Abstract
The aims of the current study were to assess whether sodium dodecyl sulphate–agarose gel electrophoresis (SDS-AGE) and high-resolution electrophoresis (HRE) can identify dogs with a urinary protein-to-creatinine ratio (UPC ratio) >0.2 and whether HRE can provide preliminary information about the type of proteinuria, using SDS-AGE as a reference method. HRE and SDS-AGE were conducted on 87 urine samples classified according to the International Renal Interest Society as non-proteinuric (NP; UPC ratio: <0.20; 32/87), borderline proteinuric (BP; UPC ratio: 0.21–0.50; 15/87), or proteinuric (P; UPC ratio: >0.51; 40/87). SDS-AGE and HRE were positive in 14 out of 32 and 3 out of 32 NP samples and in 52 out of 55 and 40 out of 55 samples with a UPC ratio >0.20, respectively. The concordance between HRE or SDS and UPC ratio was comparable (κ = 0.59; κ = 0.55). However, specificity (90%) and positive likelihood ratio (7.76) were higher for HRE than for SDS-AGE (56% and 2.16) while sensitivity was lower (73% vs. 94%). The analysis of HRE results revealed that a percentage of albumin >41.4% and an albumin/α1-globulin ratio (alb/α1 ratio) >1.46 can identify samples classified by SDS-AGE as affected by glomerular proteinuria while a percentage of α1-globulin >40.8% and an alb/α1 ratio <0.84 can identify samples classified by SDS-AGE as affected by tubular proteinuria. In conclusion, both SDS-AGE and HRE could misclassify samples with a UPC ratio higher or lower than 0.20. Therefore, UPC ratio must always be determined before conducting these tests. The percentage of albumin and α1-globulin or the alb/α1 ratio determined by HRE can provide preliminary information about the origin of proteinuria.
Keywords
Introduction
In dogs, a late diagnosis of kidney disease is associated with poor prognosis, since in most cases potentially irreversible damage develops. Proteinuria, in turn, affects the progression of the nephropathy and in some cases, such as in glomerulonephritis, is the first and only tangible sign of the disease. 15 Dogs should be screened for proteinuria as part of the annual wellness visit if they belong to breeds that have a high risk of renal diseases (e.g., Sharpei) or if they live in endemic areas of diseases that could cause glomerular injury (e.g., leishmaniasis). Specifically, “high-risk” dogs should be screened every 6 months. 5,6 An early and accurate recognition of proteinuria is essential to provide appropriate patient care. Therefore, quantitative analysis of urinary proteins is a fundamental step in early diagnosis and subsequent monitoring of renal disease in dogs. Although kidney biopsy is the most accurate and sensitive method to identify the type and localization of kidney damage, in some comparative studies, renal diseases can be suspected by measuring the concentration of single urinary markers such as albumin, C-reactive protein, retinol-binding protein, and N-acetyl-b-D-glucosaminidase. 19 Nevertheless, information on multiple proteins in a single urine sample can be achieved using electrophoretic techniques. Specifically, it was found that sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE), a noninvasive method to localize the origin of urinary proteins based on their molecular weight, provides a diagnostic sensitivity comparable to results obtained by kidney biopsy (Brown JS, Nabity MB, Brock R, et al.: 2010, Comparison of urine sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) with renal histological findings and clinicopathologic data in dogs with renal diseases. Vet Clin Pathol 39:556. Abstract). SDS-PAGE is considered the gold standard method for qualitative analysis of urinary proteins. After the use of SDS, all the proteins are denatured and negatively charged, and as a consequence, they migrate in polyacrylamide gel electrophoresis based on their molecular weight, thus allowing the differentiation of glomerular versus tubular proteinuria. 17 Although the specificity of SDS-PAGE is low (Brown JS et al.: 2010) and its correlation with the severity of renal damage is moderate, 6 SDS-PAGE is considered a very sensitive tool to discriminate between glomerular, tubular, or mixed proteinuria, and it is commonly used to monitor renal patients and to assess therapeutic responses, as in human medicine. In routine practice, however, SDS–agarose gel electrophoresis (SDS-AGE) is preferred to SDS-PAGE for its superior ability to separate large proteins and for its lower toxicity as shown in a previous study. 22
Recently, high-resolution electrophoresis (HRE) has been proposed as an electrophoretic technique to be applied to fluids with low concentration of proteins such as urine or cerebrospinal fluid. 2,3,8,9,12,21 HRE is a zonal electrophoresis in which proteins migrate based on charge, volume, and mass. HRE differs from the standard AGE (the technique routinely used in serum) by the use of a more alkaline buffer (pH 8.6 vs. 8.5), a different barbital content (0.18% vs. 0.61%), and a different voltage per hour (40 V/hr vs. 36 V/hr) at 20°C. These different analytical conditions lead to a better separation of globulins compared with standard electrophoretic techniques. In addition, HRE can be fully automated and is less time-consuming, more standardized, and less expensive than SDS-AGE. Bands on the gel are transformed by the software in peaks corresponding to groups of proteins characterized by similar mass, volume, and weight; the percentage of area under the peaks is calculated by specific software. To the authors’ knowledge, HRE has never been tested on canine urine samples, except for searching monoclonal gammopathies, as in human beings. 8
Both SDS-AGE and HRE can be applied to unconcentrated urine, 4,16 avoiding the possible interference of urine salt particles that induce technical problems when concentrated urine are analyzed 10 if not removed with special procedures. 14 Nevertheless, the 2 methods are based on a different analytical principle: SDS-AGE identifies single proteins, while HRE, as any other zonal electrophoresis, provides rough information on “clusters” of proteins characterized by a peculiar electrophoretic motility. This means that proteins with different size can have the same electrophoretic motility. Just as an example, both transferrin and β2-microglobulin that in SDS-AGE are respectively suggestive of glomerular and tubular damage based on their molecular weight (80 kDa and 12 kDa, respectively) share the same electrophoretic motility in the β-region of HRE. 20 From a practical point of view, however, the purpose of qualitative analysis of proteinuria is to identify glomerular, tubular, or mixed patterns rather than to identify single proteins, except in specific conditions such as multiple myeloma. 12 From this perspective, the detection of glomerular or tubular HRE patterns, independently on the proteins migrating in each fraction, would have a diagnostic utility.
The aims of the current study were 1) to evaluate the analytical and diagnostic performances of 2 different electrophoretic techniques, namely SDS-AGE and HRE, and in particular their ability to provide positive results only in dogs with urinary protein/creatinine ratio higher than 0.2; and 2) to verify whether the type of electrophoretograms obtained with HRE, a quicker and less-expensive method than SDS-AGE, can provide preliminary information about the type of proteinuria.
Material and methods
Samples and pre-processing
The current study was performed on 87 urine clinical samples of privately owned dogs of different age, sex, and breed (42 male, 23 female, and 22 of unknown gender), submitted to the authors’ laboratory for routine check-up visits or for diagnostic purposes. Samples were collected under informed consent by the owners according to the regulation of the Institutional Animal Care Committee (Comitato Etico per la Tutela degli Animali) of the University of Milan (Milan, Italy), and were included in the study irrespective of the availability of information concerning the presence or absence of renal diseases. However, data regarding the concentration of creatinine in plasma and the diseases suspected on a clinical basis were recorded.
All urine samples were collected by ultrasound-guided cystocentesis, using a 5- or 10-ml syringe with a 21-gauge needle, and put into sterile blood collection tubes. a Gross appearance, color, and transparency were immediately recorded after collection, followed, in 40 cases, by the determination of urine specific gravity (USG) by a manual refractometer. b Only samples without macroscopical abnormalities were included in the study.
Examination of the sediment was performed in all samples immediately after sampling or within 24 hr of storage at 4°C, as follows. After centrifugation (160 × g, 10 min) of 5.0 ml of urine, 4.5 ml of the supernatant was transferred in another plain tube to perform biochemical analysis and electrophoresis: specifically, after addition of 1% sodium azide at a rate of 0.1 ml per ml of urine supernatants were frozen at −20°C to be analyzed within 1 month. The pellet was re-suspended in the remaining 0.5 ml of supernatant. One drop of the re-suspended pellet was placed on a glass slide, coverslipped, and examined by light microscopy at 10× and 40× magnification. Only samples with “inactive” sediment (>5 red or white blood cells per high power field, presence of bacteriuria, spermaturia, or crystalluria) were included in the study.
Quantitative analysis of proteinuria
Using an automatic analyzer, c the urinary protein concentration was determined by the pyrogallol method using a commercially available reagent, d while creatinine was measured using the modified Jaffe method on samples diluted 1:20 (v:v). The urinary protein-to-creatinine (UPC) ratio was then calculated with the formula: UPC ratio = protein (mg/dl)/creatinine (mg/dl). Samples were then classified as non-proteinuric (NP; UPC ratio: <0.20), borderline proteinuric (BP; UPC ratio: 0.21–0.50), or proteinuric (P; UPC ratio: >0.51) according to the International Renal Interest Society (IRIS) classification. 5 For further statistical evaluation (see below), BP and P samples were considered as a single group of samples from dogs with a grade of proteinuria that according to the IRIS classification requires additional investigation or clinical intervention (BP + P).
Qualitative analysis of proteinuria
After thawing, samples were processed in a single session with both HRE and SDS-AGE techniques using commercially available kits e,f on automated electrophoretic equipment g with a scanner for the acquisition of gels and with a specific software h for the analysis of the results. Specifically, SDS-AGE was conducted as follows. Each urine sample was mixed (4:1 v/v) with the diluents included in the kit containing SDS and bromophenol blue. The instrument was then prepared by mounting the sponges embedded with the appropriate buffer (pH 8.5) on the electrodes and by placing the gel on the migration chamber after removal of excess buffer. Five ml of urine diluted as above were placed into dug wells of the gel and the procedure was started using the specific program (Proteinuria 1*5). Migration takes place under conditions of constant power (10 W) at 20°C to accumulate 60 Volt per hour (V/hr; approximately 15 min). The gel was then transferred to the staining module and automatically stained with acid violet, washed, treated with glycerin, and dried. Gels were then visually analyzed. Samples with weak or barely visible bands were considered negative, while among the positive samples, the presence of proteins with molecular weight (MW) greater than or equal to that of albumin was considered as glomerular and the presence of proteins with MW less than that of albumin, as tubular. If proteins with higher and lower MW than albumin were present, then proteinuria was considered mixed (M). If albumin was the only visible band, patients were considered as albuminuric.
The details of the HRE were as follows. Ten µl of undiluted urine were placed in each well of the applicator. The applicator was then inserted into the moist storage chamber and left for 5 min to spread the samples in the teeth of the applicator. The instrument was then prepared by mounting the sponges embedded with the appropriate buffer (pH 8.6) on the electrodes and by placing the gel on the migration chamber after removal of excess buffer. The applicator was then removed from the moist storage chamber, placed in the mobile frame in the migration chamber, and the instrument was started using the specific program (7/15HR3). Migrations occurred under conditions of constant voltage (225 V) at 20°C to accumulate 75 V/hr (approximately 10 min). After migrations, the gel was automatically dried at 60°C for 9 min. The gel was then transferred in the staining module and automatically stained with acid violet or amidoschwartz, washed, treated with glycerin, and dried. Gels were then acquired with the scanner, which performed the densitometric analysis and generated the electrophoretograms. Samples were considered as negative when no bands were visible by gross examination or when no uniform peaks were generated during densitometric analysis of scanned gels (see the Results section for description of negative densitograms). In positive samples, the densitometric analysis provided an electrophoretogram for each lane and calculated the percentage of area of the corresponding electrophoretic fractions (albumin, α1-, α2-, β-, and γ-globulin) for each electrophoretic peak.
Statistical analysis
Statistical analyses were performed in an Excel i spreadsheet using the Analyse-it software. j Since one of the aims of the current study was to assess whether SDS-AGE and HRE are able to correctly classify dogs, which, according to the IRIS classification, 5 were borderline proteinuric or proteinuric (UPC ratio: >0.2), BP + P samples were considered as “positive” and samples with UPC <0.2 were considered as “negative.” Results were categorized as follows: true positive (UPC ratio: >0.2) were “positive” samples that showed visible electrophoretic bands; false negative (UPC ratio: >0.2) were “positive” samples that did not show visible electrophoretic bands; true negative (UPC ratio: <0.2) were “negative” samples that did not show visible electrophoretic bands; and false positive (UPC ratio: <0.2) were “negative” samples that showed visible electrophoretic bands. Based on these values, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), accuracy, and positive likelihood ratio (LR+) were calculated.
For SDS-AGE, the Pearson’s χ2 test was used to investigate the presence of differences between the proportion of false-positive and false-negative samples with high and low USG. Moreover, the concordance between the positivity of SDS-AGE and of HRE, as well as between the positivity of each of the 2 tests with the presence of a sufficient amount of urinary protein to classify the dog as BP or P according to the IRIS classification (e.g., UPC ratio: > 0.2), was evaluated by determining the Cohen’s κ coefficient. The degree of concordance was then classified as suggested by a previous study 11 as follows: (κ <0.00: poor; κ = 0.00−0.20: slight; κ = 0.21−0.40: fair; κ = 0.41−0.60: moderate; κ = 0.61−0.80: substantial; κ = 0.81−1.00: almost perfect).
The presence of significant differences between the percentages of each electrophoretic fraction detected by HRE in the 43 proteinuric samples that provided positive bands in both methods and that were classified by SDS-AGE as affected by glomerular, tubular, or mixed proteinuria was assessed using an nonparametric analysis of variance test (Kruskal–Wallis test) followed by the Bonferroni test to assess the differences between groups.
For HRE, the percentage of albumin and α1-globulins and the albumin-to-α1-globulin ratio (alb/α1), were used to design receiver operating characteristic (ROC) curves by plotting the true-positive rate (sensitivity) with that of false positives (1 – specificity) and by calculating the area under the curve (AUC). The ROC curves were also used to identify the cut-off value that maximizes their diagnostic performance for both glomerular and tubular proteinuria (i.e., the value characterized by equally important sensitivity and specificity).
Results
Characteristics of electrophoretic profiles
According to the IRIS classification 5 of 87 dogs, 32 dogs were classified as NP, 15 were classified as BP, and 40 were classified as P. Using this classification system, 55 out of 87 dogs had a grade of proteinuria that requires additional investigation or clinical intervention (BP + P). The plasma creatinine concentration was within the reference interval of the laboratory (<1.5 mg/dl) in 31 NP dogs, 11 BP dogs, and 10 P dogs, slightly increased (1.5−3.0 mg/dl) in 1 NP dog, 2 BP dogs, and 7 P dogs, moderately increased (3.0−5.0 mg/dl) in 2 BP dogs and 6 P dogs, and severely increased (>5.0 mg/dl) in 17 P dogs. Non-proteinuric dogs were clinically healthy or had pathophysiological conditions not commonly associated with proteinuria (i.e., gastrointestinal, neurological, orthopedic disorders, inflammation, and neoplasia). Leishmaniasis, glomerulonephritis of different type and severity, metabolic diseases (Cushing’s disease, diabetes mellitus), and/or renal amyloidosis were the diseases most frequently diagnosed in BP or P dogs. No significant differences (P = 0.254) in the UPC ratio was recorded in male dogs (0.87 ± 1.02; median: 0.53) compared with female dogs (0.97 ± 2.39; median: 0.33).
Results of SDS-AGE were always clearly interpretable in terms of visualization of the bands, although in samples with a low protein content, bands were often weak and, conversely, samples with high protein content had prominent and sometimes overlapping bands or smeared on the gels (Fig. 1). The densitometric analysis of HRE automatically provided peaks corresponding to the bands (Fig. 2). When no bands were visible on the gels or when impurities (e.g., dust, stain precipitates) were present on negative gels, the densitometric analysis generated irregular profiles not corresponding to evident peaks (Fig. 3).
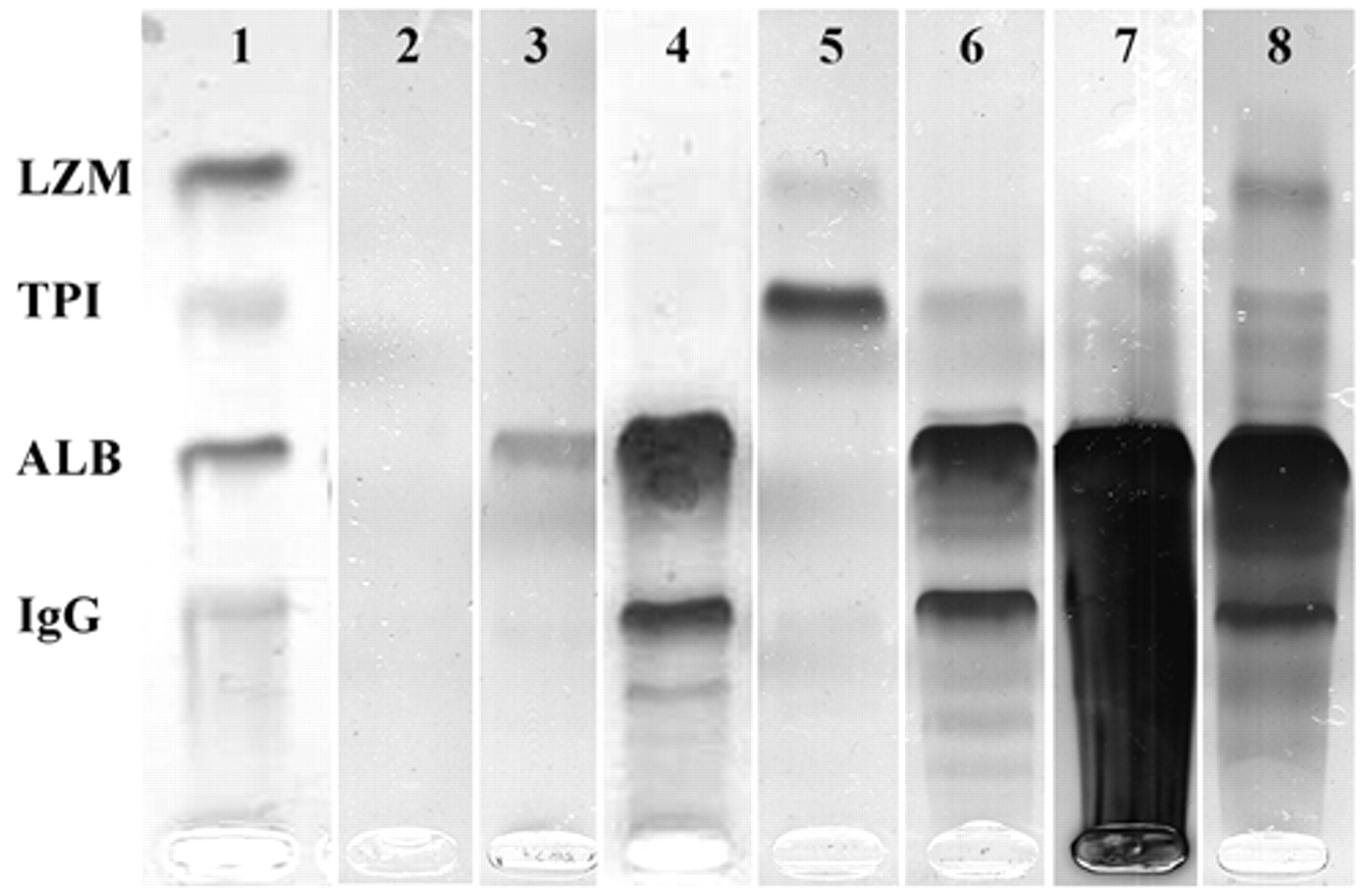
Example of sodium dodecyl sulphate–agarose gel electrophoresis (SDS-AGE) electrophoresis. Lanes are displayed as follows: lane 1: molecular weight control (MWC) where known proteins are underlined (LZM = lysozyme 15 kDa; TPI = triose phosphate isomerase [27 kDa]; ALB = albumin [66 kDa]; IgG = immunoglobulin G [150 kDa]); lane 2 = negative sample (no bands are visible); lane 3 = sample classified as albuminuric (only the albumin band is visible); lane 4 = sample with a glomerular pattern (only proteins with MW higher than albumin are present); lane 5 = sample with tubular pattern (only proteins with MW lower than albumin are visible; lane 6 = sample with a mixed pattern (bands with MW higher than albumin are clearly visible but weak bands with MW lower than albumin can also be detected); lane 7 = example of the pre-analytical artifact due to the high content of protein in the sample (proteins are so abundant that smear on the lane, thus hampering the identification of bands); lane 8 = same sample than in lane 7 after dilution 1:1 (v:v): although the intensity of some bands (e.g., albumin) is still intense, the sample can be easily classified as mixed (both proteins with MW higher than albumin and lower than albumin are detectable).
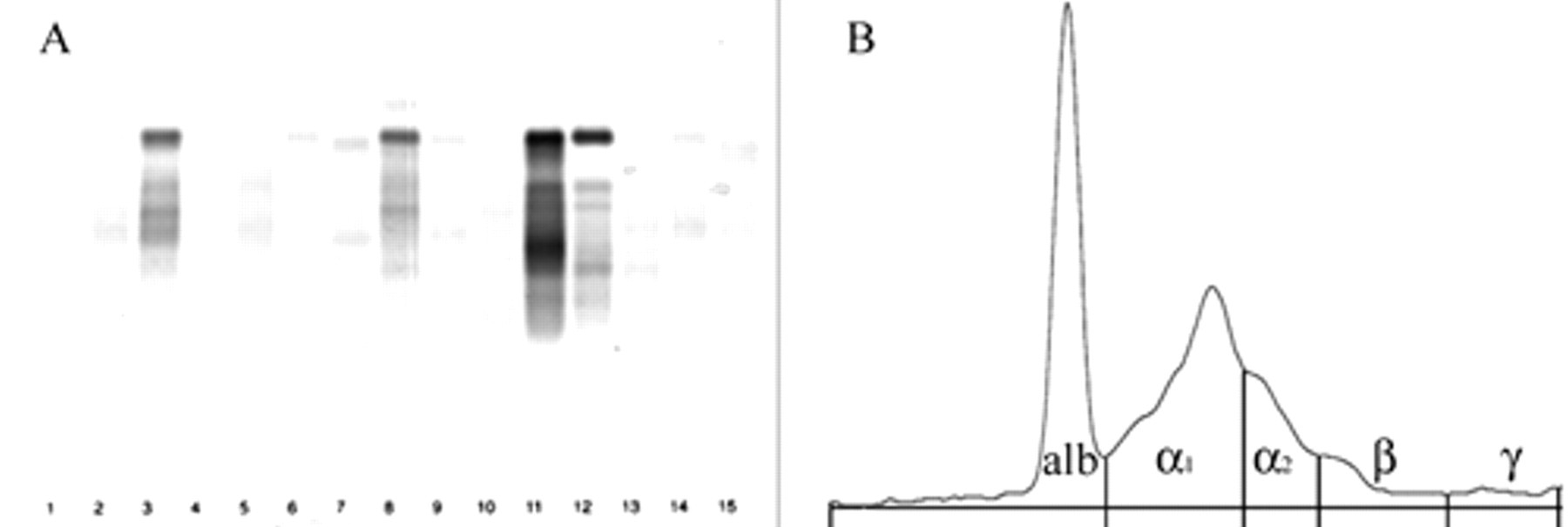
Electrophoretograms obtained using high-resolution electrophoresis (HRE).
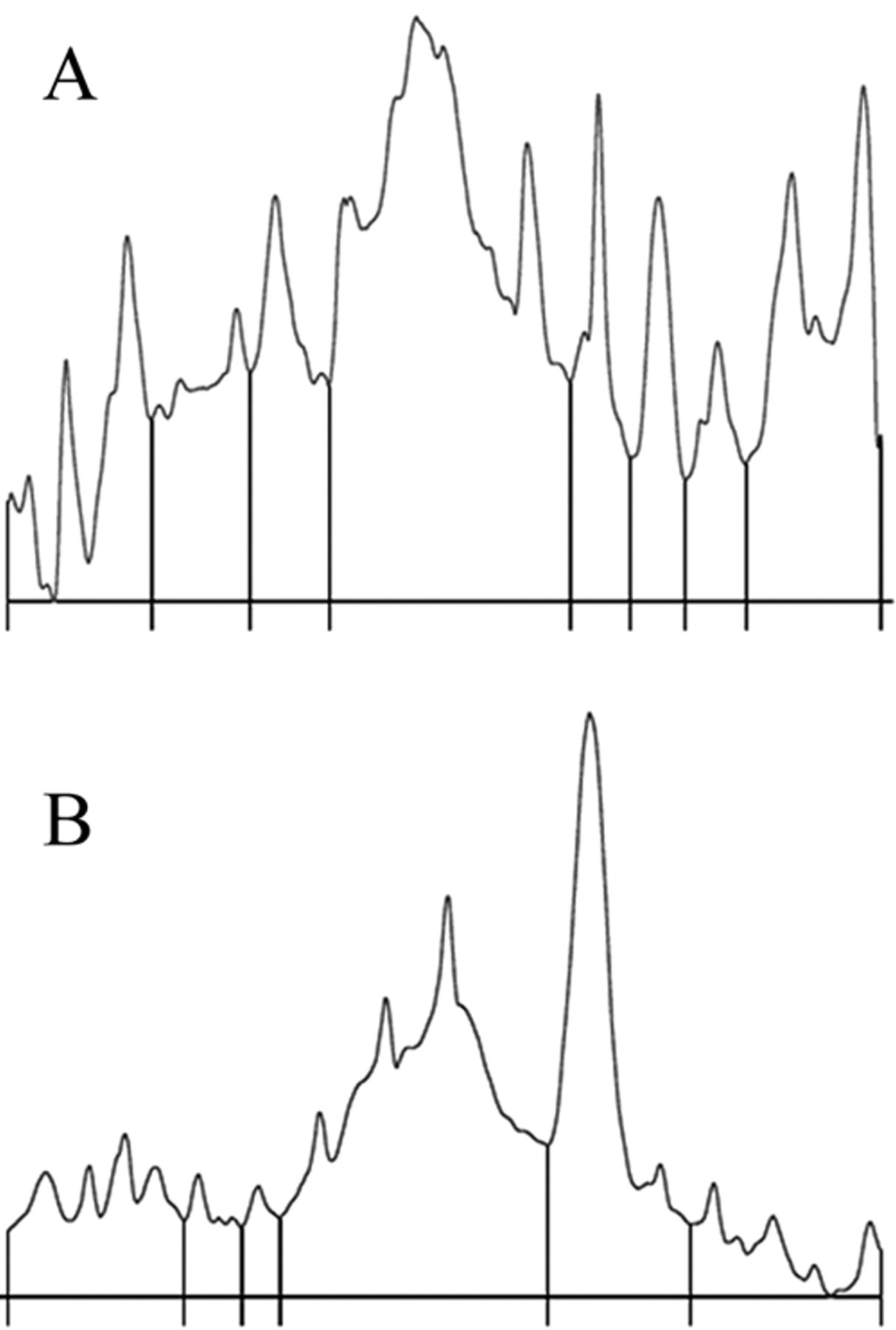
High-resolution electrophoresis electrophoretogram of a negative sample (
Ability of SDS-AGE and HRE to detect borderline proteinuric or proteinuric dogs
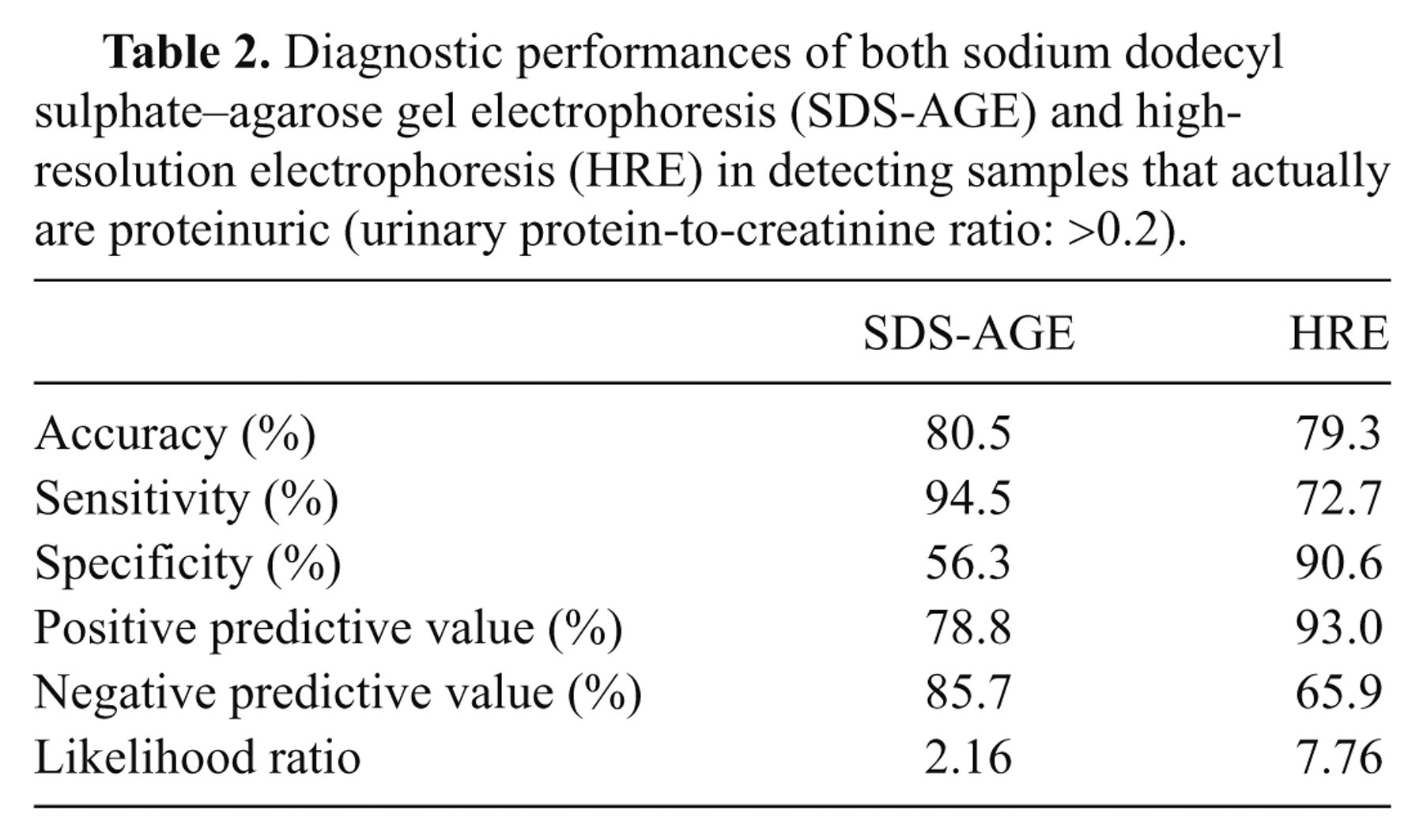
As shown in Table 1, the large majority of samples with a UPC ratio >0.2 had evident bands in SDS-AGE, but false-positive results were frequently found among samples with a UPC ratio <0.2. The proportion of false positive (10/24, 41.7%) was significantly higher (P = 0.049) in concentrated urine (USG: >1.030) than in diluted urine (USG: <1.030, 2/16, 12.5%). Conversely, HRE yielded a lower rate of false positive but a higher rate of false-negative results. As a consequence, the 2 electrophoretic tests had a moderate degree of concordance with each other (κ = 0.45, 95% CI = 0.30−0.61, P < 0.001) and with the actual proteinuric status of the patients (SDS-AGE: κ = 0.52, 95% CI = 0.33–0.70, P < 0.001; HRE: κ = 0.59, 95% CI = 0.42−0.75, P < 0.001). In addition, sensitivity and NPV were higher for SDS-AGE than for HRE while specificity, PPV, and LR+ were higher for HRE (Table 2).
Distribution of electrophoretic results in non-proteinuric (NP), borderline proteinuric (BP), and proteinuric (P) samples.*
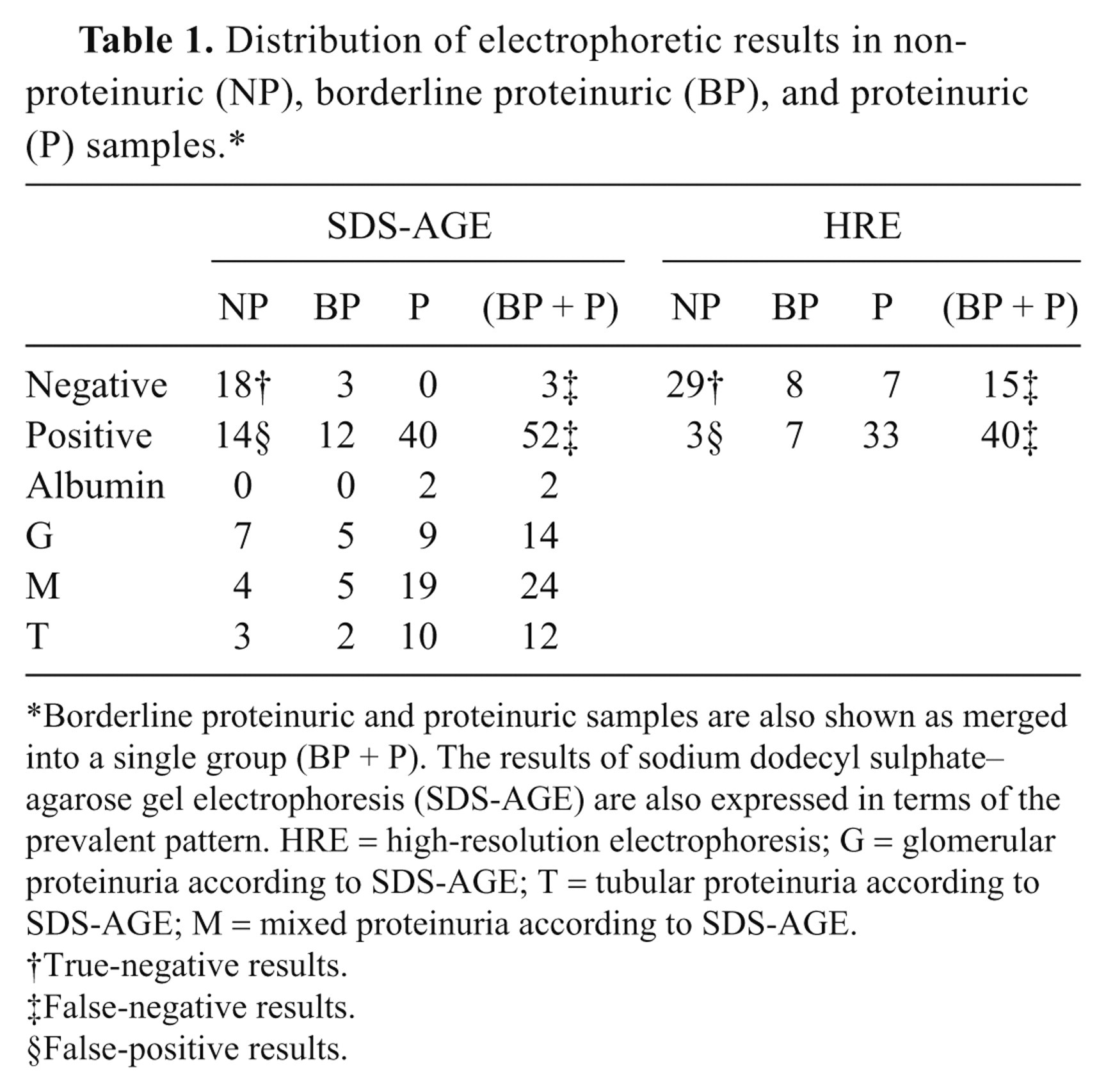
Borderline proteinuric and proteinuric samples are also shown as merged into a single group (BP + P). The results of sodium dodecyl sulphate–agarose gel electrophoresis (SDS-AGE) are also expressed in terms of the prevalent pattern. HRE = high-resolution electrophoresis; G = glomerular proteinuria according to SDS-AGE; T = tubular proteinuria according to SDS-AGE; M = mixed proteinuria according to SDS-AGE.
True-negative results.
False-negative results.
False-positive results.
Diagnostic performances of both sodium dodecyl sulphate–agarose gel electrophoresis (SDS-AGE) and high-resolution electrophoresis (HRE) in detecting samples that actually are proteinuric (urinary protein-to-creatinine ratio: >0.2).
Ability of HRE to detect the type of proteinuria
Using HRE, samples classified as affected by glomerular proteinuria by SDS-AGE had a significantly higher percentage of albumin compared with samples with mixed or tubular proteinuria and a significantly lower percentage of α1-globulins compared with samples with tubular proteinuria (Table 3). As a consequence, the alb/α1 ratio was significantly higher in dogs with glomerular proteinuria than in dogs with mixed or tubular proteinuria, and the distribution of values regarding alb/α1 ratio did not evidence a substantial overlap of values between samples with glomerular and tubular proteinuria (Fig. 4).
Mean values ± standard deviation recorded by high-resolution electrophoresis in samples classified as albuminuric (N = 2), glomerular (N = 11), mixed (N = 20), or tubular (N = 10) according to the results of sodium dodecyl sulphate–agarose gel electrophoresis.*
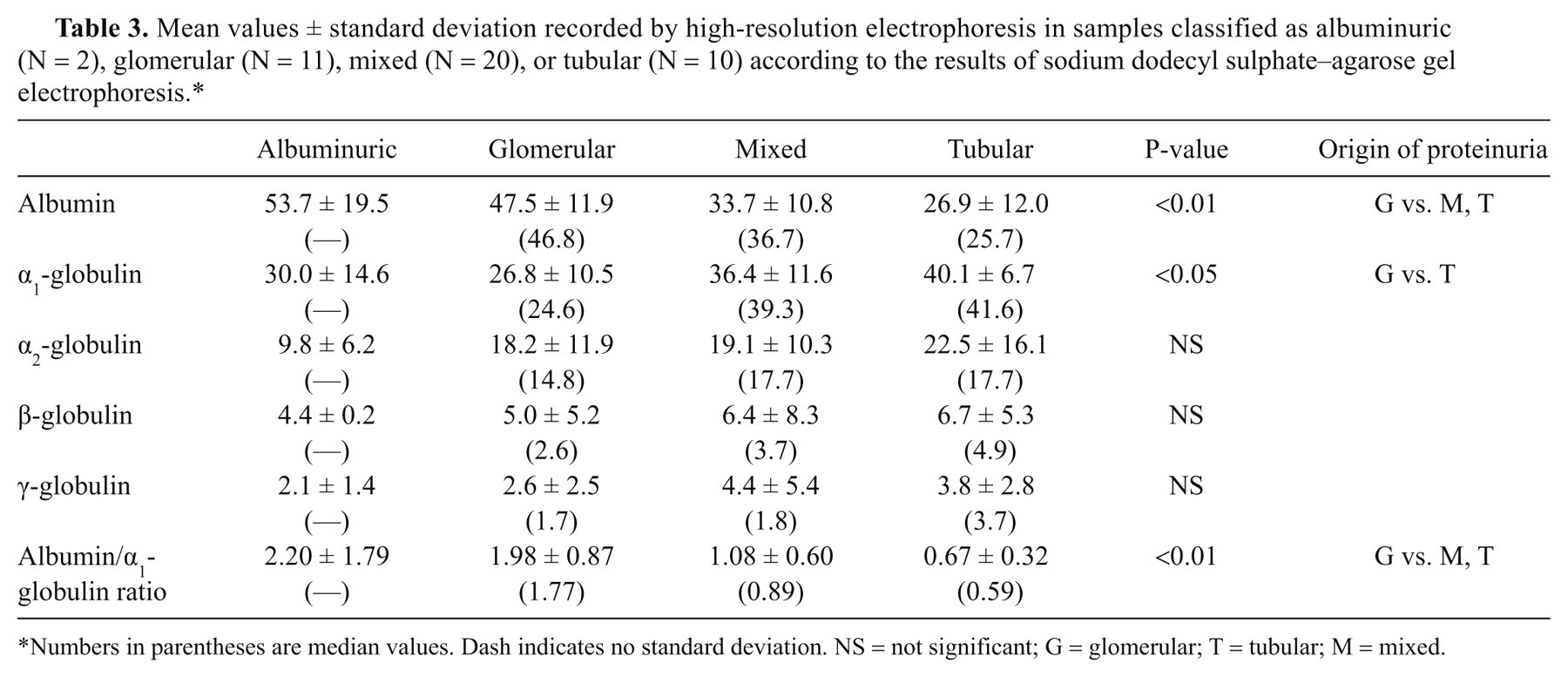
Numbers in parentheses are median values. Dash indicates no standard deviation. NS = not significant; G = glomerular; T = tubular; M = mixed.
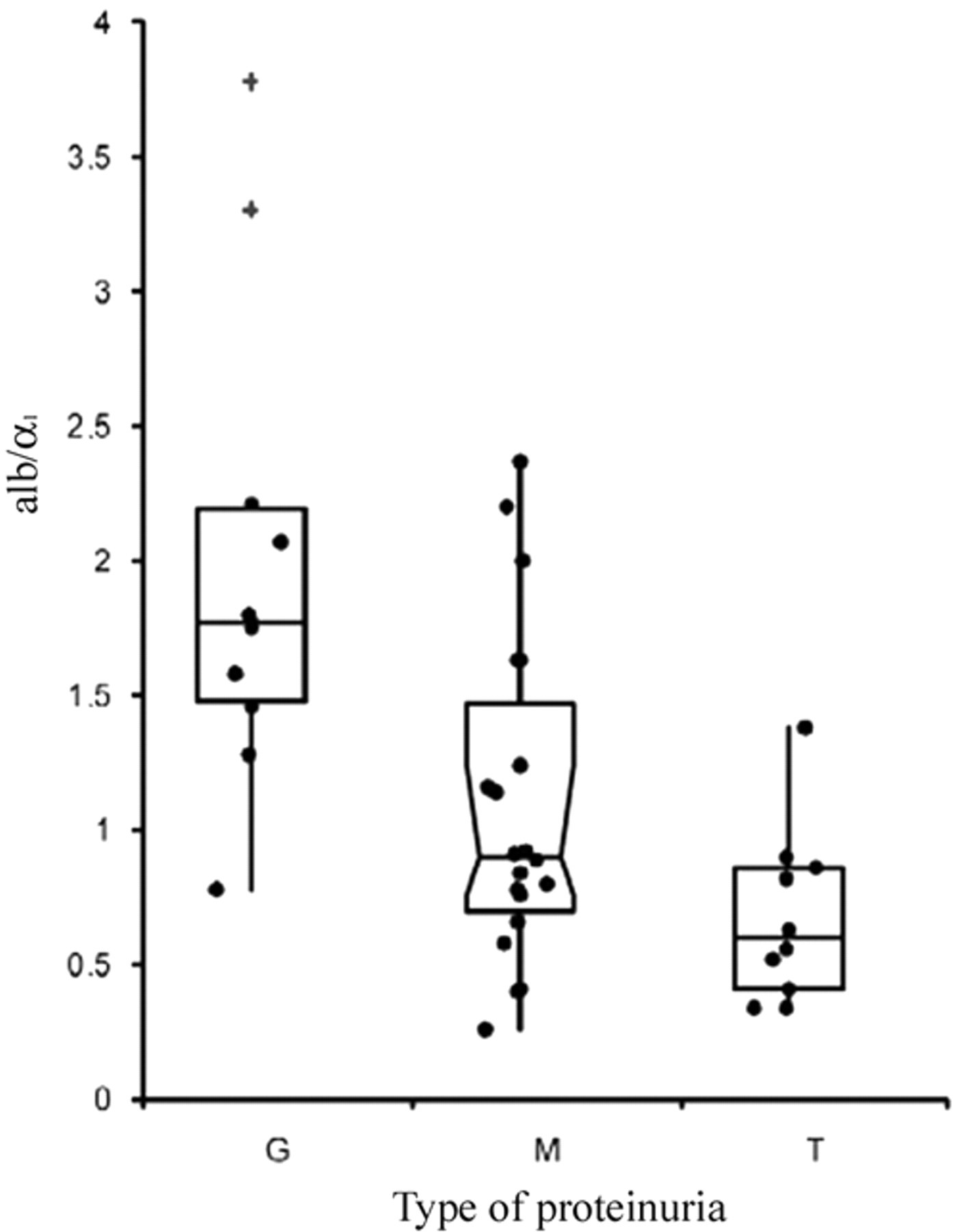
Distribution of values of albumin-to-α1 globulin (alb/α1) ratio in samples with glomerular (G), tubular (T), or mixed (M) proteinuria, as classified by sodium dodecyl sulphate–agarose gel electrophoresis. Box and whiskers represent the I–III interquartile range (IQR), vertical lines extend up to the limit for classifying values as outliers (1.5 × IQR), and horizontal lines correspond to the median value. The symbol “+” indicates values classified as “near outliers” (lower than 3.0 × IQR).
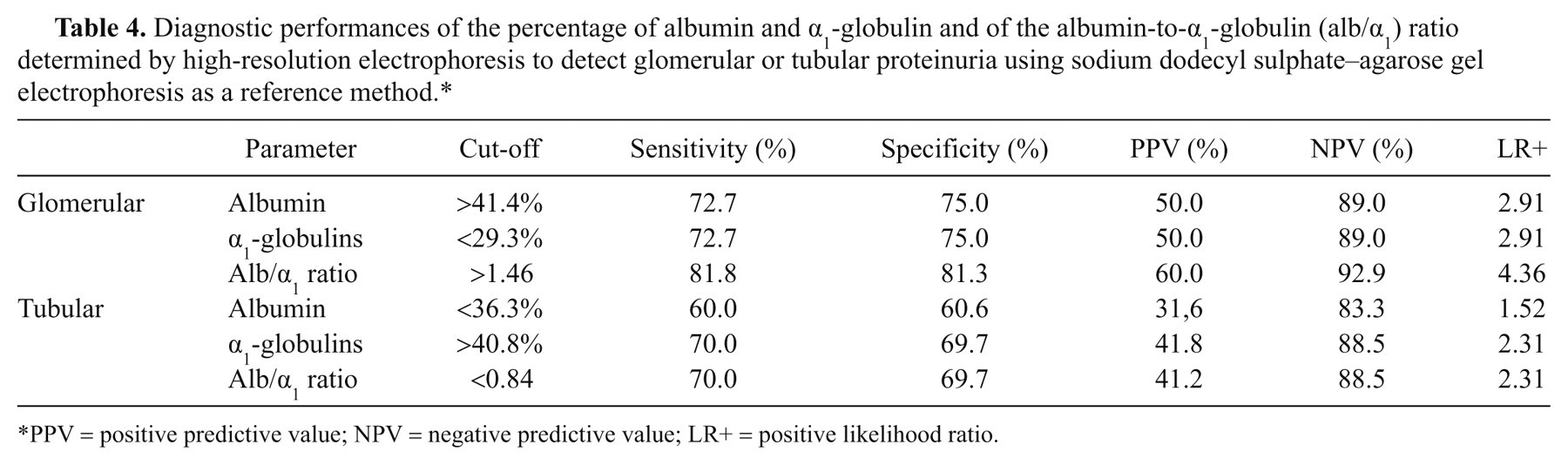
The ROC curves (Fig. 5) revealed that a high percentage of albumin (AUC = 0.79, 95% CI = 0.62−0.95, P < 0.001), a low percentage of α1-globulins (AUC = 0.76, 95% CI = 0.58−0.95, P = 0.002), and a high alb/α1 ratio (AUC = 0.84, 95% CI = 0.71−0.97, P < 0.001) can discriminate glomerular proteinuria at cut-off values reported in Table 4. The ROC curves designed for tubular proteinuria (Fig. 5B) evidenced similar AUCs for low percentages of albumin (0.75, 95% CI = 0.58–0.92, P = 0.002), high percentages of α1-globulins (0.67, 95% CI = 0.51−0.84, P = 0.018), and a low alb/α1 ratio (AUC = 0.81, 95% CI = 0.67−0.95, P < 0.001), but the cut-off values with the best diagnostic performances (Table 4) had moderate diagnostic performances.
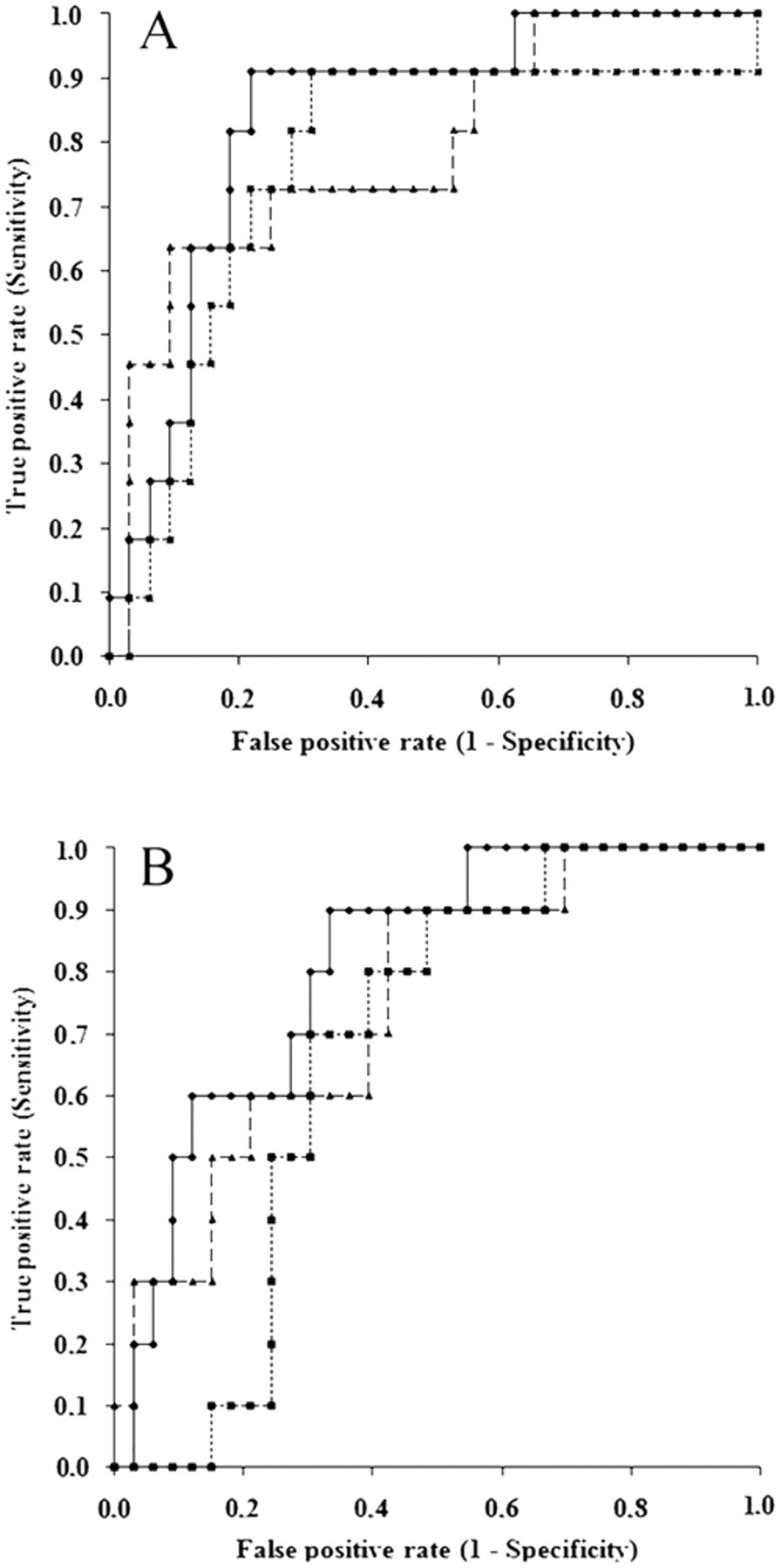
Comparison of receiver operating characteristic curves designed to (
Diagnostic performances of the percentage of albumin and α1-globulin and of the albumin-to-α1-globulin (alb/α1) ratio determined by high-resolution electrophoresis to detect glomerular or tubular proteinuria using sodium dodecyl sulphate–agarose gel electrophoresis as a reference method.*
PPV = positive predictive value; NPV = negative predictive value; LR+ = positive likelihood ratio.
Discussion
The quantitative analysis of proteinuria by the measurement of the UPC ratio is a mandatory step to classify proteinuria in dogs, allowing a correct management of proteinuric patients. 13 SDS-AGE has been previously validated in proteinuric dogs, 22 but no information is available on the performances of SDS-AGE on samples with a UPC ratio <0.2, that actually contain proteins but at a concentration that, according to the clinical classification proposed by the IRIS, does not require further investigation or therapeutic intervention. To the authors’ knowledge, HRE has never been tested on canine urine samples except for the diagnosis of gammopathies. 8
In the first part of the current study, the ability of both electrophoretic techniques to identify dogs with a grade of proteinuria that according to the IRIS classification requires additional investigation or clinical intervention (BP + P) was evaluated. To this aim, dogs were initially classified in 3 stages (NP, BP, and P) of the IRIS classification. 5 As expected, samples with a high UPC ratio frequently had high plasma creatinine values, likely due to renal diseases. The study design was focused on the analytical properties of electrophoretic techniques rather than on the diagnostic significance of increased UPC ratio: it is thus not possible to explain the occasional presence of proteinuric samples in dogs with normal or slightly increased creatinine values, but the more likely interpretation is that these dogs had physiological or pre-renal proteinuria.
The BP and P samples were then merged in a single group of dogs with a UPC ratio >0.2 to split data into 2 categories, thus allowing to enumerate false-negative and true-negative, and false-positive and true-positive, results. The rationale for including BP samples in a single group with P samples was based on the recommendation of both IRIS 5 and American College of Veterinary Internal Medicine (ACVIM) 13 to monitor and investigate patients with borderline proteinuria. Using this dichotomization, the 2 techniques had a moderate degree of agreement and similar accuracy but this was basically due to an opposite ability to classify patients: HRE is highly specific but suffers from a poor sensitivity while SDS-AGE has a high sensitivity and a low specificity. The high frequency of false-positive results with SDS-AGE is basically due to the high analytical sensitivity of the technique, which detects small amounts of protein (up to 1–10 ng protein/sample using highly sensitive stains such as silver stain). On one hand, this might suggest that the threshold defined by the IRIS classification could be inadequate and also that dogs with a UPC ratio <0.2 should be considered proteinuric when tubular or glomerular proteins are detected by SDS-AGE. Unfortunately, according to the IRIS classification and ACVIM guidelines, 5,13 dogs with a UPC ratio <0.2 included in the present study were not investigated through kidney biopsies and/or they were not monitored during the follow up, but these aspects merit further investigation. On the other hand, the high rate of false-positive results may depend on the concentration of urine, as evidenced by the comparison of results in samples with high or low USG; ultimately, highly concentrated urine contains more proteins than diluted urine with the same UPC ratio. The measurement of USG is thus mandatory to correctly interpret SDS-AGE, and future studies should define the cut-off values of USG that minimize the risk of detecting positive bands in dogs with a UPC ratio <0.2.
As a consequence of the performances mentioned above, the probability that a negative SDS-AGE actually corresponds to a NP sample is high (more than 85%) whereas the probability that a positive sample actually has a UPC ratio >0.2 is lower than 80%. This means that approximately 20% of dogs with a UPC ratio <0.2 could be considered affected from glomerular, tubular, or mixed proteinuria if SDS-AGE is conducted without information on the UPC ratio. The low likelihood ratio means that in the case SDS-AGE is positive is only about two times more likely that the UPC ratio is >0.2 than that the UPC ratio is <0.2. By contrast, a positive result in HRE likely correlates to a sample with a UPC ratio >0.2, while negative results do not necessarily indicate that the sample is NP because the probability of this event is “only” approximately 66%.
Based on these results, neither of the 2 methods can replace the determination of proteinuria by UPC ratio, and both tests must be conducted only after classifying dogs as BP or P based on the UPC ratio. Nevertheless, given its LR+, a positive HRE can be confidently interpreted as associated with a UPC ratio >0.2 while, based on the Bayesian approach, 1,18 a positive SDS-AGE can be considered associated with a UPC ratio >0.2 only in the case of a high pre-test probability of this event (e.g., dogs with leishmaniasis, amyloidosis, or with history of a persistently high UPC ratio).
The second part of the study, focusing on the ability of HRE to provide preliminary information about the origin of proteinuria, revealed a high albumin percentage in glomerular forms and a high α1-globulin percentage in tubular forms. This suggests that HRE could be a useful and inexpensive screening tool to predict SDS-AGE results. Nevertheless, at the cut-offs characterized by the best diagnostic performances, sensitivity, specificity and PPV are not optimal and likelihood ratios are low. 7 The high NPV is not clinically useful; for example, a low percentage of albumin or a high percentage of α1-globulins correspond to a probability of 90% of not having glomerular or tubular proteinuria, respectively, but this does not clarify the true type of proteinuria. As expected, the diagnostic performances were better using the alb/α1 ratio that increased the difference between values recorded in samples with glomerular and tubular proteinuria. The increased alb/α1 ratio appears useful to identify glomerular proteinuria (due to the relatively high LR+). To a lesser extent, the decreased alb/α1 ratio can identify tubular proteinuria, and values between the 2 cut-offs could suggest mixed proteinuria.
In conclusion, both SDS-AGE and HRE have a limited capacity to correctly identify patients with a UPC ratio >0.2. In particular, SDS-AGE is poorly specific, especially in samples with high USG, and HRE is poorly sensitive. The evaluation of UPC ratio and USG are mandatory before running qualitative analysis of proteinuria. Conversely, HRE could be a cheap and rapid preliminary screening test in the case of elevated UPC ratio when the detection of high or low alb/α1 ratio may be suggestive of glomerular or tubular proteinuria, respectively. These results encourage further prospective studies aimed to assess the clinical application of HRE and of the clinical significance of altered alb/α1 ratio in field conditions.
Footnotes
a.
Venosafe® (VF-109SURI), Terumo Italia, Roma, Italy.
b.
HR-160, Optika SRL, Ponteranica, Bergamo, Italy.
c.
Cobas Mira, Roche Diagnostic, Basel, Switzerland.
d.
Fluitest USP, Analyticon Biotechnologies, Lichtenfels, Germany.
e.
Hydragel 15 HR, Sebia Italia SRL, Bagno a Ripoli, Firenze, Italy.
f.
Hydragel 5 Proteinuria, Sebia Italia SRL, Bagno a Ripoli, Firenze, Italy.
g.
Hydrasis, Sebia Italia SRL, Bagno a Ripoli, Firenze, Italy.
h.
Phoresis, Sebia Italia SRL, Bagno a Ripoli, Firenze, Italy.
i.
Microsoft Office Excel 2007, Microsoft Corp., Redmond, WA.
j.
Analyse-it version 2.21, Analyse-it Software Ltd., Leeds, United Kingdom.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
