Abstract
Swarms or blooms of jellyfish are increasingly problematic and can result in high mortality rates of farmed fish. Small species of jellyfish, such as Phialella quadrata (13 mm in diameter), are capable of passing through the mesh of sea cages and being sucked into the mouth of fish during respiration. Results of the current study show that the initial damage to gills of farmed Atlantic salmon, likely produced by nematocyst-derived toxins from the jellyfish, was compounded by secondary bacterial infection with Tenacibaculum maritimum. Results also demonstrate that these filamentous bacteria were present on the mouth of the jellyfish and that their DNA sequences were almost identical to those of bacteria present on the salmon gills. This suggests that the bacterial lesions were not the result of an opportunistic infection of damaged tissue, as previously thought. Instead, P. quadrata is probably acting as a vector for this particular bacterial pathogen, and it is the first time that evidence to support such a link has been presented. No prior literature describing the presence of bacteria associated with jellyfish, except studies about their decay, could be found. It is not known if all jellyfish of this and other species carry similar bacteria or the relationship to each other. Their source, the role they play under other circumstances, and indeed whether the jellyfish were themselves diseased are also not known. The high proteolytic capabilities of T. maritimum mean that partially digested gill tissues were readily available to the jellyfish, which rely heavily on intracellular digestion for their nutrition.
Introduction
Environmentally linked mass mortality of sea-caged fish is an ever-present risk for the aquaculture industry. 13 In Scotland, many such events have in recent years been linked to planktonic organisms, such as jellyfish. These may occur at high densities, “blooms,” and cause high mortality rates in intensively stocked fish due to oxygen depletion or the effects of their toxins. 32 Swarms of larger oceanic species of jellyfish are often involved, but smaller species, such as Phialella, have also been implicated. These neritic species are limited to coastal areas by their bipartite life cycle that includes a fixed hydroid stage that requires a seabed in relatively shallow waters. 31 Phialella quadrata has already been implicated in sea-caged salmon mortality in Scotland, 8 and it has been shown that this species can inhabit inshore waters of the British Isles almost year-round, 6,31 suggesting that it is not constrained to 1 generation per year.
The main mechanism whereby a species such as Phialella harms fish is through the stinging cells or nematocysts that can be found on the tentacles and possibly elsewhere.7 At 13 mm in diameter, P. quadrata are small enough to pass easily through the mesh of the nets in which the fish are held and subsequently to be sucked in through the mouth of the fish during respiration. Thus, gills are a major target. Jellyfish toxins are considered to be some of the most potent of all venoms. 14 The specific toxins of P. quadrata have not yet been studied, but in general, jellyfish toxins can cause cytotoxic, neurotoxic, cardiotoxic, hemolytic, dermatonecrotic, immunogenic, and inflammatory effects. 17,35 Despite this broad pathogenic range, cytolytic and/or hemolytic effects appear to be common to all nematocyst venoms. 14 When the fish does not die directly from the immediate effects of the toxins, it can succumb within a few hours from respiratory failure or later from secondary bacterial infections on the body and the gills caused by opportunistic bacteria such as flavobacteria and vibrios. 30
Bacterial diseases of the gills in salmon are much less common in saltwater than in freshwater, but they do occur. Tenacibaculum maritimum is 1 species that has been regularly incriminated in gill disease out-breaks, 12 although little is known of predisposing factors or source(s) of infection. Lesions are generally restricted to lamellae and filaments and include severe necrosis and even infarction. 11
The present case investigates an outbreak of gill disease in sea-caged salmon in the Shetland Isles, Scotland. Lesions from fish sampled early in the outbreak were determined by histopathology to be associated mainly with the jellyfish P. quadrata, but later on, they were found to be associated with filamentous bacteria morphologically similar to T. maritimum. The close association between the 2 types of lesions suggested that there was a link between the jellyfish and the bacteria. This study describes the gill lesions and confirms the identity of the bacteria in the gill lesions as T. maritimum. Moreover, it shows the presence of morphologically similar bacteria on the manubrium (mouth) of the jellyfish and identifies them also as T. maritimum. Sequencing phylogenetically relevant regions of bacterial DNA from the 2 sources, gill and jellyfish, showed that they were virtually identical, suggesting that the jellyfish were acting as vectors for this pathogen. To the authors' knowledge, this is the first time that such a link has been documented. Moreover, this is the first report of salmonid tenacibaculosis in Scotland.
Materials and methods
Case history
During July and August 2008, many small jellyfish were reported swarming around salmon cages in a sea-site off the Shetland Isles. They were identified as the neritic species P. quadrata (Forbes 1848) based on the following morphologic characteristics: leptomedusae with a flattened bell of 1- to 15-mm wide, fairly thick, clear jelly and a small stomach with 4 short slightly folded mouth lips; gonads on the 4 radial canals being distal to the stomach, near, but not touching, the ring canal and with a median groove; 16 to 32 hollow marginal tentacles, no marginal cirri or condyli, and 8 closed, adradial, marginal vesicles with concretions.
Along with this jellyfish bloom, mortality in caged salmon was also reported. Many fish were seen to be lethargic and hanging at the water surface. Gross lesions included the presence of increased mucus and hemorrhage on gills; later, white patches on the gill arches were observed. Routine histopathologic samples were taken from affected moribund fish on a number of occasions in July, August, and September. Some jellyfish were also collected from within as well as outside affected cages and fixed in either 10% buffered formalin or 70% alcohol. By November, total losses of fish were approximately 75%.
Bacterial cultures
An isolate of T. maritimum (NCIMB 2163) from the National Collection of Industrial and Marine Bacteria (Aberdeen, Scotland) was used for comparative scanning electron microscope observations and as a positive control for the polymerase chain reaction (PCR) assays. The bacteria were recovered from lyophilized stocks into 10 ml of marine cytophaga broth and incubated at 22°C for 3 days. Purity checks were performed by inoculating a marine cytophaga agar plate with a loopful of the broth culture using the streak plate method to obtain a bacterial plate culture. The culture was incubated at 22°C for 2-3 days to obtain bacterial growth.
Histopathology of fish samples
Tissues from anesthetized moribund fish were sampled into 10% buffered formalin. After at least 24 hr of fixation at room temperature, tissues were routinely processed through to paraffin wax blocks and sections were stained with hematoxylin and eosin (HE).
Scanning electron microscopy
After the light microscopic observations, a wax-embedded gill sample was dewaxed by transferring it into 3 changes of pure xylene, each for 1 hr, and then rehydrating in a descending series of ethanol solutions. Some jellyfish preserved in ethanol were also rehydrated in a descending series of ethanol solutions. All samples were processed according to a previously described method. 21 Finally, samples were air-dried at room temperature, mounted on stubs, and sputter coated with gold.
Samples and DNA extractions
Approximately 20 jellyfish samples were processed in a single batch for DNA extraction. a Two formalin-fixed, wax-embedded gill samples from 2 fish were also selected, because these were seen in HE sections to have high numbers of bacteria.
Purification of total DNA from formalin-fixed and ethanol-fixed jellyfish was achieved using a kit a and following the manufacturer's protocol. Gills from formalin-fixed, wax-embedded samples were processed in a similar manner using the same extraction method but this time adapted for wax-embedded material. 10 Finally, bacterial DNA from the T. maritimum isolate (NCIMB 2136) was extracted 33 with minor modifications, 10 and this was used as the positive control for the primer specific PCR. The total DNA concentration of each sample was measured using a spectrophotometer. b All DNA samples were then stored in aliquots at −20°C until required.
Primers and PCR performed on the fish and jellyfish samples
Two sets of 16S ribosomal RNA (rRNA) primers were used; a set of universal primers (20F) conserved in all 16S rRNA was used in the primary PCR reactions 37 designated 20F (5′-AGAGTTTGATCATGGCTCAG-3′) and 1500R (5′-GGTTACCTTGTTACGACTT-3′). Secondary PCR used 16S rRNA-specific primers 36 for T. maritimum designated MAR1 (5′-AATGGCATCGTTTTAAA-3′) and MAR2 (5′-CGCTCTCTGTTGCCAGA-3′) designed to give a 1,088-base pair (bp) product.
The PCR amplifications were performed using a commercially available kit. c Each 25 μl reaction in the primary PCR contained 1 μM of each primer 20F and 1500R and 500 ng of sample DNA, with the rest of the 25 μl volume made up of sterile distilled water. Reaction mixtures were amplified using a DNA thermocycler. d The cycling conditions were identical to those described. 9 For the primary PCR, an initial preheating cycle at 95°C for 4 min was performed followed by 25 cycles of 95°C for 1 min, 55°C for 1 min, 72°C for 90 sec, and a final step of 72°C for 5 min. This PCR was performed twice only for the jellyfish samples, in which 1 μl of the first-round PCR product was removed and added to new PCR beads c containing 1 μM of each primer 20F and 1500R in a final volume made up to 25 μl with sterile distilled water. The PCR reactions were performed following thermocycling conditions as described. 9 The second-round PCR samples were then used for the species-specific PCR. The PCR products for all reactions were visualized on a 1.5% (w/v) agarose gel containing ethidium bromide.
Sequencing
Nucleotide sequencing was used to confirm the identity of the final PCR products. Each sample was purified in duplicate or triplicate using a DNA and gel band purification kit e following the manufacturer's protocol and sequenced f following the manufacturer's protocol. All the sequences obtained were compared and contrasted with other 16S rRNA sequences in the EMBL database by BLAST analysis. 1
Results
Histopathology and scanning electron microscope observations
All tissues were examined, but significant histo-pathologic findings were restricted largely to the gills. Changes involved the respiratory surfaces but notably also the gill arch, and they varied in severity from moderate to marked. On the arch epithelium, superficial lesions were characterized by multifocal acute ballooning degeneration, by spongiosis and hydropic degeneration, and by necrosis (Fig. 1). The inflammatory response was variable but was composed mainly of neutrophils. Most samples were also characterized by the presence of marked hypereosinophilia of basement membrane and the superficial subepithelial layer, suggesting collagen denaturation. This was usually accompanied by proteinaceous edema and neutrophils. In some cases, the gill rakers were severely affected with necrosis, complete sloughing of epithelium, and severe acute inflammation (Fig. 2). In some sections, a few jellyfish were apposed to the arch epithelium, and fine basophilic filaments emanating from the jellyfish appeared to penetrate deep into the epithelium (Fig. 3). Finally, mats of filamentous bacteria were observed on the severely ulcerated gill arch and gill rakers in a number of samples (Fig. 4). Changes to the respiratory epithelium were lamellar epithelial degeneration and necrosis, exfoliation of epithelial cells, lamellar fusion, congestion, and an infiltration of inflammatory cells, leading to overall lamellar thickening. Bacteria were present in abundance on the lips in all jellyfish samples (Fig. 5A, 5B). They were long and thin at 0.2 μm wide and therefore considered to be filamentous.
Tenacibaculum maritimum PCR results
Four samples were positive by primary PCR after 30 amplifications. These corresponded to the jellyfish sample, 2 gill extracts, and the positive controls. However, only the positive control produced a clear band of approximately 1,500 bp. Accordingly, the primary PCR product was used as a template for a second eubacterial amplification. This produced a clear band at the correct molecular weight for the jellyfish and the positive control but not for the gills. A nested PCR reaction, however, gave positive bands at the correct molecular weight (1,088 bp) for gills, jellyfish, and positive controls. No amplification products were observed in any of the negative control samples.
Bacterial sequencing
From multiple sequencing of both ends of the PCR amplicon, followed by contig assembly, g a robust 936-bp consensus sequence was produced for the positive control, as well as for gill and jellyfish samples. BLASTn analyses of positive control and gill consensus sequences produced a 100% match to the T. maritimum sequence with EMBL accession number AB078057.1 20 from base positions 233 to 1,168. Similar analysis of the jellyfish consensus sequence matched at 935 of 936 bases (99.9% match), the only mismatch being a C to T transition at base position 362 in the published sequence. 20
Discussion
The findings show that the jellyfish were carrying filamentous bacteria and that at least some of these were T. maritimum. It is not known where the bacteria on the gill arch originated, whether from the water or from the jellyfish, but the close association between the jellyfish and the lesions on the gill arch suggests that the bacterial lesions were not merely the result of secondary infection of an exposed and vulnerable tissue. The findings also suggest that the jellyfish were responsible for carrying bacteria to the gill arch and infecting with T. maritimum the tissue into which they were injecting toxins. In a species that has been shown to exhibit genetic variability, 3 the virtually identical gene sequences of the T. maritimum from gill and jellyfish (a single bp difference) support this suggestion. In future outbreaks, sampling of fresh gills and jellyfish would allow recovery of bacteria and direct comparison of isolates.
There is little doubt that the observed mortality in the salmon can be attributed to the gill lesions, but it is less certain whether these were due to the direct action of the jellyfish, the subsequent bacterial infection, or some combination of both. The severe inflammatory response that accompanied the major lesions of ballooning degeneration and denaturation (necrobiosis) of basement membrane and superficial dermal collagen suggests the possibility of complement activation due to injected jellyfish toxins, but these lesions by themselves would not necessarily have led to respiratory compromise, unless inflammatory cytokines and/or jellyfish toxins were carried by the circulation into the filaments and lamellae, with resulting changes in blood flow. By contrast, the observed lamellar epithelial changes would probably lead to turbulence, increased diffusion distance, and hypoxemia. Tenacibaculum maritimum is known to possess aggressive proteolytic enzymes, 26 so colonization of the gills by these bacteria would rapidly lead to further severe gill damage.
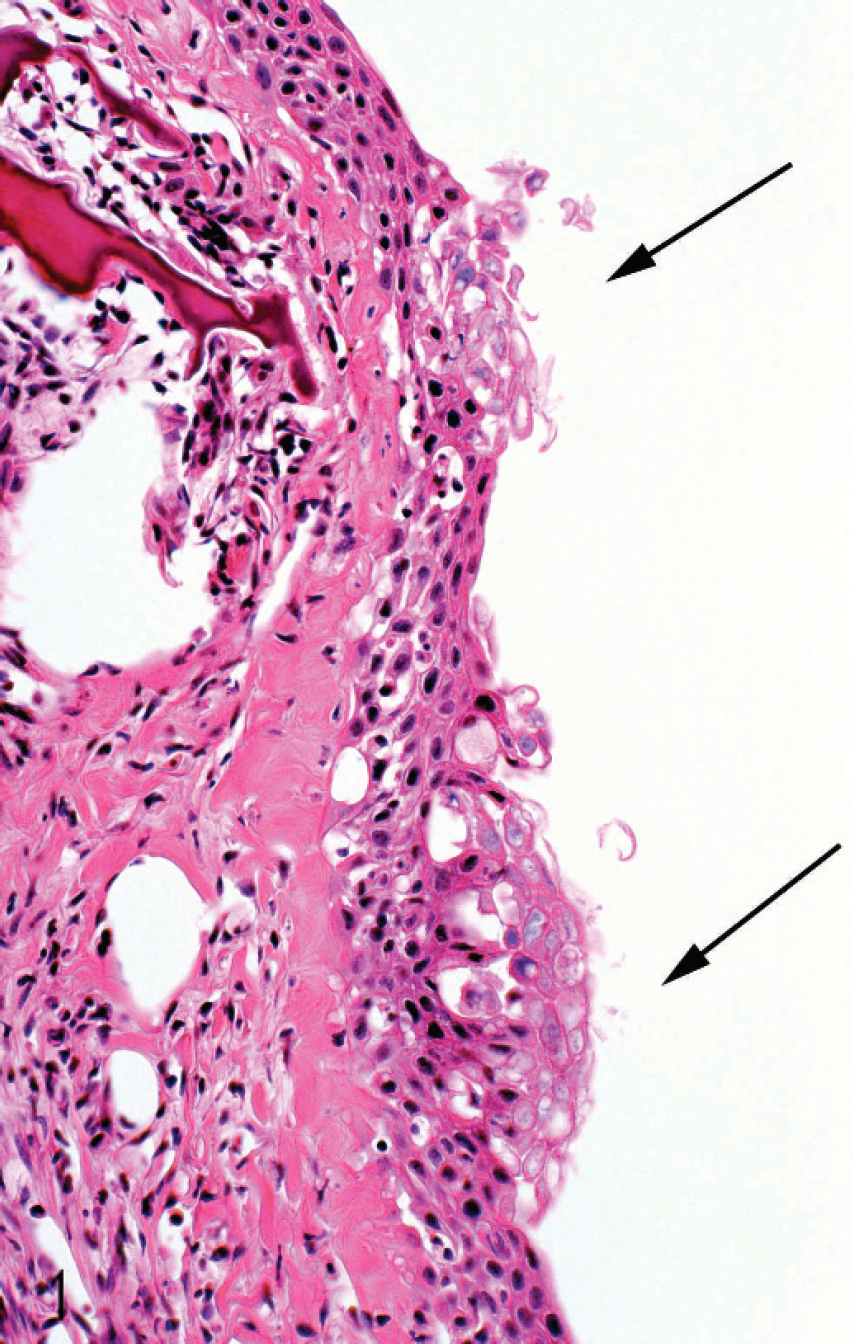
Micrograph of section of gill arch from moribund salmon showing 2 foci of acute hydropic and ballooning degeneration of arch epithelium (arrows). Hematoxylin and eosin.
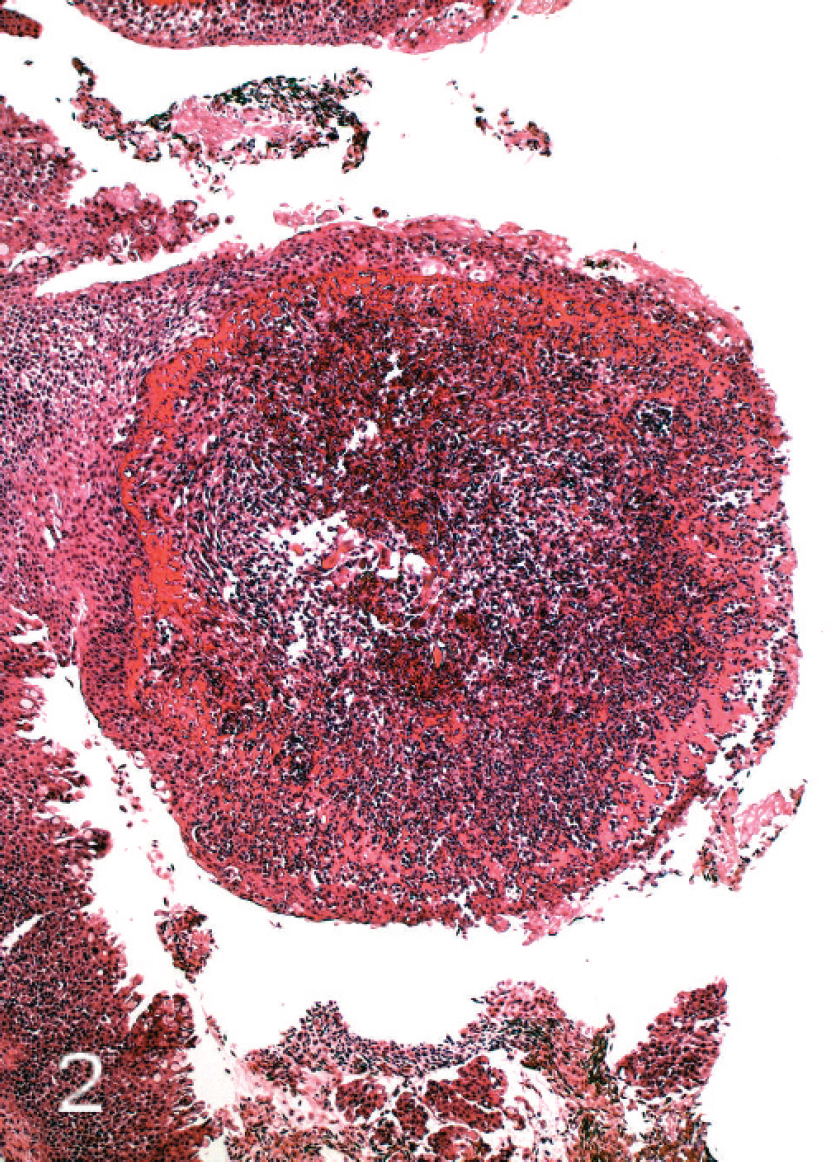
Micrograph of severely affected gill raker showing complete loss of epithelium and marked necrosis. The pronounced hypereosinophilia represents necrobiosis of basement membrane and superficial dermal collagen; in addition, there is pronounced inflammatory cell invasion. Hematoxylin and eosin.
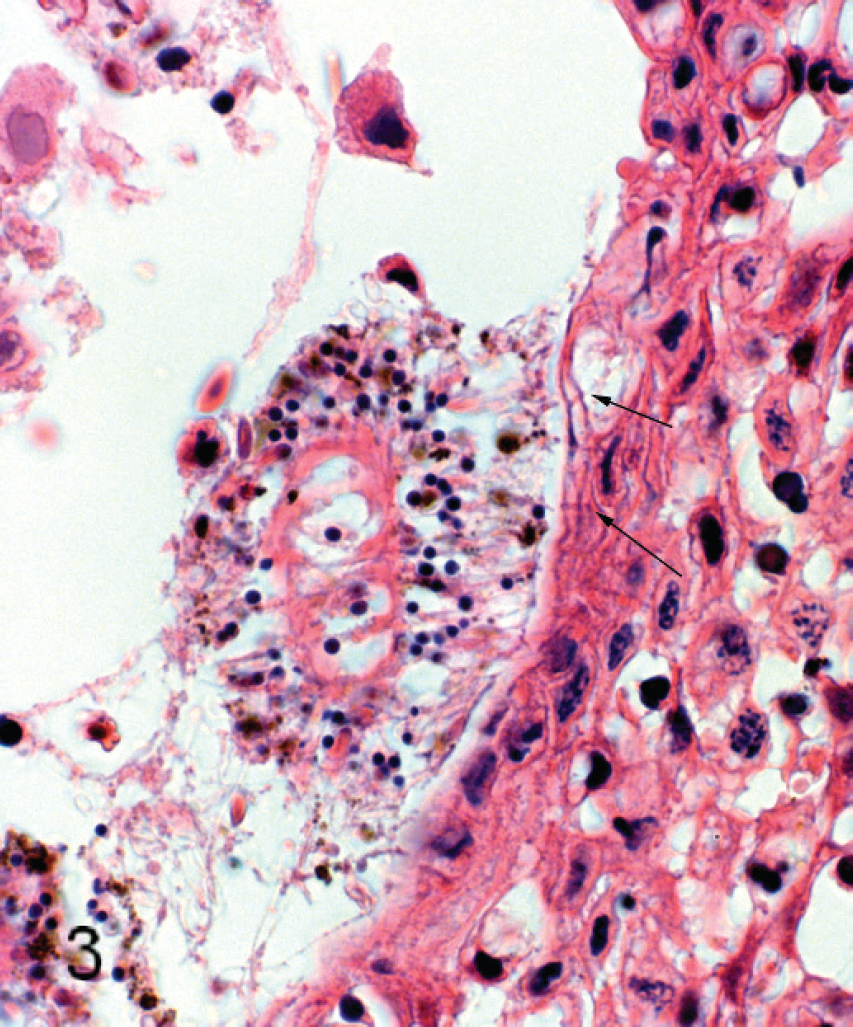
Micrograph of salmon gill arch showing jellyfish apposed to epithelium. Basophilic filaments (possibly discharged nematocyst tubes) from the jellyfish penetrate the spongiotic epithelium (arrows). Hematoxylin and eosin.
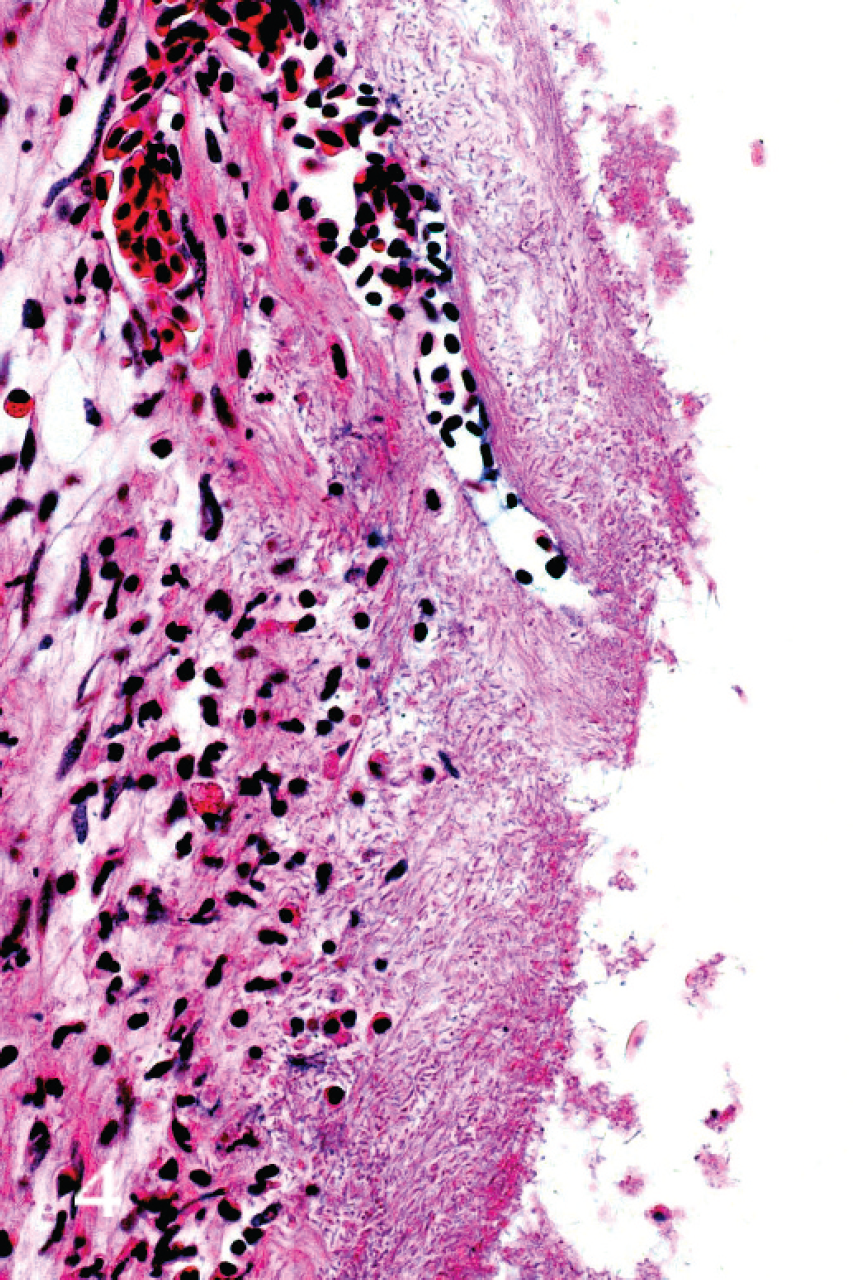
Micrograph of gill arch from moribund salmon showing severely ulcerated epithelium. Large numbers of filamentous bacteria are present throughout. Hematoxylin and eosin.
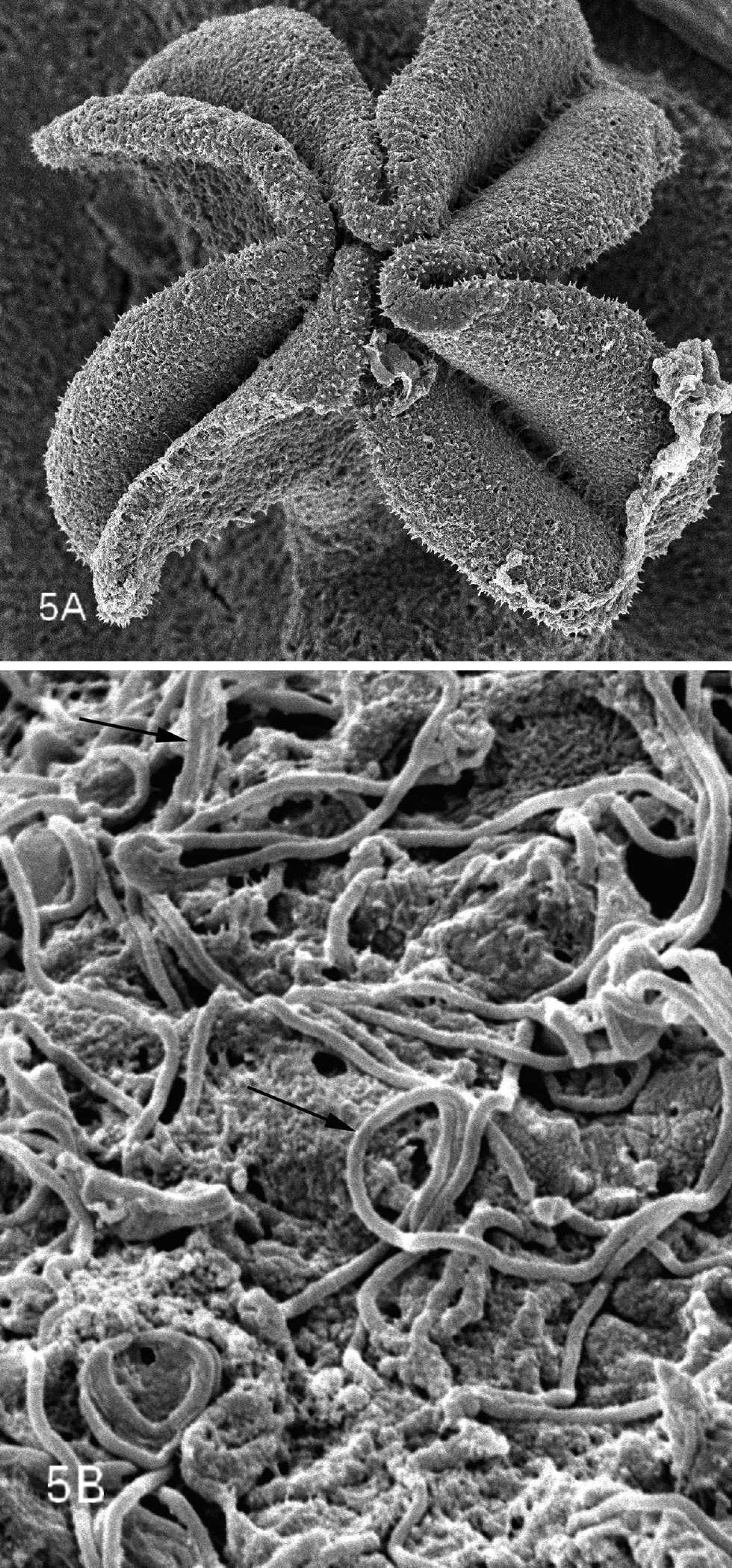
Low- and high-power scanning electron micrographs of mouth region (
Tenacibaculum maritimum (formerly Flexibacter maritimus) is the causative agent of marine tenacibaculosis in fish. Tenacibaculosis is primarily a skin infection causing ulcerative dermatitis 12 in a range of commercially important species worldwide. 2 The bacteria have also been isolated from the gills in salmonids, where a necrotizing branchitis occurs, although the arch is not usually involved, as it was in this case. 12 This serious pathogen has already been isolated from Atlantic salmon in 3 countries: Canada, Spain, and Australia. 12,24,26 Tenacibaculosis has been reported previously in Scotland but only from Dover sole (Solea solea). 18 The present study constitutes the first report of tenacibaculosis in Atlantic salmon in Scotland. It also suggests that jellyfish may act as a reservoir of infection for farmed fish, especially for this bacterium, which has been shown to survive poorly in seawater alone.
The results raise several interesting questions. First, what is the relationship between the bacteria and the jellyfish? Are they a pathogen of the jellyfish, or are they present in a symbiotic or commensal capacity? Jellyfish do not have a “typical” digestive tract but instead variably complex gastric cavities and canals. These are usually ciliated and provided with secretory cells that release exoenzymes. Predigestion in the gastric cavities is followed by endocytosis of food particles and intracellular digestion in the gastrodermal cells. In the case of a small and simple medusa, such as P. quadrata, there is only a simple elastic stomach cavity linked to the radial and circular canals. 4 The presence of a population of proteolytic enzyme-producing bacteria on their mouths could therefore aid in predigestion of prey; in this case, the salmon gills were possibly the target. It is not known whether all P. quadrata harbor these bacteria or whether this is peculiar to this particular case. Nor do the authors of the present study know their source; maybe the salmon themselves supplied the bacteria for the jellyfish!
Bacteria have long been known to be associated with feeding, gastrodermal, and tentacle cells and mucosal secretions in other cnidarians, notably coral polyps. 22 Coral mucus harbors specific populations of bacteria. It is likely that such bacteria play a role in cnidarian immune defenses as well as in their nutrition. However, no prior literature describing the presence of bacteria associated with medusae could be found, other than some studies concerned with their decay. As major predators in all marine ecosystems, jellyfish provide body surfaces, mucous secretions, and egestion and excretion products that are probably substrates for the activities of marine bacteria. Do other species of jellyfish carry such bacteria, and how do they manage to resist becoming themselves damaged by their proteolytic or other enzymes?
Aside from moving the cages or fish to unaffected water, there are few practical suggestions for ameliorating mortality in sea-caged salmon once a bloom of jellyfish occurs. The results of the current study do suggest, however, that approaches aimed at reducing bacteria on the gills may help to prevent one consequence of jellyfish attack, namely tenacibaculosis. Incorporating antibiotics in the feed may not be practicable, because fish often stop feeding once jellyfish appear, so topical bath disinfection is possibly the only avenue open.
In recent years, there has been increasing concern about the potential causal relationship between humans' impact on the oceans and large concentrations of jellyfish. 25,29 Overfishing and resulting depletion of top predator fish communities are estimated to be 90% in coastal regions worldwide. 19 Many of these commercially important fish feed on the same plankton food as jellyfish, 4,5,28 and their dramatic decline would therefore probably improve conditions for jellyfish and open up food resources. 13 More food availability for the life stages of jellyfish (hydroids and medusae) has been correlated with an enhanced number of jellyfish that are budded from polyps, 34 as well as an increase in sexual reproduction. 16 Increased food availability for jellyfish can also be caused by coastal marine eutrophication, which can lead to an increase in populations of zooplankton. 23 Moreover, eutrophication is often associated with low dissolved oxygen levels and increased turbidity, conditions more favorable to jellyfish than to energetic visual predators, such as fish. 29 Finally, oceanic warming, caused by climate change, is incriminated in the increase in jellyfish numbers. It has been shown that warmer temperatures increase asexual reproduction and the ratio of jellyfish to polyps in some hydrozoan species. 27 Temperatures of the oceans are expected to rise during the next decades, 15 and it would seem, therefore, that a more gelatinous future can be anticipated.
Acknowledgements
The authors thank Drs. John Taggart and Gavin McKinley for help with sequencing.
Footnotes
a.
DNeasy™, Qiagen, Crawley, UK.
b.
Nanodrop ND-1000, Labtech International, East Sussex, UK.
c.
Ready-To-Go™ PCR beads, Amersham Pharmacia Biotech, Amersham, UK.
d.
Biometra T gradient™, Goettingen, Germany.
e.
Illustra GFX™ PCR, GE Healthcare, Little Chalfont, Buckinghamshire, UK.
f.
Beckman Coulter, High Wycombe, UK.
g.
Seqman II, DNAStar Inc., Madison, WI.
