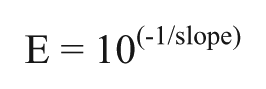Abstract
The objective of the current study was to investigate if the presence of colostral-derived immunoglobulin G (IgG) in blood is an inhibitor of diagnostic polymerase chain reaction (PCR) for detection of Bovine viral diarrhea virus (BVDV). Eleven precolostral and 11 postcolostral blood samples in ethylenediamine tetra-acetic acid (EDTA) anticoagulant as well as serum samples were collected from 11 Holstein bull calves. Calves were fed 3 liters of colostrum once, by oroesophageal tubing. Postcolostral, blood, and serum samples were collected at 48 hr of age. Serum IgG concentrations were determined in the precolostral and postcolostral serum samples using radial immunodiffusion. The blood samples (precolostral and postcolostral) were spiked with BVDV, and 2 diagnostic PCR extraction methods were applied to each sample. The extraction and amplification efficiencies of the 2 PCR methods on the precolostral and postcolostral EDTA blood samples were evaluated. Two of the 11 calves had inadequate passive transfer of colostral immunoglobulins at 48 hr of age based on the serum IgG concentrations. All blood samples from calves were negative for BVDV prior to the spiking with the virus. Evaluation of the 2 different methods among 3 different virus concentrations demonstrated that there was no difference in extraction or amplification efficiency in precolostral and postcolostral samples. The results of this study suggest that bovine IgG is not an inhibitor of PCR used for detection of BVDV in cattle. The methods used in the current study are acceptable for PCR detection of BVDV in cattle.
Infection by Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) has a negative economic impact and is associated with reduced fertility, abortions, reduced growth, and production losses in the cattle industry.10,19 The virus is transmitted horizontally and vertically among herds,10,20 and has the ability to infect the fetus during specific stages of the gestation period to create life-long virus-shedding calves (persistently infected calves).4,15 Herd-level economic losses due to infection with virulent strains of BVDV are reported to range from US$40,000 to 100,000. 3 Population level economic losses due to BVDV have been estimated at US$10–40 million.12,13 Costs associated with eradication of BVDV in Demark were estimated at US$27 million. 6 Herd-level prevalence of persistent infection by BVDV in beef cattle in the United States has been estimated at <1%. 22
Real-time polymerase chain reaction (PCR) has been utilized as one of the diagnostic methods of choice to detect BVDV-infected animals due to its high sensitivity and the fast turnaround time of results. 11 Real-time quantitative reverse transcription PCR (qRT-PCR) technology includes 2 procedural steps, RNA extraction and PCR amplification including the reverse transcription of RNA. 11 The overall efficiency of the PCR amplification step is influenced by the efficiency of the nucleic acid extraction step, with failure to eliminate an inhibitor during the extraction stage prior to the PCR reaction leading to potential false-negative results. Known PCR inhibitors include heme, ethylenediamine tetra-acetic acid (EDTA), and immunoglobulin G (IgG).1,2,8,14,18 The importance of IgG as a PCR inhibitor has been described in studies on human blood samples. 2 In those studies, blood was separated into fractions, and the importance of IgG as a RT-PCR inhibitor was demonstrated using purified plasma IgG. 2 Similar studies with either fractionated and purified IgG or naturally occurring immunoglobulins have not been replicated in domestic animal samples.
Calves are born with minimal measurable immunoglobulins in their blood and receive maternal antibodies from the ingestion of colostrum. 7 Maximum IgG concentrations are measured in serum 48 hr after ingestion of colostrum to determine adequacy of passive transfer of colostral immunoglobulins. 7 The hypothesis put forward was that presence of naturally acquired IgG in calf blood samples following ingestion of colostrum would inhibit qRT-PCR, which is commonly used for detection of BVDV in a diagnostic laboratory setting. Evaluation of blood from newborn calves before and after ingestion of colostrum provided the optimum design for testing the proposed hypothesis. The applied research objective of the current study was to determine if presence of IgG in bovine blood affects the diagnostic value of the qRT-PCR for detection of BVDV resulting in false-negative test results in the diagnostic laboratory setting using current PCR assays.
The research study was approved by the University of California, Davis Animal Care and Use Committee. Eleven Holstein bull calves between 2 and 4 hr of age were enrolled in the study. Calves were obtained from a single dairy farm. Adult cows on the farm were vaccinated annually with a multivalent modified live respiratory vaccine (Bovine herpesvirus 1, Bovine viral diarrhea 1 and 2, Bovine parainfluenza virus 3, and Bovine respiratory syncytial virus strains). Cows born to eutocia were immediately separated from the dam after parturition to prevent nursing. Calves were raised at the University of California, Davis. Calves were identified using ear tags and housed individually in calf hutches. Blood samples were collected in 10-ml tubes containing EDTA a and serum samples were collected in 10-ml blood-collecting tubes containing no anticoagulant b prior to ingestion of colostrum for qRT-PCR analysis for BVDV and for serum IgG determination, respectively. Three liters of stored pooled colostrum (with similar IgG concentrations) was fed by oroesophageal tubing to each calf, once. Serum and EDTA blood samples were collected from calves at 48 hr of age for serum IgG determination and BVDV analysis, respectively. Serum (precolostral and 48 hr) was frozen at −20°C for subsequent IgG determination using single radial immunodiffusion. Serum IgG determination and BVDV qRT-PCR on blood samples were performed at the California Animal Health and Food Safety Laboratory (Davis, CA).
A cytopathic BVDV-1b strain (TGAC) c originating from a clinical case and propagated in cell culture was used for inoculation. The BVDV titer calculated by a previously described method 17 was 7.94 × 106 TCID50 (50% tissue culture infectious dose) per ml of the BVDV TGAC stock culture. Quantitative serum IgG determination was performed using radial immunodiffusion according to the manufacturer’s recommendations. d Only animals with corresponding serum samples determined to have a serum IgG concentration <184 mg/dl prior to ingestion of colostrum and >300 mg/dl after ingestion of colostrum were enrolled in the study.
All blood samples collected from the calves prior to ingestion of colostrum were tested for presence of BVDV using real-time PCR. A one-step real-time RT-PCR was performed using a commercial real-time PCR e kit according to manufacturer’s recommendations. f Only blood samples negative for BVDV based on the real-time PCR were included in the study.
A 10-fold dilution series of BVDV TGAC cell culture was performed using BVDV-negative tested bovine blood. Virus aliquots were extracted via a commercial extraction kit g and subjected to PCR analysis. Low, medium, and high threshold cycle (Ct) values were determined representing the diagnostic range of the assay. The correspondent viral dilution (10−1, 10−3, and 10−5) was used for the preparation of the sample aliquots.
Precolostral and postcolostral EDTA blood samples were spiked with each of the BVDV virus dilutions. For the preparation of 10−1 dilutions, 160 μl of TGAC cell culture was added to 1,440 μl of blood sample. For the 10−3 dilution, 16 μl of the 10−1 TGAC dilution was added to 1,584 μl of blood sample; for the preparation of the 10−5 dilution, 16 μl of the 10−3 dilution was pipetted into 1,584 μl of blood sample. All samples were vortexed for 8–10 sec to ensure thorough mixing of virus and blood. Each spiked sample was aliquoted into 3 equal amounts, which were stored at −20°C until use to minimize intra- and interassay variation. Samples were assayed following 1 day of storage at −20°C. Once samples were thawed, they were processed on the same day to ensure similar sample handling conditions for all aliquots.
Two different methods of RNA extraction (methods A g and B h ), both utilizing magnetic bead–based protocols, were compared. In both procedures, 1 μl of an internal control i was added to each sample before starting the extraction process. Whereas method A was used according to the manufacturer’s recommendations, changes were necessary to improve the uneven lysis of the red blood cells by method B. The following sequence of steps were used for method B: after pipetting 20 μl of bead mix into a 96-well plate, 50 μl of blood was added and vigorously agitated using a shaker j for 3 min. Thereafter, 130 μl of lysis binding solution was added, followed by manual mixing, and then agitated using an orbital shaker (550 relative centrifugal force; 3 min). The remainder of the procedure was performed according to the manufacturer’s recommendation. Three experimental runs for each of the 3 aliquots were performed a day apart for each extraction method and for both precolostral and postcolostral blood samples.
Variability in Ct values among test results for each extraction method and the internal control was determined using coefficient of variation. 9 Amplification efficiencies (E) between the methods were determined using the gradient (slopes) of the curves generated by plotting the Ct values against the logarithmic concentration of the DNA with the following formula 16 :
Comparison of amplification efficiencies within each method in blood samples before and after feeding colostrum containing the different virus dilutions were determined by testing the differences between the slopes of the curves generated by plotting the Ct values against the logarithmic concentration of the DNA using a t-test. 23 Likewise, comparison of amplification efficiencies between the 2 extraction methods was performed by testing the differences between the slopes of the curves generated by plotting the Ct values against the logarithmic concentration of the DNA using at-test. 23 For all comparisons of the slopes, P < 0.05 was considered significant.
Mean ± standard error serum IgG concentration prior to ingestion of colostrum and after ingestion of colostrum was 100.4 ± 8.7 mg/dl and 1617 ± 58.2 mg/dl, respectively. Serum IgG concentration in the calves prior to ingestion of colostrum was consistent with previous reports. 7 Two calves (2/11, 18%) had inadequate ingestion of colostral immunoglobulins as indicated by serum IgG concentration of <1,000 mg/dl at 48 hr of age. 5 Blood samples in EDTA collected from all 11 calves prior to ingestion of colostrum and after ingestion of colostrum were negative for BVDV based on the real-time PCR. The coefficient of variation (<10%) of the Ct values indicated that both extraction methods demonstrated acceptable precision. Based on the single regression models generated, there was no difference in amplification efficiency between either extraction method in precolostral and postcolostral blood samples (P > 0.05).
The main objective of the current study was to determine if IgG is a potential inhibitor of PCR amplification in laboratory assays currently used to detect BVDV. Results of the present study indicate that IgG does not act as an inhibitor of PCR amplification during detection of BVDV in bovine calves. In this study, blood samples were spiked with BVDV virus. The potential for differences in extraction efficiency of BVDV following use of spiked samples rather than samples from naturally infected animals does exist. Further studies would be required to evaluate these differences. In addition, in this study, naturally acquired colostral-derived IgG was evaluated as an inhibitor of PCR, in contrast to previous studies, 2 which evaluated purified human IgG as an inhibitor of PCR. The differences between fractionated and purified IgG and naturally occurring IgG could potentially explain the lack of inhibition in the present study. Colostral-derived IgG was specifically utilized to mimic the blood specimens that would be routinely submitted to a diagnostic laboratory for detection of BVDV, including samples from neonates that have ingested colostrum. There was no difference in efficiency of the extraction and amplification methods used for calf blood samples prior to and after ingestion of colostrum. This suggests that both methods are acceptable for routine detection of BVDV in blood samples. It is important to note that the highest dilution (lowest concentration of virus) of the virus used in the study was 10−5 and that the virus was not serially diluted to nondetectable levels. The study focus was on samples with detectable virus as needed for evaluation of the extraction and amplification efficiency of the methods.
Cattle can be infected with both cytopathic and noncytopathic biotype strains of BVDV. 21 A cytopathic strain of BVDV was chosen to spike the blood samples in the present study; however, the BVDV PCR method used detects both cytopathic and noncytopathic strains of the virus. Bovine viral diarrhea was specifically selected for investigation in the current study for 2 reasons. First, infections with BVDV have profound negative economic impact on cattle production, and second, bovine serum samples for BVDV diagnostic testing are routinely submitted to laboratories. The results of this basic and applied study support recommendations concerning PCR testing to eliminate persistently infected cattle from herds. It should be noted that other potential PCR inhibitors or cumulative effects of potential inhibitors reported in previous studies1,8,14,18 were not investigated in the current study, and more in-depth studies are warranted on PCR inhibitors in veterinary diagnostic testing of samples. In conclusion, both methods utilized in the current study for testing the presence of BVDV in blood samples are acceptable. The presence of colostral-derived IgG in blood samples does not inhibit PCR used for detecting presence of BVDV in cattle.
Footnotes
a.
EDTA blood collection tubes, BD, Franklin Lakes, NJ.
b.
Serum blood collection tubes, BD, Franklin Lakes, NJ.
c.
Generously provided by Dr. Julia Ridpath, Ames, IA.
d.
Radial immunodiffusion plates and kit, Triple J Farms, Bellingham, WA.
e.
VetMAX-Gold BVDV Detection Kit, Life Technologies, Carlsbad, CA.
f.
ABI 7500fast PCR system, Life Technologies, Carlsbad, CA.
g.
MagMax-96 for Microarrays (AM1839), Life Technologies, Carlsbad, CA.
h.
MagMax96Viral RNA Isolation well kit, Life Technologies, Carlsbad, CA.
i.
TaqMan BVDV and Xeno RNA controls, Life Technologies, Carlsbad, CA.
j.
LabLine orbital shaker, Barnstead, Melrose Park, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.