Abstract
The goal of this retrospective study was to have a comprehensive picture of the β-hemolytic streptococci of horses including tissue/organ distributions and susceptibility patterns against specific antimicrobials between January 1, 2000 and December 31, 2010. A total of 2,497 β-hemolytic streptococci were isolated from 2,391 cases, of which Streptococcus equi subsp. zooepidemicus was the most frequent isolate (72.0%). Other species isolated were Streptococcus dysgalactia subsp. equisimilis (21.3%), Streptococcus equi subsp. equi (5.8%), and unidentified β-hemolytic streptococci (0.9%). As expected, S. equi was mostly isolated from lymph node abscesses and the respiratory tract in foals and adult horses. Streptococcus equi subsp. zooepidemicus and S. equisimilis were mostly isolated from placenta, fetal tissues, and genital tract of horses; S. zooepidemicus and S. equisimilis were also recovered in significant numbers from a number of other organs including lung, liver, brain, kidney, and joints, indicating a much broader tissue tropism than S. equi. In addition, more than 1 Streptococcus spp. was recovered in 106 cases, indicating the co-existence of these bacteria in some horses. This data also suggested that S. equisimilis is a major bacterial agent of horses, contrary to present knowledge. Based on Kirby-Bauer antimicrobial susceptibility data, streptococci were found to be generally susceptible to cephalothin, erythromycin, nitrofurantoin, penicillin, and ticarcillin and clavulanate. Resistance to antimicrobials has not developed over the years, except for gentamicin and tetracycline against S. equisimilis.
Streptococcus equi subsp. equi, Streptococcus dysgalactia subsp. equisimilis, and Streptococcus equi subsp. zooepidemicus are 3 major β-hemolytic Streptococcus spp. that cause severe and economically important diseases in horses. Streptococcus equi subsp. equi is the causative agent of strangles, a highly contagious infection of the upper respiratory tract and associated with lymph nodes of horses2,22 and a host-restricted pathogen of horses. 11 The guttural pouch is commonly infected during early stages of infection with S. equi, and a small percentage of infected horses may continue to harbor bacteria, presenting a risk of infection for other susceptible horses. 22 Streptococcus equi subsp. zooepidemicus is considered to be a mucosal commensal of the oral cavity, pharynx, and respiratory tract of horses, 22 causes disease as an opportunistic pathogen of the respiratory tract and reproductive tract (endometritis) in horses,21,22 and has been reported as the most common bacteria causing placentitis in mares. 12 This species has also been reported to cause bacteremia, meningitis, and arthritis in human beings.6,7 Streptococcus dysgalactia subsp. equisimilis, on the other hand, was considered to be an infrequent bacterium isolated from horses, 22 and has been reported in horses from aborted placenta, 12 less frequently from abscessed lymph nodes 2 and the upper respiratory system. 14 This species is also an important pathogen of human beings, causing bacteremia, pneumonia, and endocarditis. 3
Even though there are a number of publications regarding β-hemolytic streptococci in horses, comprehensive and exhaustive studies are limited. These publications describe studies either with a limited number of samples or addressing only a single type of organ/system/disease making it difficult to make conclusions regarding which organs and tissues are infected.8,12,14-16,19 In the case of S. equisimilis, information is very limited, and comprehensive epidemiological data is not available. 15 The goal of the present study was to have a large-scale picture of these important horse pathogens in terms of tissue and/or organ distributions and susceptibility patterns against specific antimicrobials from the years 2000 to 2010.
A retrospectively review was conducted on 3,587 cases from which β-hemolytic streptococci were isolated. Of those cases, 2,391 contained full age information of horses, and the isolates from these cases were included in the organ and/or tissue distributions of β-hemolytic streptococci. The antimicrobial susceptibility study contained all the isolates recovered from 3,587 cases, regardless of age information. The cases were submitted to the University of Kentucky Veterinary Diagnostic Laboratory (Lexington, Kentucky) from equine fetuses, foals, and adult horses between January 1, 2000 and December 31, 2010. During routine necropsy of fetal cases, lung, liver, stomach content, and placenta were the usual specimens harvested and cultured. Aborted fetuses and 1-day-old newborns were considered as fetus. Animals from 2 days to 2 years old were classified as foals. Any horse 2 years or older was classified as an adult horse. In foal and adult horse necropsy cases, lung, liver, intestines, and any other organ and/or tissue affected were cultured as a separate specimen based on pathologists’ recommendations. The remaining cases were submitted by veterinary practitioners from clinical cases and included nasal swabs and washes, guttural pouch and nasopharyngeal swabs, lower respiratory system (trachea, lung, pleura, and thorax cavity), lymph node abscesses, liver, abdominal area (lymph nodes and abdominal swabs), kidney, brain, intestine, umbilical cord, spleen, joints, wounds, milk, bladder, cornea, bone, heart, implants, incision site tissue, muscle, scrotum, testicle, thymus, tendon, skin, and udder. Swabs from the genital tract (vagina, cervix, and uterus) were submitted for pre-breeding screening or from mares with clinical uterine infections.
For the isolation of bacteria, specimens were cultured on blood agar and eosin methylene blue agar plates at 37°C in 5–10% CO2, for a minimum of 24 hr. If the specimen was from a likely contaminated site such as placenta and nasal swabs, a Columbia colistin and nalidixic acid (CNA) plate with blood was also inoculated. The plates were examined for pathogenic bacteria and were incubated for an additional 24 hr at 37°C in aerobic incubators and examined again for pathogenic bacteria. The criteria used for reporting a microorganism was the isolation of the microorganism in pure culture or significant numbers from specimens (as the predominate microorganism), as previously described. 12 Beta-hemolytic streptococci isolates were identified by using colony morphology, dark-field examination, β-hemolysis on the blood agar and CNA plates, and conventional biochemical tests, including catalase, litmus milk, lactose, maltose, sorbitol, sucrose, and trehalose. Any unidentified β-hemolytic streptococcus (UBHS) was named as β-hemolytic Streptococcus spp.
Of 2,391 cases analyzed, a total of 2,497 β-hemolytic streptococci isolates were identified (Table 1). In 106 cases, more than 1 Streptococcus spp. was recovered, mostly combination of S. zooepidemicus and S. equisimilis (data not shown). Streptococcus spp. isolated in fetuses, foals, and adult horses were S. zooepidemicus (72.0%), S. equisimilis (21.3%), S. equi (5.8%), and UBHS (0.9%; Table 1).
The distribution of beta-hemolytic streptococci isolates among fetuses, foals, and adult horses between 2000 and 2010.*
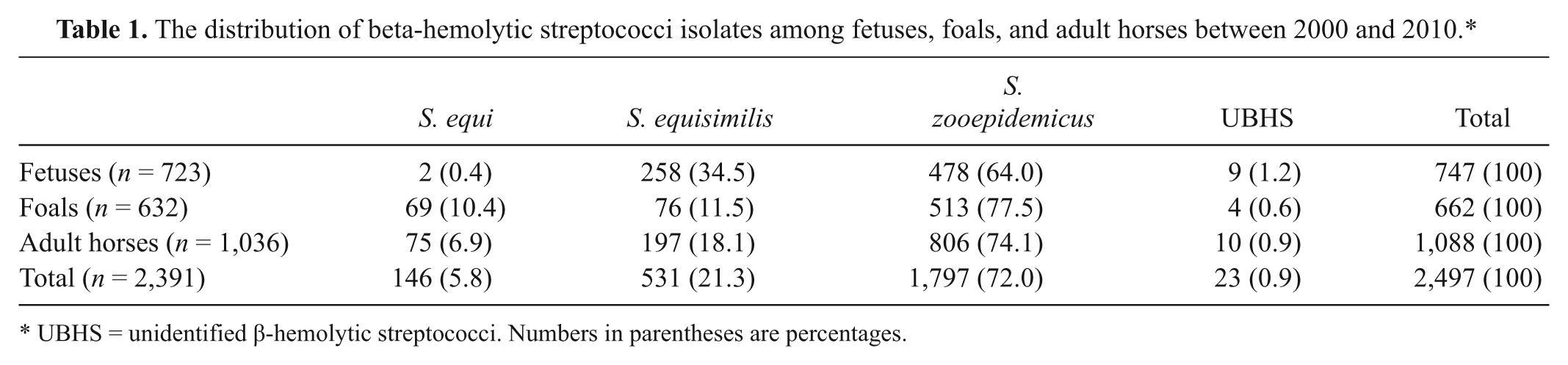
UBHS = unidentified β-hemolytic streptococci. Numbers in parentheses are percentages.
When sites of isolations were examined, placenta was the most productive specimen for isolation of S. zooepidemicus associated with fetuses (Table 2). Lung, stomach content, and liver also contained high number of S. zooepidemicus, indicating the presence of S. zooepidemicus in more than 1 tissue/organ. In foals, S. zooepidemicus were recovered from several organs indicating a broader tissue tropism. In adult horses, the genital tract was the most prominent area to isolate S. zooepidemicus. Streptococcus dysgalactia subsp. equisimilis were recovered considerable in fetuses, foals, and adult horses, with a similar tissue distribution to S. zooepidemicus (Table 2). On the other hand, S. equi were mostly recovered from lymph node abscesses, the lower respiratory system, nasal samples, and guttural pouches. Interestingly, in 2 cases, S. equi isolates were also recovered from fetal tissues and placentas.
Beta-hemolytic streptococci historical data between 2000 and 2010: organism and organ and/or tissue distributions.*
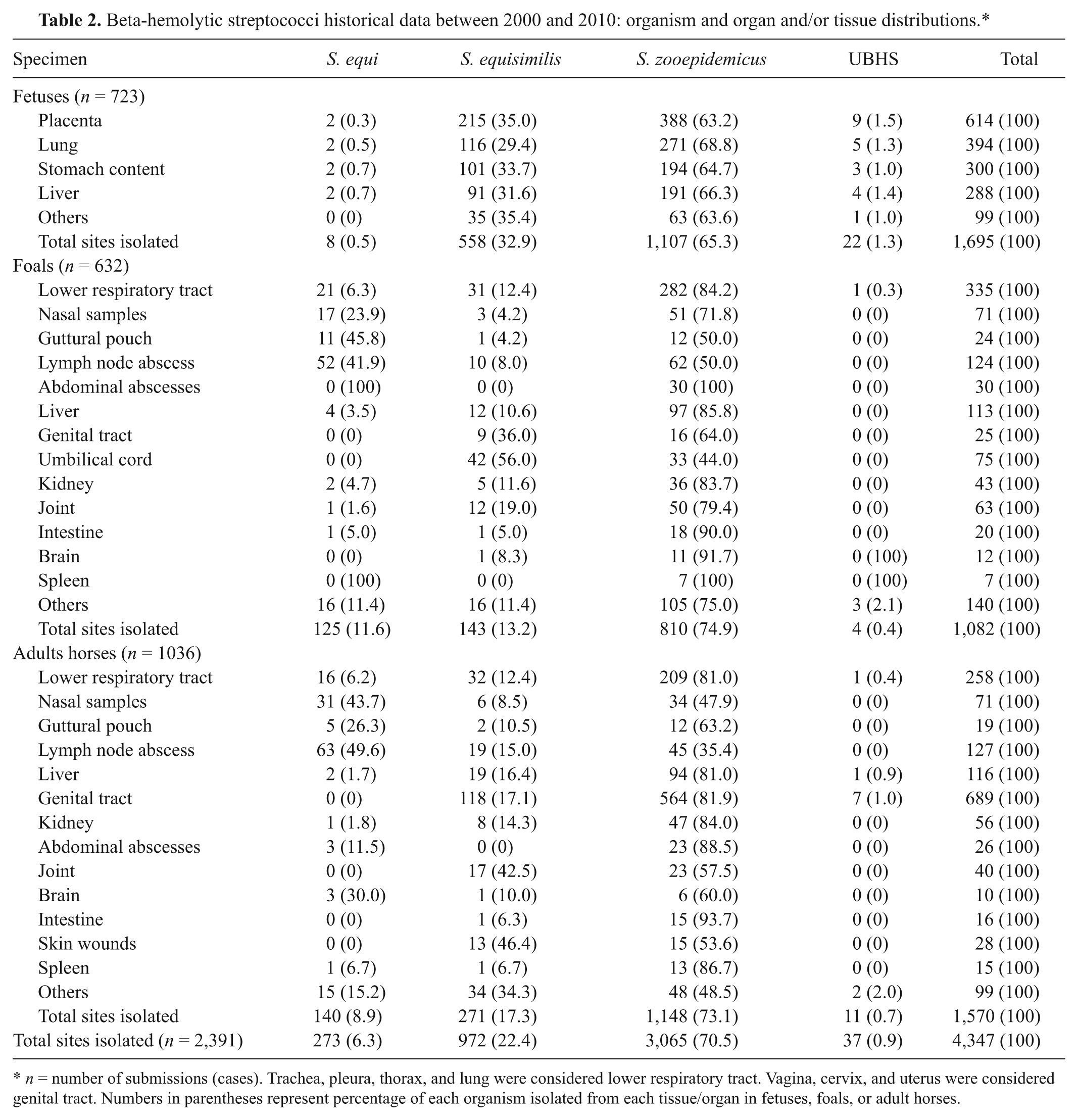
n = number of submissions (cases). Trachea, pleura, thorax, and lung were considered lower respiratory tract. Vagina, cervix, and uterus were considered genital tract. Numbers in parentheses represent percentage of each organism isolated from each tissue/organ in fetuses, foals, or adult horses.
A total of 240 S. equi, 833 S. equisimilis, 2893 S. zooepidemicus, and 34 UBHS isolates were tested for antimicrobial susceptibility by using Kirby–Bauer disk diffusion method (Table 3), including the isolates recovered from the horses that did not have age information. Antimicrobial susceptibility testing was performed following Clinical and Laboratory Standards Institute (CLSI) criteria. Bacitracin, cephalothin, erythromycin, nitrofurantoin, gentamicin, novobiocin, penicillin, sulpha drugs, tetracycline, ticarcillin and clavulanate, and trimethoprim and sulfadiazine were used. Sizes of the zones of inhibition were measured and interpreted as susceptible, intermediate, or resistant. Sizes of zones of susceptible and resistant in millimeters were as follows: bacitracin (≥13, ≤8), cephalothin (≥18, ≤14), erythromycin (≥21, ≤15), nitrofurantoin (≥17, ≤14), gentamicin (≥15, ≤12), novobiocin (≥17, ≤14) penicillin (≥28, ≤19), sulpha drugs (≥17, ≤12), tetracycline (≥23, ≤18), ticarcillin and clavulanate (≥15, ≤14), and trimethoprim and sulfamethoxazole (≥16, ≤10). Any sizes between these values were reported as intermediate. Antimicrobial susceptibilities of Escherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27853), Staphylococcus aureus (ATCC 25923), and Streptococcus pneumoniae (ATCC 49619) were performed weekly as quality control for the antimicrobial susceptibility testing method per CLSI. Mantel–Haenszel chi-square statistical method was used to detect any antimicrobial resistance over the 11-year period.
Beta-hemolytic streptococci historical data between 2000 and 2010: organism and antimicrobial susceptibility distribution (%).*
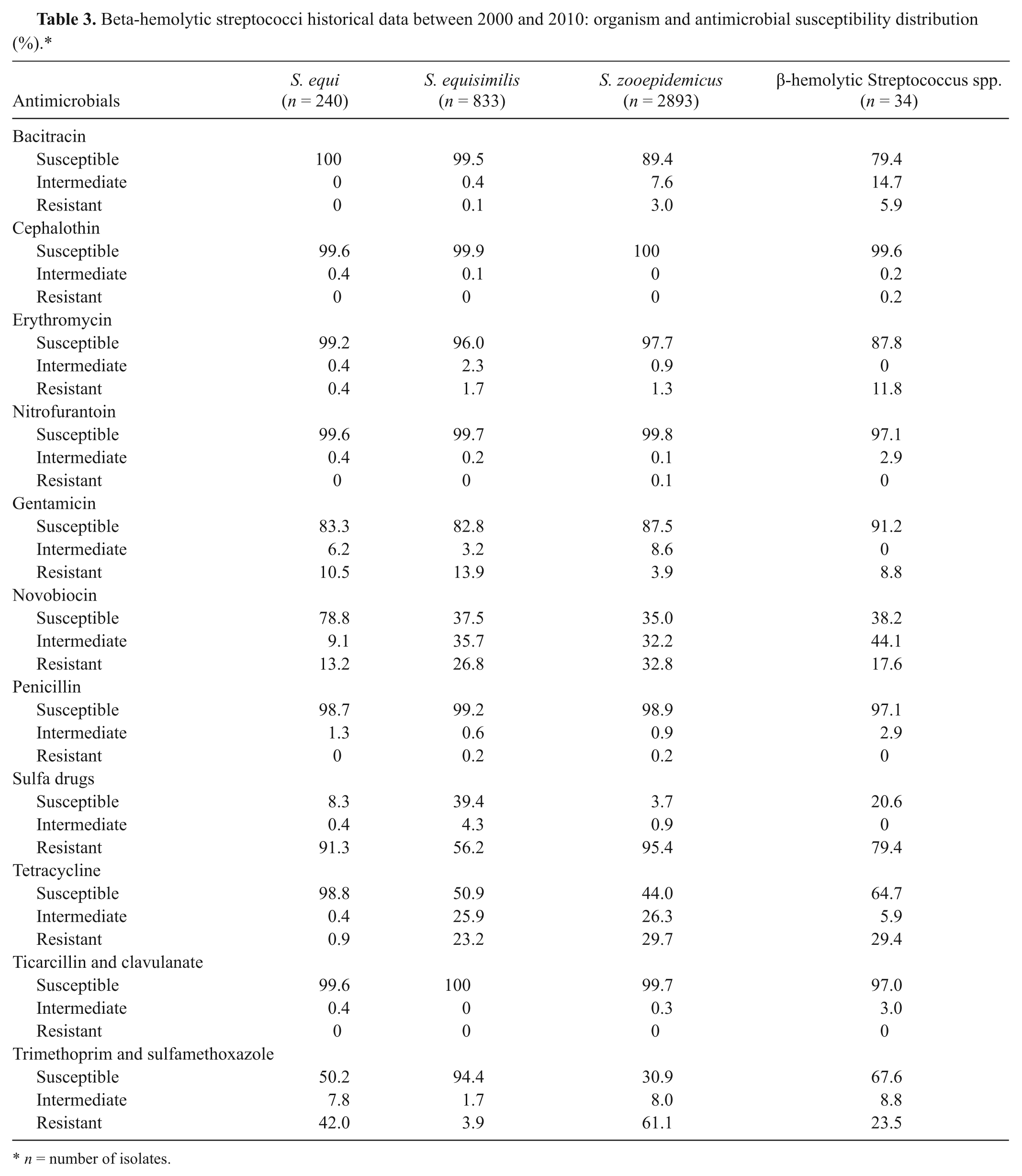
n = number of isolates.
In general, streptococci were susceptible to cephalothin, erythromycin, nitrofurantoin, penicillin, and ticarcillin and clavulanate (Table 3). Gentamicin, tetracycline, novobiocin, and bacitracin showed lesser percentage of susceptibility against streptococci. The resistance to gentamicin and tetracycline for S. equisimilis developed over the years (P < 0.0001 and P < 0.0002, respectively; Table 4). Resistance to other antimicrobials for S. equisimilis and, resistance to all antimicrobials against S. equi, S. zooepidemicus, and UBHS did not develop over the years (data not shown).
Development of gentamicin and tetracycline resistance for S. equisimilis isolates recovered from horses between 2000 and 2010.
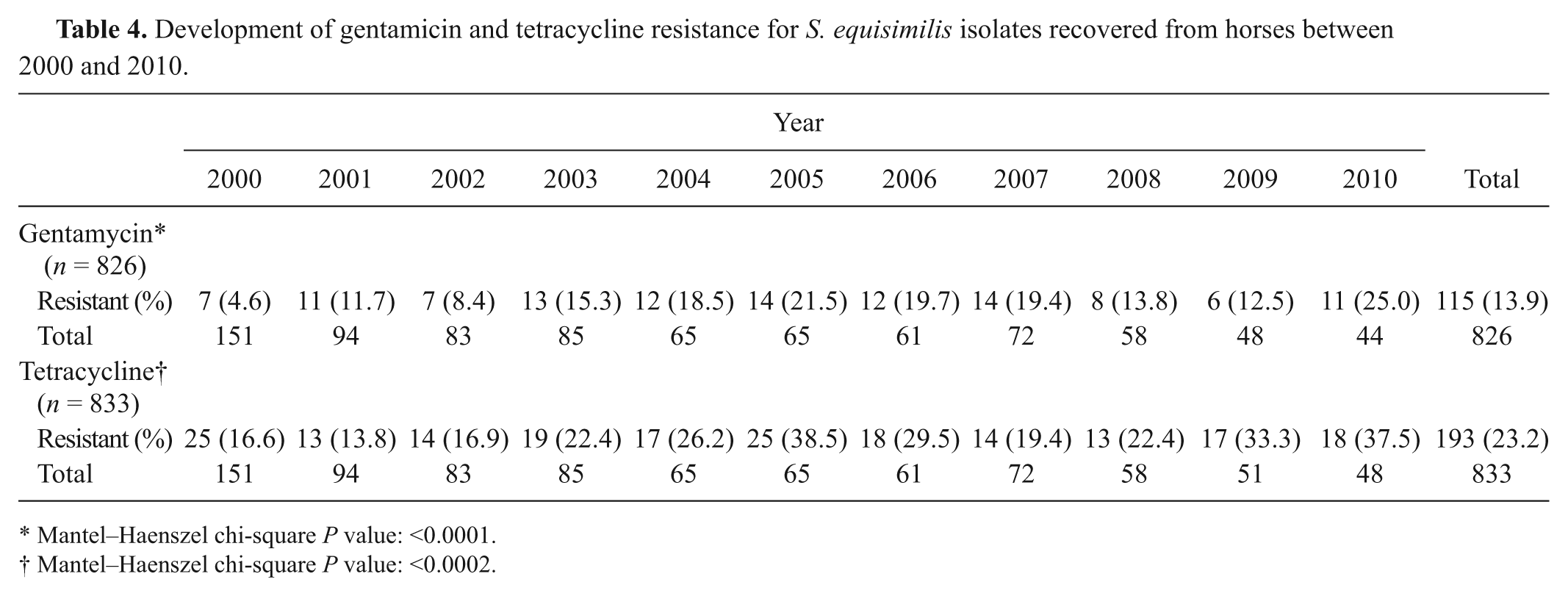
Mantel–Haenszel chi-square P value: <0.0001.
Mantel–Haenszel chi-square P value: <0.0002.
The current retrospective study of β-hemolytic Streptococcus spp. in horses in terms of tissue and/or organ distributions and susceptibility patterns against specific antimicrobials from 2000 to 2010 was extensive, involving 2,391 cases from which 2,497 Streptococcus isolates were recovered. Interestingly in 106 cases, more than 1 Streptococcus spp. was isolated. This information could be important to ascertain how different species may play a role or interact in pathogenesis. Currently, it is not known if the presence of multiple streptococci in a particular animal (or a single tissue) represents a super infection or if a particular species plays the primary role and the other one is just a secondary infection, part of the normal flora or contamination. The co-existence of different streptococci in the same host may facilitate lateral gene transfer which has an important role in the generation of genetic diversity within streptococci. 5
As expected, S. equi was seen almost exclusively only in foals and adult horses. Streptococcus equi subsp. equi was mostly recovered from lymph node abscesses, the lower respiratory system, nasal samples, and guttural pouches in foals. This is in agreement with the existing scientific literature.2,9,21 In adults, the frequency of S. equi in guttural pouches was lower (only 5 cases), but remained as a major isolate not only in lymph node abscesses but also in the upper and lower respiratory tract. Interestingly, S. equi was isolated in 2 abortion cases, which appears to be a novel finding. It is known that strangles is an invasive type infection and it is likely that these 2 cases were the results of severe strangles infections.
In the present study, S. zooepidemicus was the most frequently isolated β-hemolytic streptococci, confirming previous studies.4,22 Streptococcus equi subsp. zooepidemicus was mostly recovered from fetus, placenta, and genital system of horses, which suggests that this organism causes reproductive system diseases, as reported earlier.4,8,12,22 However, S. zooepidemicus was also isolated considerably from lymph nodes, respiratory system, joints, and other organs of foals and adult horses, indicating this organism has a much broad tissue tropism. Streptococcus zooepidemicus is considered as an opportunistic bacteria and present in upper and lower respiratory system and guttural pouches in healthy horses. This may explain opportunistic infections when animals are exposed to stressful conditions. 22
Interestingly, there was very little information regarding S. equisimilis in the peer-reviewed scientific literature. The taxonomic nomenclature of this organism has changed several times due to frequent isolation from human specimens and the animal species of origin. 23 This agent causes several clinical syndromes in human beings including bacteremia, arthritis, osteomyelitis, pleuropulmonary infections, peritonitis, intra-abdominal and epidural abscesses, meningitis, endocarditis, puerperal septicemia, neonatal infections, necrotizing fascitis, myositis, and streptococcal toxic-like syndrome.3,17 Early studies on S. equisimilis in horses raised very little concern, except in the case of uterine abscesses. 9 Later, S. equisimilis was reported to be an infrequent cause of lymphadenitis and placentitis 22 in horses. In a retrospective study to better characterize the etiology and pathology of equine placentitis, it was demonstrated that S. equisimilis is the seventh most common microorganism isolated from equine placentitis cases (4.7% of the total isolates). 12 A study published in 2007 described the isolation of this bacterium from horses with chronic upper respiratory disease 14 and from nasopharyngeal swabs. 15 All of these publications were based on either a limited number of isolates or addressing only a single system, which did not paint a full picture of S. equisimilis. Furthermore, many laboratories do not differentiate β-hemolytic streptococci. This might explain why there is not very much information in the scientific literature.
A novel finding in the present study that extends the existing scientific body of knowledge18,22 was that S. equisimilis was the second most frequent isolate, representing 21.3% of all cases. The higher number of isolations in placenta than other fetal tissues (for both S. zooepidemicus and S. equisimilis; Table 2) strongly suggests that both S. zooepidemicus and S. equisimilis infections originate from placenta and may cause ascending infections. It is interesting to note that, out of 972 sites where S. equisimilis was recovered, fetal tissues (558), umbilical cord (42) and genital tracts of both foals and adult horses (127) comprise 74.8% of total sites isolated for S. equisimilis. This data strongly suggests that S. equisimilis is mostly a reproductive system agent. There is only limited information available on the virulence factors of S. equisimilis, and the pathogenesis of S. equisimilis in horses is poorly understood. 22 However, the results of the present study show that both S. equisimilis and S. zooepidemicus have an ability to invade sterile organs such as brain, kidney, and joints suggesting they have similar pathogenesis and tissue tropism. The newly sequenced whole genome of S. equisimilis (GenBank accession no. 486410 ) and S. zooepidemicus 1 will accelerate the understanding of virulence factors and pathogenesis of these organisms in horses.
The antimicrobial susceptibility testing results of the present study revealed that cephalothin, erythromycin, nitrofurantoin, penicillin, and ticarcillin and clavulanate are generally efficacious against a number of streptococci, in agreement with previous studies.2,20 Practitioners can employ these products in their treatment regimens with a high level of confidence prior to the availability of antibiotic sensitivity test results. Finally, susceptibility patterns did not change over the years except for S. equisimilis against gentamicin and tetracycline, indicating that limited drug resistance has developed. It is not known why resistance has developed for only gentamicin and tetracycline against S. equisimilis. In an earlier study, 20 an increase for S. zooepidemicus (n = 13 isolates) was reported but resistance to any other antimicrobials was not reported. In conclusion, the current study showed that 1) S. equisimilis was a major β-hemolytic streptococci of horses, contrary to present knowledge; 2) S. zooepidemicus and S. equisimilis were isolated mostly in fetuses and genital system of adult horses and had a broad and similar tissue tropism; 3) more than 1 species of β-hemolytic streptococci could be present in some of horses; and 4) gentamicin and tetracycline resistance was observed against S. equisimilis over the years.
Footnotes
Acknowledgements
The authors thank Drs. Mike Donahue and Alan Loynachan for critical review of the manuscript, Jackie Smith and Anthony Cattoi for the extraction of data from UKVDL electronic archives, and Xia Yu for statistical analysis. This manuscript was given permission by Associate Dean for Research, University of Kentucky, College of Agriculture (11-15-076).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
