Abstract
Intracellular organisms in the endothelial cells of several organs of an adult great blue heron (Ardea herodias) were identified as a yeast in the family Saccharomycetales based on ultrastructural morphology and sequence data from the ribosomal RNA operon. Morphologically similar organisms of unknown identity have been described previously in Muscovy (Cairina moschata) and domestic (Anas platyrhynchos domestica) ducks.
Outbreaks of disease with high mortality in farmed Muascovy (Cairina moschata) and domestic (Anas platyrhynchos domestica) ducks in Canada and the United Kingdom have been associated with an unidentified intracellular organism. 3,4,6 Disease outbreaks were associated with access to a pond or spring. Clinical signs included lameness, weakness, ataxia, and respiratory distress. At necropsy, the lungs were edematous and coelomic effusion, pericardial effusion, and fibrin tags were present. 6 Microscopic lesions included pulmonary edema, expansion of interlobular septa with edema and inflammatory cells, and distention of endothelial cells with many 1–2-μm basophilic organisms. Fewer intracellular organisms were variably present in endothelial cells in the liver, spleen, intestine, kidney, heart, and brain. Organisms usually were not associated with inflammation; however, hepatitis, myocarditis, pericarditis, and encephalitis were seen in some birds. No significant pathogens were isolated by routine bacterial and fungal culture.
These intracellular organisms have been described as protozoa, 4 bacteria, 3 and, most recently, yeast-like organisms based on ultrastructural studies, which showed budding of the organism and positive staining with periodic acid–Schiff reaction (PAS). 6 Attempts at fungal culture resulted in a sparse growth of a yeast-like organism; however, subcultures were not viable. The identification and pathological features of a morphologically similar yeast organism in the endothelial cells of a great blue heron (Ardea herodias) are described in the present report.
An adult female great blue heron was found in Moose Jaw, Saskatchewan (50° 20′ N, 105° 33′ W), with an open compound fracture of the right wing. The heron was submitted to the Wildlife and Exotic Animal Clinic at the Western College of Veterinary Medicine, University of Saskatchewan (Saskatoon, Saskatchewan, Canada). The fracture was inoperable, and the distal wing was amputated at the metacarpus under general anesthesia. Postoperatively, the heron was maintained indoors for 5 weeks and treated with 5 mg/kg enrofloxacin and 0.1 mg/kg meloxicam daily. These medications were discontinued on day 36 of treatment. Complete blood cell counts were performed on days 1, 21, 35, and 39 and were evaluated relative to the International Species Information System 2002. On days 1 and 21, there was a left shift, which was marked by day 39. Toxic heterophil changes were present on days 21 and 39. At day 35, there was a mild heteropenia. The heron was found dead on day 40 and was submitted to the Canadian Cooperative Wildlife Health Centre (Saskatoon, Saskatchewan, Canada) for postmortem examination.
At necropsy, the heron was very thin with no subcutaneous, mesenteric, or coronary fat. One to two milliliters of yellow tenacious fluid containing fibrin strands was present in the coelomic and pericardial cavities. The intestines were adhered but were easily separated. The lungs were dark red, and fluid was released on cut section. Representative tissue samples were collected in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned at 6 μm, and stained with hematoxylin and eosin (HE) and by PAS reaction. Selected tissue sections were also stained with Perls technique. Transmission electron microscopy was performed on lung tissue reprocessed from a paraffin-embedded, HE-stained slide. 1
Standard aerobic bacterial culture was performed on lung, spleen, and kidney. Fungal culture was attempted by applying a lung tissue homogenate to Sabouraud agar and blood agar. In addition, lung tissue homogenate was incubated in trypticase soy broth, brain heart infusion broth, or Sabouraud broth, with and without discs of penicillin and gentamycin, followed by culture on Sabouraud and blood agars. Agar plates were incubated at 26°C, 37°C, and 42°C.
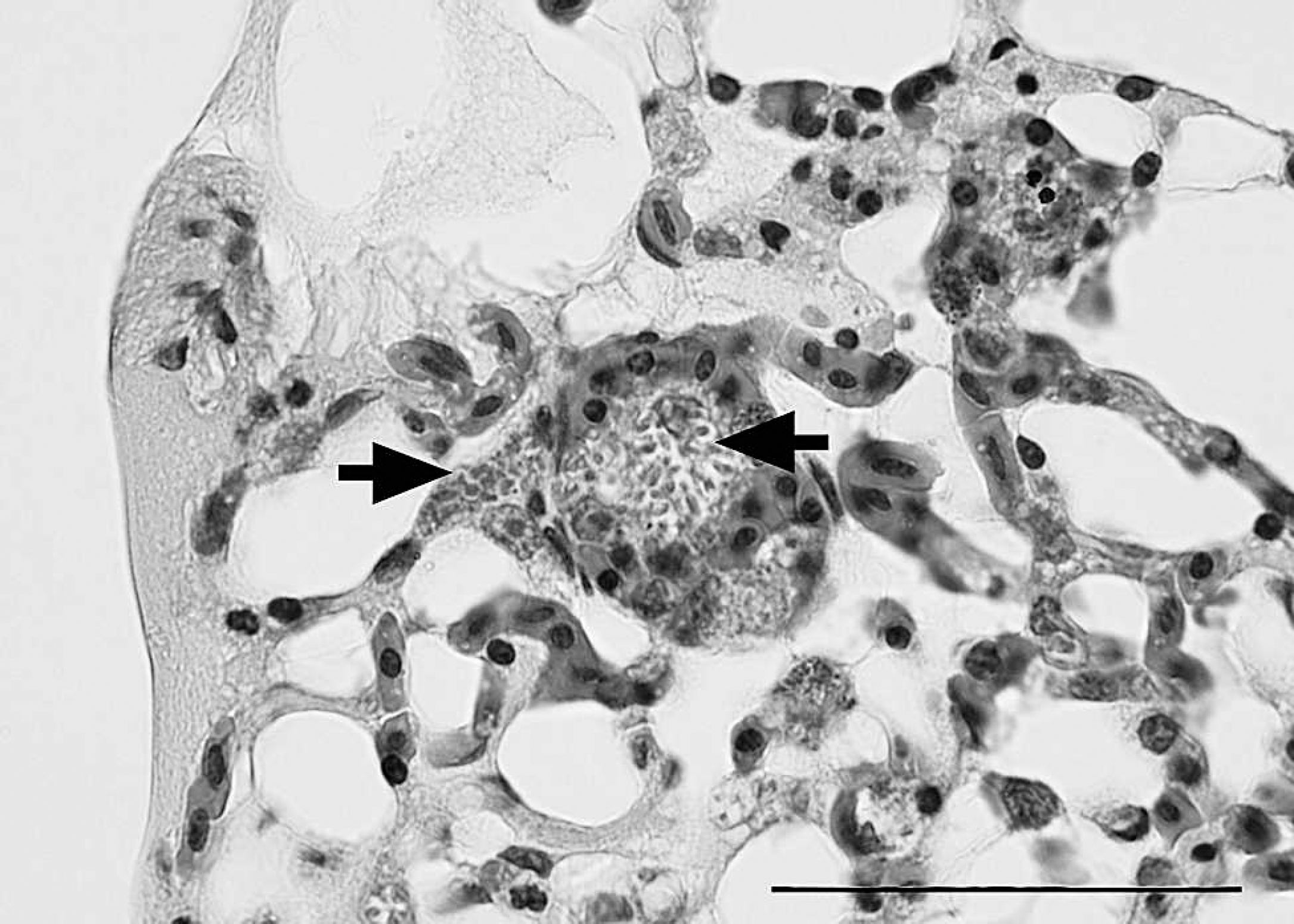
Lung, great blue heron (Ardea herodias). Septa are distended by variable numbers of yeasts within endothelial cells. Organisms (arrows) are round to oval, approximately 1 μm x 2 μm, with a lucent cell wall and a basophilic core. Hematoxylin and eosin. Bar = 50 μm.
Frozen lung tissue from the heron was homogenized, and routine DNA extraction was performed. Separate polymerase chain reaction (PCR) assays were carried out using standard fungal primers for ITS1-5.8SrRNA-ITS2 (primers ITS1 and ITS4), 9 18S ribosomal RNA (rRNA; primers NS5 and NS8), 9 and 26S rRNA (primers NL-1 and NL-4). 5 Amplicons were visualized by electrophoretic separation on a 1.5% agarose gel. Direct sequencing of purified PCR products was done, and sequence analysis included a BLASTn search (http://blast.ncbi.nlm.nih.gov/) of the National Center for Biotechnology Information GenBank database as well as phylogenetic analysis.
Microscopically, parabronchi were filled with pale, eosinophilic material, which was interpreted as proteinaceous fluid. Many endothelial cells in the alveolar septa and small vessels were expanded by basophilic round to oval structures measuring approximately 1 μm x 2 μm (Fig. 1). The cell wall of the organisms was positive by PAS reaction. The organisms were not associated with an inflammatory response, but occasional lymphoplasmacytic perivascular aggregates and low to moderate numbers of circulating heterophils were present. Air sacs were expanded by fibrin and mononuclear cells. Myocardial degeneration and necrosis were frequently associated with the intra-endothelial organisms. These areas were characterized by multiple irregularly shaped areas in the myocardium in which cardiomyocytes were vacuolated with loss of cross striations. Cellular nuclei were pyknotic or absent. Occasional foci of hemorrhage and mononuclear cell infiltrates were also observed. Rare endothelial cells in the brain, liver, and skeletal muscle were expanded by intracellular organisms without associated lesions.
Incidental findings included random, acute hepatic necrosis often associated with degenerative parasitic eggs (presumably of schistosome origin), and foci of lymphocytes, plasma cells, and macrophages, particularly in periportal areas of the liver. Kupffer cells were prominent within sinusoids and frequently contained golden brown pigment, which stained blue by Perls technique and were interpreted as hemosiderin. Moderate to severe ureteritis was associated with many trematode-type eggs in the wall of the ureter.
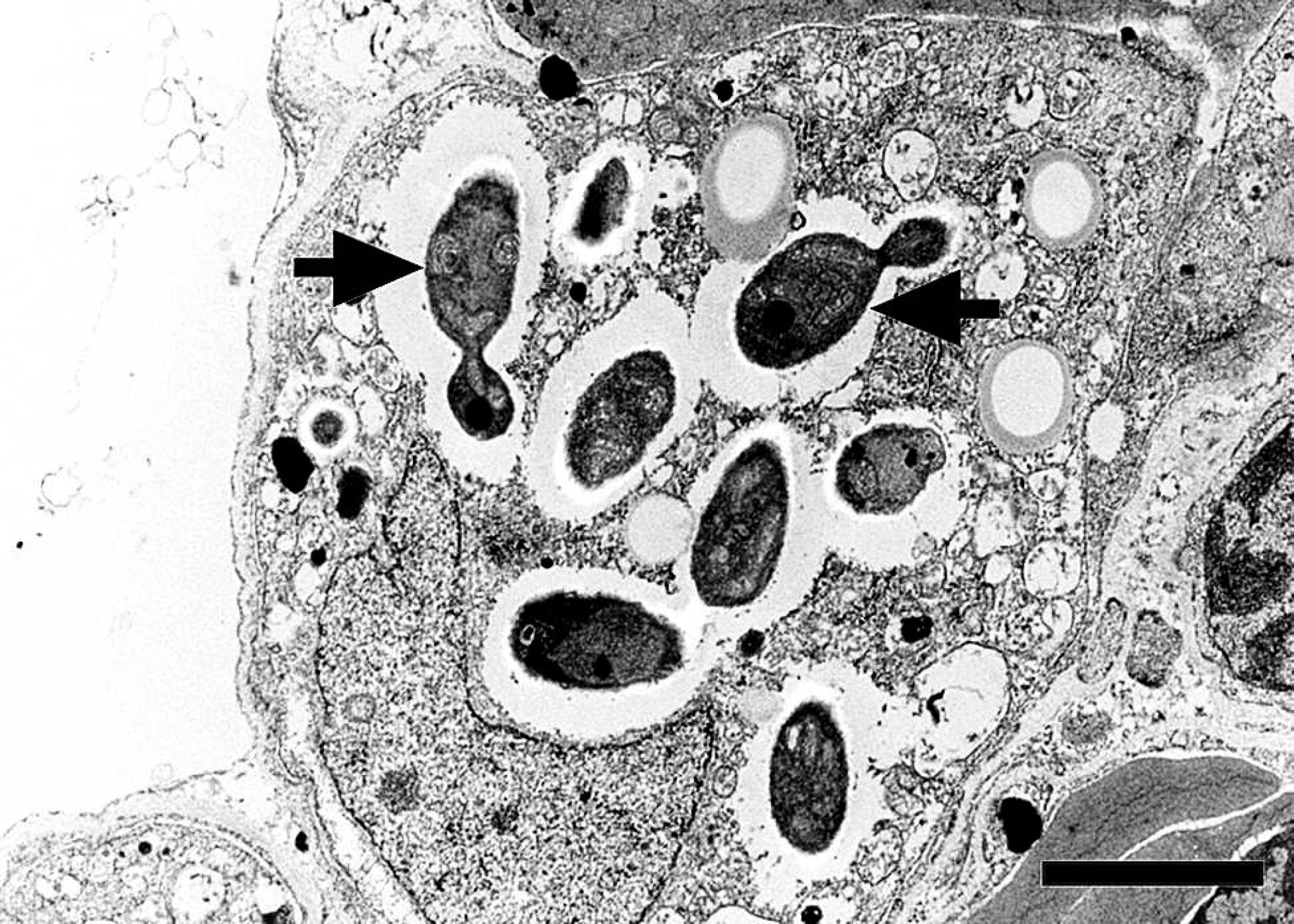
Transmission electron micrograph, lung, great blue heron (Ardea herodias). Numerous round to oval yeasts are present, some of which are budding (arrows) within the cytoplasm of an endothelial cell. Bar = 2 μm.
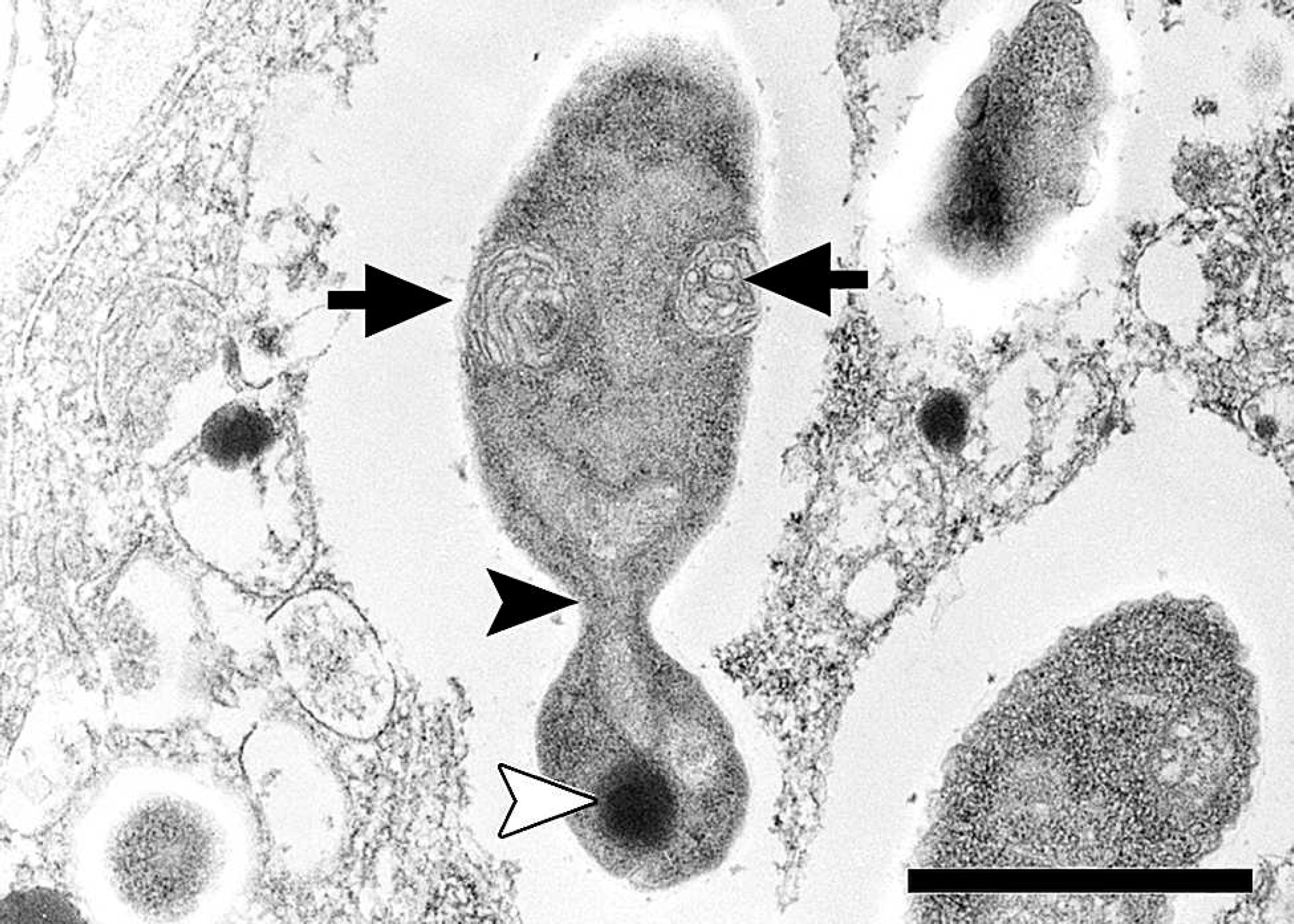
Transmission electron micrograph, lung, great blue heron (Ardea herodias). A budding yeast contains mitochondria (arrows), a nucleus (black arrowhead), and a dense body (white arrowhead). Bar = 1 μm.
The only bacterium isolated from the lung, spleen, and kidney was a sparse growth of Escherichia coli. No fungal organisms were cultured; however, the cell walls of the organisms in sections stained by PAS reaction were positive, which is consistent with glycogen as seen in fungi. Ultrastructurally, 10–100 organisms were counted within most infected endothelial cells. Some organisms had apical budding (Fig. 2). Organisms were surrounded by a lucent space and contained mitochondria, dense bodies, and a nucleus (Fig. 3). Sequences of regions of the rRNA operon from the yeast in this heron were deposited in GenBank (accession nos. FJ848337, FJ848338) and were not identical to previously published sequences. Maximum sequence matches of 90% were observed as representatives of various saccharomycetes genera, including Candida sp. Phylogenetic analysis supported placement of the organism in the Saccharomycetales family of yeasts. Identification to the genus level is problematic because of inconsistencies and lack of resolution in ascomycetous yeast phylogenies based solely on rRNA sequences. 8
The yeast organism in the heron of the current study is similar in morphology and tissue distribution to organisms previously described in Muscovy ducks, 3,4,6 and it is similar in size to unrelated pathogenic dimorphic fungi, such as Histoplasma capsulatum. 2 It is likely that this organism has a mycelial stage in the environment. 2,7 Disease in Muscovy ducks is strongly associated with aquatic environments, and a waterborne stage in the life cycle seems likely. Infection and death of Muscovy ducks occurred when the birds had access to water that was also visited by wild birds. 4 Similar organisms have been observed in a number of waterbird species from Saskatchewan, including American white pelicans (Pelecanus erythrorhynchos), lesser scaup (Aythya affinis), and Pekin ducks (Anas platyrhynchos domestica; G. Wobeser, unpublished data, 1990). Organisms in the aforementioned cases were present predominantly within endothelial cells in the lungs, and fewer organisms were present in other organs, including heart, liver, spleen, and brain.
The ultimate cause of death of the heron in the present study is unclear. The heron appeared to deteriorate rapidly on cessation of antibiotic treatment, suggesting bacterial septicemia. The scant growth of E. coli from the lung, spleen, and kidney on bacterial culture may have been associated with the antibiotic treatment. The significance of the intra-endothelial organisms is unknown; however, numbers of these organisms were highest in the lungs, and the organisms may have resulted in endothelial injury and pulmonary edema. Additionally, areas of coagulative myocardial necrosis corresponded to sites of intra-endothelial organisms. Many infected endothelial cells were distended with organisms, which might have led to capillary obstruction and ischemia.
Acknowledgements. The authors would like to thank Dr. Denni-Lynn Parker for submitting this interesting case to the Canadian Cooperative Wildlife Health Centre and for information on the case history, Dr. Manuel Chirino for his attempts to culture this yeast, and Dr. Moira Kerr for interpretation of the clinical pathology data.
