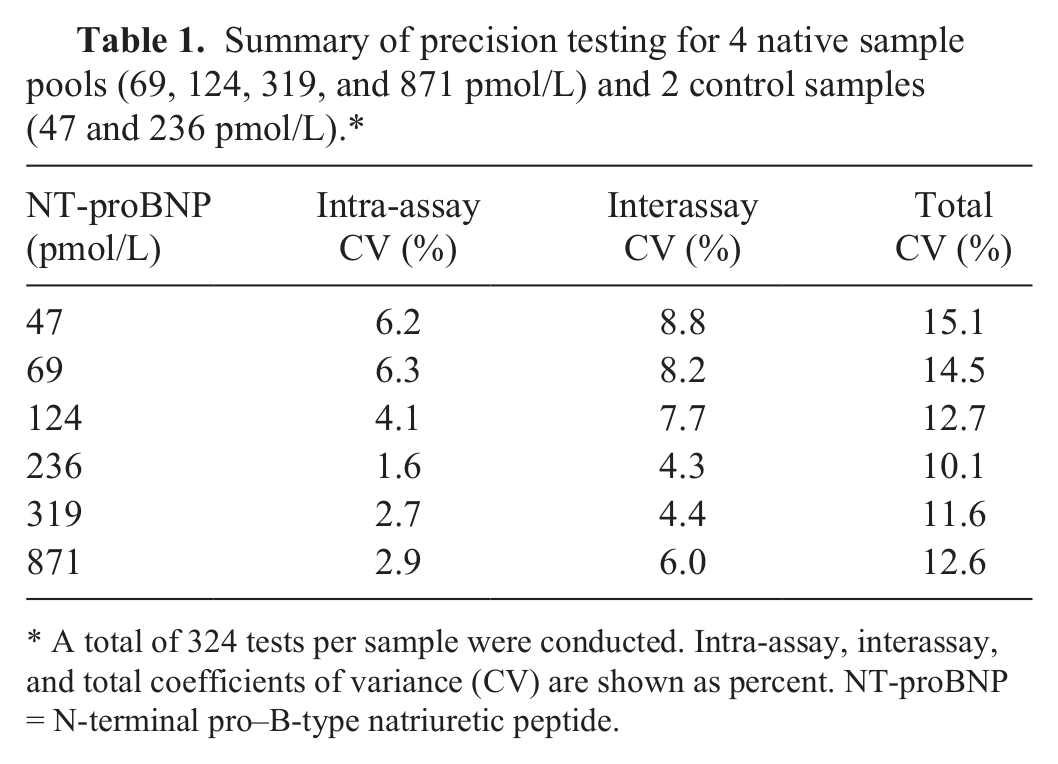
A total of 324 tests per sample were conducted. Intra-assay, interassay, and total coefficients of variance (CV) are shown as percent. NT-proBNP = N-terminal pro–B-type natriuretic peptide; CV = coefficient of variation.
Comparison of 317 matched feline serum and EDTA plasma samples on 2 independent lots of the assay resulted in similar NT-proBNP doses reported by both lots for both sample types (Fig. 3). Deming regression correlation for EDTA plasma was 0.9945 (slope 1.11) and for serum 0.9955 (slope 1.11).
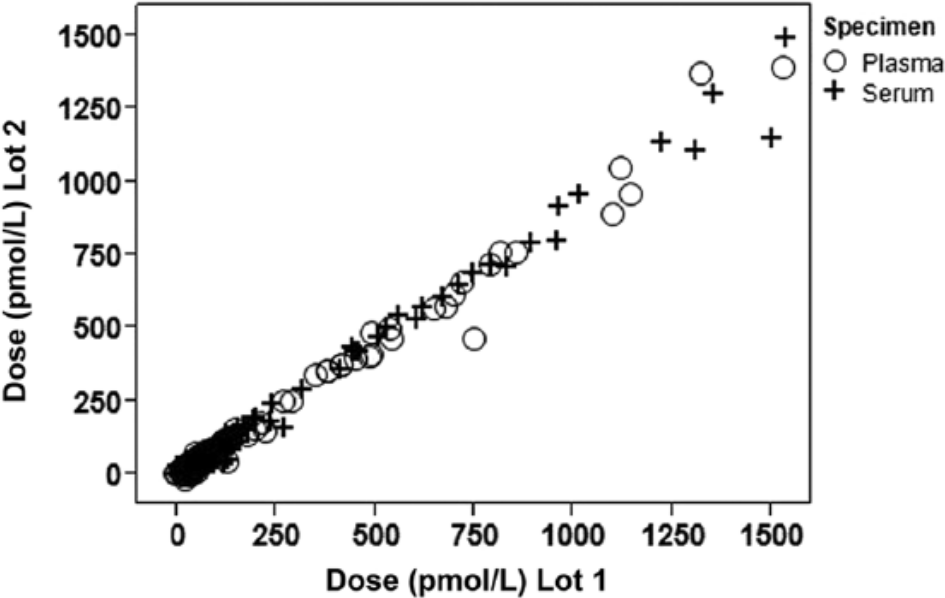
To examine assay repeatability, 317 matched feline serum (+) and ethylenediamine tetra-acetic acid (EDTA) plasma (○) samples were tested on 2 independent lots of the feline N-terminal pro–B-type natriuretic peptide (NT-proBNP) immunoassay. Scatter plot with NT-proBNP values (pmol/l) from lot 1 plotted against NT-proBNP values from lot 2 is shown. Deming regression correlation for EDTA plasma was 0.9945 (slope 1.11) and for serum 0.9955 (slope 1.11).
The analysis of interference from hemoglobin, lipid, and bilirubin (Fig. 4) showed statistically significant difference between the unspiked high NT-proBNP dose serum sample and samples with spiked hemoglobin (P = 0.0453, mean bias: –3.3 pmol/l), lipid (P = 0.001, mean bias: 30.3), or bilirubin (P = 0.0249, mean bias: −15.1 pmol/l). No statistically significant differences were observed between the unspiked low NT-proBNP dose serum sample and samples with spiked hemoglobin (P = 0.2406, mean bias: –2.1 pmol/l), lipid (P = 0.1167, mean bias: 2.0 pmol/l) or bilirubin (P = 0.5022, mean bias: 2.2 pmol/l).
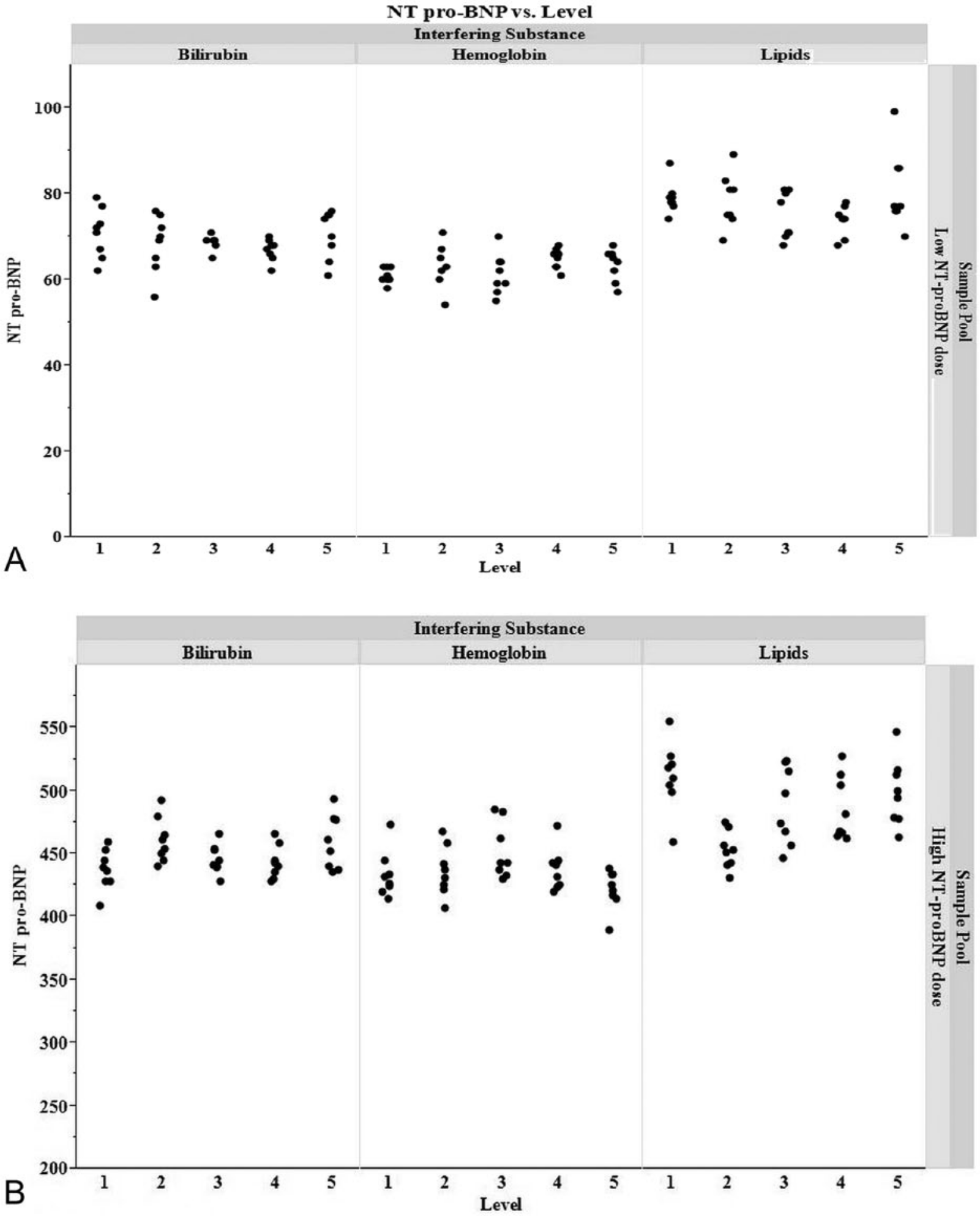
Bilirubin, hemoglobin, and lipids were spiked into a low N-terminal pro–B-type natriuretic peptide (NT-proBNP; 64 pmol/l) and high NT-proBNP (521 pmol/l) feline sample pool at increasing levels. Results of the low-dose samples are shown in the top graph (a); the high-dose samples are summarized on the bottom graph (b). Bilirubin levels for the low-dose sample are 1) unspiked, 0.28 mg/dl; 2) 4.28 mg/dl; 3) 8.82 mg/dl; 4) 13.29 mg/dl; 5) 17.35 mg/dl. Hemoglobin levels for the low-dose samples are 1) unspiked, 30 mg/dl; 2) 160 mg/dl; 3) 300 mg/dl; 4) 430 mg/dl; 5) 555 mg/dl. Lipid levels for the low-dose samples are 1) unspiked, 0.27 optical density at 660 nm (OD660); 2) 1.9 OD660; 3) 3.53 OD660; 4) 5.15 OD660; 5) 6.67 OD660. Bilirubin levels for the high-dose sample are 1) unspiked, 0.23 mg/dl; 2) 4.67 mg/dl; 3) 8.63 mg/dl; 4) 13.19 mg/dl; 5) 17.68 mg/dl. Hemoglobin levels for the high-dose samples are 1) unspiked, 30 mg/dl; 2) 170 mg/dl; 3) 300 mg/dl; 4) 430 mg/dl; 5) 560 mg/dl. Lipid levels for the high-dose samples are 1) unspiked, 0.25 OD660; 2) 1.88 OD660; 3) 3.41 OD660; 4) 4.93 OD660; 5) 6.44 OD660.
Stability of NT-proBNP
Integrity of NT-proBNP was tested with a population of 92 feline serum samples stored without specialized protease inhibitor tubes. The mean dose percent recovery for samples held at 4°C for 24 hr was 100% ± 7.1%, after 48 hr was 99.1% ± 7.1%, and 99.1% ± 7.9% after 72 hr (Table 2). Samples held for 7 days (168 hr) at 4°C had a mean dose percent recovery of 94.4% ± 10.0%. The mean dose percent recovery for samples held at 25°C for 24 hr was 91.9% ± 13.5%, after 48 hr was 85.6% ± 17.8%, and 80.0% ± 21.0% after 72 hr. Statistically significant differences (P
Percent of baseline N-terminal pro–B-type natriuretic peptide (NT-proBNP) dose recovery from time = 0 for 92 native feline serum samples stored at 4°C for 24, 48, 72, and 168 hr or 25°C for 24, 48, and 72 hr.*
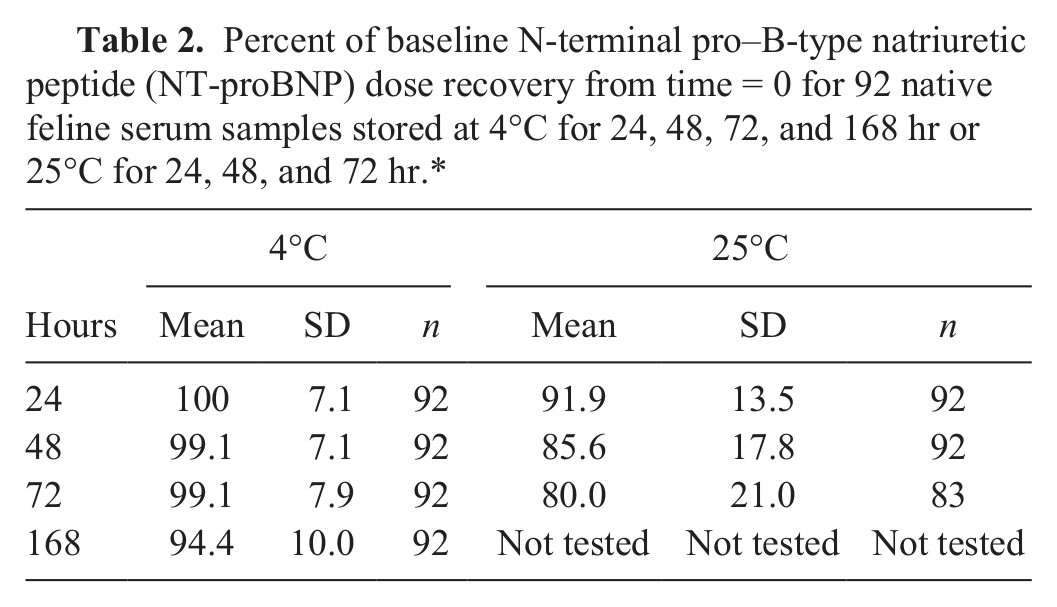
Because of insufficient volume, only 83 samples were tested at the 25°C 72 hr time point. Means and standard deviations are shown. SD = standard deviation; n = number of samples tested.
Method comparison
Testing a clinically characterized population of 66 feline samples on the first- and second-generation feline NT-proBNP assays revealed no statistically significant difference in the reported NT-proBNP values (P = 0.1992) with a mean bias of 34 pmol/l (Fig. 5). ROC analysis performed to assess the ability of both assays to differentiate normal cats from those with OCM resulted in areas under the curve (AUC) for the first- and second-generation assay of 0.8454 and 0.8505, respectively, with the observed difference determined not to be statistically significant (P = 0.9395) by the Hanley–McNeil test (data not shown). Median NT-proBNP dose values were significantly higher (P
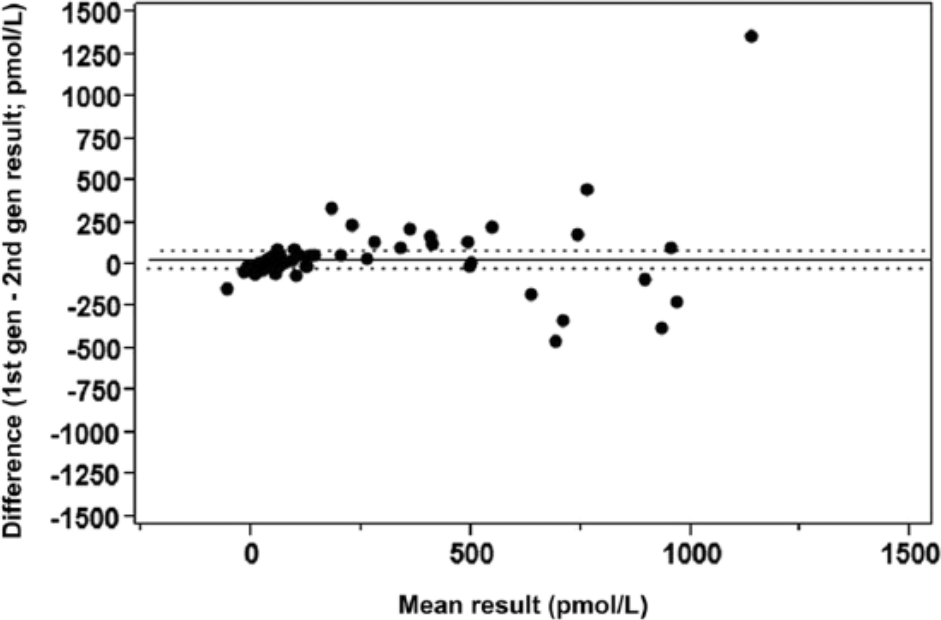
Bland–Altman difference plot comparing N-terminal pro–B-type natriuretic peptide (NT-proBNP) dose results of 66 feline samples tested on the first- and second-generation feline NT-proBNP assays. The overall mean bias is 34 pmol/l, which is not statistically significant (P = 0.1992). The solid horizontal line indicates the mean bias with 95% confidence intervals as dotted lines above and below.
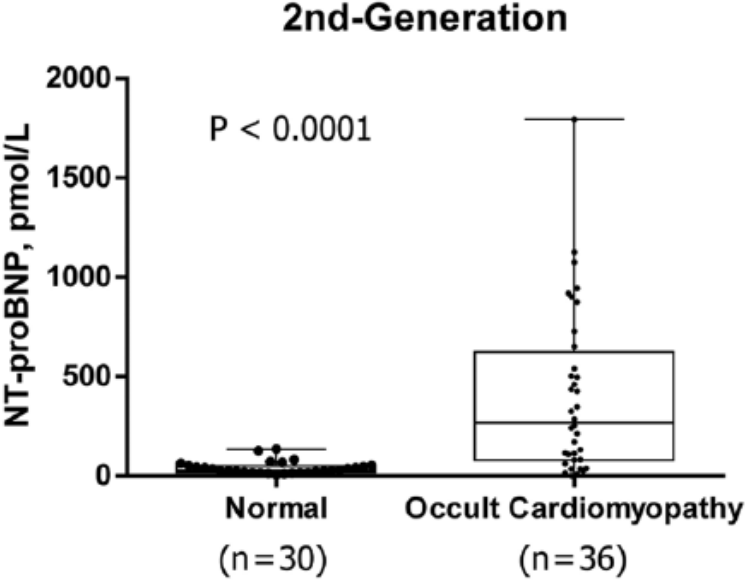
Box and whiskers plots indicating the second-generation feline N-terminal pro–B-type natriuretic peptide (NT-proBNP) immunoassay retains the ability to differentiate between normal cats and cats with occult cardiomyopathy (OCM). Median NT-proBNP dose values were significantly higher (Mann–Whitney test, P < 0.0001) for cats with OCM (270 pmol/l, interquartile range [IQR] 83–569 pmol/l) than normal cats (28 pmol/l, IQR 19–51 pmol/l).
Discussion
The results of our evaluation indicate that the performance of a second-generation feline NT-proBNP immunoassay with improved precision and accuracy will more effectively measure NT-proBNP, while retaining the clinical utility of the assay. The ability to differentiate normal cats from those with OCM by measuring levels of NT-proBNP has been preserved with the second-generation assay. While no significant bias was observed, the levels of NT-proBNP for individual animals may be reported differently by the 2 assays. If cats are being monitored over time for NT-proBNP, it would therefore be important to establish new baseline levels for individual cats.
The improved precision and accuracy may support the use of the assay for monitoring levels of NT-proBNP over time in cats at risk of cardiac disease. The level of biological or longitudinal variation of NT-proBNP in individual cats is not known. Further study is needed to support the clinical utility of monitoring NT-proBNP levels for diagnostic or treatment applications. In addition, the impact of alternating between EDTA plasma and serum for longitudinal monitoring is not known, therefore a consistent sample type would be recommended to minimize sample type as a source of potential variation in this application.
No statistically significant differences from baseline were observed for a sample with low levels of NT-proBNP to which varying levels of hemoglobin, lipids, and bilirubin had been added. Significant impact of varying levels of hemoglobin, lipids, and bilirubin on measured levels of NT-proBNP was observed for a sample with high levels of NT-proBNP. However, the mean bias was between –15.1 and 30.3 pmol/l, was not directional across multiple levels of added substance, and was consistent with what would be expected due to normal assay variation. Normal assay variation for our experiment may be estimated by 2 control samples tested, control A (n = 6 replicates, mean dose: 49.5 pmol/l, SD: 7.9 pmol/l) and control C (n = 6 replicates, mean dose: 500.5 pmol/l, SD: 45.5 pmol/l). For all, the mean bias observed was within 1 or 2 SDs of the mean dose value for the baseline sample (SD for baseline hemoglobin sample: 18.5 pmol/l, for baseline lipid sample: 27.3 pmol/l, and for baseline bilirubin sample: 15.8 pmol/l), supporting the hypothesis that any bias observed is likely due to normal assay variation and not the substances tested. The impact on the assay for levels of hemoglobin, lipids, and bilirubin higher than those tested is not known.
The second-generation assay supports the use of both serum and EDTA plasma samples. Stability of the NT-proBNP analyte as measured by percent baseline recovery is greater than 94% for serum samples held for 168 hr (7 days) at 4°C and greater than 80% for samples held at 25°C for 72 hr. As a statistically significant decline in recovery is observed at 25°C for 72 hr, when possible, samples should be submitted on ice for evaluation, or some underreported values of the analyte may occur with individual samples. In addition, the effect on analyte integrity and subsequent impact to reported levels of NT-proBNP at temperatures higher than 25°C is not known. The impact on NT-proBNP integrity of a sample cycling between 4°C and higher temperatures has also not been established. Limited evaluation of NT-proBNP analyte integrity in EDTA plasma samples has been done. However, when a small population of matched EDTA plasma and serum samples was compared for 72 hr, no statistically significant difference was observed, indicating that NT-proBNP stability is similar in both sample types.
The second-generation feline NT-proBNP assay contains anti-feline NT-proBNP capture and detection antibodies distinct from the first-generation assay. These antibodies target epitopes directed to the N-terminal portion of NT-proBNP. NT-proBNP stability studies suggest that the N-terminal epitopes are more stable than those in the first-generation assay7,8 where declines in the median NT-proBNP concentration were observed over time in the absence of a protease inhibitor tube. 6
The second-generation feline NT-proBNP immunoassay accurately measures levels of NT-proBNP, which are often elevated in cats with cardiac disease, and the use of this test can improve diagnostic accuracy. 11 However, levels of NT-proBNP may also be elevated due to concurrent diseases such as hypertension, hyperthyroidism, and severe azotemia. 9 The use of the feline NT-proBNP immunoassay, therefore, should be included as part of a more comprehensive diagnostic work-up for cats at risk for cardiac disease. The improved assay, using routine sample submission, should facilitate regular inclusion of NT-proBNP measurement in the clinical setting.
Footnotes
Acknowledgements
The authors acknowledge Drs. Mark Oyama and Maggie Machen (University of Pennsylvania School of Veterinary Medicine) and Dr. Sonya Gordon (Texas A&M University) for supplying the population of feline samples for method comparison studies.
Authors’ contributions
CA Mainville contributed to conception and design of the study, contributed to analysis and interpretation of data, drafted the manuscript, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. GH Clark, KJ Esty, WM Foster, JL Hanscom, KJ Hebert, and HR Lyons contributed to design of the study, contributed to acquisition and analysis of data, and critically revised the manuscript.
a.
Feline Cardiopet proBNP test kit, IDEXX Laboratories Inc., Westbrook, ME.
b.
Immulon, Thermo Fisher Scientific, Waltham, MA.
c.
Roche Molecular Biochemicals, Indianapolis, IN.
d.
Seramun Diagnostica GmbH, Heidesee, Germany.
e.
Excel, Microsoft Corp., Redmond, WA.
f.
Analyse-it Software Ltd., Leeds, UK.
g.
JMP v. 10.0, SAS Institute Inc., Cary, NC.
h.
HemoCue Plasma/Low Hb System, HemoCue America, Brea, CA.
i.
Bovine hemoglobin, Sigma-Aldrich, St. Louis, MO.
j.
Beckman DU 640B, Beckman Coulter Inc., Brea, CA.
k.
Intralipid 30% IV solution, VWR International, Radnor, PA.
l.
Vitros 350, Ortho-Clinical Diagnostics Inc., Rochester, NY.
m.
Ditaurobilirubin disodium salt, JBL Scientific Inc., San Luis Obispo, CA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of IDEXX Laboratories Inc., Westbrook, Maine.
Funding
The author(s) received no financial support for research, authorship, and/or publication of this article.
