Abstract
A simple and accurate method for quantifying sucrose in equine serum that can be applied to sucrose permeability testing in the horse was developed and validated using gas chromatography with flame ionization detection. The assay provided an acceptable degree of linearity, accuracy, and precision at concentrations of sucrose as low as 2.34 μmol/l and as high as 20.45 μmol/l. Percentage recovery of sucrose from serum ranged from 89% to 102%; repeatability and intermediate precision (relative standard deviation) ranged from 3.6% to 6.7% and 4.1% to 9.3%, respectively. The limit of detection was 0.73 μmol/l. No interfering peaks were observed except lactose, which gave 2 peaks, one of which overlapped partially with sucrose. To evaluate the suitability of the method for quantifying sucrose in serum samples from horses with naturally occurring gastric ulceration, 10 horses with and without naturally occurring gastric ulceration were subjected to sucrose permeability testing. All horses demonstrated an increase in serum sucrose concentration over time following oral administration of sucrose; however, the increase from baseline was significant for horses with gastric ulceration at 45 min (P = 0.0082) and 90 min (P = 0.0082) when compared with healthy horses. It was concluded that gas chromatography with flame ionization detection is a valid method for quantifying sucrose in equine serum and can be applied directly to the analysis of sucrose in equine serum as part of a larger validation study aimed at developing a blood test for the diagnosis of gastric ulcers in horses.
Introduction
Gastric ulcers can develop in horses of all breeds and ages, with up to 100% of racehorses and 58% of other performance horses found to have ulcers on endoscopic examinations.5,8,16–18,22,29,31 To date, detection of ulcers by endoscopic examination of the stomach (gastroscopy) is the only reliable antemortem method for definitive diagnosis of gastric ulcers in horses. Endoscopy is precise, sensitive, and relatively easy to perform; however, it is unsuitable as a screening test because it is expensive, time consuming, and usually only available in specialized centers. 13
In rats, rabbits, dogs, and people, permeation of sucrose across the gastric mucosa has been demonstrated to be a reliable marker of gastroduodenal permeability and may be a useful alternative to gastroscopy for diagnosis of gastric ulcers.19,20,27 Sucrose is inexpensive, nontoxic, and specific due to its cleavage in the small intestine. Healthy gastrointestinal mucosa is relatively impermeable to sucrose. 27 After sucrose is emptied into the proximal portion of the small intestine, it is rapidly hydrolyzed to its monosaccharide units by the brush border enzyme sucrase–isomaltase. 23 This occurs even in the face of severe small intestinal disease. 20 Sucrose is cleared from blood via the urine, where it is concentrated. It is not metabolized after permeation, and the body does not produce it. 32 Consequently, increased amounts of sucrose in blood or urine after an oral dose would suggest gastrointestinal damage oral (proximal) to the site where sucrose is normally digested.
Based on the premise that sucrose permeability can be used to detect the presence and severity of gastric ulcers in other species, quantitation of sucrose in urine has been reported to be a reliable screening method for detecting gastric ulcers in horses 21 ; however, the technical difficulties associated with collection of urine from the horse has limited the practicality of this test. To make the test more practical, quantitation of sucrose in equine blood using a similar approach has been suggested. 13 In a preliminary feasibility study involving a small group of horses, 13 it was demonstrated that quantitation of sucrose in equine blood may be a useful alternative test for identifying horses with endoscopically visible gastric ulceration and has potential as a noninvasive method for screening and monitoring horses engaged in racing training and other performance-related disciplines. We aim to develop and validate this simple blood test in a large group of horses with and without naturally occurring gastric ulcers, and believe that the development of an easy and affordable laboratory method for quantifying sucrose in serum is essential if the blood test is to have any practical value.
Sucrose has been determined and quantified in biological samples by several methods, including enzymatic assays,14,33 high-performance liquid chromatography (HPLC)–mass spectrometry,6,7,13 HPLC pulsed amperometric detector, 26 HPLC fluorescence detector, 15 and gas chromatography with mass spectrometry. 24 Gas chromatography with flame ionization detection (GC-FID) has been used for quantifying sucrose in urine1,9; however, to the authors’ knowledge, a GC-FID method for quantifying sucrose in serum has not been published.
The use of GC-FID for quantitation of sucrose in the sugar industry is well described2,3,10; however, GC-FID quantitation of sucrose in serum is more challenging because serum is a complex matrix containing other sugars, lipids, amino acids, and proteins; all of which affect resolution and sensitivity of the assay. Despite these challenges, GC-FID is accurate and can be used to measure minute amounts of a substance, an attribute that suits its applicability for quantitation of sucrose in equine serum, where expected concentrations of sucrose are low. 14 Furthermore, GC-FID is comparably inexpensive, and the equipment is relatively widespread, making it an ideal analytical method for developing a practical and affordable diagnostic test. The objective of the current study was to develop and validate a GC-FID method for quantitation of sucrose in serum that is simple, rapid, sensitive, precise, accurate, and specific, and can be applied to sucrose permeability testing in the horse.
Materials and methods
Animal welfare
The animal welfare in the study was evaluated and accepted by the ethical committee of the University of Helsinki. Informed consent from the owner, or the trainer acting as an agent for the owner, was obtained at the time of enrollment in the study.
Sucrose permeability testing and gastroscopy
Ten horses with and without naturally occurring gastric ulceration were subjected to sucrose permeability testing. Owners were asked to withhold food from their horses for 16 hr and water for 6 hr prior to sucrose testing. On the morning after fasting, blood samples (20 ml) were collected in vacuumed clot tubes from the jugular vein and then the horses were sedated with a combination of detomidine hydrochloride (3 mg) a and butorphanol (0.6 mg) b ; andan endoscopic evaluation of the stomach was performed to determine the presence or absence of gastric ulceration. Gastric ulcer severity was scored using a published 4-point ulcer scoring system 4 (Table 1).
Four-point gastric ulcer scoring system 4 used in the current study.

Following blood collection, the serum was separated by centrifugation (10 min at 2,000 × g) and then stored in a freezer at −80°C. Following endoscopy, 500 g of sucrose c was administered as a 10% solution via nasogastric tube to each horse. Serial blood samples (20 ml) were then collected by venipuncture from the jugular vein at 45 and 90 min after administration of sucrose. These time points were chosen based on data from a previous study, which indicated that peak sucrose concentrations occur approximately between 45 and 90 min after sucrose administration. 13
Collection of sucrose-free serum
Blood samples (20 ml) were collected in vacuumed clot tubes from the jugular vein of healthy horses that had been fasted for 16 hr. The serum was be separated by centrifugation (10 min at 2,000 × g) and was then pooled and stored in a freezer at −80°C. In order to ensure that the serum was sucrose free, a sample from each horse was analyzed for sucrose prior to pooling and was discarded if any sucrose was detected.
Preparation of solutions
Standard preparation
Stock solutions of analytical grade sucrose d and trehalose e (29.2 and 26.4 mmol/l, respectively) were prepared independently by dissolving each compound in ultrapure water. f Working solutions were made by diluting the stock solutions 1:100 in water. Stock and working solutions were stored in a refrigerator at 4°C. Standards were prepared in sucrose-free serum by spiking sucrose to yield final concentrations of 2.34, 2.92, 5.84, 8.76, 14.61, and 20.45 μmol/l; and trehalose (26.4 μmol/l) was used as an internal standard. Standards were prepared as samples, and they were made weekly.
Sample preparation
Depending on the application, test samples were prepared by spiking known concentrations of sucrose in sucrose-free serum or they were obtained from horses that had been subjected to sucrose permeability testing. Prior to analysis, test samples were thawed and mixed using a vortex. Two hundred microliters of test sample was transferred to a centrifugation tube, and 20 μl of a working solution of trehalose was added as an internal standard. Proteins were precipitated by adding 0.6 ml of acetonitrile g –water (90:10), vortexing, and then centrifuging for 10 min at 10,000 × g. The resulting supernatant was evaporated to dryness in a vacuum evaporator at 60°C. The dried residues were purged with nitrogen for 10 sec, dissolved in 0.1 ml of anhydrous pyridine, h and then sonicated for 5 min. Sucrose and trehalose were converted to their trimethylsilyl (TMS) derivatives by adding 0.1 ml of N-trimethylsilylimidazole (TMSI). i The vials were capped tightly and heated at 100°C for 1 hr. After reaction, the samples were centrifuged for 5 min at 1,711 × g at 4°C. An aliquot of the mixture was then transferred to a new autosampler vial, and the samples were analyzed immediately.
Instrumentation and acquisition parameters
Gas chromatography was performed using a GC instrument j equipped with a FID and autosampler. k Aliquots(1 μl) of the mixture were injected by utilizing a single tapered glass liner fitted to an injector into a 2.5 m × 0.320 mm inner diameter (ID) retention gap (uncoated precolumn) in pulsed splitless mode at a pressure of 80 psi for 0.4 min. The retention gap was attached to a 30 m × 0.320 mm ID fused-silica capillary column, coated with 0.25-μm thickness HP-5 stationary phase. l Initial oven temperature 95°C was held for 2 min, then increased to 205°C at a rate of 30°C/min, held at 205°C for 3 min, and finally increased 1°C/min to 250°C. The injector and detector temperatures were set at 230°C and 300°C, respectively. The purge flow rate was 60 ml/min, and the purge valve was turned on after 0.5 min. Helium was used as a carrier gas for the mobile phase at a constant flow rate of 4.8 ml/min. Hydrogen (45 ml/min) and synthetic air (400 ml/min) were used as auxiliary gases, and helium (30 ml/min) was used as the make-up gas for the FID. The column was backflushed at 300°C for 10 void volumes after every run to prevent ghost peaks from previous runs.
Validation
The method was validated according to the guidelines of the International Conference on Harmonisation (ICH) for validation of analytical methods.11,12 Validation criteria included determination of specificity, linearity, range, accuracy, precision, detection limit, quantitation limit, robustness, and system suitability.
Specificity and linearity
The assay was compared for resolution of carbohydrates (fructose, glucose, lactose, and maltose), which are likely to be present in equine serum. The linearity of the method was assessed across the expected range of the analytical procedure by comparing the recovery of serial dilutions of sucrose in serum relative to the internal standard, trehalose. The calibration curves were constructed with 7 concentrations of sucrose ranging from 2.34 to 20.45 μmol/l. The integrated peak areas of sucrose were normalized by dividing them by the peak area of trehalose, and the ratios were plotted against the concentrations. Linearity was evaluated by linear regression analysis, calculated using the least squares regression method.
Range and accuracy
The specified range of the analytical procedure was derived from the linearity studies by confirming that the assay provided an acceptable degree of linearity, accuracy, and precision when applied to serum samples containing amounts of sucrose within or at the extremes of the specified range. The accuracy of the assay was established across the specified range by spiking 3 known concentrations of sucrose (2.92, 8.76, and 20.45 μmol/l) in sucrose-free serum and analyzing each sample a minimum of 3 times to compare the measured and actual values. Accuracy was reported as the percentage recovery of the known added amount of sucrose in each sample.
Precision (repeatability and intermediate precision)
The precision of the assay was established across the specified range by spiking 3 known concentrations of sucrose (2.92, 8.76, and 20.45 μmol/l) in sucrose-free serum and analyzing each sample a minimum of 3 times in 1 day (repeatability or intra-assay variability) and a minimum of 3 different days (intermediate precision or inter-assay variability). The precision was expressed as the relative standard deviation (RSD%).
Detection and quantitation limits
The detection limit was defined as the minimum concentration of sucrose in equine serum that resulted in a peak height of 3 times that of baseline noise. The quantitation limit was defined as the minimum concentration of sucrose that resulted in a peak height of 10 times that of baseline noise.
Robustness
To evaluate the stability of sucrose during storage, sucrose concentration was measured in duplicate equine serum samples 1) immediately (i.e., serum was separated, and then frozen immediately); 2) following storage of whole blood for 6 and 18 hr at room temperature; 3) following storage of serum for 24 hr at room temperature and at 4°C; 4) following storage of serum for 48 hr at 4°C; and 5) following storage of serum for 72 hr at 4°C. Results were expressed as the percentage recovery (%).
System suitability testing
To evaluate the suitability of the method for quantifying sucrose in serum samples from horses with naturally occurring gastric ulceration, 10 horses with and without endoscopically visible gastric ulceration were subjected to sucrose permeability testing. The method was considered to be suitable for sucrose permeability testing if 1) sucrose could be quantified in the serum of horses that had been subjected to permeability testing and 2) if there was a significant difference in sucrose concentrations when comparing horses with and without naturally occurring gastric ulceration following administration of sucrose.
Statistical analysis
Commercial software m was used to integrate the chromatographic peaks, and statistical software n was used for thestatistical analysis. Comparisons between horses with and without gastric ulceration for individual time points were made using a 2-sample Wilcoxon rank-sum test for nonparametric data. A P value <0.05 was considered significant. All data given are means ± standard deviation (SD).
Results
Chromatogram
A typical chromatogram obtained with the sucrose and trehalose standard is demonstrated in Figure 1A. Sucrose was identified by comparison of the retention time of TMS ether derivative with standard compounds. The TMS derivatives of sucrose and trehalose gave 1 peak of each. The retention times were 37.3 and 42.7 min, respectively, with good resolution.
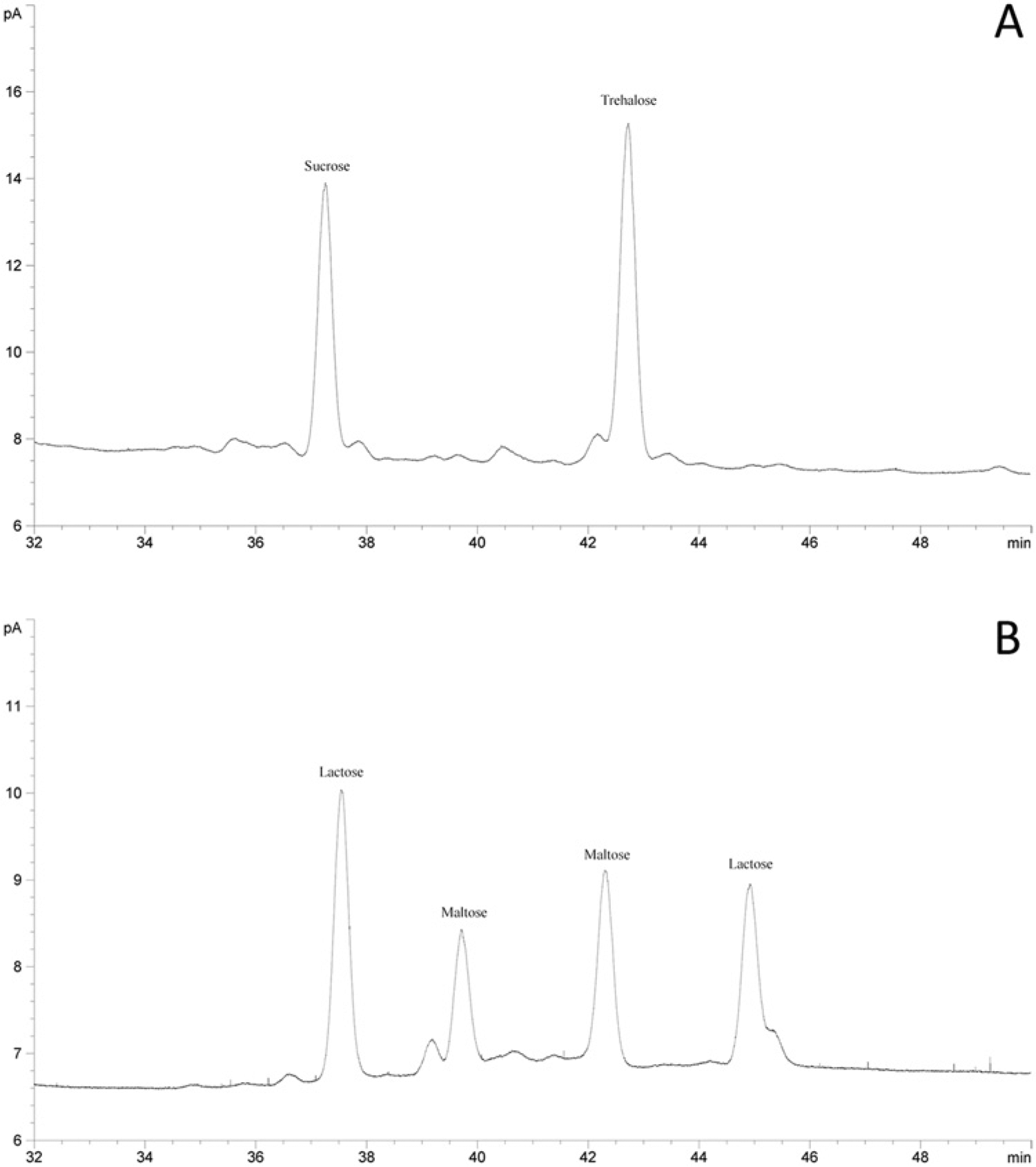
Method validation
Specificity and linearity
The assay was compared for resolution of carbohydrates (fructose, glucose, lactose, and maltose), which are likely to be present in equine serum. No interfering peaks were observed except lactose, which gave several peaks, one of which overlapped partially with sucrose (Fig. 1B). The calibration curve was linear over the concentration range. The linearity parameters are summarized in Table 2.
Detection properties of gas chromatography (GC)–flame ionization detection (FID) for sucrose in equine serum.

Range, accuracy, and precision (repeatability and intermediate precision)
The assay provided an acceptable degree of linearity, accuracy, and precision at concentrations of sucrose as low as 2.34 μmol/l and as high as 20.45 μmol/l. Percentage recovery of sucrose from serum was 89–102%. Repeatability and intermediate precision (RSD%) ranged from 3.6% to 6.7% and 4.1% to 9.3%, respectively.
Detection limit, quantitation limit, and robustness
The minimum concentration of sucrose that could be reliably detected in equine serum was 0.73 μmol/l (signal-to-noise ratio of 3:1); the minimum concentration of sucrose that could be quantitatively determined was 2.34 μmol/l (signal-to-noise ratio 10:1; Table 2). The assay was precise under a variety of storage conditions, with percentage recovery ranging from 72% to 105% (Table 3).
Robustness of gas chromatography (GC)–flame ionization detection (FID) for detection of sucrose in equine serum when exposed to different storage conditions.

System suitability
Ten horses with and without gastric ulceration were subjected to sucrose permeability testing. Five horses had endoscopic evidence of gastric ulceration, and 5 horses had normal stomachs. The median ulcer score for horses with gastric ulceration was 3 (range: 3–4), and lesions were distributed in the glandular and nonglandular mucosa in all cases. On analysis of the serum samples, all horses demonstrated an increase in serum sucrose concentration over time following oral administration of sucrose. The increase from baseline was significant for horses with gastric ulceration at 45 min (P = 0.082) and 90 min (P = 0.082) when compared with healthy horses (Fig. 2). The mean ± SD serum sucrose concentration at 45 min was 2.57 ± 0.32 μmol/l for horses with no ulceration (n = 5), and 11.16 ± 3.52 μmol/l for horses with ulceration (n = 5). The mean ± SD serum sucrose concentration at 90 min was 2.97 ± 0.97 μmol/l for horses with no ulceration (n = 5), and 12.75 ± 4.68 μmol/l for horses with ulceration (n = 5). Peak serum sucrose concentrations occurred 90 min after administration of sucrose.
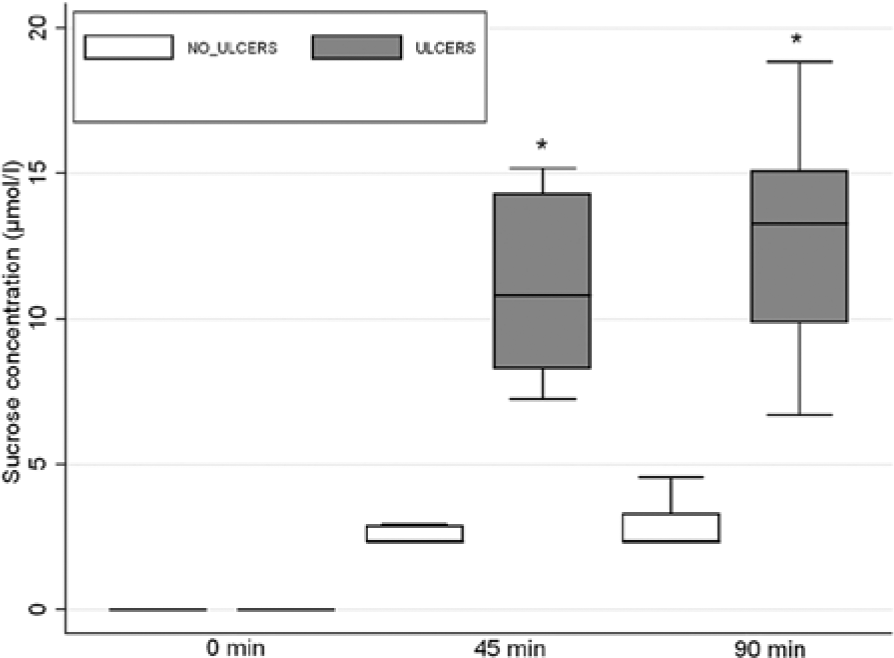
Box and whisker plot of serum sucrose concentrations from horses with (n = 5) and without (n = 5) gastric ulceration at 0, 45, and 90 min after administration of 500 g of sucrose via nasogastric intubation. Horses with gastric ulceration (shaded boxes) had significantly greater concentrations of sucrose detectable in the peripheral circulation than horses with no ulceration (nonshaded boxes) at 45 and 90 min (*P < 0.05).
Discussion
The development of a valid, cost-effective method for quantifying sucrose in equine serum is the first step in a larger project to develop and validate a blood test for detecting gastric ulcers in horses. 13 The GC-FID method was developed and optimized as a quantitative test intended to measure minute concentrations of sucrose present in equine serum, and its validity for this purpose was tested using the ICH guidelines for validation of analytical procedures.11,12 The results of the current study indicate that the GC-FID method is valid and can be applied to the assessment of gastrointestinal permeability in the horse.
Method development and optimization
The development of a GC-FID method for determination of sucrose in equine serum was based on the innate chemical properties of sucrose and the fact that low concentrations of sucrose needed to be identified and quantified in equine serum. Standards and selected test samples were prepared in sucrose-free serum that was obtained by pooling serum from healthy horses that had been fasted for 16 hr. The average half-empty time (t1/2) for solid phase and liquid phase emptying of the equine stomach is 3.8 and 1.5 hr, respectively,25,28 and the approximate half-life of sucrose in equine serum is 90 min (unpublished data). Thus, a 16-hr fast should have allowed ample time for passage of ingesta from the stomach into the small intestine, and urinary excretion of any food-derived sucrose that may have permeated across the gastric mucosa, therefore ensuring that the serum was free of sucrose prior to pooling. The final concentrations for the sucrose standards were selected based on data from a previous study, which indicated that the analytical range for sucrose in serum for the purposes of permeability testing was approximately 2.34–20.45 μmol/l. 13
The samples were quantified using the peak area ratio between sucrose and the internal standard, trehalose. This method of quantification was used because it has been reported to be more reproducible than the use of absolute peak area for sucrose. 24 Trehalose was used as an internal standard because 1) it does not normally exist in equine serum, 2) its retention time differs significantly from sucrose, and 3) like sucrose, it is a nonreducing sugar and thus can be derivatized without oximation.
Different precipitation solvents (methanol, ethanol, acetone, and mixture of acetonitrile–water) were tested. Recovery of sucrose from serum was best when a mixture of acetonitrile–water (90:10) was used.
Sucrose is a highly polar, nonvolatile substance, thus it requires chemical derivatization before GC analysis. When analyzing with GC under normal circumstances, reducing sugars have to be transformed to an oxime before TMS derivatization to decrease the number of chromatographic peaks. However, because sucrose is a non-reducing sugar, it can be directly derivatized without oximation, a process which has been reported to cause the degradation of sucrose. 11
Trimethylsilylimidazole is a well-described silylating agent used for quantitative analysis of sucrose by GC in the sugar industry (Nurok D, Reardon TJ: 1975, Quantitative determination of sugars in factory products by gas chromatography using open tubular columns. Proceedings of The South African Sugar Technologists’ Association, pp. 94–97), and was used as a silylating agent for derivatization of sucrose in this method. Trimethylsilylimidazole reacts quickly and smoothly with hydroxyls and carboxylic acids and is thus easy and quick to prepare. One potential problem with TMSI was that it crystallized due to humidity in the air and caused blockage of the syringe. This made the injection technique unrepeatable. The problem was resolved by washing the syringe immediately after every injection with acetonitrile and pyridine. Precision of the method may be improved further by using an extra underivatized standard to monitor the repeatability of the injections. Silylating reagents also contaminate the FID and may cause a loss of sensitivity. During method development, the FID was cleaned after every 60 runs to prevent this problem, but later it was determined that even after 100 runs, there did not appear to be any significant loss of sensitivity.
Different derivatization circumstances were tested. The derivatization was found to be complete after 1 hr at 100°C, measured as the FID response. Following derivatization, the samples were analyzed by GC immediately, but because of the long run times, the last samples to be analyzed were left standing in the autosampler tray at room temperature for up to 24 hr. A control sample was therefore injected prior to running the samples and immediately after the last sample was finished to confirm the stability of TMS derivates. No significant degradation of TMS derivates was observed despite this unavoidable delay in analysis.
The pulsed splitless injection technique was used to shorten the analyte residence time in the hot injection port, resulting in lower analyte degradation and adsorption in the inlet. The glass injection liner was replaced after every 60 injections. Different temperature programs were investigated for the GC oven, and a final temperature program with a total run time of 53.7 min was selected based on the best possible resolution.
Method validation
Validation of the method was based on ICH guidelines11,12 and included determination of the specificity, range of linearity, accuracy, precision, detection limit, quantitation limit, robustness, and system suitability of the assay. Specificity of the assay was demonstrated by comparing the resolution of sucrose with various carbohydrates that are likely to be present in equine serum. No interfering peaks were observed except lactose. This interference is unlikely to affect the specificity of the assay when determining sucrose concentration in adult horses, as lactose is not a normal component of an adult horse’s diet. Lactose peak interference may be a problem when determining sucrose concentrations in foal serum however, as foals drink milk that contains high concentrations of lactose. Some of this lactose may permeate across a damaged gastric mucosa and accumulate in the serum, where it will interfere with sucrose and reduce assay specificity. This problem can be solved by fasting foals for at least 6 hr prior to sucrose testing to ensure adequate time for excretion of any milk-derived lactose that may have permeated across the gastric mucosa. 30
The GC-FID method was demonstrated to be linear in the range of 2.34–20.45 μmol/l. This range constituted 80–120% of the sucrose concentration expected in clinical cases. 13
Accuracy and precision testing demonstrated a high degree of reproducibility. Using the GC-FID method, it appears that accuracy at low concentrations of sucrose is improved when compared with comparable data, where percentage recovery of sucrose at low concentrations in canine serum was only 56.7%. 25
The robustness study demonstrated that the methods performance remained unchanged despite variations in storage conditions. This is important, as there may be a substantial lag phase between blood collection and analysis under field conditions, and stability of sucrose in whole blood at room temperature is imperative for method validity.
System suitability testing using a small number of horses with and without gastric ulceration demonstrated that the GC-FID method was able to quantify sucrose in horses with gastric ulcers and that there appeared to be a significant association between sucrose concentration and the presence of gastric ulceration (P < 0.05). The data indicates that sucrose can be quantified in the serum of horses that have been subjected to sucrose permeability testing using the GC-FID method. Furthermore, there appears to be a significant difference in serum sucrose concentrations at 45 and 90 min when comparing healthy horses with horses that have endoscopic evidence of gastric ulceration. It is important, however, to realize that the small sample size in the current study makes interpretation of the significance of these results difficult. The purpose of this small sample set was to demonstrate that the GC-FID method is suitable for sucrose permeability testing in horses, and conclusions on the validity of the sucrose permeability test should not be inferred on the basis of this data.
In conclusion, the results of the present study indicate that the GC-FID assay for the determination of sucrose in serum is a valid method and can be applied to the assessment of gastrointestinal permeability in the horse. Further studies to determine the diagnostic accuracy of the blood test and to define cutoff values in a large group of horses with and without naturally occurring gastric ulcers are planned.
Footnotes
Acknowledgements
The authors would like to thank Professors Sandy Love, Noah Cohen, and Allen Rousell for their support, guidance, and vision; and Dr. Thomas Becker for his assistance with the statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
a.
Domosedan, Elanco Animal Health, Basingstoke, Hampshire, United Kingdom.
b.
Butador, Chanelle Animal Health UK, Hungerford, Berkshire, United Kingdom.
c.
Kidesokeri 530, Sucros Oy, Kantvik, Finland.
d.
α-
e.
f.
Milli-Q Gradient purification system, EMD Millipore Corp., Billerica, MA.
g.
HPLC-grade acetonitrile, LabScan, Gliwice, Poland.
h.
Anhydrous pyridine, Sigma-Aldrich, St. Louis, MO.
i.
N-trimethylsilylimidazole (TMSI), Pierce Biotechnology Inc., Rockford, IL.
j.
Agilent 7890A, Agilent Technologies Co. Ltd, Beijing, China.
k.
Agilent 7683B, Agilent Technologies Co. Ltd, Beijing, China.
l.
J&W Scientific, Folsom, CA.
m.
GC ChemStation, Agilent Technologies Co. Ltd, Böblingen, Germany.
n.
Stata11, StataCorp LP, College Station, TX.
