Abstract
The molecular characterization of a strain of Canine minute virus (CnMV) associated with neonatal death is reported. Three newborn puppies of a litter of Jack Russell terrier dogs died after displaying systemic disease, whereas 2 surviving puppies showed no clinical signs with the exception of transient cardiac abnormalities that were evident by electrocardiography. Necropsy of 1 dead puppy revealed severe lesions in the internal organs. A strain of Canine minute virus was detected in tissue samples collected from the puppy, and virus circulation was demonstrated by molecular or serological testing in the dam, puppies of the same litter, and other puppies in the same kennel. By sequence and phylogenetic analysis of the gene encoding for the VP2 capsid protein, the strain circulating in the kennel was found to be related to recent Asian CnMV isolates. Continuous molecular surveillance for CnMV in kennels, shelters, and rescue centers would expand the knowledge base on the epidemiological and pathogenetic features of CnMV, which has been known for several decades but still poorly understood.
Canine minute virus (CnMV), formerly known as Minute virus of canines or Canine parvovirus type 1, is an autonomous parvovirus of dogs that is genetically and antigenically unrelated to Canine parvovirus type 2, the common agent of canine hemorrhagic gastroenteritis. 5 Recent studies have shown that CnMV is more closely related to Bovine parvovirus and human bocaviruses, and now has been included in the new genus Bocavirus of the family Parvoviridae. 23 Canine minute virus has been isolated from healthy dogs, as well as from cases of reproductive failure. Different outcomes of CnMV infection in pregnant females depend on the time of infection during pregnancy, with embryo death/resorption and stillbirths/birth of weak pups occurring during infections in early and late gestation, respectively. 7 Recently, CnMV was reported to be associated with neurological disease in dogs of various ages 15 and to severe gastroenteritis in an elderly dog. 24 Natural outbreaks of CnMV-associated neonatal mortality have been reported only sporadically, and last reports date back to the 1990s.2,18,20,25 Puppies infected less than 4 weeks of age often had mild or vague symptoms preceding their rapid death; others displayed depression, loss of appetite, acute myocarditis, respiratory distress, and/or enteritis. 7 A certain role in CnMV pathogenesis may be played by virus-induced immunosuppression due to reduction of monocyte phagocytosis. 3 The present study reports the clinical, pathological, and molecular findings of a case of neonatal mortality associated with CnMV in a breeding kennel in Italy.
The current case was observed in a litter of Jack Russell terrier dogs housed in a kennel in the Apulia region, Italy. In July 2011, a 3-year-old female whelped 6 puppies, 3 female (F1–3) and 3 male (M1–3), the last one being stillborn. Of the remaining 5 animals, puppies M2, F3, and F2 displayed a systemic disease characterized by dyspnea and sudden death at 12, 20, and 23 days of age, respectively. The 2 surviving puppies (F1 and M1) showed no clinical signs, with the exception of a slight alteration of the ST segment by electrocardiography, which disappeared at the subsequent examination performed 10 days later. Puppy M2 (285/11) was submitted to the authors’ laboratory for diagnostic investigation. At postmortem examination, the puppy displayed severe pneumonia, catarrhal enteritis, and liver and myocardial degeneration with the presence of areas of necrosis. Tissue fragments were collected for virological and bacteriological testing. In addition, serum and rectal and oropharyngeal swabs were collected from the dam, from puppies F1 and M1, and from 6 other puppies in the same kennel.
Tissue samples were homogenized (50% w/v) in Dulbecco minimal essential medium (D-MEM) using a tissue lyser, a whereas rectal and oropharyngeal swabs were homogenized (10% w/v) in D-MEM by vortexing. After RNA b and DNA c purification, all samples were tested by polymerase chain reaction (PCR) for the main viral pathogens of dogs: Canid herpesvirus 1, 4 CnMV, 3 canine coronaviruses,12,13 reoviruses, 6 rotaviruses, 16 caliciviruses, 21 Canine parvovirus type 2,8-10 Canine adenovirus types 1 and 2, 19 and Canine distemper virus. 14 Standard procedures were carried out on respiratory specimens for isolation of the most common bacteria responsible for dog mortality. Samples were plated out on 5% sheep blood agar and cultured aerobically at 37°C for 24 hr for detection of aerobic pathogens. Bacteria were identified by standard biochemical procedures and analytical profile index. d A CnMV strain was detected by means of PCR in the tissues of puppy M2 as well as in the rectal swabs of two 3-month-old puppies of 2 different litters (Fig. 1). However, CnMV DNA was not detected in the rectal swabs of either surviving puppies at the moment of sampling. No other infectious disease agents were detected by molecular or traditional assays in the tested samples.
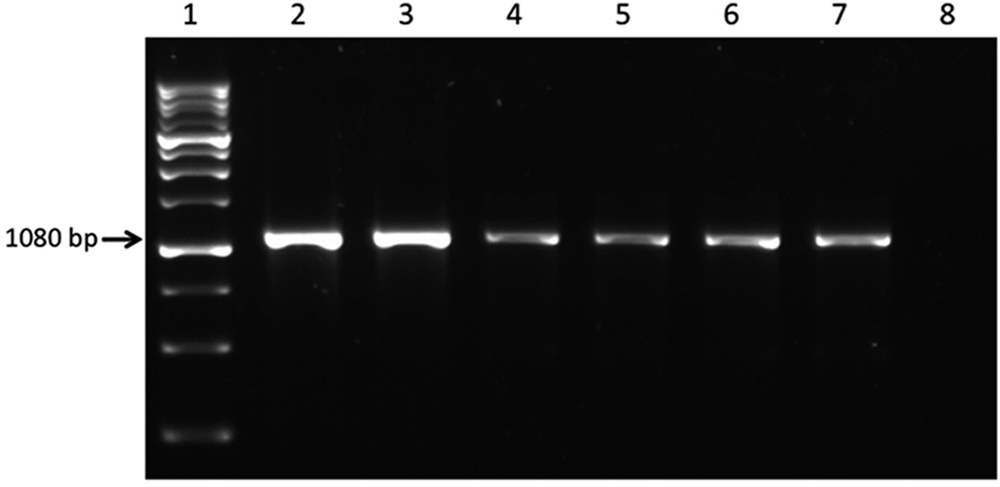
Gel electrophoresis of products obtained from the Canine minute virus (CnMV) polymerase chain reaction assay. Lane 1: markerg; lane 2: positive control (CnMV strain GA3)f; lanes 3 through 5: tissue samples (heart, lung, gut) from the dead puppy M2; lanes 6, 7: rectal swabs from 2 puppies of the same kennel; lane 8: negative control (heart from a CnMV-negative puppy).
Canine minute virus–positive samples were homogenized in Eagle minimal essential medium (E-MEM), treated with antibiotics (penicillin: 5,000 IU/ml; streptomycin: 2,500 μg/ml) at 37°C for 30 min, and inoculated onto freshly trypsinized Walter Reed canine cells (WRCC) and Madin–Darby canine kidney (MDCK) cells. The inoculated cells were observed daily for the occurrence of cytopathic effect. After 3 days of incubation, the cells were tested by the direct fluorescent antibody test (FAT) for detection of CnMV antigens and also stained with hematoxylin and eosin (HE) for detection of inclusion bodies. All attempts to isolate the virus from the PCR-positive samples were unsuccessful, as shown by the negative results obtained by FAT and HE staining. Three consecutive passages, each with an incubation period of 7 days, were carried out prior to considering the virus isolation as negative.
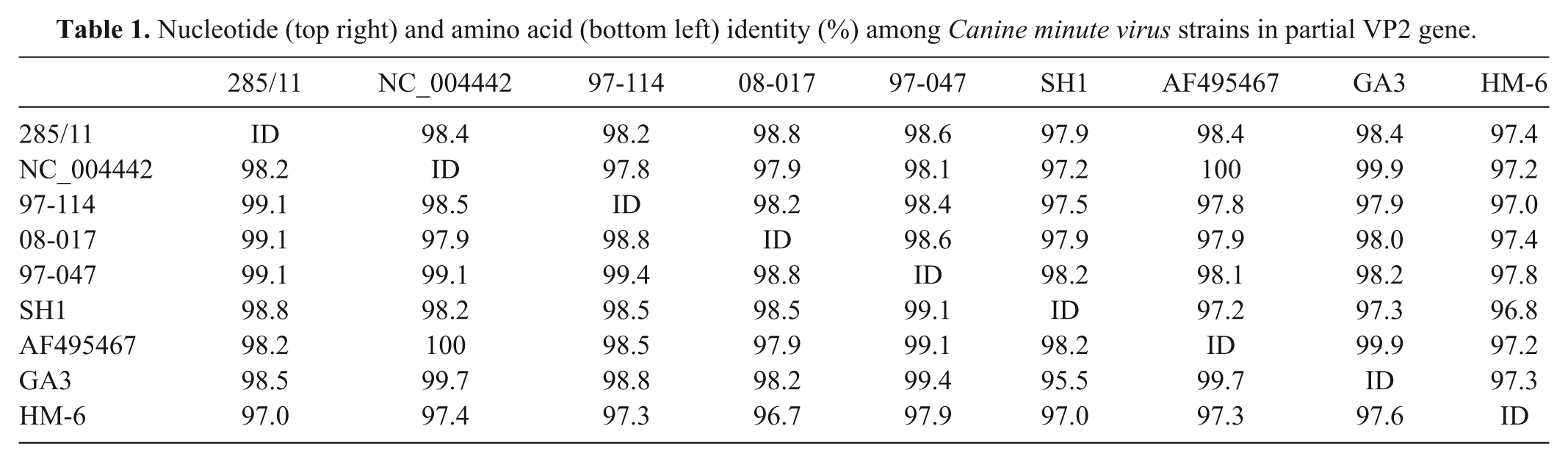
The VP2 protein gene fragment amplified by PCR from the organs of puppy M2 and from the rectal swabs of 2 puppies of the same kennel were determined by direct sequencing, e and consensus sequences were obtained using the contig assembly program option of the BioEdit software package. 17 The obtained sequences were aligned with reference sequences using the ClustalW tool of the European Molecular Biology Laboratory (http://www.ebi.ac.uk). Sequence analysis of a 1,005-nt fragment of the VP2 gene showed that the 3 CnMV strains detected in the kennel were identical; the virus recovered from the dead puppy (strain 285/11) was used for subsequent comparison with reference sequences. GenBank accession number JQ612703 was assigned to the VP2 sequence obtained from the prototype strain. By sequence analysis, strain 285/11 was found to be mainly related to strain 08-017, which had been isolated from an elderly dog with severe gastroenteritis in Japan, 24 displaying a 98.8% nucleotide identity with this isolate (Table 1). The relatedness to the Japanese isolates was also evident at the protein level (amino acid identity of 99.1%). Phylogeny inferred on the nucleotide sequence using the Mega 4.1 software 26 and the neighbor-joining method showed that the Italian CnMV clustered with Asian isolates, albeit with low bootstrap values, and was clearly distinct from the newly discovered Canine bocavirus 22 (Fig. 2).
Nucleotide (top right) and amino acid (bottom left) identity (%) among Canine minute virus strains in partial VP2 gene.
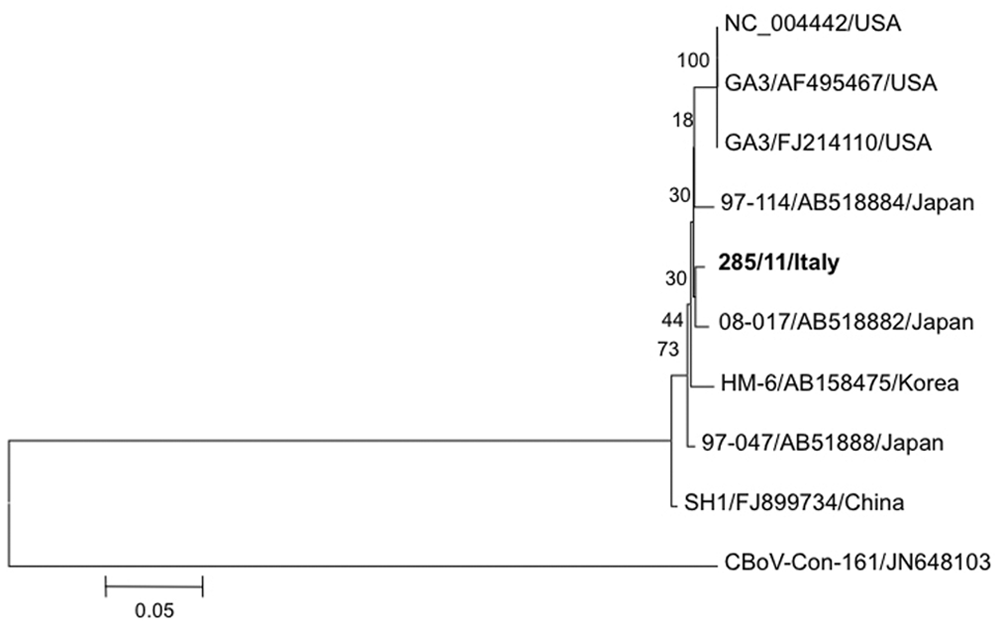
Neighbor-joining tree based on a 1,005-nucleotide fragment of the VP2 gene of Canine minute virus (CnMV) strains. For phylogenetic tree construction, CnMV sequences listed in Table 1 and the distantly related Canine bocavirus (CBoV) strain Con-161 were used. GenBank accession numbers are indicated for each strain. The scale bar represents the estimated numbers of nucleotide substitutions per site; bootstrap values were calculated and are indicated at each node.
Detection of CnMV antibodies in the canine serum samples was carried out using an indirect fluorescent antibody test (IFAT) with strain MVC/GA32,f as antigen, as previously described. 25 Canine minute virus–specific antibodies were detected in the sera of the dam (titer of 1:100), of the 2 surviving puppies (titers of 1:200 and 1:400), and of additional 2 puppies of the same kennel (titers of 1:100 and 1:200).
The present study reports the detection and molecular characterization of a CnMV strain associated with neonatal death. The virus was proven to be circulating in the kennel through detection of viral DNA in puppies of different litters and/or of specific antibodies in the dam, in the puppies of the same litter, and in those of other litters. The fact that the 2 surviving puppies tested negative for CnMV in both rectal and oropharyngeal swabs may be due to the recovery from the infection at the time when the animals were sampled. This hypothesis was supported by the detection of high titers of CnMV antibodies in these puppies. Unfortunately, no histopathological study was possible due to inappropriate storage and subsequent freezing of the dead puppy before submission to the laboratory; however, by sequence and phylogenetic analyses a strict relatedness of the Italian virus to CnMV strains recently isolated in East Asia was found. The present case report corroborates previous observations that CnMV can act as an infectious agent of neonatal mortality in dogs. 7 Both CnMV and Canid herpesvirus 1 may cause reproductive failure and puppy mortality, but the latter virus has been isolated with more frequency from outbreaks of neonatal death.7,11 The paucity of reports of the CnMV-associated disease may reflect failure to submit dead neonatal puppies to laboratory diagnosis, especially when death involves single members of a litter. This may possibly explain why there are no recent reports of CnMV-associated death in breeding kennels. At the same time, the ability of the virus to cause inapparent or subclinical infections in older animals may interfere with proper assessment of the impact of CnMV circulation in breeding kennels and rescue shelters. The pathobiology of CnMV appears to be still evolving, as further demonstrated by the 2011 report of this virus in the cerebrospinal fluid of adult dogs with neurological problems 15 and in the 2010 report of this virus in feces of an elderly dog with severe gastroenteritis. 24 Therefore, considering the lack of sequence data present in the publicly accessible databases, a more extensive molecular surveillance for CnMV in kennels, shelters, and rescue centers will help expand the knowledge base on the epidemiological and pathogenetic features of CnMV, which has been known since 1967 1 but still poorly understood.
Footnotes
a.
Tissue lyser, Qiagen SpA, Milan, Italy.
b.
QIAamp Viral RNA Mini Kit, Qiagen SpA, Milan, Italy.
c.
DNeasy Tissue Kit, Qiagen SpA, Milan, Italy.
d.
API, BioMérieux Italia SpA, Rome, Italy.
e.
BaseClear BV, Leiden, The Netherlands.
f.
Kindly provided by Dr. L. E. Carmichael, James A. Baker Institute for Animal Health, Cornell University, Ithaca, NY.
g.
GeneRuler 1kb DNA Ladder, MBI Fermentas GmbH, St. Leon-Rot, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Italian Ministry of Health, Ricerca corrente 2009, project IZS VE 21/09 RC “Definizione di una procedura validata per la selezione di cani per programmi di Interventi Assistiti dagli Animali (IAA).”
