Abstract
The objective of the current retrospective study was to describe naturally occurring type A botulism in horses in the United States. In the past 10 years, the Botulism Laboratory at the University of Pennsylvania's School of Veterinary Medicine has identified 3 isolated cases and 8 outbreaks of type A botulism in horses via samples positive for Clostridium botulinum type A toxin or spores using the mouse bioassay test. Additional information was obtained by review of submission forms and by telephone or email interviews. Almost all type A cases and outbreaks occurred in the western United States, with Oregon and Idaho overrepresented. Type A toxin was identified in only 1 outbreak; all other identified cases and outbreaks were positive for spores but not preformed toxin. Reported clinical signs included progressive muscle weakness, recumbency, decreased tail and/or tongue tone, dysphagia, respiratory distress, and death. Isolated cases involved foals ≤1 month of age; outbreaks involved horses ≥11 months. One hundred and nineteen horses were potentially exposed to the toxin source; 54 out of 119 showed signs of botulism, and 49 out of 54 affected horses were confirmed dead. The number of horses affected per outbreak ranged from 2 to 24. The source of infection was confirmed to be hay or silage in 6 out of 8 outbreaks and was unknown in 2 out of 8 outbreaks. The present report is the first description of outbreaks of type A botulism in horses and has important implications for prevention and treatment. Based on these findings, type A botulism should be considered in suspect cases of equine botulism in the western United States.
Introduction
On a weight basis, the Clostridium botulinum neurotoxin (BoNT) is the most potent and lethal toxin known to mankind. 10 Although BoNT has never been used successfully as a bioterrorism agent, its potential for this use has generated significant interest and research into the disease it produces, its mechanism of action, and strategies for its detection, treatment, and prevention. 3 Natural intoxication has been well-documented to occur in people and animals that have the misfortune of being exposed to BoNT, primarily through contaminated food. 17 There are 8 different serotypes (A–G, with C-α and C-β subgroups) of C. botulinum, a spore-forming, anaerobic, Gram-positive bacterium, 12 each of which is distinguished by the unique properties of its toxin. These toxins are antigenically different and act specifically on various parts of the synaptic vesicle docking and fusion apparatus to prevent release of acetylcholine. 25 Regardless of botulinum toxin type, the end result of intoxication is the same: progressive flaccid paralysis and cranial nerve deficits such as dysphagia, weak tongue tone, weak eyelid tone, and slow pupillary light reflexes. 26 These signs, collectively known as botulism, have been recognized in many different species, including human beings, farm animals, companion animals, and birds. 17
Botulism in horses can be categorized in 2 ways; the first is by the C. botulinum serotype involved. Certain serotypes are prevalent in certain geographic regions; in the United States, type B is endemic in mid-Atlantic states and Kentucky, while type A is thought to occur in the western states. 24,32 In the United States, reports indicate that horses are affected most commonly by type B (>85% of cases) and occasionally by types C and A. 32 Confirmed cases of types D–G have not been reported in horses. 32 Equine botulism can also be categorized by route of acquisition of the disease. Horses with botulism almost always acquire the disease in 1 of 3 ways: ingestion of preformed toxin with food by adult horses (known as food-borne botulism), ingestion of C. botulinum spores that subsequently germinate in the gastrointestinal tract of foals and elaborate toxin (known as toxicoinfectious botulism), or contamination of wounds with C. botulinum and subsequent growth of the bacteria with toxin release (known as wound botulism). 32 One case of iatrogenic botulism during an experimental trial using botulinum toxin type B in horses has been reported, but this is not a common means of infection. 1 Of the 3 major routes of infection, toxicoinfectious (in foals) and food-borne (in adults) are most common. 32
Cases and outbreaks of equine botulism have been associated with specific routes of acquisition, age groups, geographic regions, and serotypes. Toxico-infectious botulism is most commonly seen in foals in the United States in the mid-Atlantic region and Kentucky and almost always involves C. botulinum type B. 24 Food-borne botulism in adult horses most commonly results from spoiled or moldy forage (hay, haylage, or silage) contaminated with C. botulinum type B, and several outbreaks have been re-ported. 11,22,33 Clostridium botulinum type C has been reported less frequently in adult horses, with forage contaminated by carrion implicated as the cause in most cases. 14
Based on known serotype prevalence in a geographic region, hypothesized route of exposure, and, potentially, on clinical signs, equine practitioners can make an educated guess as to the most likely serotype affecting a horse with suspected botulism. However, the authors believe that distinction between serotypes must be made in the diagnostic laboratory as clinical signs may not be specific for type, and limited information is currently available regarding potential differences between types. Type B is the most common cause of equine botulism, so the classic description of signs is based on disease caused by type B and includes generalized muscle weakness; dyspha-gia; decreased tail, eyelid, and tongue tone; mydriasis; prolonged pupillary light reflexes; recumbency; respiratory failure; and death. 32 Two reports of type C botulism in horses describe differences between witnessed signs and those reported for type B; one mentions prominent mydriasis early in clinical disease and labored breathing with prominent abdominal lift later in clinical disease, 31 while in the other case, the affected horses did not demonstrate dysphagia. 14
To the authors' knowledge, descriptions of outbreaks of C. botulinum type A in horses have not been published. Therefore, potential differences in epidemiology and clinical signs have not been reported. This fact is somewhat surprising given the predominance of C. botulinum type A in human botulism. 6 The purpose of the present manuscript is to describe the epidemiology, clinical signs, and other salient features of equine botulism caused by C. botulinum type A.
Materials and methods
Records of test results for samples submitted to the University of Pennsylvania's School of Veterinary Medicine Botulism Laboratory (Kennett Square, PA) during the last decade (1998–2008) were reviewed retrospectively. The inclusion criteria was a suspected equine case or outbreak of botulism with at least 1 sample positive for C. botulinum type A toxin or spores using the mouse bioassay test. Suspected cases were defined as horses of any age that displayed 1 or more signs suggestive of botulism (progressive weakness, decreased tongue tone, decreased tail tone, dysphagia, recumbency, respiratory distress, decreased pupillary light reflexes) for which no other disease was confirmed. Outbreaks were defined as 2 or more cases attributable to the same toxin source that occurred at the same time on the same farm. Once horses meeting the inclusion criteria were identified, additional information was obtained by review of submission forms and by telephone or email interviews with referring veterinarians and owners.
The mouse bioassay was performed following Centers for Disease Control guidelines. 6 Samples included forage (hay, haylage, or silage), gastrointestinal contents, and feces. In brief, 2–5 g of sample were diluted 1:2 (w/v) with sterile gelatin phosphate buffer (0.2% gelatin, 0.4% Na2PO4; pH 6.4), homogenized, and then refrigerated at 4°C for at least 3 hr to allow elution of toxin (if present) from solid material. The sample was then divided into 2 aliquots, one of which was used to detect preformed toxin and one of which was used to detect C. botulinum spores. The first aliquot was centrifuged at 12,000 × g for 10 min, and the supernatant, containing any preformed toxin, was collected and injected intraperitoneally into 2 Swiss–Webster mice weighing 20–30 g each (0.4 ml/mouse). A 1ml sample from the second aliquot of original sample was inoculated into a tube containing chopped meat–glucose–starch (CMGS) media. The tube was degassed prior to inoculation by boiling for 10 min while vented and then cooling while sealed. The samples in CMGS media were transferred to an anaerobic chamber (88% nitrogen, 7% hydrogen, 5% carbon dioxide) at 37°C for 4–7 days. Culture sample tubes were vented for 10 min to allow gas equilibration and then sealed. After the incubation period, the culture samples were vortexed to remove toxin from the chopped meat particles, and then the liquid portion was transferred into tubes containing 4 ml of gelatin phosphate buffer. The resulting mixture was centrifuged at 12,000 × g for 10 min, and the supernatant, containing any toxin elaborated by C. botulinum during the culture period, was collected and injected intraperitoneally into 2 mice (0.4 ml/mouse).
Inoculated mice were carefully observed for signs of botulism, including labored abdominal breathing, limb weakness, respiratory distress, and death. Mice that showed signs compatible with botulism were euthanized in a carbon dioxide chamber for humane reasons. Development of signs after inoculation with supernatant from the first aliquot (which was not cultured) indicated presence of preformed toxin in the original sample. Development of signs after inoculation with supernatant from the second aliquot (which had been cultured) indicated presence of C. botulinum in the original sample.
Clostridium botulinum serotype was determined by adding 0.25 ml of serotype-specific monovalent antitoxins (most commonly types B, A, and/or C) to 1-ml aliquots of samples prior to inoculation. The mixtures were vortexed, and 0.4 ml of the mixture was injected into each of the 2 mice. If mice inoculated with sample showed signs of botulism but mice inoculated with a specific sample and antitoxin mixture did not, the C. botulinum serotype was determined to be the same as the protective antitoxin. Mice surviving the 4-day bioassay were humanely euthanized in a carbon dioxide chamber.
Results
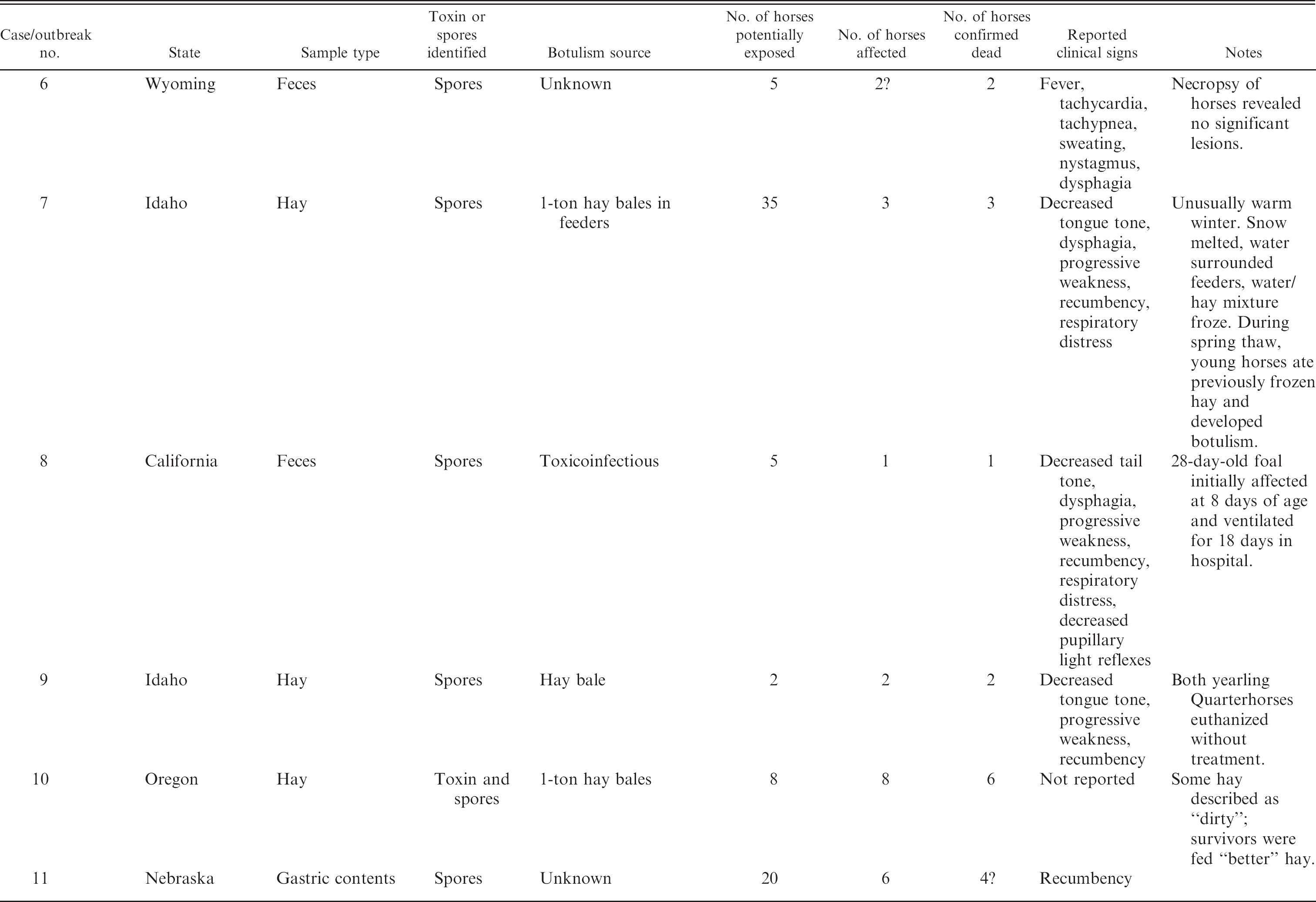
Three isolated cases and 8 outbreaks were identified (Table 1). Samples testing positive for botulism included forage (hay or silage) for 6 cases, gastrointestinal contents for 3 cases, feces for 3 cases, and an umbilical abscess for 1 case. The isolated cases were foals in the first month of life. Wound botulism was identified in a 20-day-old Thoroughbred colt from Florida (no. 3) with an umbilical remnant infection positive for C. botulinum type A spores. The other 2 cases were a 24-day-old foal from Washington (no. 2) with a short duration of illness and a 28-day-old foal from California (no. 8) affected at 8 days of age. Both foals presumably had toxicoinfectious botulism based on fecal samples positive for type A spores. All foals were treated with botulism antitoxin (but the type of antitoxin and whether it was monovalent type B or pentavalent types ABCDE was not always known), antibiotics, and supportive care. Despite treatment, which was prolonged and intensive for the California foal (18 days on a ventilator), none of the foals survived.
Outbreaks occurred on variably sized farms located in the northwestern to central United States: 2 in Oregon, 3 in Idaho, and 1 each in Montana, Wyoming, and Nebraska. The number of horses per farm potentially exposed to the presumed toxin source ranged from 2 to 40 (mean: 14.9, median: 6.5). The number of horses per farm displaying clinical signs of botulism ranged from 2 to 24, yielding a case attack rate of 9–100%, with a mean of 67.4% and a median of 80%. The number of horses that died or were euthanized per farm ranged from 2 to 24, with a case fatality rate of 66.7–100% (mean: 90.2%, median: 100%). The source of botulism toxin was identified as forage (hay or silage) in 6 out of 8 outbreaks but was unknown in 2 out of 8 outbreaks. All affected horses showed some combination of the following clinical signs: progressive weakness, recumbency, dysphagia, respiratory distress, decreased tail tone, decreased tongue tone, or mydriasis and/or decreased pupillary light reflexes. In several outbreaks, some of the horses were found dead without showing premonitory signs.
Information regarding the circumstances surrounding the outbreaks was available for 7 out of 8 farms. The largest outbreak (no. 1) involved 40 horses on pasture in Oregon in late February. The farm owners had spread large (∼1 ton) bluegrass hay bales on the ground along a creek in the pasture, which they had done previously with no problems. However, the owners reported unusually warm and wet weather conditions for that particular February and stated that the hay became wet and matted. After the first horse showed signs of botulism, 20 horses (including the sentinel case) died within 3 days, and 4 more horses died over the next 6 days. The 16 other horses on the same pasture did not show any signs. This outbreak was the first and only time botulism occurred on the farm; the owners no longer place hay by the creek. A similar scenario occurred in Idaho (no. 7), involving 35 horses on pasture in early spring. The horses were fed 1-ton square bales of hay in large square feeders. The owner reported an unusually warm January during which the snow melted and water accumulated around the feeders, mixing with the hay on the ground prior to the ground freezing again. The owner noted that during the early spring thaw, old hay surfaced and the younger horses started eating it. Soon afterwards, 3 of the younger horses developed botulism and died within 24 hr. According to the owner, this was the second occurrence of type A botulism on the farm; a similar outbreak had occurred 10 years previously.
An initially spoiled forage source, rather than an abnormal weather pattern causing spoilage, was implicated in all the other outbreaks for which information was available. These forage sources were often noticeably wet or moldy and were described as follows: a new 1-ton bale of alfalfa hay with areas of moisture and mold; a 3-year-old silage bale; a bale of hay with a pH of 5.6; and 10 tons of approximately 1-ton square bales piled 3 rows high with “bad” hay on top that appeared moldy, muddy, and wet.
In 6 of the 8 outbreaks, type A C. botulinum spores were identified in forage sources. In only 1 of these outbreaks (no. 10) was preformed type A toxin also identified in the forage. In 2 of these 6 outbreaks (nos. 1 and 5), type A spores were identified in gastrointestinal contents as well as in forage. In 2 of the 8 outbreaks, fecal or gastrointestinal samples were positive for type A spores but the source of botulism was unknown.
Most of the affected horses in the outbreaks did not receive any treatment and died or were euthanized within days because of the severity of their clinical signs. A few affected horses were treated supportively, but none survived. Only 2 were known to have received antitoxin, but the type of antitoxin was unknown (type B vs. types A–E). A total of 54 horses were affected in the 8 outbreaks, and 49 were confirmed dead based on information obtained at the time of sample submission or during follow-up interviews. The remaining 5 horses were alive at the time of sample submission but were lost to follow-up; it is unknown whether they subsequently died or survived. Therefore, the overall survival rate of affected horses may be as low as 0% or as high as 9.3%.
Reported cases and outbreaks of type A botulism in horses.
Discussion
As a result of mandatory reporting, information about human botulism in the United States is readily available. In human outbreaks, there is a distinct geographic distribution of botulinum toxin types. There were 167 type A outbreaks from 1950 to 1996, and 86.2% (144) occurred west of the Mississippi River, with California, Washington, Oregon, and Colorado accounting for 58.7%. 6 Conversely, of the 61 type B outbreaks reported during this period, 60.7% (37) occurred in eastern states. 6 Most (83.6%) type E outbreaks occurred in Alaska. 6 These outbreak findings are in concurrence with soil surveys that demonstrated a predominance of type A spores in soil samples from western states and a predominance of type B spores in soils in the northeast and central states, 18 while type E spores have been found in marine life and in sediment from the Pacific Northwest and the Great Lakes. 5,30 Although the C. botulinum serotypes showed geographic differences, no differences in neurologic symptoms and signs were reported for toxin types. 6
Unfortunately, information regarding equine botulism in the United States is relatively limited; 9 reports describing naturally occurring cases of type B or C botulism were identified. Three reports described individual cases of toxicoinfectious or wound botu-lism. 4,16,19 Two reports described case series of type B toxicoinfectious botulism in foals from Kentucky and the mid-Atlantic region, 27,34 and 2 described outbreaks of type B food-borne botulism in groups of horses living in states east of the Mississippi River (North Carolina and Tennessee). 13,33 Finally, 2 reports described outbreaks of type C botulism in groups of horses in western states (California and Arizona) linked to carrion contamination of feed or feeding areas. 14,23
Based on the above reports and the experience of the authors of expert reviews, several conclusions about the epidemiology of equine botulism in the United States have been made. No case reports of type A have been published, but previous manuscripts have mentioned its occurrence in the western states. 24,32 Horses are affected most commonly by type B botulism, which is endemic in the eastern United States (particularly in the mid-Atlantic states and Kentucky), but only occasionally affected by types C and A. 32 Clostridium botulinum type B may cause toxicoinfectious botulism in foals or food-borne outbreaks in adult horses associated with spoiled or improperly fermented forages, in which C. botulinum may proliferate and produce toxin. 32 Type C has most commonly caused food-borne outbreaks in adult horses associated with carrion contamination of feed after C. botulinum has proliferated and produced toxin in decomposing animal carcasses. 23,24 Confirmed cases of types D–G have not been reported in horses. 32 As mentioned previously, reported clinical signs of botulism in horses are generally similar regardless of causative serotype, and the potential differences reported for type C have not been consistent among reports.
To the authors' knowledge, the current study is the first to provide information on type A botulism in horses and indicates that type A botulism may be more common in horses than previously recognized, that its geographic distribution is similar to what is reported for humans, and that its route of acquisition is similar to type B botulism. With the exception of an isolated case in Florida (no. 3) and a case in Ohio that occurred prior to this study period (unpublished data from the authors' laboratory), all cases and outbreaks occurred west of the Mississippi River in 7 contiguous states (Oregon, Idaho, Montana, Wyoming, Washington, California, and Nebraska). When known, the toxin source was forage (food-borne) or soil (toxicoinfectious), similar to type B botulism.
Additional information regarding forage sources involved in outbreaks may help to identify risk factors for type A botulism in the western United States. Environmental factors that favor C. botulinum spore germination and toxin production include low acidity (pH > 5), low oxygen, and high water content. 29 Two of the outbreaks (nos. 1 and 7) were attributed by the farm owners to abnormal weather patterns that caused forage to become unusually wet for prolonged periods of time. The implicated forage was often described as wet or moldy in the other 4 outbreaks reportedly caused by spoiled forage. Additionally, the forage source was often large bales of hay, which previous reports have listed as a risk factor for botulism in horses. 13
The reported differences in geographic incidence of type A compared with type B may assist equine practitioners to form an educated guess as to the toxin type affecting suspected clinical cases. Because of similarity in clinical signs, it remains difficult to predict toxin type based on clinical examination, though reports have suggested that horses with type C botulism may have more prominent mydriasis, more labored breathing, or less dysphagia. 14,31 These findings may help separate type C cases from type A or B cases prior to laboratory confirmation, particularly if carrion contamination of feedstuffs is confirmed or suspected. No differences in clinical signs for the type A cases described in the current report were evident when compared with previous reports of type B cases. Although accurate determination of toxin types requires laboratory testing, data from the current study indicates that practitioners in the western United States should be suspicious of type A botulism, whereas practitioners in the eastern United States should continue to be more suspicious of type B botulism.
The importance of determining toxin type is a result of the fact that antitoxin administration is considered a mainstay of therapy. 34 Antitoxin for horses is available currently in 2 forms, a polyvalent antitoxin against types A–E a and a monovalent antitoxin against type B. b The monovalent type B antitoxin is significantly less expensive than the polyvalent antitoxin and therefore may be the treatment of choice in type B outbreaks. However, in regions where type A botulism is common, the polyvalent antitoxin would obviously be preferred, as there is no significant cross-protection between types. Currently, cases of equine botulism usually progress much more quickly than results can be obtained from the mouse bioassay, forcing practitioners to treat suspect cases without the benefit of diagnostic testing results. An additional reason that practitioners should be aware of the geographic prevalence of type A and type B is that only a type B toxoid vaccine is commercially available for horses and is not cross-protective against type A botulism. 32
Another difference that may be significant between C. botulinum types A and B is case fatality rate. In human beings, type A produces more severe and fatal disease than type B, with a faster onset of paralysis, increased likelihood of requiring intubation, and longer time to recovery. 9,12 It is difficult to calculate accurate case fatality rates for adult horses with different types of botulism. Unlike in human medicine, there is no mandatory reporting, and many cases of equine botulism are never reported in the veterinary literature as evidenced by the discrepancy in the number of positive samples confirmed at the Botulism Laboratory at the University of Pennsylvania's School of Veterinary Medicine and the number of reported cases. The majority of available publications involve larger outbreaks and more severely affected horses, with inconsistent treatment protocols. Most of the adult horses in the present study did not receive antitoxin products, and it is unknown how the case fatality rate would have changed with treatment. These limitations aside, it is the authors' impression that type A outbreaks may have higher case fatality rates than types B or C. The mean case fatality rate in the outbreaks described in the current study was approximately 90%, with a median case fatality rate of 100%. One large outbreak involving type C had roughly an 80% case fatality rate, 14 whereas the cumulative case fatality rate in 3 type B outbreaks was about 70%. 11,22,33 It is possible that the case fatality rate in the present study is even higher than reported. As mentioned earlier, only 5 affected horses potentially survived (in other words, only 5 affected horses were alive at time of sample submission). These horses were lost to follow-up and cannot be confirmed to have died or survived. If some or all of these horses died after sample submission, it is possible that the overall case fatality rate could be as high as 100%.
The 90% fatality rate appears very high when compared with human data. Over the period of 1950–1996, the case fatality rate for type A botulism in human beings was 17%, and in the period limited to 1990–1996, it was 6.7%. 6 There are multiple explanations for this discrepancy. Early symptoms of human botulism include double vision, blurred vision, drooping eyelids, slurred speech, difficulty swallowing, dry mouth, and muscle weakness (Centers for Disease Control and Prevention: 2008, Botulism. Available at: http://www.cdc.gov/nczved/dfbmd/disease_listing/botulism_gi.html. Accessed August 24, 2008). In horses, these signs may be missed until the disease progresses to severe paresis of pharyngeal, laryngeal, and appendicular muscles. Therefore, recognition and treatment are likely delayed until more severe disease is present. Even if disease is recognized early, treatment may be prohibitively expensive or unavailable for a particular owner or horse. The cost of 1 dose of polyvalent antitoxin (sufficient for treatment of 1 horse) is approximately $2,500; in outbreak situations, the owner may be unable or unwilling to treat all affected horses. Furthermore, most equine veterinarians do not keep botulism antitoxin in stock and therefore would have to wait for overnight shipment prior to administration. Finally, hypoventilation is much harder to manage in adult horses than in human beings or even foals. Mechanical ventilation has contributed significantly to improved survival rates in foals; the published survival rate for treated foals with botulism (all type B when known) has risen to 96% in the most recent retrospective study, likely a result of both improved intensive care and early administration of botulism antitoxin. 34 Unfortunately, mechanical ventilation is usually not an option for adult horses with botulism because of the high complication rate, expense, and labor involved.
The current study highlights the importance of determining toxin type quickly, which remains difficult with currently available tests. The mouse bioassay, the gold standard for diagnosis, has a lag time of days to weeks. The best way to confirm botulism intoxication for a particular animal would be detection of BoNT in the blood or gastrointestinal contents. However, confirmation of botulinum intoxication in horses is difficult for several reasons: Horses are highly sensitive to BoNT and require smaller amounts of toxin to cause clinical signs than do other species 13 ; toxin present in circulation is quickly bound to receptors and internalized in motor endplates, thus becoming undetectable 13 ; and toxin remaining in the gastrointestinal tract is likely degraded by microbial organisms and their enzymes. 2 Consequently, preformed toxin is rarely found in serum or gastrointestinal contents of adult horses, as levels are usually below the detection limits of the mouse bioassay. More commonly, the diagnosis of botulism is formed when BoNT and/or C. botulinum spores are detected in forage material, with the assumption that the horse(s) consumed them. 13 Identifying C. botulinum spores in gastrointestinal contents or feces is presumptive evidence of botulism as these organisms are not considered part of the normal flora and therefore should be considered significant if compatible clinical signs are present. 32 In the present study, type A BoNT was only detected in 1 forage sample; all other cases and outbreaks were considered positive on the basis of detection of type A spores in forage, fecal, or gastrointestinal samples along with compatible clinical signs of botulism.
Currently, the mouse bioassay remains the only accepted method for detection and identification of BoNT by the Centers for Disease Control. 6 Isolation of C. botulinum from feces or gastrointestinal contents is considered good confirmatory evidence since C. botulinum is rarely, if ever, encountered in human specimens in the absence of botulism. 6,8 The identity of the organism is established using 6 criteria: lipase reaction, Gram stain, observation of spores, determination of anaerobic requirement for growth, demonstration of toxigenicity, and identification of toxin by specific neutralization. 6 The last 2 criteria require use of the mouse bioassay. The 2 major problems with the mouse bioassay are its lag time (minimum of 1 day if preformed toxin is present; minimum of 4 days if spores are present) and its live animal use. Many additional diagnostic tests for botulism have been developed recently with the goals of decreasing test time, maintaining sensitivity and specificity, and reducing animal use. Description of these tests is beyond the scope of this paper; interested readers are referred to a recent review. 15 In brief, multiple immunoassays, such as enzyme-linked immunosor-bent assays and endopeptidase assays, have been developed for toxin detection. Also, many different molecular detection protocols using polymerase chain reaction (PCR) to detect the BoNT gene have been described. To date, all tests other than the mouse bioassay are considered investigational in human medicine. 6 These tests are relevant to veterinary samples as well and in fact, the application of PCR in the diagnosis of equine 28 and bovine 7,20,21 botulism has been described. However, there are no laboratories in the United States currently performing PCR on veterinary botulism samples on a commercial basis, and development of such a test remains a future goal.
Although the current study provides important information regarding type A botulism in horses, it has limitations. The retrospective nature made some information unavailable or incomplete for some cases, and not all owners or veterinarians were available for follow-up. As cases occurred in 8 different states, many different veterinarians (and owners) were responsible for evaluating the horses and submitting samples and information. Also, individual samples from each affected horse were not submitted or tested. Horses were included in the current study if they displayed signs suggestive of botulism at the same time and on the same farm where feed samples or other horses were positive for type A botulism.
In conclusion, type A botulism appears to be more common in horses than previously assumed. Type A botulism produces similar signs to type B botulism, the most common form, and is also similar in that the toxin source is generally spoiled forage. However, its geographic predilection is different, as type A botulism should be suspected west of the Mississippi River (particularly in the northwest), while type B botulism should be suspected east of the Mississippi River (particularly in Kentucky and the mid-Atlantic region). Clostridium botulinum type A produces severe disease and may have a higher case fatality rate in horses than types B and C. Equine practitioners should be aware of this regional variation in types and subsequent importance of choosing the appropriate antitoxin product as well as the high case fatality rate in untreated horses. Further studies are warranted to characterize more fully the different types of equine botulism, and a faster, validated diagnostic test for determining toxin type is needed.
Footnotes
a.
Botulism polyvalent antitoxin, Botulism Laboratory, New Bolton Center, Kennett Square, PA.
b.
Equiplas B®, Plasvacc USA Inc., Templeton, CA.
