Abstract
Cryptococcal meningitis and encephalitis is reported in a 5-year-old bull. The clinical signs included multifocal neurological deficits manifested by hypermetria, ataxia, depression, circling, impaired vision, head pressing, low head carriage, wide-based stance, and falling to the side or backwards. At necropsy, 6 cavities with a gelatinous content were observed in the brain and brain stem, and the meninges were thickened and off white. Numerous yeasts, with a nonstaining capsule, and associated mild inflammatory reaction were observed histologically. Specimens for culture were not collected at necropsy, but the identity of the yeast in tissue sections as Cryptococcus neoformans var. grubii was supported by immunohistochemistry.
Cryptococcosis is a granulomatous disease of domestic and wild animals as well as human beings, and is caused by 2 fungal species: Cryptococcus neoformans, with the varieties grubii (serotype A) and neoformans (serotype D); and Cryptococcus gattii (serotypes B and C). 2 Cryptococcus neoformans is commonly isolated from avian excreta, particularly that of pigeons, occurs worldwide, and primarily infects immunocompromised patients. 2 Cryptococcus gattii primarily mainly infects otherwise healthy persons and was widely regarded to be restricted to subtropical regions; however, it has also been isolated in Canada, 20 the Pacific Northwest region of the United States, 10 and Europe, 21 thus showing that it can also occur in temperate climates.
The classical route of inoculation of cryptococcosis is respiratory, by the inhalation of infectious particles. In human beings, the agent can cause symptomatic or asymptomatic pulmonary infection followed by invasion of the central nervous system by the blood (King JW, DeWitt ML: 2007, Cryptococcosis. Available at http://emedicine.medscape.com/article/215354-overview. Accessed April 18, 2010). 2 Although both species infect the central nervous system, C. gattii appears to invade the brain parenchyma more commonly than C. neoformans (King JW, DeWitt ML: 2007, Cryptococcosis). 2 Furthermore, in C. gattii infections, pulmonary lesions are more likely than in C. neoformans infections. 2 The infection can disseminate to include other organs causing cutaneous manifestations, osteomyelitis, septic arthritis, chorioretinitis, myocarditis, hepatitis, peritonitis, renal abscesses, prostatitis, myositis, and gastroenteritis (King JW, DeWitt ML: 2007, Cryptococcosis).
Cryptococcosis in animals can be due to both C. gattii and C. neoformans.1,3,10,20 It is common in cats and less frequently in dogs, causing rhinitis, skin lesions mainly of the nose and head, meningitis, ocular lesions, and systemic disease. 3 In horses, cryptococcosis may cause rhinitis, 14 pneumonia, 13 meningitis, 19 encephalitis, 4 abortion with mycotic placentitis, and fetal pneumonia, 15 and disseminated infection. 9 Cryptococcosis of the skin is reported in a donkey. 6 In goats, cryptococcosis may cause mastitis, 18 pulmonary infection and meningoencephalitis 1 ; in cattle, mastitis 11 and pulmonary infections 5 ; in sheep, mastitis 16 and rhinitis 17 ; and in water buffalo, 12 the only form of cryptococcosis reported is mastitis.
The aim of the current work is to report a case of bovine cryptococcal meningoencephalitis. The disease affected a 5-year-old Brown Swiss bull from a dairy farm in the semiarid region of the state of Paraíba in northeastern Brazil. Clinical signs were hypermetria, ataxia, depression, circling, impaired vision, head pressing, low head carriage, and wide-based stance. Falling to the side or backwards was induced by the head rising test (raising the head of the animal for approximately 60 sec and then suddenly releasing it). The farmer reported that the animal had been affected for several days. Eight days after its arrival at the Veterinary Hospital (Patos, Paraíba, Brazil), the bull became recumbent and was euthanized.
At necropsy, the meninges of the brain stem and cerebellum were thickened and off white with a cloudy appearance. The brain was asymmetric with the left hemisphere increased in volume. The cerebellum was compressed and displaced to the right. The whole brain was fixed in 10% formalin. After fixation, coronal sections of the brain were asymmetric showing cystic cavitations with viscous, gelatinous, sometimes translucent content. A spongy granular tissue was observed in the border of the lesions or within the lesions. Six cavities were observed. Lesion 1 (Fig. 1A), measuring 3.5 cm in its largest diameter and 1.5 cm craniocaudal, was located in the left temporal and piriform lobes. The rostral part of the lesion was compressing the thalamus and extended to the level of the basal nuclei. Lesion 2 (Fig. 1A) was located in the right parietal lobe. It extended to the cingulate gyrus and compressed the hippocampus, and measured 3 cm in diameter and 2.5 cm craniocaudal. Lesion 3 (Fig. 1A, 1D), with a diameter of 2 cm and measuring 1.5 cm craniocaudal, was located in the right occipital lobe. Lesion 4 (Fig. 1A) was located in the mesencephalon, above the mesencephalic aqueduct, and was 1 cm in diameter and 1 cm craniocaudal. Lesions 5 and 6 (Fig 1C) were similar, located in the right and left frontal lobes, respectively, and measuring 0.9 cm and 1 cm in diameter. Lesion 5 communicated with the meninges.
A spongy granular tissue was observed over the ependymal epithelium of the rostral ventral horn of the left lateral ventricle (Fig. 1B). A similar, but smaller lesion was observed in the right lateral ventricle (Fig. 1B). Single raised rough plaques with a diameter of approximately 0.5 cm × 1 cm were observed in the lateral wall of both lateral ventricles.
Samples of the central nervous system and other tissues were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 4–6 µm, and stained with hematoxylin and eosin. Selected sections were also stained with periodic acid–Schiff and alcian blue. Upon histologic examination, the cavities within the nervous tissue appeared spongy with nearly absent inflammatory reaction (Fig. 2A, 2B). The most prominent lesion was the presence of numerous yeasts with a nonstaining capsule giving the classic “soap bubble” appearance to the lesion (Fig. 2). These structures were also observed in the meninges (Fig. 2B) and ependyma. Occasionally, a fibrillar material was observed around the yeasts (Fig. 2C). Very few mononuclear cells were observed within the lesions (Fig. 2A, 2B). Perivascular cuffing of lymphocytes, plasma cells, and macrophages was occasionally observed in the nervous tissue around the lesions or in the meninges and ependyma. No significant lesions were observed in the lung, nasal cavity, and other tissues.
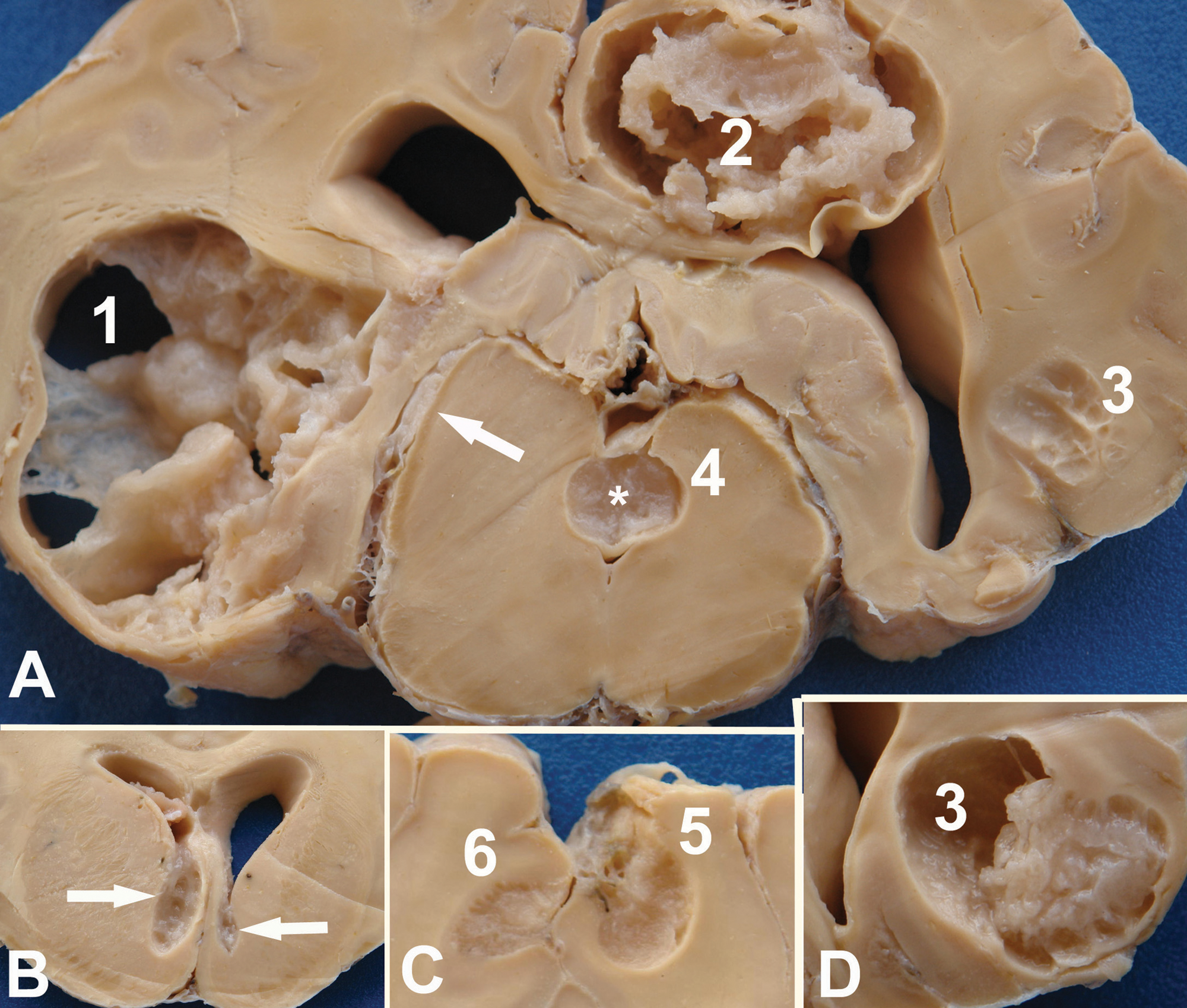
Bovine; brain, coronal section.
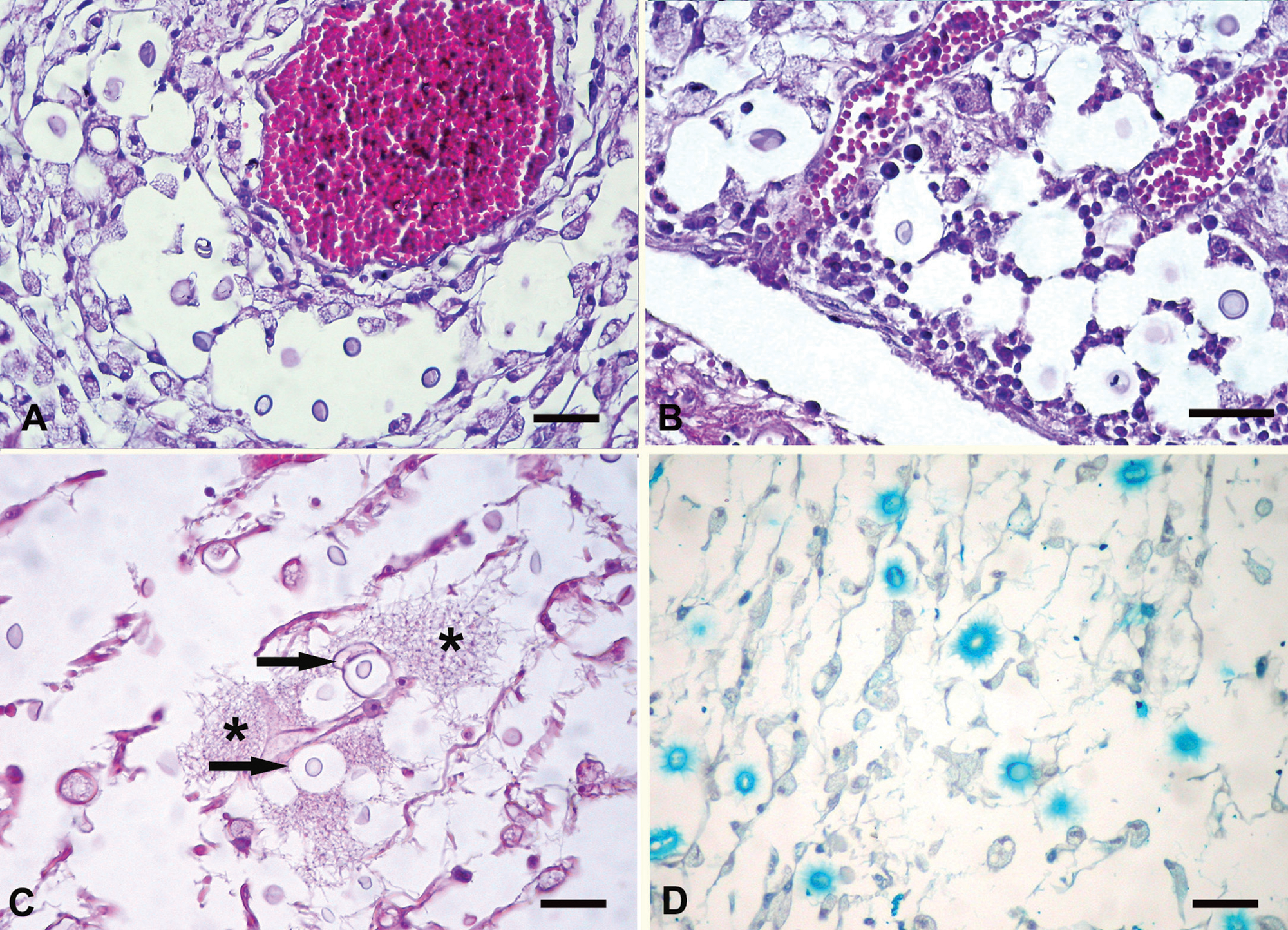
Bovine. Brain (
Immunohistochemical staining of brain sections was performed by a method reported previously 8 to determine Cryptococcus species using mAbs 471, 302, and F10F5. 7 Briefly, the method utilized 5-µm sections cut onto silane-coated slides and microwave antigen retrieval by boiling in antigen retrieval buffer a for 5 min. A 3% peroxidase block was cooled slowly to room temperature, then incubated for 60 min with primary antibodies, 60 min with secondary anti-mouse antibody, followed by 30 min with streptavidin–biotin link, b followed by 5 min incubation with 3,3′-diaminobenzidine chromogen. a The immunohistochemical labeling was performed in an autostainer. a Strong positive labeling of cryptococcal capsule within the brain sections was evident with mAbs 471, 302, and F10F5. c Cryptococcal capsule was not labeled with negative antibody isotype control (immunoglobulin G1) or in other negative antibody controls. The specimens acted as their own positive tissue controls. The specificity of the anti-cryptococcal capsular antibodies has previously been described.7,8 Briefly, mAb 471 is an anti-capsular antibody that positively labels all members of the C. neoformans–C. gattii species complex, mAb 302 positively labels C. neoformans var. grubii and C. neoformans var. neoformans, while mAb F10F5 positively labels C. neoformans var. grubii and C. gattii (serotype B; T. Kozel, personal communication, 2005). The findings were consistent with an identification of C. neoformans var. grubii.
The immunohistochemical panel, as with all such panels, is reliant on the specificity of the antibodies and the capacity to retrieve antigenic sites. A 2010 study has demonstrated that there is a proportion of C. gattii molecular type VGII that are inappropriately, weakly, and nonspecifically labeled with mAb 302. 8 In the current study, labeling with mAb 302 was very strong, and, therefore, interpreted as positive specific labeling.
Clinical signs were of a widespread multifocal disease of the brain, which was confirmed at necropsy. Lesions located in the cerebrum were responsible for signs of depression, circling, impaired vision, and head carriage. Ataxia was probably due to the lesion in the mesencephalon or to the compression of the brain stem by the cerebral lesions. Signs of wide-based stance, hypermetria, and falling after the head rising test were due to cerebellar compression. The mild inflammatory reaction on histologic examination suggests that clinical signs are due to compression of the brain tissues by the lesions.
In the bovine case presented herein, location of lesions around blood vessels in the brain tissue suggests that meningitis and ependymitis are probably the initial lesions on the nervous system, followed by invasion of the brain throughout the Virchow–Robin space. On histologic examination, the most striking feature was the absence of a consistent inflammatory reaction within and around the lesions, suggesting an absence of immunologic reaction of the host to the yeast. A cryptococcal polysaccharide capsule has antiphagocytic properties and may be immunosuppressive. The antiphagocytic properties of the capsule block recognition of the yeast by phagocytes and inhibit leukocyte migration into the area of fungal replication (King JW, DeWitt ML: 2007, Cryptococcosis). Another possibility is that the absence of significant inflammatory reaction was due to an immunocompromised host; however, there is no other evidence to support this. Damage of the nervous tissue was likely due to tissue distortion secondary to the expanding fungal burden. In human beings, extensive inflammation or fibrosis is also rare. The characteristic lesion of cryptococcosis consists of a cystic cluster of yeast with no well-defined inflammatory response. Well-formed granulomas are generally absent (King JW, DeWitt ML: 2007, Cryptococcosis).
In the current case, it is not possible to speculate further about the pathogenesis and relationship of this case to the global epidemiology of cryptococcosis caused by the 8 molecular types of the C. neoformans species complex. 2 Because of the type of specimens available, immunohistochemistry was used for identification of the fungus causing disease in the bull in the present study. While the method clearly confirmed the identification of the infecting fungus as a member of the C. neoformans species complex, and the strong labeling with the antibodies utilized was consistent with the infecting strain being C. neoformans var. grubii based on the known specificities of these antibodies, the specimens available were not suitable for culture or molecular typing and so further confirmation of this identification could not be made.
Footnotes
a.
Dako Denmark A/S, Glostrup, Denmark.
b.
LSAB+ System HRP K0690, Dako Denmark A/S, Glostrup, Denmark.
c.
Kindly supplied by Prof. Thomas Kozel, Department of Microbiology and Immunology, School of Medicine, University of Nevada, Reno, NV.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
