Abstract
Vascular mineralization (siderocalcinosis) in the brain of horses has been usually assumed to be an incidental age-related finding with no clinic significance. In the present study, eight 15–32-year-old horses of different breeds with cerebral siderocalcinosis were studied. Four of these horses had acute and severe central nervous system clinical signs of unknown etiology, 2 horses had neurological signs of known cause, and 2 horses did not have neurological signs. Gross examination of the brains in 4 animals revealed symmetrical foci of malacia in the cerebellar white matter. Histologically, moderate to severe mineralization of blood vessels and parenchyma were observed in all 8 horses, occasionally associated with necrosis of the adjacent tissue. Some horses were tested by virus isolation, polymerase chain reaction, immunohistochemistry, and serology to investigate Rabies virus; West Nile virus; Equid herpesvirus 1 and 4; Eastern, Western, Venezuelan, and Saint Louis encephalitis virus; and Sarcocystis neurona infection. These tests were negative in all samples analyzed. Brain cholinesterase activity and heavy metal screening were also unremarkable. The significance of the vascular and parenchymal mineralization in the brains of some of these horses remains undetermined. However, the severity of the lesions observed in the brains of some of the animals in the present study, coupled with the negative results for other common causes of neurological disease in horses, suggests a possible relationship between siderocalcinosis and the clinical signs observed.
Dystrophic mineralization of the wall of brain and meningeal blood vessels has been reported in human beings, monkeys, mice, dogs, cows, horses, and cats.1,2,6,10,14,15 In horses, mineralization has been described in 26% and 29% of aged animals, most commonly in the corpus striatum (internal capsule) and cerebellar white matter, respectively.1,6 In the few cases in which the composition of the mineral has been described, it consisted of a mucopolysaccharide matrix with variable composition of mineral salts, mostly calcium and iron, but also smaller amounts of salts of aluminum, phosphorus, and zinc. Because of this mineral composition, the lesion has been also called siderocalcinosis.1,12,14
In spite of the relatively frequent observation of siderocalcinosis in horses, the significance of mineralization remains undetermined. Siderocalcinosis in horses is usually assumed to be an incidental age-related finding with no clinic significance; however, vascular occlusion associated with contiguous areas of ischemic necrosis has been described, albeit rarely.11,12 These areas of necrosis were characterized by loss of parenchyma and shrunken, angular, and hypereosinophilic neurons, associated with minimal inflammatory response consisting of astrocytosis and lymphoplasmacytic infiltration.
The current study describes brain mineralization in 8 aged horses, 6 of which had neurological clinical signs. The cause of neurological disease in 4 of the horses could not be determined after a comprehensive diagnostic work up.
Eight horses with cerebrovascular mineralization were selected from the archives of the San Bernardino Branch of the California Animal Health and Food Safety Laboratory System (CAHFS). The animals were divided into 2 groups of 4 horses each. Group A included 4 horses (horses 1–4) with central nervous system (CNS) clinical signs (Table 1). The neurological signs described in the clinical history reported by the submitting veterinarians were variable and included circling, lethargy, cortical blindness, seizures, nystagmus, dysmetria, tremors, ataxia, head tilt, and/or recumbency. Group B included 2 animals with ataxia due to spinal cord hemangiosarcoma and idiopathic compressive myelopathy (5 and 6, respectively), and 2 animals without neurological clinical signs (7 and 8). The horses included mares, stallions, and geldings between 15 and 32 years of age, with an average of 25 and 22.3 years of age for groups A and B, respectively. Several breeds were represented. The 8 studied horses originated from 8 properties in Southern California and were submitted for postmortem examination to CAHFS San Bernardino between 2004 and 2007. During this period, 1,622 horses were submitted for necropsy and 173 (10.6%) were older than 15 years.
Age, breed, sex, and clinical and gross pathological finding in the 8 horses used in the current study.*
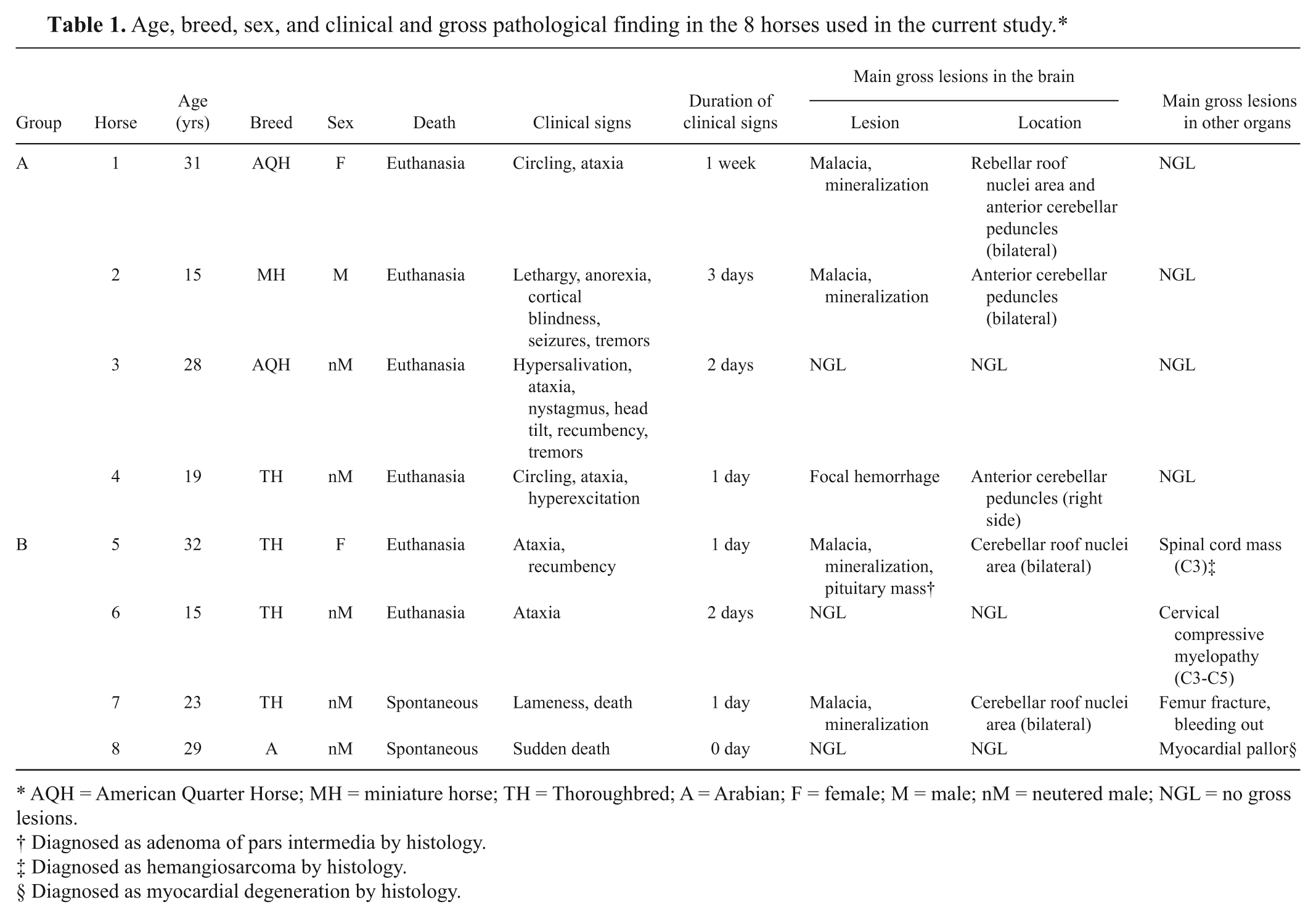
AQH = American Quarter Horse; MH = miniature horse; TH = Thoroughbred; A = Arabian; F = female; M = male; nM = neutered male; NGL = no gross lesions.
Diagnosed as adenoma of pars intermedia by histology.
Diagnosed as hemangiosarcoma by histology.
Diagnosed as myocardial degeneration by histology.
A complete necropsy was performed on all horses within 8–24 hr of death; whole brains, and samples of spinal cord, lung, heart, liver, kidney, spleen, urinary bladder, muscle, and small and large intestine were fixed by immersion in 10% buffered formalin. After a minimum of 24 hr, the brains were sliced into approximately 0.5 cm thick sections and fixed in fresh formalin for at least 7 more days. Sections for microscopic examination were taken from cortex, corpus striatum, thalamus, midbrain at the level of rostral colliculi, pons, cerebellar peduncles, cerebellum, and medulla at the level of obex.
After fixation, the tissues were processed routinely for the production of 4-µm-thick sections and stained with hematoxylin and eosin. Selected sections from brain were also stained with periodic acid–Schiff (PAS), Von Kossa, Perls Prussian blue, phosphotungstic acid–hematoxylin (PTAH), Congo red, and Masson trichrome. The sections were examined by light microscopy, and the vascular mineralization was classified as grade 1 (mild sporadic capillary mineralization), grade 2 (moderate mineralization of a moderate number of capillaries, venules, and arterioles), or grade 3 (severe mineralization of large numbers of capillaries, venules, and arterioles, with occlusion of the vascular lumen in some vessels). Parenchymal necrosis surrounding vascular mineralization was also classified in 3 grades depending on the extension (1: mild, 2: moderate, 3: severe).
Selected brain sections from all 8 horses were processed by an avidin–biotin complex immunoperoxidase method using primary antibodies against Equid herpesvirus 1 and 4 and Sarcocystis neurona.4,16 Rabies testing was performed on brains of 3 horses (horses 1, 3, and 4) by a direct fluorescent antibody test (FAT) at the San Bernardino County Public Health Laboratory, following the Centers for Disease Control protocol (http://www.cdc.gov/rabies/pdf/RabiesDFASPv2.pdf). Serum samples from horses 1, 3, and 4 were tested for antibodies to West Nile virus (WNV); Eastern, Western, Venezuelan, and Saint Louis encephalitis virus; and Sarcocystis neurona following protocols of the World Organization for Animal Health (http://web.oie.int/eng/normes/MMANUAL/A_Index.htm). Polymerase chain reaction (PCR) for WNV was performed on pooled CNS tissue (medulla and lumbar spinal cord) of horses 2–4 as previously described. 8 Similar CNS tissue from horses 2 and 3 were collected into virus transport medium and stored at −80°C until processed for virus isolation according to standard operating procedures of the School of Veterinary Medicine at Cornell University (Ithaca, NY). Cecal content from horses 2 and 6 was cultured on blood agar and selenite broth at 37°C under aerobic conditions for 72 hr. Brain cholinesterase activity was determined in horses 3 and 8 following the modified Ellman method. 13 A heavy metal screening was performed on liver and kidney of horses 5 and 8 by inductively coupled argon plasma emission spectrometry. This screen included lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, and cadmium.
A summary of the main gross lesions found in the horses is presented in Table 1. Bilateral and roughly symmetrical, 0.5–3 cm in diameter, poorly demarcated, and irregularly shaped dark brown malacic foci were observed in the cerebellum of 2 animals from group A (horses 1 and 2) and 2 animals from group B (5 and 7; Fig. 1). One horse of group A (4) presented a unilateral, 0.7 cm in diameter, well-demarcated hemorrhagic focus in the right anterior cerebellar peduncle. Other extra-cerebral lesions of horses from group B were hemorrhagic cervical mass in the spinal cord and a small mass in the pituitary gland (5), cervical compressive myelopathy (6), acute femoral fracture with tearing of the large blood vessels of the area and bleeding out (7), and focally extensive area of myocardial pallor affecting the free wall of the left ventricle of the heart (8). No other significant gross lesions were observed in any of the horses examined.
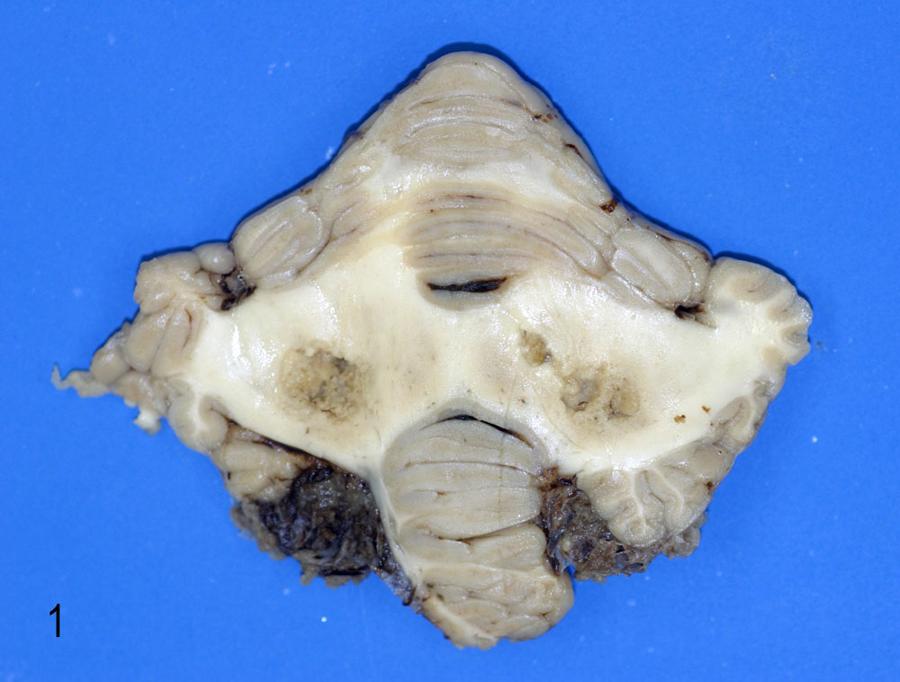
Cerebellum; horse 1. Bilateral and symmetric malacic foci affecting cerebellar white matter in the area of the cerebellar roof nuclei.
The most prevalent microscopic lesion observed in the CNS of these horses was mineralization of venules, arterioles, and/or capillaries of the cerebellar peduncles, cerebellar white matter, cerebellar roof nuclei (i.e., dentatus and interpositus), and internal capsule (Table 2, Fig. 2). The tunica media of affected arterioles and venules presented variable degrees of mineral deposition, which stained positively in every case with PAS, Von Kossa, and Perls, confirming the presence of mucopolysaccharides, calcium, and iron salts. Most vessels also showed deposition of an acidophilic, fibrillar, and concentric proteinaceous material with a few spindle cells in the subintimal space. This proteinaceous material was stained blue with Masson trichrome in all cases (Fig. 3). In most vessels, the lumen was partly to completely occluded by mineral deposits. In horses with macroscopic lesions, there was minimal inflammatory reaction adjacent to areas of parenchymal loss, which consisted of mild astrocytosis and lymphocytic infiltration. However, in 3 horses from group A (horses 1, 2, 4) and 2 from group B (5 and 7), a few necrotic neurons were observed adjacent to mineralized blood vessels in the cerebellar white matter and internal capsule. In addition, 1 animal from group A (4) and 3 from group B (5, 7, and 8) had small foci of parenchymal necrosis in the interpositus and dentate cerebellar roof nuclei. Neuronal necrosis was characterized by angular and poorly defined borders, chromatolysis, homogeneous and strongly acidophilic cytoplasm and karyorrhectic or pyknotic nuclei (Fig. 4). In horse 4 there was a focal, well-defined, hemorrhagic focus in the anterior cerebellar peduncle close to the vestibular nucleus. In 3 animals from group A (1, 3, 4) and B (5, 7, 8) moderate accumulation of lipofuscin was observed in the cytoplasm of neurons in most brain stem nuclei. The PTAH and Congo red stains were negative in all cases.
Distribution and severity of vascular mineralization and necrosis in the brain of 8 horses.
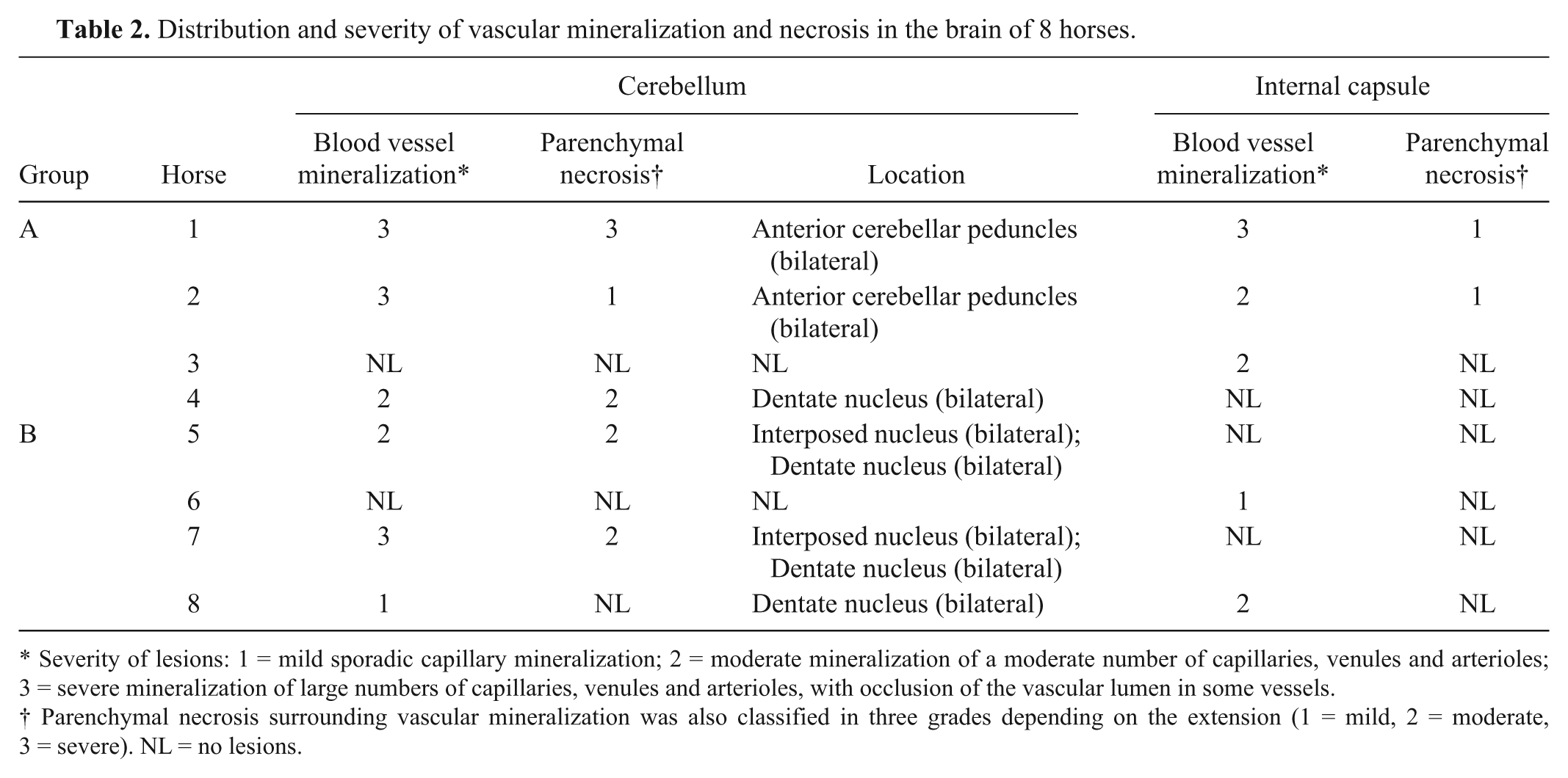
Severity of lesions: 1 = mild sporadic capillary mineralization; 2 = moderate mineralization of a moderate number of capillaries, venules and arterioles; 3 = severe mineralization of large numbers of capillaries, venules and arterioles, with occlusion of the vascular lumen in some vessels.
Parenchymal necrosis surrounding vascular mineralization was also classified in three grades depending on the extension (1 = mild, 2 = moderate, 3 = severe). NL = no lesions.
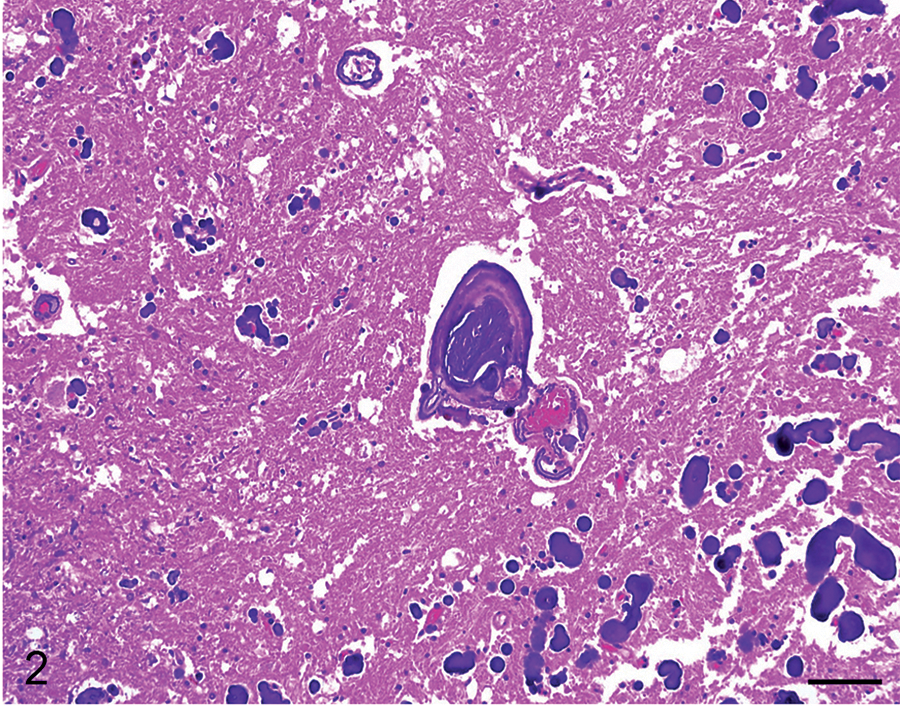
Cerebellum; horse 1. In the periphery of the malacic focus, the wall of small blood vessels and capillaries show variable mineralization, sometimes occluding the vascular lumen (grade 3 of vascular mineralization). Hematoxylin and eosin. Bar = 100 µm.
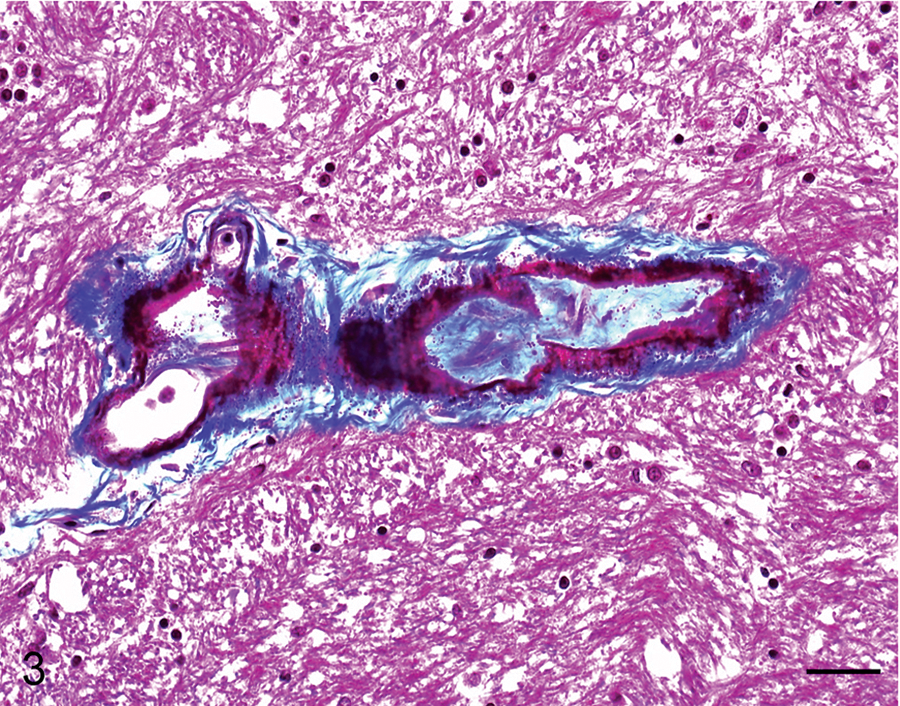
Cerebellum; horse 8. Some of the mineralized blood vessels show deposition of fibrillar and concentric collagenous material with a few spindle cells in the subintimal space. Masson trichrome. Bar = 50 µm.
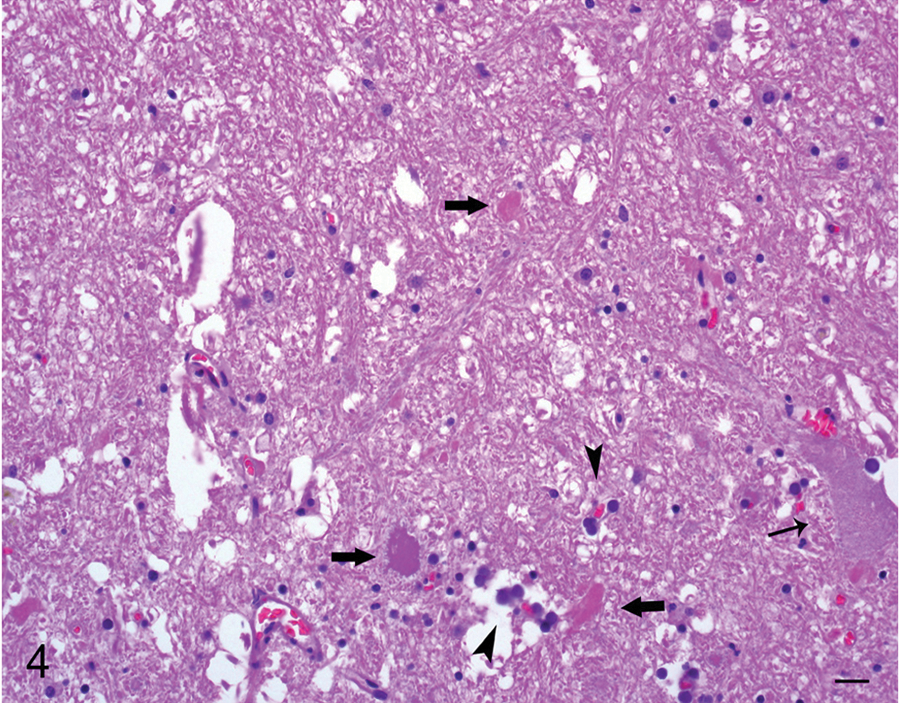
Cerebellum; horse 1. Adjacent to malacic foci, a few necrotic neurons with angular and poorly defined borders, shrunken and strongly acidophilic cytoplasm, and karyolitic nuclei are observed (thick arrows) intermingled with mineralized capillaries (arrowheads) and viable, nonaffected neurons (thin arrow). Hematoxylin and eosin. Bar = 20 µm.
The severity of blood vessel mineralization and parenchymal necrosis in each location were averaged for groups A and B and then a total average for each lesion per group was calculated. The average of lesion severity for blood vessel mineralization in groups A and B was 1.8 and 0.9, respectively (2.1 and 1.3 in the cerebellar peduncles, 1.5 and 0.5 in the internal capsule). For parenchymal necrosis, the averages were 1 and 0.5, respectively (1.5 and 1 in cerebellar peduncles, 0.5 and 0 in internal capsule).
In addition to the changes of mineralization in the brain, the spinal cord mass of horse 5 was diagnosed histologically as a hemangiosarcoma. This horse also had an adenoma of the pars intermedia in the pituitary gland. Horse 6 presented degenerative histological lesions consistent with compressive myelopathy in the spinal cord at C3–C5 level. No other significant histological abnormalities were observed in any other tissue of these horses examined. Immunohistochemistry, FAT for rabies, serological tests, PCR, bacterial cultures, virus isolation and toxicological analyses were negative or unremarkable in all the animals.
Brain vascular mineralization in horses was already described with similar anatomical locations but with minimal or no inflammatory or glial reaction.12,14 Rarely, areas of ischemic necrosis with neovascularization were reported close to mineralized vessels.1,11,12 In the present study, malacic areas associated with siderocalcinosis were observed in both groups of horses. The gradual deposition of concentric layers of mineral salts could have produced a slow vascular occlusion with consequent ischemic damage of surrounding nervous parenchyma and minimal inflammatory or glial reaction. It is noteworthy to stress that inflammation was minimal in all cases. It is possible that this was consequence of slow progression of the lesions.
The localization, cause, and pathogenesis of brain vascular mineralization in horses are poorly understood. Similar mineralization has been observed in the basal ganglia of human beings. In human beings, these lesions have been associated with age and a great variety of factors, including, among others, abnormal calcium metabolism, neuropsychiatric and neurodegenerative diseases, familial syndromes and infective, and inflammatory, toxic, and hypoxic disorders. 5 Brain mineralization in children and adult human beings has also been associated with dysregulation of several bone growth and ossification genes. 9 High levels of these minerals found in basal ganglia may be a consequence of anoxia or a microvasculopathy. 3 However, although vascular degenerative changes and mineralization have been described in the brain of ageing horses, it was not clear if these were age related or associated with a specific dysfunction in calcium metabolism. 1 The cause of mineralization of the brain in the horses in the current study could not be determined.
The clinical significance of the brain changes in the horses in the present study remains undetermined. In human beings, basal ganglia calcification appears to be an incidental finding in most cases, but in a small proportion of patients it has been associated with clinical manifestations. 5 In horses and cows, vascular mineralization in the corpus striatum and cerebellar white matter has been traditionally considered an age-related incidental finding without clinic significance.2,6,14 In the current study, however, the severity of lesions in the 4 horses with clinical neurological signs of undetermined etiology (group A) was higher than in horses in which the etiology was determined or in the 2 animals without neurological clinical abnormalities (group B). It is therefore tempting to speculate that the siderocalcinosis of the brain was responsible for at least some of the clinical signs observed in the animals with clinical neurological signs of undetermined etiology. Hence, it can be hypothesized that chronic progression of this vascular mineralization and associated malacic lesions were compensated by the rest of unaffected CNS until a certain threshold was crossed and the (sudden) appearance of the neurological signs was triggered. In addition, some of the neurological symptoms reported in these horses could be associated with the location of vascular lesions. The cerebellum coordinates all somatic motor activity, and the cerebellar peduncles receive afferent sensory fibers from the muscles and generate efferent fibers to the command centers of the pyramidal and extrapyramidal systems. Also, fibers originating in the cerebellar roof nuclei (i.e., dentate nucleus and interpositus) link these nuclei with the thalamus and red nucleus. Therefore, lesions located in these areas could produce a “pontocerebellar” syndrome due to the interference with feedback pathways between the cerebellum and the higher motor neurons. 7 Pontocerebellar syndrome is usually characterized by dysmetria, tremor, and hypotonia, 2 of which (ataxia/dysmetria and tremors) were observed in the horses in the current study. It is important to stress, however, that the descriptions of the clinical signs for the horses in the present study were provided by the submitting veterinarians, none of whom was a neurologist. The interpretation of a possible correlation between clinical signs and anatomical location of the lesions should therefore be considered in light of these facts.
The present study provides a new insight about the pathological implications of siderocalcinosis in the brain of horses, a lesion usually considered age-related and of nonclinical significance. Severe cerebellar malacic lesions and possible related neurological signs in a low number of aged horses are described. Further research is needed, including a larger number of animals, with exhaustive neurological clinical examination and postmortem studies, to determinate the pathogenesis and possible clinical consequences of this lesion.
Footnotes
Acknowledgements
The authors are grateful to Drs. Rick Allen from San Bernardino Public Health Laboratory (rabies testing), and Michelle Davidson (serology), Beate Crossly (PCR), Robert Poppenga, and Birgit Puschner (toxicology) from California Animal Health and Food Safety Davis, for their analyses and technical support. The authors thank R. Higgins from University of California, Davis for his critical review of the article. Virus isolation was conducted at the Animal Health Diagnostic Center at Cornell. The authors thank Ms. S. Fitisemanu for her excellent work and patience typing this article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received financial support from California Animal Health and Food Safety Laboratory (California Animal Health and Food Safety, University of California, Davis) and Generalitat de Catalunya (travel grant for Dr. J Martínez).
