Abstract
The present report describes a spontaneously developing, highly infiltrative, and expansile neoplasm in the mandible of a 9-month-old female Sprague Dawley rat. The lesion consisted of abundant odontogenic epithelium with induction of osteodentin in the adjacent mesenchyme and was classified therefore as an odontoameloblastoma. Odontoameloblastomas are reported uncommonly in domestic animals, and the histologic and histochemical features of this neoplasm are reported herein along with a review of the veterinary literature.
Odontoameloblastoma is an uncommon oral neoplasm characterized by odontogenic epithelium resembling that of an ameloblastoma with the additional features of odontogenic ectomesenchyme and induction of mineralized dental tissues. 20 Odontoameloblastomas are locally aggressive, infiltrative neoplasms but do not metastasize. 7 Diagnosis of odontoameloblastoma is straightforward when mature tooth structures are present; however, the presence of small islands and thin seams of atubular dentin or osteodentin, particularly in animals with continuously erupting teeth, should not be overlooked and support the diagnosis of odontoameloblastoma over ameloblastoma.
A 9-month-old, female Sprague Dawley rat from an institutional breeding colony was euthanized due to the presence of a large mandibular mass. On gross examination, the left mandible was surrounded and expanded by a 2-cm wide, highly infiltrative mass (Fig. 1A). The lesion extended beyond the rostral margin of the mandible, the native oral epithelium was ulcerated, and the exposed surface was tan and friable. On cut surface, the mass was diffusely yellow to tan with friable and purulent material near the oral cavity, and deeper aspects of the mass were hard and gritty. Other organs and tissues were unremarkable. The mandible and samples of all major organs were fixed in 10% neutral buffered formalin for 24 hr, at which time peripheral sections of soft tissue from the mass and all other tissues were processed routinely and embedded in paraffin. Decalcified sections of the mandible and associated tumor were sectioned and processed 8 days later. Tissue sections were cut to 5 mm and stained with hematoxylin and eosin, Gomori trichrome, periodic acid—methenamine silver, and periodic acid—Schiff reaction. For immunohistochemistry, 3-μm sections were treated with 3% hydrogen peroxide to inhibit endogenous peroxidase and then pretreated with Tris—ethylenediamine tetra-acetic acid (cytokeratin) or citrate buffer (vimentin) in a steam bath for antigen retrieval. Immunostaining was performed using an automated cell staining system a with a commercial chromogen b and hematoxylin c counterstain. The primary antibodies used were mouse monoclonal anti-cytokeratin d (1:100) and mouse monoclonal anti-vimentin e (1:500), with a goat anti-mouse secondary antibody f and horseradish peroxidase—streptavidin conjugate. g
Histologic examination revealed effacement of native mandibular bone by a well-demarcated, nonencapsulated, and highly infiltrative mass of neoplastic epithelial cells arranged in islands and anastomosing cords and trabeculae with a palisading peripheral layer of densely packed, tall, columnar cells. These columnar cells were further characterized by apically located, oval nuclei and prominent basilar cytoplasmic clearing consistent with odontogenic epithelium (Fig. 1B). Centrally, neoplastic foci were composed of stellate cells with prominent intercellular bridges. The mitotic rate within the palisading layer was high with 1 to 5 mitotic figures per high-power field (400×) and a total of 25 noted in the evaluation of 10 high-power fields. Scattered, individual apoptotic cells were present within the cords of palisading cells. In multiple foci, centers of groups of neoplastic cells were replaced by variable amounts of eosinophilic, granular fluid admixed with cellular debris and small numbers of degenerating neutrophils or macro-phages. Along the basilar aspects of the palisading columnar cells were streams of densely packed fusiform cells that were often separated from the epithelial cells by variably thick, wedge-shaped foci of homogeneous, brightly eosinophilic, extracellular matrix material consistent with atubular dentin (Fig. 1B). This matrix material was often present within invaginations into neoplastic foci, frequently contained individual polygonal to fusiform cells within lacunae (osteodentin), and was associated with a shift in appearance of the adjacent mesenchymal component from fusiform to plump cuboidal or short columnar, consistent with odontogenic ectomesenchyme (Fig. 1C). Periodic acid—Schiff reaction revealed that the polarized, neoplastic odontogenic epithelial cells were separated from the mesenchymal stroma by a prominent basement membrane and that this membrane was absent in areas where eosinophilic matrix material was present. This matrix material was homogeneous and pale green to blue with Gomori trichrome staining (Fig. 1D), which is consistent with collagen, 4 the major organic component of dentin. 23 Immunohistochemical staining revealed that the palisading columnar cells were cytokeratin positive (Fig. 1E) and vimentin negative (Fig. 1F), whereas the adjacent mesenchymal component was cytokeratin negative (Fig. 1E) and vimentin positive (Fig. 1F), consistent with ameloblasts and odontogenic ectomesenchyme, respectively. Cells within the central regions of the neoplastic islands were variably positive for both cytokeratin and vimentin. At the surface of the neoplasm, there was loss of the overlying epithelium; extensive necrosis; and replacement of ulcerated tissues by numerous viable and degenerate neutrophils and macrophages and fewer lymphocytes admixed with necrotic bone fragments, cellular and karyorrhectic debris, basophilic granular material (mineral), plant material, and myriad colonies of coccoid bacteria. Histologic lesions in other tissues consisted of moderate, splenic lymphofollicular hyperplasia and mild, random, necropurulent hepatitis.
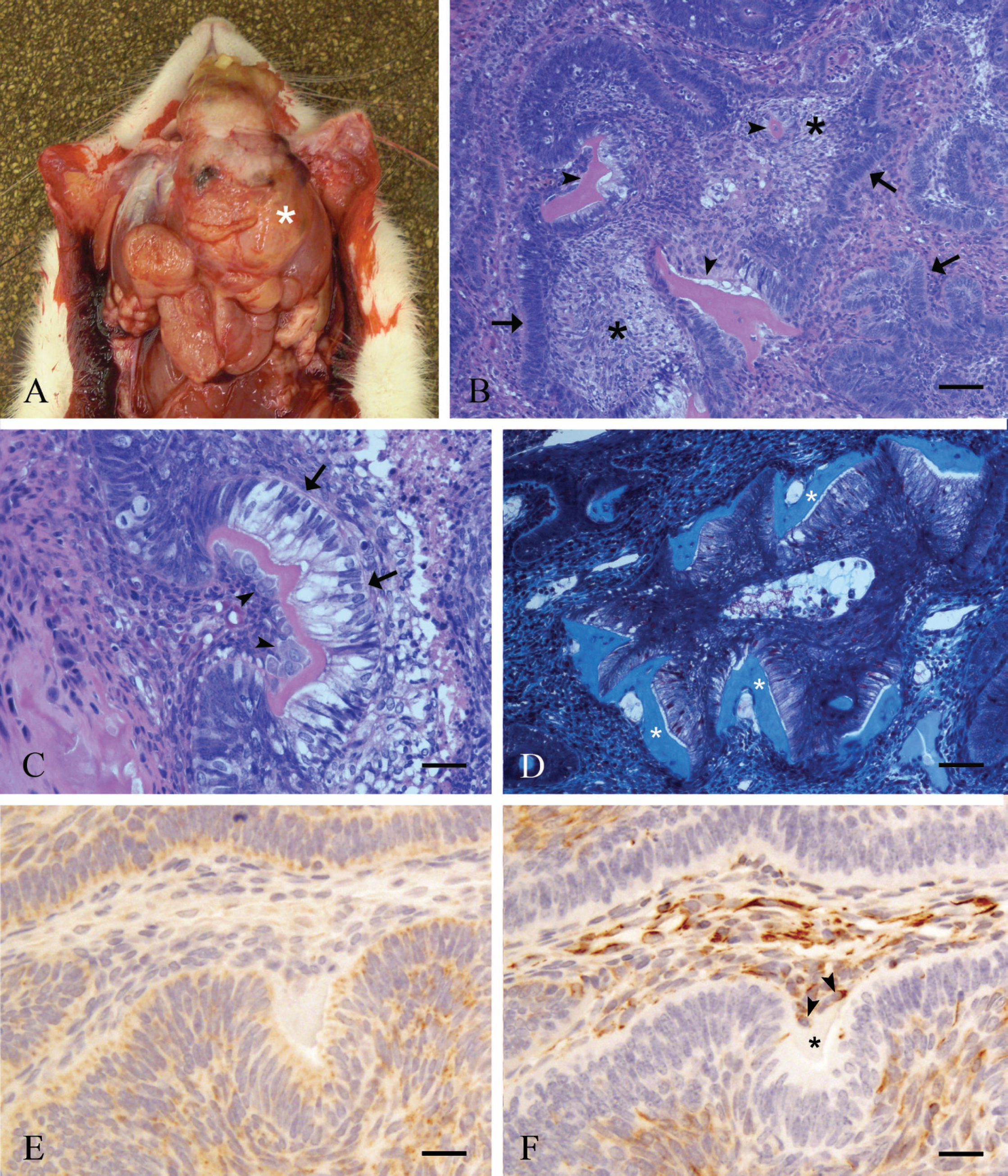
Macroscopic, histopathologic, and immunohistochemical features of a mandibular neoplasm from a female Sprague Dawley rat.
Much of the odontogenic epithelial component of this neoplasm was consistent with the follicular variant of ameloblastoma; however, by definition, ameloblastomas are noninductive neoplasms. 7,14,25 During odontogenesis, ameloblasts degrade their underlying basement membrane and achieve cell-to-cell contact with odontoblasts, whereby their interaction induces the production of dentin. 28 In the neoplasm described in the present report, there was locally extensive loss of basement membrane at and adjacent to foci of eosinophilic matrix material representative of such induction. The histologic and histochemical features of the eosinophilic matrix material in the context of its presence adjacent to an ameloblastic neoplasm are most consistent with osteodentin. Osteodentin is rapidly formed, tertiary dentin that often entraps odontoblasts with few recognizable dentin tubules, giving it a morphologic appearance similar to bone. 13,23 The formation of metaplastic osteodentin in the pulp cavity of rat incisors is a recognized reaction mechanism in toxicologic studies. 15 The combination of a predominantly ameloblastic neoplasm with features of odontogenic ectomesenchyme and mineralized dental tissues in a single lesion thus warrants the diagnosis of odontoameloblastoma. 20
Previous histologic classification schemes for odontogenic neoplasms in domestic animals have referred to lesions with features similar to odontoameloblastoma as ameloblastic odontomas, 12,28 and the World Health Organization classification of rodent tumors 7 lists the 2 terms as synonymous; however, ameloblastic odontoma is not listed as a tumor classification in the 1992 World Health Organization classification of odontogenic neoplasms. 14 Additionally, in a review of the nomenclature of odontogenic tumors in animals, 11 the author suggests that the term “ameloblastic odontoma” no longer be used and that the pathologic behavior implied by the terms “ameloblastoma” and “odontoma” be considered when incorporating these terms into a diagnosis. Accordingly, the authors have chosen to use the term “odontoameloblastoma” for the description of this infiltrative and productive neoplasm. Ameloblastic odontoma and odontoameloblastoma have been reported in a variety of mammalian species, including dog, 16,21 cat, 24 horse, 18,22 cow, 5,28 sheep, 8 llama, 27 nonhuman primate, 2,3,6,26,30 rat, 1,10,29 and mouse. 19
Odontogenic tumors are uncommon in all species, but animals with continuously erupting teeth, such as rodents, would appear to be at greater risk for developing these lesions, because the odontogenic tissues in these animals remain functional throughout life. The rat incisor is commonly evaluated in toxicologic studies, 15 and knowledge of dysplastic and neoplastic lesions that may arise in the rat incisor is highly relevant to veterinary pathologists. In the rat, lesions arising from the mandible with features of odontogenic epithelium and mineralized dental tissues have been reported as either spontaneous 1,10 or chemically induced tumors 29 and were diagnosed as ameloblastic odontoma. In these cases, some of the tumors arose along the mandibular margin without extensive infiltration and had well-developed dental structures, whereas other lesions effaced the mandible and had extensive variability in the character of mineralized dental tissues. The pathologic behavior of these reported lesions varies dramatically, and some tumors appear more consistent with complex or compound odontomas while others appear consistent with odontoameloblastoma. As further evidence of confusion in the nomenclature, the term “ameloblastoma” was used to characterize infiltrative neoplasms identified in the mandible of 2 Sprague Dawley rats 17 and a young female Wistar rat 9 ; however, the descriptions and photomicrographs from these cases depict features of mesenchymal induction similar to that in the present report and would probably have been designated as odontoameloblastoma using the criteria listed in recently published references.
In summary, odontoameloblastoma is an uncommon odontogenic neoplasm characterized by odontogenic epithelium, odontogenic ectomesenchyme, and mineralized dental tissues within a single, locally invasive mass. The true incidence of this lesion is unclear, given the inconsistent use of the term “ameloblastic odontoma” in describing odontogenic neoplasms with similar features in various animal species and human beings. The term “odontoameloblastoma” should be used to describe lesions with the clinical and odontogenic epithelial features of an ameloblastoma that also have induction of dental hard tissues, including atubular dentin and osteodentin.
Footnotes
a.
i6000™, BioGenex Laboratories Inc., San Ramon, CA.
b.
NovaRED™, Vector Laboratories Inc., Burlingame, CA.
c.
Shandon Harris hematoxylin, Thermo Fisher Scientific, Pittsburgh, PA.
d.
MNF116 clone, Dako North America Inc., Carpinteria, CA.
e.
V9 clone, Dako North America Inc., Carpinteria, CA.
f.
MultiLink®, BioGenex Laboratories Inc., San Ramon, CA.
g.
Zymed, Invitrogen Corp., Carlsbad, CA.
