Abstract
Since 2006, an unprecedented epidemic of highly pathogenic
Porcine reproductive and respiratory syndrome (PRRS) is a severe viral disease characterized by reproductive failure (stillborn, mummified, and weak-born piglets) and respiratory disease in young and growing pigs. The syndrome is caused by the PRRS virus (PRRSV), a member of a group of enveloped, positive-strand RNA viruses, classified in the family
In 2006, the so called high fever disease caused by a highly pathogenic PRRSV (HP-PRRSV) broke out in southern China. This disease affected over 2,000,000 pigs and caused 400,000 fatal cases in more than 10 provinces.6-8,13 The pigs infected by HP-PRRSV had the following atypical PRRS symptoms: neurological symptoms (e.g., shivering, lameness, and opisthotonus), high fever (40–42°C), and an erythematous blanching rash. 14 Numerous adult pigs and some pregnant sows died as a result of the HP-PRRSV infection in these outbreaks. “High fever” disease is characterized by initial viremia, then the distribution and replication of HP-PRRSV in multiple organs.10,12,13 The encephalitis was characterized by lymphohistiocytic perivascular cuffing, gliosis, and less often mild vasculitis.3,4 Although the lymphohistiocytic encephalitis had been reported in pigs, the encephalitis with marked nervous symptoms caused by the HP-PRRSV infection has not been previously investigated. 12
In the current study, brain tissues from the pigs inoculated with HP-PRRSV were investigated through reverse transcription polymerase chain reaction (RT-PCR), histopathology, and transmission electron microscopy. The pathological characters of encephalitis induced by HP-PRRSV and the process of this virus invading the brain tissue are described.
Ten PRRSV-free pigs (8–10 weeks old) were kept under good hygienic condition in Yueyang City, Hunan Province, China. The pigs were housed in separated rooms and divided into 2 groups (5 pigs per group). The pigs in the experimental group were inoculated intranasally with 105 TCID50 HP-PRRSV strain WUH1 (GenBank accession no. EU187484.1) grown in primary porcine alveolar macrophages in 4 ml of phosphate buffered saline (PBS). The pigs in the control group were inoculated intranasally with 4 ml of PBS. 1 Clinical signs were recorded daily. All pigs were euthanized and necropsied at 14 days postinoculation, and the brains were collected for investigation. The results showed that no illness was observed in the 5 pigs of the control group. In the experimental group, 4 pigs had the following clinical signs: inappetence, lethargy, high and continuous fever (40–42°C), hemorrhages in the skin, shivering, and lameness. The other pig in the experimental group did not have clinical signs. No deaths occurred during the experimental period.
The brains were analyzed for virus by RT-PCR as previously described. 8 Four sick pigs were positive for PRRSV by RT-PCR and were negative in the healthy inoculated pig and control group (data not shown; Fig. 2).
For histopathology, brains were fixed in 10% neutral buffered formalin for 48 hr. The tissues were routinely processed through a series of alcohols and xylene, embedded in paraffin, and then cut at 5 µm, deparaffinized, and stained with hematoxylin and eosin. Transmission electron microscope (TEM) was performed on other portions of the brains. The gray matter of the brain was cut into 2-mm 3 pieces then fixed in 2% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.0) for 12 hr. After rinsing in 0.1 M sodium phosphate buffer (pH 7.0) 3 times at room temperature (20–25°C), the samples were postfixed in 2% osmium tetroxide in 0.1 M sodium phosphate buffer for 1.5 hr. Then, the sections were dehydrated in a graded series of acetone, infiltrated and embedded in epoxy resin SPI-812, and polymerized at 60°C. Ultrathin sections (60–70 nm in thickness) were cut with a diamond knife and ultramicrotome, a mounted on slotted and formvar-coated grids, and stained with 5% uranyl acetate and 5% lead citrate.
Histological examination of the brain tissues of sick pigs showed marked lymphohistiocytic perivascular cuffing and gliosis with shrunken basophilic neurons (Fig. 1). By TEM, some red blood cells were outside of the capillaries, and the capillaries were completely detached from the neuropil (Fig. 3A). Numerous virions were accumulating around the Golgi apparatus in endothelial cells. Virions 25–40 nm in diameter clustered around the
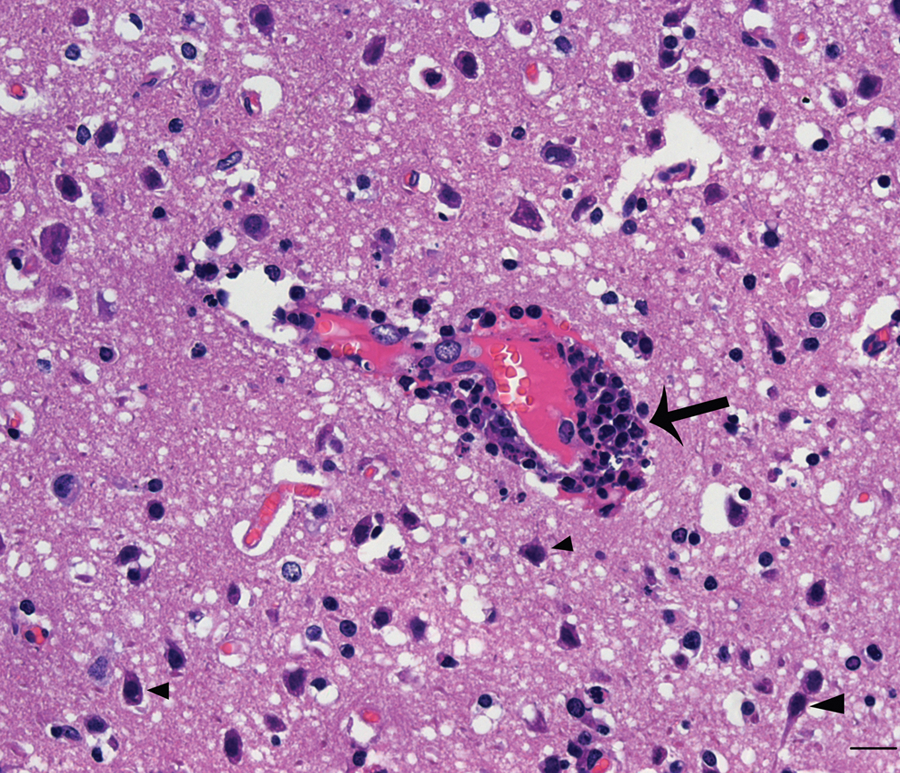
Pig. Photomicrograph shows perivascular cuffing (arrow) and the shrinking neurons (arrowhead) in the brain. Hematoxylin and eosin. Bar = 20 µm.
Moreover, vesicles contained some enveloped virions in the cytoplasm of the neuron (Fig. 4B). The perikaryons shrank into an irregular shape and detached from the neuropil. The electron density of perikaryons was apparently higher than around the neuropil (Fig. 4A, 4C). The nuclei of the neurons became oval, and the chromatin was condensed into an osmiophilic mass with thickened nuclear membranes (Fig. 4C). The cristae were partially obscure and lost in the mitochondria. The matrixes of mitochondria disappeared or were empty (Fig. 4A, 4C). The swollen endoplasmic reticulum (ER) and the expanded Golgi apparatus formed a complex labyrinthine vacuolation in the cytoplasm of the neurons.
Necrotic changes were found in 2 types of neuroglial cells: astrocytes and oligodendrocytes. The necrotic cells had condensation of the nuclear chromatin and disruption of nuclear membranes (Fig. 5A). The neuropil entered the cytoplasm area of astrocyte due to the disruption and loss of their cytoplasm membranes (Fig. 5B). There were more severe necrotic changes in astrocytes, including the complete loss of cytoplasm, the loss of nuclear matrix, and only some fragments of nuclear membrane remained (Fig. 5C).
As a multisystemic disease, PRRSV infection is characterized initially by viremia with subsequent virus distribution and replication in multiple organs, causing interstitial pneumonia, lymphadenopathy, myocarditis, and encephalitis.3,4,9,10 Lymphohistiocytic encephalitis with gliosis and severe necrotizing vasculitis had been demonstrated by histologic sections of brain tissues in weanling pigs infected by PRRSV. 13 The lymphohistiocytic perivascular cuffing and gliosis were also present in the brain tissues of pigs infected by HP-PRRSV (Fig. 2) in the current study.
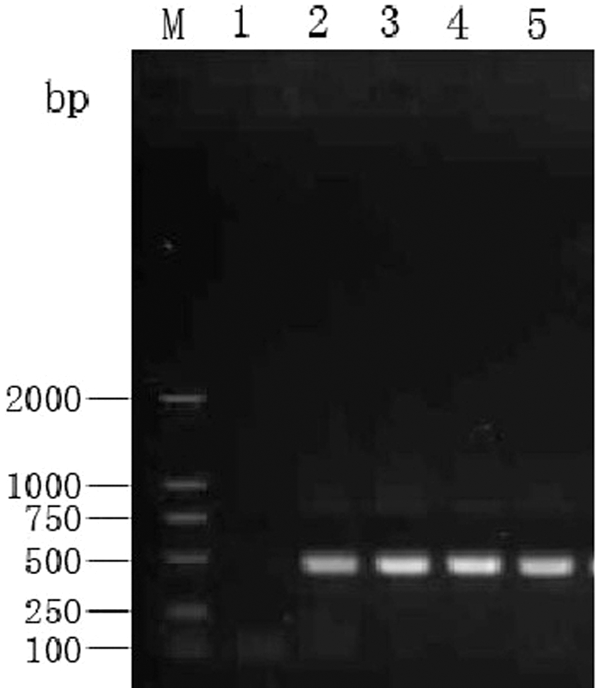
Reverse transcription polymerase chain reaction detection of
Several experimental infections have suggested that some strains of PRRSV are more likely to induce encephalitis than others. 4 The antigen of PRRSV was detected in the brain tissue of a weanling pig by immunohistochemical stain. 12 The HP-PRRSV antigen was also found in the brains of the “high fever” disease pigs. 13 These facts suggest that HP-PRRSV has the ability to invade brain tissue. The following findings provide further evidence that HP-PRRSV invaded into the brain tissue and caused the nonsuppurative encephalitis: nucleic acid of HP-PRRSV was detected in brain tissue (Fig. 2), the immunohistochemical stain was positive (data not shown), virions of 25–40 nm in diameter became enveloped virions of 50–65 nm in diameter through the Golgi apparatus in the capillary endothelial cells (Fig. 3B, 3D), the enveloped virions were also in cell vesicles of neuron cytoplasm (Fig. 4B), and the morphology and morphogenesis of these virions are identical to that reported for the PRRSV virion. 11
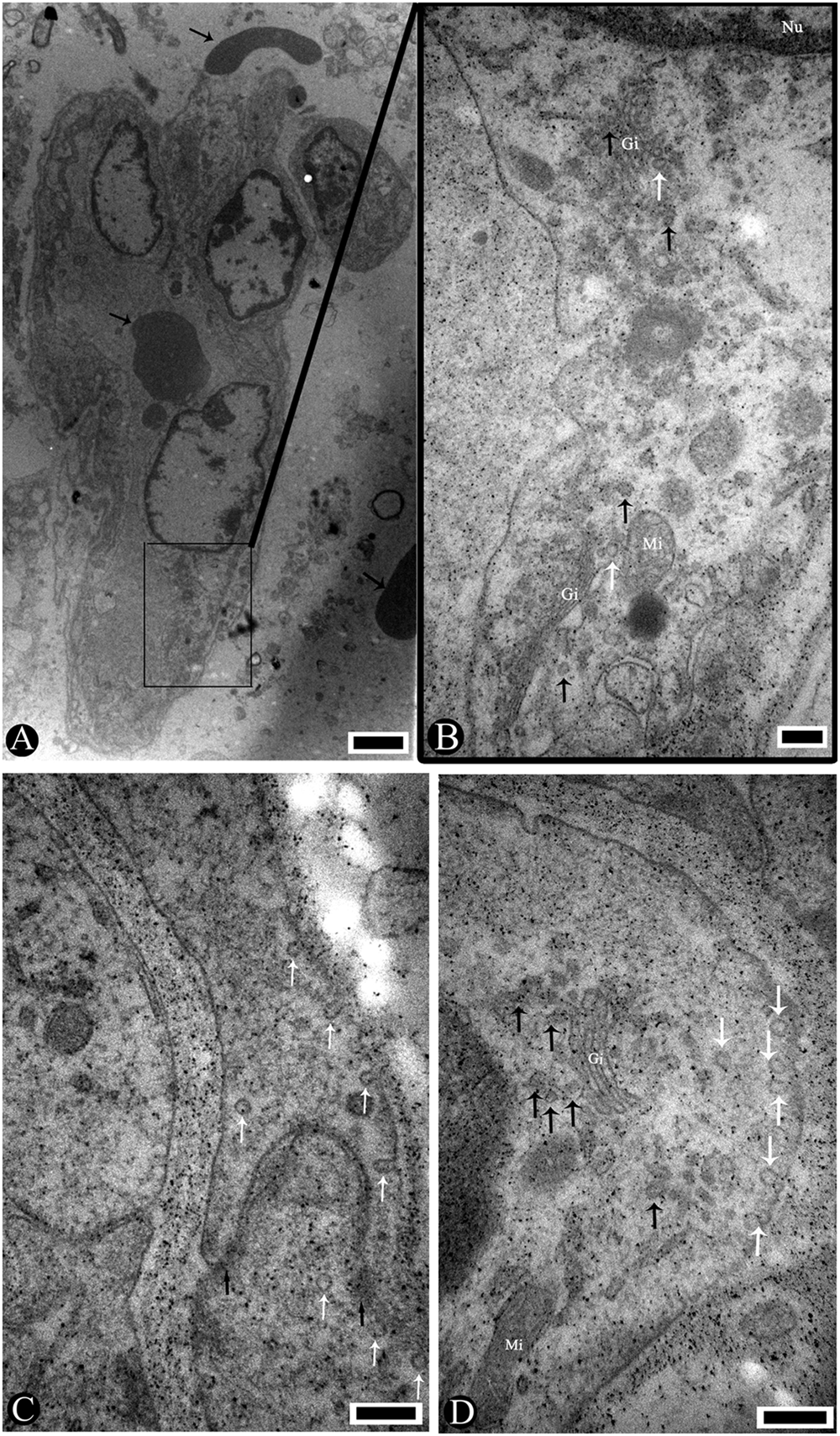
Pig. Electron micrographs showing a capillary endothelial cell infected by
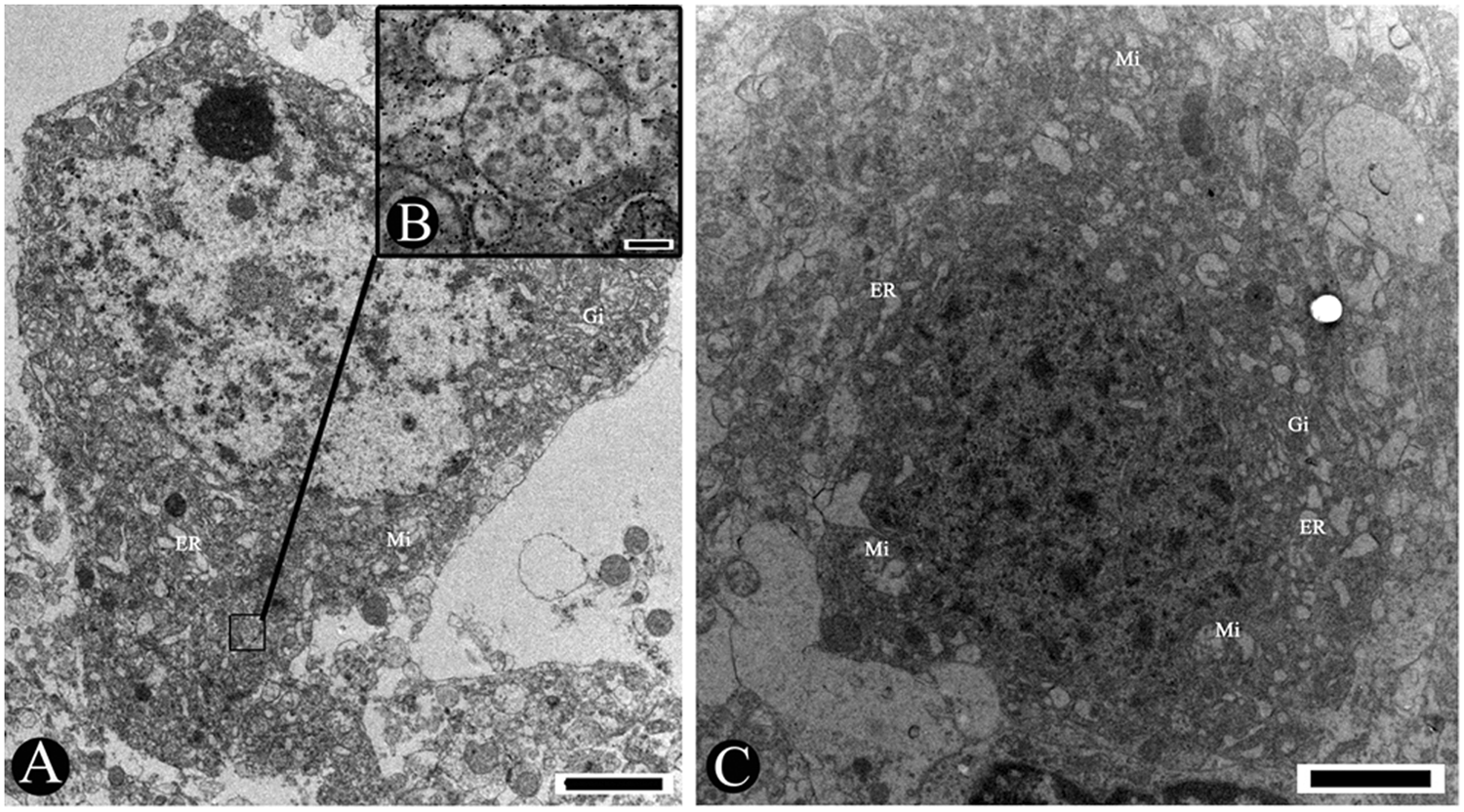
Pig. Transmission electron micrographs showing the changes in neurons induced by
Four pathways of virus entrance into the nervous system have been reported in human or animal brain diseases. One pathway is involved in the blood-brain barrier (BBB) and the capillary endothelial cells.
14
The brain capillary endothelial cells are the major component of the BBB. The BBB is a highly selective barrier that strictly regulates the exchange of nutrients, ions, inflammatory cells, and pathogens between the blood and the brain.
5
However, some RNA viruses such as lentiviruses and
The HP-PRRSV strain can cause nonsuppurative encephalitis in pigs (Fig. 1). Shrinkage of perikaryons with high electron density and condensation of the nuclear chromatin were present in neurons (Figs. 4A, 4C) and virions in the neuron (Fig. 4B). Both the disruption of the cytoplasm membrane and nuclear membranes and the loss of cytoplasm and nuclear matrix were observed in the neuroglial cells (Fig. 5). Cellular damage in the central nervous system can undoubtedly cause the neurological symptoms in HP-PRRSV–infected pigs.
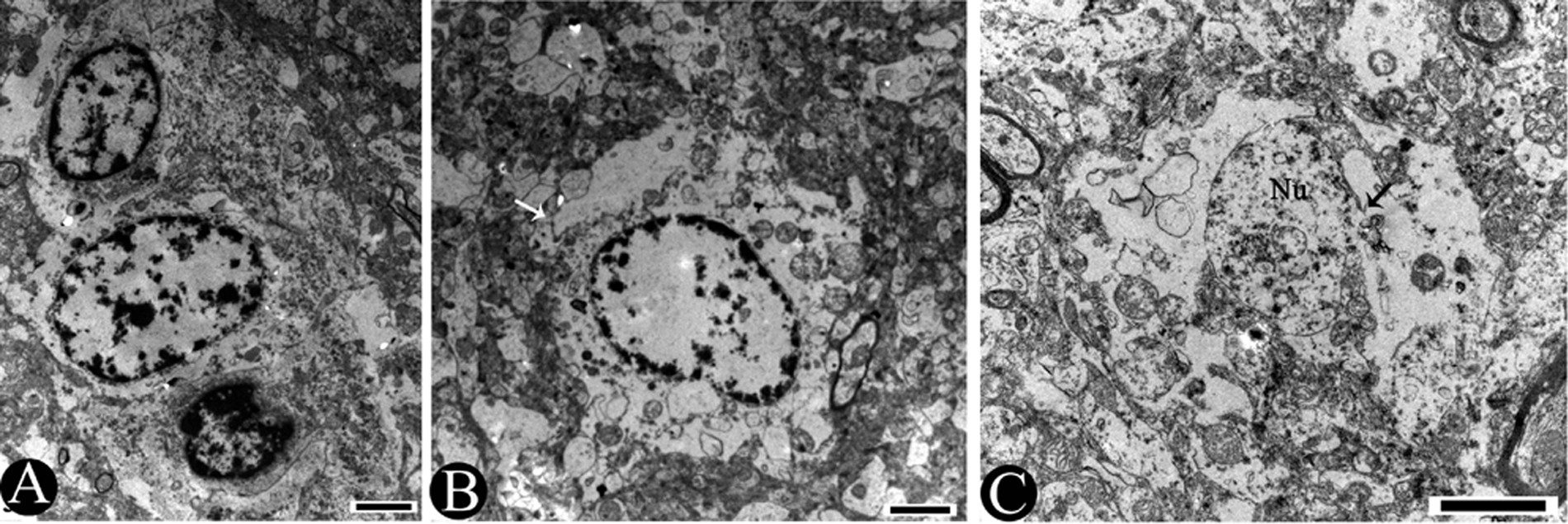
Pig. Transmission electron micrographs showing the changes of neuroglial cells.
In conclusion, HP-PRRSV can infect the capillary endothelial cell in the brain and subsequently cross the BBB, then damage neurons and neuroglial cells. These findings clarify the pathogenesis of nonsuppurative encephalitis and the neurologic signs.
Footnotes
Acknowledgements
Jianbo Cao and Bin Li contributed equally to this work.
a.
Leica UC6 ultramicrotome, Leica Microsystems GmbH, Wetzlar, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the State 863 High Technology R&D Project of China (2011AA10A208), the Genetically Modified Organisms Breeding Major Projects (2009ZX08009-142B), and the National Natural Sciences Foundation of China (31001066, 30972189).
