Abstract
An 8-year-old male rhesus macaque (Macaca mulatta) presented with unilateral enlargement of the left mandible. Radiographs revealed a marked expansion of the left mandible with a multilocular radiolucent mass with abundant osteolysis. The mass was grossly firm, fleshy, and gelatinous on the cut surface. Histologically, the mass was locally infiltrative and composed of neoplastic epithelial and mesenchymal components that stained positive for cytokeratin and vimentin, respectively. Occasional densely spherical condensations of fibroblasts resembling the cap stage of odontogenesis were present in the mesenchyma. Immunohistochemical staining with Ki-67, S-100, and CD34 indicated that both epithelial and mesenchymal components of the neoplasm had low proliferation. Alcian blue, periodic acid–Schiff, and trichrome stains showed an immature stromal component with no collagen formation. Based on the clinical, histologic, and immunophenotypic features, the tumor was identified as a locally infiltrative ameloblastic fibroma.
Ameloblastic fibroma (AF) is a rare odontogenic tumor in which both the odontogenic epithelial and ectomesenchymal elements are neoplastic.5,29 In human beings, AF occurs most commonly in the first 2 decades of life, with an incidence of 1.5–4.5% of all jaw tumors.16,31 Ameloblastic fibroma is a benign, slowly growing, expansile tumor that commonly affects the posterior mandible.3,31 Radiographically, AF is usually small in size (≤4 cm in diameter) with a unilocular lucent lesion but may develop into a multilocular lesion as it gets larger (>4 cm in diameter).3,32 Histologically, the epithelial element is usually similar to ameloblastoma, but the mesenchyma is an immature cell-rich myxoid tissue with an embryonic appearance. Dental hard tissue is not a part of AF.17,29
Ameloblastic fibroma in animals has been reported rarely in cattle, 7 a dog, 14 and cats. 19 Controversially, most cases originally diagnosed as AF in cats9,21,34 were renamed feline inductive odontogenic tumor (FIOT), a unique odontogenic tumor in cats that has most of the histological features of AF but a locally infiltrative nature and forms mesenchymal spherical nodules resembling the cap stage of odontogenesis.6,8,23 It is unclear whether AF and FIOT are stages in the evolution of a single type of lesion or represent separate entities. Feline inductive odontogenic tumor is unique to cats and is not documented in other species, likely due to the extreme rarity of odontogenic neoplasms.
The current article describes the clinical, radiographic, gross, and histological features of an odontogenic tumor with similar histological features of both AF and FIOT in a rhesus macaque (Macaca mulatta). In addition, to predict the biological behavior and to help classify this tumor, the expressions of cytokeratin, vimentin, Ki-67, cluster of differentiation (CD)34, and S-100 were evaluated by immunohistochemistry. Mesenchymal components were analyzed by trichrome, periodic acid–Schiff (PAS), and alcian blue stains.
An 8-year-old male India-origin rhesus macaque presented with a large firm mass over the left mandible. On oral examination, the left mandible was markedly expanded by a mass that caused the lingual displacement of the left mandibular dental arcade. The overlying gingival mucosa was multifocally ulcerated with formation of draining tracts. All other oral structures were normal. Normal bacterial flora was cultured from ulcerated areas. Hematology showed a mild neutrophilic leukocytosis (total white blood cell count = 12,850 and 87.1% of neutrophils). All remaining parameters of the complete blood count and serum chemistry were within reference ranges. Radiographs revealed a marked expansion of the left mandible with a multilocular radiolucent mass with abundant bone lysis and a cystlike structure in the frontal sinus (Fig. 1a). The animal was observed and treated for 4 months. No enlargement of the mass was noted during that period of time. Euthanasia was elected due to the lack of responsiveness to antibiotic treatment and deteriorating physical condition.
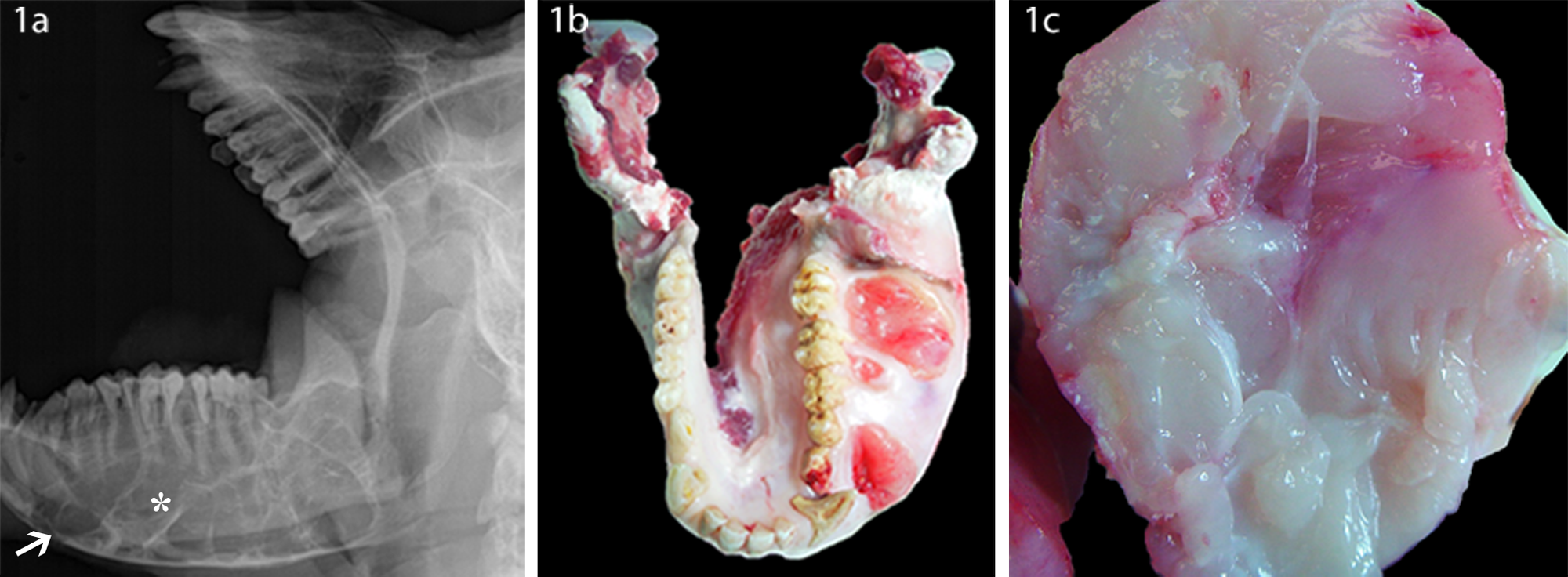
India-origin Rhesus macaque (Macaca mulatta): dental radiograph (
Grossly, the whole left mandible was markedly expanded by a firm mass, 9 cm × 6 cm × 7 cm, producing an asymmetrical jaw (Fig. 1b). Multifocal gingival ulcers were noted. On cross section, the mass was multilobulated, pale white to pink, fleshy, and gelatinous with many tiny gray gritty foci (Fig. 1c). Mandibular bone loss was evident, especially at the center of the mass. Sparse spicules of trabecular bone were noted at the periphery of the mass. The right mandible and maxilla were unaffected.
Histopathologically, the normal tissue architecture of the left mandibular bone was effaced and expanded by a demarcated, partially encapsulated, and highly infiltrative neoplasm composed of both proliferative odontogenic epithelial and mesenchymal components. Both neoplastic components infiltrated into, separated, and replaced the trabecular bone. The epithelial component formed islands, cords, and branching and anastomosing strands that were separated by a large amount of myxoid mesenchymal tissue (Fig. 2a). The basal epithelial cells in the islands and cords were often but not always elongate and peripherally palisaded on a thin layer of eosinophilic membrane. These cells had distinct cell borders, basal clear cytoplasm, and apically elongated hyperchromatic nuclei with 1–2 distinct nucleoli (inner enamel epithelium). The cells in the centers of the islands and cords were usually loosely arranged as small spindle to stellate cells (stellate reticulum) with prominent intercellular spaces (Fig. 2b). However, groups of small, round, and poorly differentiated epithelial cells were noted in the islands and cords (outer enamel epithelium). These cells had indistinct cell borders, clear cytoplasm, and a round central hyperchromatic nucleus with a high nuclear to cytoplasmic ratio. Multifocally, poorly differentiated epithelial cells formed small islands and clusters without a center of stellate reticulum. Occasionally a thin layer of hyaline material surrounded these structures. Mitotic cells were extremely rare. Fibrosis was also noted in multiple areas between the alveolar bone and neoplasm and surrounding the infiltrative neoplasm (Fig. 2c).
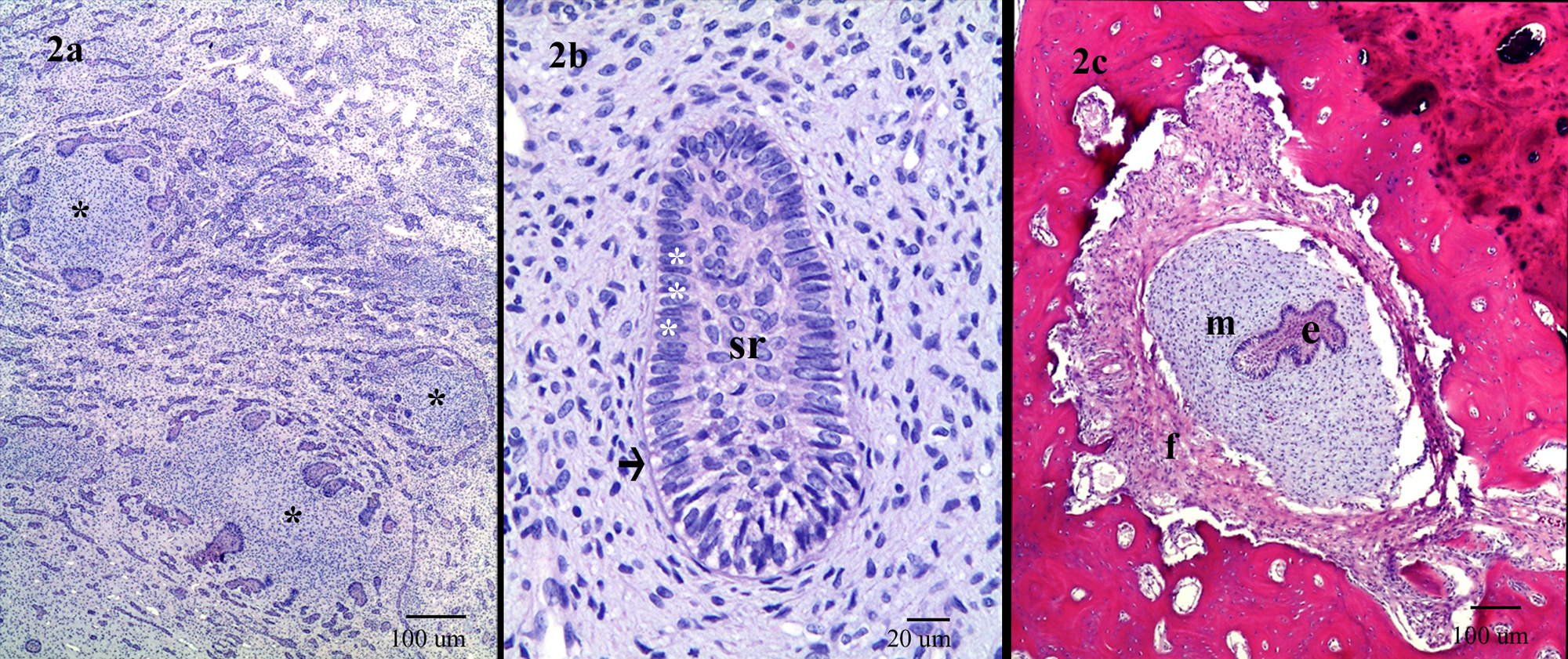
India-origin Rhesus macaque (Macaca mulatta): photomicrographs of the locally infiltrative ameloblastic fibroma. Hematoxylin and eosin.
In the areas of high cell density, the mesenchymal cells were plump, pleomorphic, and haphazardly arranged with indistinct cellular borders and pale eosinophilic, granular to vesicular cytoplasm. The nuclei were hyperchromatic and markedly pleomorphic. Occasionally, the nuclei were surrounded by a zone of clear cytoplasm. In the less dense areas, the mesenchymal cells had indistinct cell borders, abundant bluish to pale eosinophilic granular cytoplasm, and a small regular spindle-shaped nucleus. The nucleoli of all the mesenchymal cells were indistinct. Mitoses were rare, although some hyperchromatic nuclei had bizarre shapes. Multiple layers of spindle-shaped mesenchymal cells surrounded the small epithelial islands and clusters (uninteractive stage) or part of the inner enamel epithelium in large islands and cords. The mesenchymal cells adjacent to the inner enamel epithelium were cuboidal to columnar, with clear vesicular cytoplasm (formation of odontoblasts at early bud stage). Rare odontogenesis at early bell stage was noted, but dental hard tissues were not observed (Fig. 3a). There was a large cyst that was lined with 3–10 cell layers of nonkeratinized squamous cells. Mesenchymal cells occasionally formed variably sized, circular, noncollagenized, and dense cellular aggregates partially surrounded by neoplastic odontogenic epithelium that resembled the dental papilla (Figs. 2a, 3a). Multifocal areas of degeneration and/or necrosis were noted in both epithelial and ectomesenchymal components. Both blood and lymphatic vessels were congested, and there were multifocal areas of osteonecrosis, mineralization, and new bone formation.
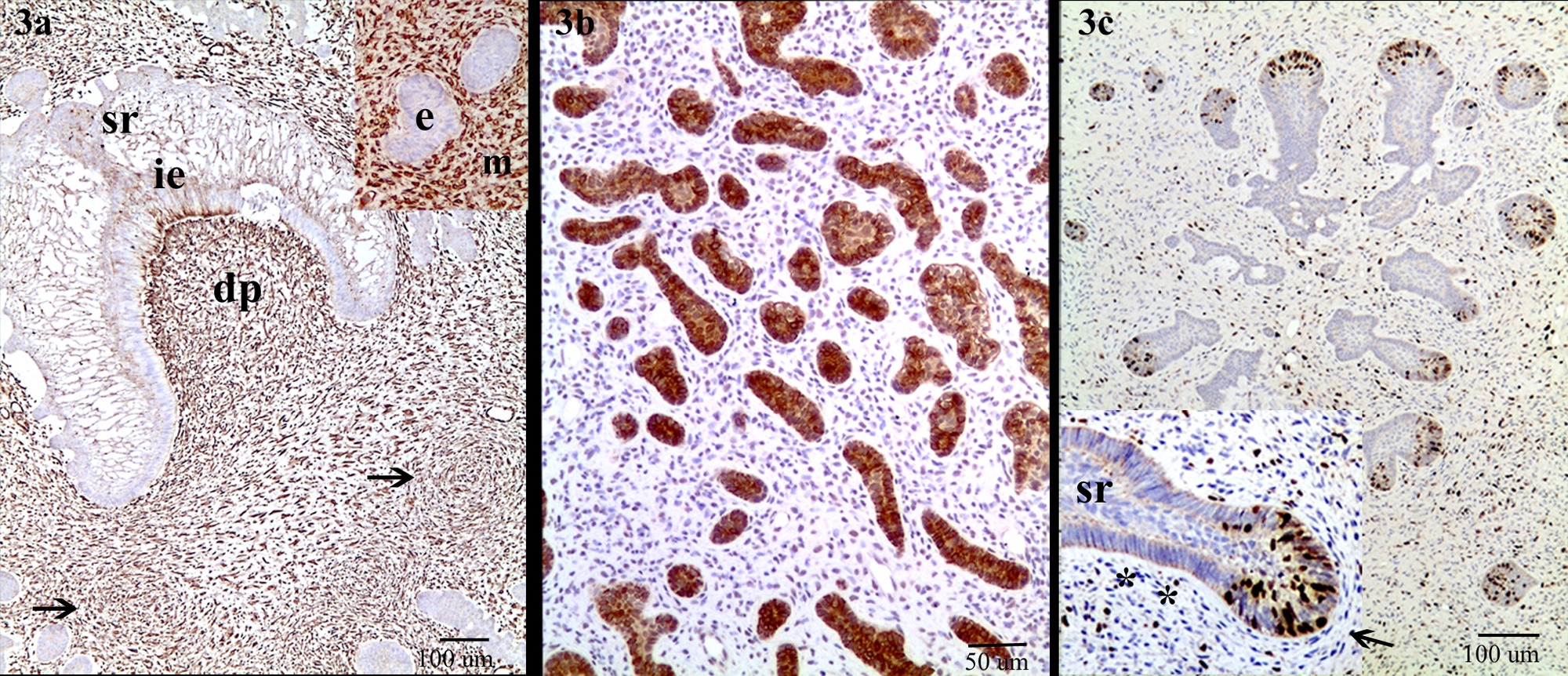
India-origin Rhesus macaque (Macaca mulatta): immunohistochemistry for vimentin (
Immunohistochemical staining was performed on 4-µm-thick, formalin-fixed, and paraffin-embedded tissue sections using a standard streptavidin–biotin method and diaminobenzidine as chromogen a detection with hematoxylin counterstain. The cytoplasms of the mesenchymal cells were strongly positive for vimentin (Fig. 3a) and negative for cytokeratin. Conversely, the odontogenic epithelial cells had strong cytoplasmic staining for cytokeratin b (1:200 dilution; Fig. 3b) but were vimentin c negative.
Odontogenic epithelial cells showed low numbers (4.37%) of Ki-67 d –positive cells, but in an interesting pattern (Fig. 3c). Most Ki-67–positive epithelial cells formed small round clusters (uninteractive stage), or in the outer enamel epithelium adjacent to the spindle mesenchymal cells in islands and cords. The columnar epithelial cells adjacent to columnar odontoblast-like cells in the same islands or cords were completely negative for Ki-67. Randomly scattered in the mesenchyma, approximately 10–15% of cells were Ki-67 positive. These cells usually had plump round or cigar-shaped nuclei. The cells with spindle-shaped nuclei were often weak to negative for expression of Ki-67.
Odontogenic epithelial cells and most mesenchymal cells were negative for S-100 e (1:2,000 dilution), but occasionally the spindle cells surrounding the epithelial islands were nuclear positive for S-100. The neoplastic epithelial and mesenchymal components were completely negative for CD34 f (1:10 dilution). Alcian blue stain (pH 2.5) revealed a large amount of intracytoplasmic bluish cytoplasm, especially in the spindle-shaped cells, which were also stained PAS positive. Trichrome stain demonstrated no mature collagen formation within the neoplastic tissue.
In nonhuman primates, odontogenic tumors are rare, and only a few cases of ameloblastic odontoma have been reported.30,35 The biphasic character of the tumor in the animal reported herein is of special interest, given the local infiltration and formation of circular, highly cellular mesenchymal aggregates resembling the cap stage of odontogenesis, a tumor type rare in the jaw of any species.
The classification of the current tumor is challenging because of a lack of comparative histopathological characteristics in the published literature. Referring to the 2003 World Health Organization’s Histological Classification of Tumors of Odontogenic Origin of Domestic Animals, odontogenic tumors are classified according to the neoplastic origin of epithelial, mesenchymal, or both. 5 Based on this guideline, this specific tumor fits into the category of “tumors of odontogenic epithelium with odontogenic mesenchyme,” which includes AF and feline inductive odontogenic tumor. Other tumors in this category, including ameloblastic fibro-odontoma, complex odontoma, and compound odontoma, were ruled out because the tumor in the present study had no dental hard tissue formation. Immunohistochemistry confirmed the biphasic proliferation in the current case, as the neoplastic epithelial cells were strongly positive for pancytokeratin but negative for vimentin. In contrast, the neoplastic mesenchymal cells were strongly positive for vimentin but negative for cytokeratin.
The tumor had many similarities to AF in human beings and animals. Ameloblastic fibroma is a slow growth tumor predominantly involving the lower jaw.27,28 Histologically, it is a well-demarcated odontogenic neoplasm composed of both epithelial components and embryonic mesenchymal stroma.5,27 The epithelial part resembles ameloblastoma, which usually forms strands, islands, and cords with a central stellate reticulum lined by peripheral palisading epithelial cells. The mesenchymal component contained an immature highly cellular myxoid tissue mimicking the dental papilla, with little collagen. Mitoses were rare, and there was no metastasis. Dental hard tissues do not form part of the histological spectrum of AF.15,20,28,29
Conversely, the tumor showed some features that differed from classical AF, as AFs are usually well demarcated and encapsulated, but the encapsulation of the current tumor was incomplete. Most important, both the neoplastic epithelial and mesenchymal components of this tumor infiltrated into and disrupted the adjacent alveolar bone. Interestingly, the neoplastic mesenchymal cells occasionally formed circular fibroblastic condensations, resembling the cap stage of odontogenesis. These histological features are not described in AFs.2,8,14
Although the infiltration into the adjacent cancellous bone is also a common feature in ameloblastoma,10,24 ameloblastoma is a tumor of odontogenic epithelium surrounded by normal mature connective stroma rather than by immature ectomesenchymal elements.15,20,28,29 The immaturity of the stromal component in the current tumor was confirmed by alcian blue and PAS stains, which demonstrated abundant glycosaminoglycans and glycogen. Trichrome stain showed no collagen formation within the neoplastic stroma.
The formation of spherical condensations of fibroblasts in the current tumor is a typical histological features of FIOT that was first reported in 1979, 6 and several cases have subsequently been reported.2,8,23 In addition, the local infiltration of neoplastic components into adjacent bone is another distinctive attribute of FIOT.8,23 In felids, FIOT is considered a tumor exclusively of young cats with no sex predilection and predominantly localized to the maxilla. Radiographically, FIOTs are uni- to multilocular lucent lesions. Histopathologically, FIOT is a nonencapsulated and poorly circumscribed expansile mass. The rate of mitosis is very low, and metastasis is not a feature of the tumor. Although noted for local bone destruction, FIOT is considered a benign tumor.2,8,23 It is noteworthy that odontogenic tumors with similar histological features in cats have previously been diagnosed as AF.9,21,34 In addition, similar histological nodular condensations of fibroblastic mesenchyma were also present in AF in human beings.1,3,27
Identification of proliferating activity in odontogenic tumors has been widely used to predict the biological behavior of odontogenic tumors in human beings.22,25 However, no studies have adequately described the proliferative activity and biological behavior of odontogenic tumor in animals, probably due to their rarity. The expression of Ki-67, a cell marker for proliferation, in both epithelial and mesenchymal components in the current tumor was low, consistent with data on AFs in human beings18,25,26 and FIOT in cats. 23 The low proliferative activity indicates a benign biological behavior of these tumors.18,23 However, the Ki-67–positive cells of this tumor had a particularly interesting distribution. Only the epithelial cells in the poorly developed stellate reticulum and outer enamel epithelium had strong nuclear staining for Ki-67. Most columnar epithelial cells (inner enamel layer) in the islands and cords were completely Ki-67 negative. This indicated a variable proliferative rate between the epithelial components, which is very similar to Ki-67 expression in ameloblastomas, in which basal epithelial cells in the outer layer and poorly developed stellate reticulum retain the character of odontogenic stem cells and have active proliferation. 25
The mesenchymal Ki-67–positive cells were randomly scattered. These cells were plump and spindloid, with myxoid cytoplasm and a large elongated or cigar-shaped nucleus. The morphology and Ki-67 expression pattern suggest that these cells are immature and actively proliferating rather than malignant. The high expression of Ki-67 in the mesenchymal component has been reported in ameloblastic fibrosarcoma, a malignant counterpart of AF with a benign epithelial component and malignant mesenchymal component.18,26 Further, CD34 expression was absent in both epithelial and mesenchymal components in the current case. Cluster of differentiation 34 is a transmembrane protein expressed on hematopoietic stem cells that is downregulated in mature cells. 33 The CD34 protein has been demonstrated in the mesenchymal component of ameloblastic fibrosarcoma. 12 Moreover, the current tumor had no evidence of metastasis to regional lymph nodes or other organs, extremely rare mitoses, and no giant cells in the mesenchyma. Metastases, high mitoses, and multinucleate giant cells are the typical histological features in malignant odontogenic tumors.15,18,29 Rare S-100 positive cells were detected in the spindle cells surrounding epithelial islands. These cells may be the odontogenic embryonic residues of neural crest-derived ectomesenchymal cells and can return to an active growth phase and may proliferate to become odontogenic tumors. 13 More recent evidence suggests that AF exhibits a high rate of recurrence within 10 years of surgical removal3,4 and that more than 30% of ameloblastic fibrosarcomas arise from recurrent AF in human beings. 11 The recurrence of FIOT has also been observed in the first month after surgery. 8 Combined, the data suggest that the current tumor has the potential to transform into a malignant tumor. However, since the animal was humanely euthanized, recurrence and malignant transformation could not be evaluated in this case.
In summary, locally infiltrative AF was the final diagnosis for this particular odontogenic tumor because of its location, multilocular radiolucency, the histopathological coexistence of both neoplastic epithelial and mesenchymal components with absence of dental hard tissue and no mature collagen formation, and low neoplastic cellular proliferative activity. These features are very similar to those reported in human AF. Meanwhile, observations suggest that the nodular condensations of fibroblastic mesenchyma may not be unique to inductive odontogenic tumor in cats (FIOT); rather, it may represent an evolutionary stage of a single type of lesion in AF.
Footnotes
Acknowledgements
The authors thank Carol Coyne, Dorothy Kuebler, Terri Rasmussen, Maury Duplantis, and Diganta Pan for their technical support, and Dr. Robert Dubielzig for the advice.
b.
FLEX Monoclonal Mouse Anti-Human Cytokeratin Clone AE1/AE3, Dako North America, Carpinteria, CA.
c.
Vimentin Concentrated and Prediluted Monoclonal Antibody, Biocare Medical LLC, Concord, CA.
d.
Monoclonal Mouse Anti-Human Ki-67 Antigen/FITC, Dako North America, Carpinteria, CA.
e.
Monoclonal Anti-S-100 (α-subunit) antibody produced in mouse, Sigma-Aldrich, St. Louis, MO.
f.
Concentrated and Prediluted Monoclonal Antibody, Biocare Medical LLC, Concord, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by National Institutes of Health grant RR00164.
