Abstract
Although Campylobacter jejuni is a common cause of ruminant abortion with great economic impact, the organism has rarely been implicated in canine pregnancy loss, with only 2 documented cases to date. In the current report, 2 cases of perinatal death in adult female Bulldogs associated with C. jejuni infection of fetoplacental organs are described. Fetuses and placentas were received at the Iowa State University Veterinary Diagnostic Laboratory (Ames, Iowa) from 3 puppies that died soon after the birth (case 1) and from an aborted fetus (case 2). Microscopic examination of tissues was generally unremarkable; however, multifocal hemorrhage and infiltrates of macrophages and neutrophils were observed in placental sections from the first case (case 1), and low to moderate numbers of degenerate neutrophils were apparent within multifocal alveoli in the fetal lung in the second case (case 2). Ancillary diagnostics for common infectious causes of reproductive failure in dogs were negative. However, C. jejuni was isolated from the submitted placentas in high numbers in both cases as well as from the fetal lungs and livers. Genotyping of the abortion isolates indicated that the isolates were distinct from each other as well as from selected canine enteric C. jejuni isolates included herein for comparison. Both abortion strains were sensitive to all 9 antimicrobials tested, except the isolate from case 2, which displayed resistance to tetracycline. These findings provide convincing evidence for the inclusion of C. jejuni culture in routine diagnostic testing for causes of canine pregnancy loss.
In addition to being a leading cause of bacterial foodborne gastroenteritis worldwide, 1 Campylobacter jejuni is one of the most frequently implicated pathogens in infectious abortions of ruminants, especially of sheep (epizootic abortion) and to a lesser extent, cattle and goats.12,14 The organism is also a rare cause of pregnancy loss in human beings. 15 Although historically C. fetus subsp. fetus has accounted for the majority of Campylobacter-associated ovine reproductive failure, C. jejuni is now the predominant abortifacient species in pregnant sheep, especially in the United States.5,12 Campylobacter jejuni is commonly present in the intestine of healthy sheep, and, in susceptible pregnant ewes, the pathogen could become bacteremic with subsequent placentitis, fetal infection, and abortion, which usually occurs in the last trimester of pregnancy. 14 Aborted ewes typically do not show any signs of clinical disease. Microscopically, aborted placentas often have evidence of acute suppurative placentitis, and the trophoblasts can be distended with intracytoplasmic organisms. 14 A mild to moderate suppurative fetal bronchopneumonia is also commonly identified. The organism can be readily cultured in large numbers from aborted placentas, fetal stomach contents, and to a lesser extent lung and liver.12,14
In dogs, Brucella canis is the major cause of bacterial abortions, and there are only 2 documented reports on Campylobacter-associated abortion to date. 11 The first case report 4 is from 1984 and describes 2 separate cases in the northwestern United States. In the first instance, which involved the premature labor of a Wheaten Terrier dam delivering 1 live and 2 dead fetuses, C. jejuni was isolated in pure culture from fetal liver and stomach contents of the aborted pup. In the second instance, Campylobacter colonies (species-level identification was not possible due to the loss of the culture during later stages of the process) were isolated from the aborted fetal lung and liver tissues of 3 poodle pups. 4 The second case report 9 is from 1994 from South Africa and describes the isolation of C. jejuni in pure culture from the vaginal discharge of 3 German Shepherd Dog dams following apparent abortions that occurred during late gestation.
Although the pathogenesis of C. jejuni–induced abortion in pregnant dogs is not clear, it is likely to involve a similar process to that in sheep (fecal–oral transmission) because both animals frequently harbor the organism in the intestine asymptomatically and C. jejuni is not normally found in the vaginal canal of either species.4,9,14 The current report documents 2 cases of C. jejuni–associated abortion in dogs from the Midwestern United States. In addition, detailed molecular and phenotypic characterization of the abortion isolates as well as time and/or region–matched canine enteritis–associated isolates are described with specific references to zoonotic potential for the public.
Case 1
In August 2012, 3 fetuses and placentas were received at the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, Iowa) from an adult female Bulldog that had presented to the referring veterinarian with frank vaginal bleeding 7 days prior to her expected whelping date. A cesarean section was performed, and 5 puppies were delivered. The 3 puppies submitted to the ISU-VDL were viable for a short period of time and then expired, 2 other puppies were delivered dead and both had amniotic sacs filled with hemorrhagic fluid. Microscopic examination of fetal tissues was unremarkable. However, histology of sections of placenta revealed multifocal dilation of lacunae with associated accumulations of fibrin and erythrocytes admixed with degenerate neutrophils and cellular and karyorrhectic debris (Fig. 1). Degeneration and necrosis of the syncytiotrophoblast layer was variably apparent in sections of the placental labyrinth. Additionally, there was mild multifocal and often perivascular infiltration of neutrophils, lymphocytes, and plasma cells within the allantoic stroma.
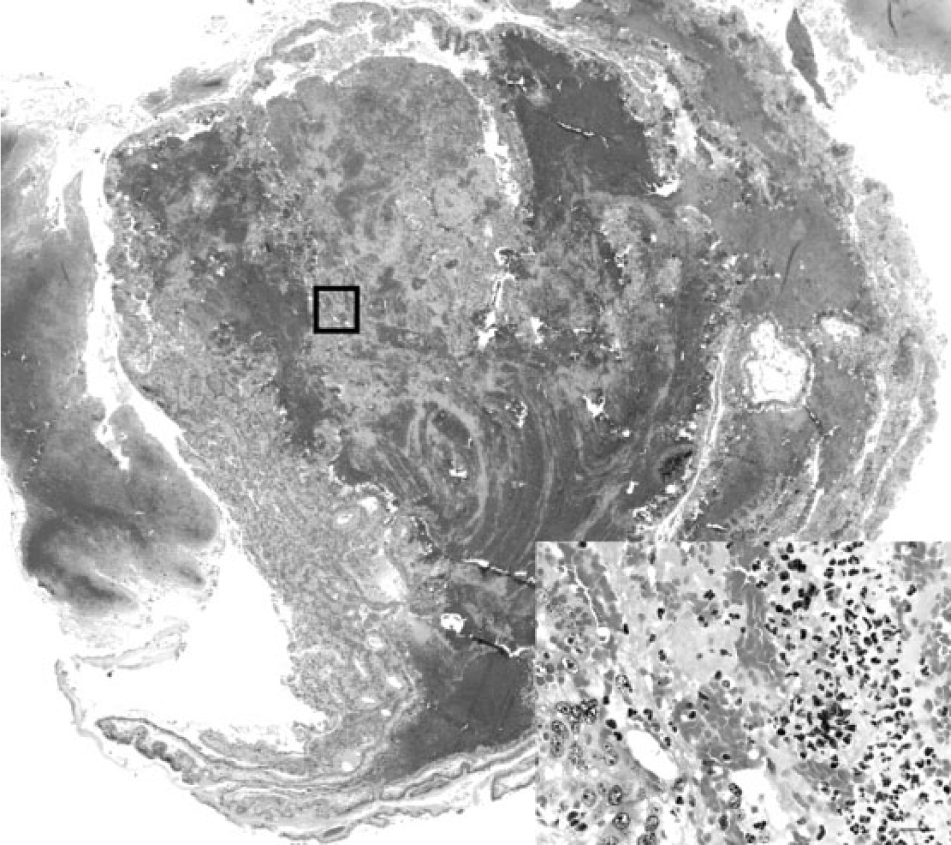
Bulldog. Photomicrograph of aborted placenta from case 1 revealing disruption of the placental architecture with accumulations of fibrin and erythrocytes admixed with cellular and karyorrhectic debris. The inset image represents the area within the box in the primary image and reveals degenerate neutrophils admixed with necrotic debris and erythrocytes. Hematoxylin and eosin. Bar = 20 µm.
Case 2
In November 2013, a single aborted fetus was received at the ISU-VDL from an adult female Bulldog that started labor 8 days prior to her expected whelping date. The dog did not appear ill or lethargic and delivered the entire litter dead. There was marked postmortem autolysis of fetal tissues that greatly impeded evaluation; however, low numbers of degenerate neutrophils were scattered throughout sections of lung. Microscopic evaluation of other fetal tissues and the small piece of placenta submitted was unremarkable.
For both cases, ancillary diagnostics for common infectious causes of reproductive failure in dogs were performed. The tests included the following: 1) a Leptospira spp.–specific polymerase chain reaction (PCR) test on aborted tissues as previously described, 16 2) a routine direct antigen fluorescent antibody test for Canid herpesvirus 1 on aborted tissues using Canid herpesvirus 1–specific polyclonal antiserum labeled with fluorescein isothiocyanate, a and 3) routine bacterial culture from the aborted tissues. The latter 2 diagnostic procedures were performed in the ISU-VDL using standard operating procedures with the bacterial culture panel including both general-purpose and selective media incubated under different atmospheric conditions and temperatures for isolation of a wide range of bacterial organisms including Brucella, Campylobacter, Escherichia coli, Salmonella, Staphylococcus, and Streptococcus spp.
The results of the diagnostics tests revealed that the samples from both cases were all negative for the infectious agents listed above with the following exception. Campylobacter-like colonies on blood agar containing selective supplements b were recovered from placentas in high numbers and from the fetal lungs and livers in low numbers after incubation for 48 hr at 37°C under microaerobic conditions. It should be pointed out that the placenta from case 2 also yielded moderate growth of mucoid E. coli. The E. coli isolate was deemed most likely a contaminant in this case as this organism can be carried in the genital tract of normal dogs 11 and was not isolated from the fetal liver or lungs whereas Campylobacter-like colonies were isolated from all 3 tissues. For confirmation and species-level identification, a single Campylobacter-like colony from each placenta was subpassaged to obtain pure cultures and subjected to dark-field microscopy for characteristic darting motility, matrix-assisted laser desorption ionization time-of-flight mass spectrometry, and PCR using established methods,8,13 which identified both isolates as C. jejuni.
Fluorescent in situ hybridization (FISH) was performed on affected paraffin-embedded placental tissues (4-µm sections on positively charged slides) using a previously described oligonucleotide probe targeting the 23S ribosomal RNA of C. jejuni. 10 The oligonucleotide probe was obtained from a commercial source c with a 5′-label of orange fluorescent d dye and was reconstituted in a commercial hybridization buffer e immediately prior to use (5 ng/µl). The FISH assay was performed using a commercially available system f as follows: 1) sections were dewaxed using a commercial dewaxing solution g and alcohol per the manufacturer’s directions, 2) sections were then treated with a commercially available enzymatic pretreatment h for 5 min followed by 5 rinses with a commercial wash solution, i 3) the diluted probe was applied and allowed to hybridize at 45°C for 16 hr, and 4) sections were rinsed, coverslipped in aqueous mounting media, and immediately read with a fluorescent microscope with a filter appropriate for the fluorescent dye. In tissues from case 1, occasional trophoblasts were observed with distended cytoplasm filled with fluorescent-labeled bacteria; however, more commonly there were high numbers of individual or clustered, curved to spiral-shaped, fluorescent-labeled bacteria varying from 0.5 to 1.0 µm in length (Fig. 2) consistent with C. jejuni. In case 2, there were occasional clusters of fluorescent-labeled bacteria within sections of placenta. In light of the culture results, histology, and positive labeling with C. jejuni–specific FISH, C. jejuni was established as the sole cause of the abortions in these cases.
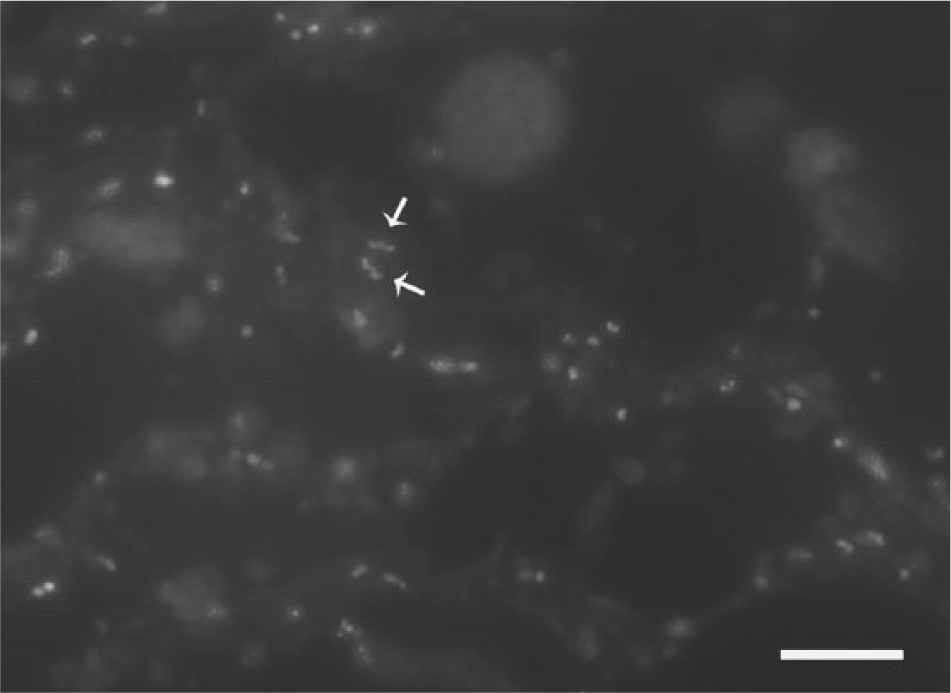
Bulldog. Photomicrograph of placenta from case 1 revealing moderate numbers of individual curved to spiral-shaped, fluorescent-labeled bacteria (arrows) varying from 0.5 to 1.0 µm in length consistent with Campylobacter jejuni. Fluorescent in situ hybridization using an oligonucleotide probe specific for C. jejuni. 10 Bar = 10 µm.
Next, additional laboratory tests for phenotypic and genotypic characterization of the abortifacient C. jejuni isolates as well as those from canine enteritis cases obtained during a 5-year period (2007–2012) within the same geographic region were performed. A standard broth microdilution test using a commercially available kit j showed that the C. jejuni isolates from dogs were generally susceptible to most of the 9 antibiotics tested (Table 1). However, the isolate from case 2 was resistant to tetracycline. Interestingly, one of the enteritis isolates (VDL06279) exhibited a multiresistance phenotype to a diverse array of antimicrobial classes. Genotyping of these isolates was performed using pulsed-field gel electrophoresis (PFGE) and multilocus sequence typing (MLST) as described previously. 12 The PFGE typing with SmaI restriction enzyme indicated that the abortion strains (Fig. 3, lanes 1 and 2) were distinct from each other and from those of enteritis strains (Fig. 3, lanes 3–9). Of note, the C. jejuni isolates from enteritis were of different subtypes although VDL26018 (lane 3) and VDL31851 (lane 8) appeared indistinguishable. In agreement with PFGE typing, MLST findings also revealed that the canine abortion and enteritis C. jejuni isolates were of different sequence types and there was no overlap among any strains (Table 2). The findings from both typing methods indicated a substantial level of genetic diversity among C. jejuni isolates from dogs analyzed in the current study. Also, it should be noted that the canine abortion C. jejuni isolates described in the current report are genotypically distinct from C. jejuni clone SA, which has been identified as the predominant cause of sheep abortions in the United States during the past decade. 12
Minimum inhibitory concentrations (µg/ml) of 9 antibiotics for 8 Campylobacter jejuni isolates from dog abortion (n = 2) and canine enteritis (n = 6) cases as determined by a microbroth dilution test.
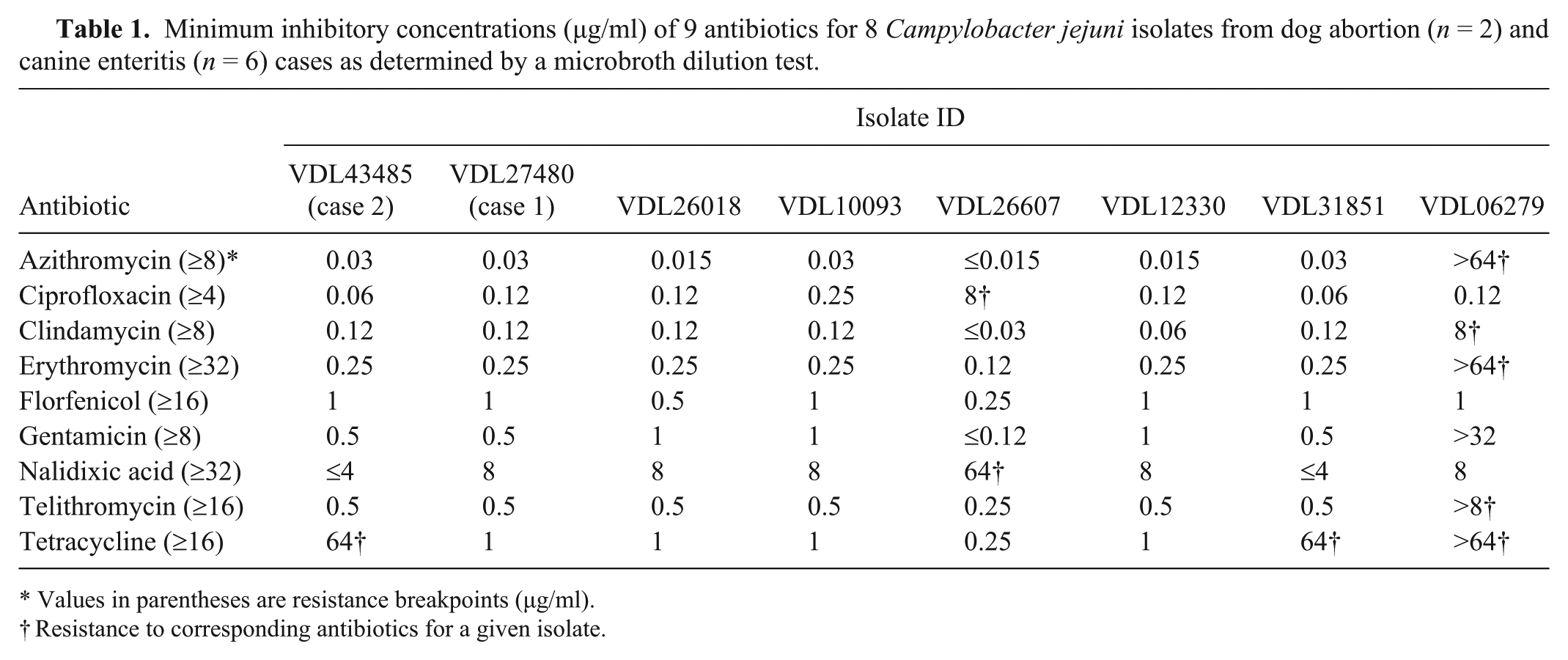
Values in parentheses are resistance breakpoints (µg/ml).
Resistance to corresponding antibiotics for a given isolate.
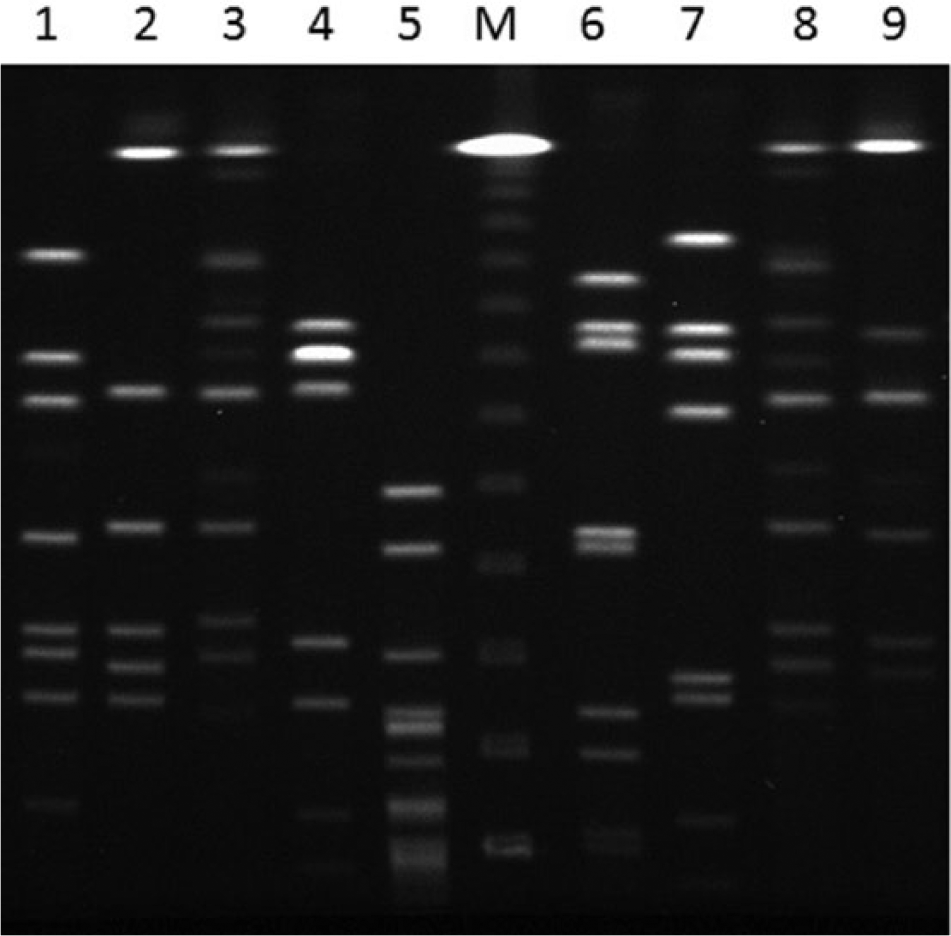
Pulsed-field gel electrophoresis analysis of Campylobacter jejuni isolates from Bulldogs using SmaI restriction enzyme. Lanes 1 (case 2) and 2 (case 1) are from abortion cases; lanes 3–9 are enteritis isolates; lane 5 is a Campylobacter sp. isolate other than C. jejuni or C. coli (species-level identification was not performed); lane M: lambda DNA ladder. k
Multilocus sequence typing (MLST) analysis of Campylobacter jejuni isolates from dogs and corresponding sequence type (ST) matches to human cases in the PubMLST database.*
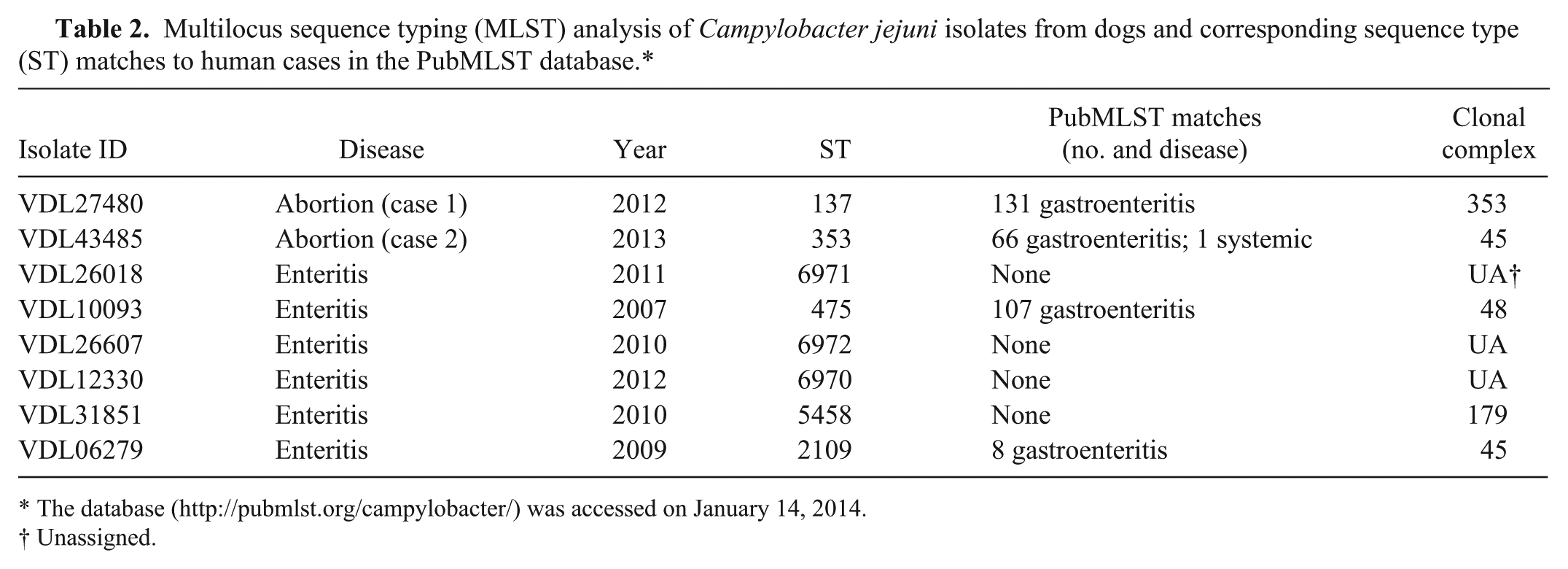
The database (http://pubmlst.org/campylobacter/) was accessed on January 14, 2014.
Unassigned.
Although a great majority of Campylobacter infections in human beings are associated with consumption of undercooked chicken and raw milk, contact with dogs has been identified as a significant risk factor and potential source for human campylobacteriosis in many epidemiological studies.6,7,17 Considering both healthy and diarrheic dogs can carry C. jejuni in the intestine,2,17 their role as a significant reservoir for human infections is not surprising. Thus, the zoonotic potential of the abortifacient canine C. jejuni isolates from the current report was further investigated with the use of the publicly available Campylobacter PubMLST database (http://pubmlst.org/campylobacter/). The C. jejuni isolates from cases 1 and 2 belong to ST-137 and ST-353, respectively (Table 2). There are many matches in the MLST database for ST-137, including over 131 cases of human gastroenteritis as well as a few cases of diarrheic dogs. Similarly, there are many C. jejuni isolates belonging to ST-353, including over 66 from human gastroenteritis and a few from healthy and diarrheic dogs (Table 2). Interestingly, one of the strains in ST-353 was isolated from blood in a case of human systemic infection. Although exact ST matches between C. jejuni isolates from canine abortions and human infections does not necessarily implicate a cause–effect relationship, it does suggest that abortifacient C. jejuni of canine origin has substantial zoonotic potential for human disease, both gastroenteritis and to a lesser extent systemic infection. Therefore, precaution should be exercised by owners and caretakers of aborting dogs as the aborted fetuses and fluids may have large numbers of C. jejuni organisms with zoonotic potential for susceptible individuals. Most Campylobacter infections in human beings are self-limiting and usually do not require antimicrobial treatment; however, macrolides are often the first choice when antibiotic therapy is indicated for severe and chronic conditions. 3 In this regard, it is important to note that one of the enteritis isolates (VDL06279) displayed high-level resistance to all macrolide group antibiotics (azithromycin, erythromycin, and telithromycin; Table 1). Considering that antibiotic-resistant Campylobacter is one of the more serious public health threats listed in a recent report (http://www.cdc.gov/drugresistance/threat-report-2013/) by the Centers for Disease Control and Prevention and the fact that canine isolate VDL06279 has the same sequence type as several human C. jejuni gastroenteritis isolates in the PubMLST database (Table 2), the potential role of dogs in zoonotic transmission of drug-resistant C. jejuni is evident.
Footnotes
Acknowledgements
The authors are grateful to the many Iowa State University Veterinary Diagnostic Laboratory staff and personnel who were engaged in the routine processing and bacterial isolation procedures associated with the cases of this report, and to Mr. Joe Brodie for his technical assistance with the fluorescent in situ hybridization procedures described herein.
a.
Canine herpesvirus type 1 (CHV-1) FITC conjugate, VMRD Inc., Pullman, WA.
b.
Skirrow Campylobacter selective supplement (Oxoid, SR0069), Fisher Scientific UK Ltd, Loughborough, Leicestershire, United Kingdom.
c.
Invitrogen Custom Oligos, Life Technologies, Carlsbad, CA.
d.
Alexa Fluor 555, Life Technologies, Carlsbad, CA.
e.
Bond hybridization solution, Leica Biosystems, Newcastle upon Tyne, United Kingdom.
f.
Leica Bond-III, Leica Biosystems, Melbourne, Australia.
g.
Bond dewax solution, Leica Biosystems, Newcastle upon Tyne, United Kingdom.
h.
Bond enzyme pretreatment kit, Leica Biosystems, Newcastle upon Tyne, United Kingdom.
i.
Bond wash solution, Leica Biosystems, Newcastle upon Tyne, United Kingdom.
j.
Sensititre Campylobacter MIC plate, Thermo Fisher Scientific, Waltham, MA.
k.
Lambda DNA ladder, Bio-Rad Laboratories, Hercules, CA.
Declaration of conflict of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
