Abstract
In horses, giant-cell tumors of soft parts are rare neoplasms, with the majority of reported cases occurring within the hind limb muscles and soft tissues in older horses. The following article documents 21 cases of equine giant-cell tumors of soft parts clinically examined within the state of Colorado from 2000 to 2007. The majority of cases occurred in male horses aged 10 years or older. Nine (43%) arose within the hind limbs. Key histologic features included numerous multinucleated giant cells and hemosiderin-laden macrophages admixed with a spindle-cell proliferation. The majority demonstrated liposarcomatous change, variable areas of necrosis and hemorrhage, and an intermediate number of mitotic figures. Immunohistochemical results demonstrated 2 distinct cell populations: vimentin-expressing neoplastic mesenchymal cells and CD18 (histiocytic marker) expressing multinucleated giant cells. These results suggest a mesenchymal origin of the neoplasm with possible recruitment of the secondary histiocytic population. Surgical excision was attempted in the majority of horses and was considered clinically complete. A recurrence of the neoplasm was documented in 1 horse and 1 mule. In 18 horses, surgical excision, regardless of margin integrity, appeared successful with no recurrence of disease documented. Unfortunately, 10 of 21 horses were lost to follow-up within approximately 3 months of surgery. Of the 11 remaining horses that were available for follow-up evaluation, there has been no evidence of metastasis. A larger case series with more controlled follow-up is necessary to evaluate malignant potential and the importance of complete surgical excision.
Giant-cell tumors of soft parts (GCTSP) are rarely reported and have been described on the carpus of a Chacma baboon (Papio porcarius), metatarsi of Syrian hamsters, on the limbs of cats, on the stifle of a mule, and in multiple subcutaneous locations in horses. 2,7 In humans, GCTSP are classified as superficial or deep. 7 Deep tumors are typically large and involve the fascia, skeletal muscle, and tendons of the thigh, and have a high metastatic potential. 7 Superficial masses are often small and occur within the subcutaneous tissue and superficial fascia, and rarely metastasize. 7 In humans, these neoplasms on gross examination and histologic examination resemble giant-cell tumors of bone. 4 The most common location in humans is on the limbs. 1 In human cases, immunohistochemistry is often positive for vimentin, CD68, and tartrate-resistant acid phosphatase (TRAP), with occasional positivity for cytokeratin and smooth-muscle actin. 4 Such tumors are typically negative for CD31, desmin, CD45, and lysozyme. 4
Giant-cell tumors of soft parts are reportedly considered relatively rare in the horse. 3 In a case series of 6 horses, solid, solitary, and subcutaneous masses with varying degrees of tissue infiltration predominated and were often located on the hind limbs. 7 Previous equine case reports indicate no age, sex, or breed predilection, although affected horses are often older, with an average age of 6.8 years (range: 3–12 years). 6,7 On gross examination, superficial GCTSP are often relatively small, firm, solitary, raised, partially lobulated, and encapsulated. 6 On the cut surface, these masses are often pale and firm, with multiple small areas of hemorrhage and necrosis. 6 Previous reports of subcutaneous nodules indicate a fair prognosis with complete surgical excision and a low metastatic potential with a high chance of a recurrence when incomplete resection is performed. 2,5–7
Current theories suggest that GCTSP may be a variant of histiocytic sarcoma. 1,4,6 Previous ultrastructural examinations also suggest a mesenchymal origin. 2 The current report documents a series of 21 cases of equine GCTSP clinically evaluated in the state of Colorado from 2000 to 2007, with partial characterization of immunohistochemistry.
Affected horses ranged in age from 3 to 23 years of age, with the majority of horses (15/21) over the age of 10 years. The breeds affected were Quarter horses (7/21), mules (4/21), Arabians (2/21), Appaloosas (2/21), Tennessee Walking Horses (2/21), Peruvian Pasos (2/21), Thoroughbreds (1/21), and Saddlebreds (1/21). The high number of Quarter horses within this series most likely demonstrates geographic differences in breed populations. The majority of cases occurred in males (13/21).
The majority of these cases of equine GCTSP (43%) occurred on the hind limbs (9/21), with the remaining occurring on the flank (3/21), abdomen (3/21), muzzle (2/17), thorax (2/21), and neck (1/21). The location of 1 case is unknown. On gross examination, these neoplasms were characterized as rapid growing (20/21), dermal (17/21), and firm (20/21), with occasional areas of fluctuance (1/21). Less commonly, masses were pedunculated and pendulous (2/21), ulcerated (2/21), subcutaneous (4/21), or intramuscular (1/21), and fixed upon palpation (2/21). The neoplasms varied in size from 2 × 11 × 1 cm up to 20 × 10 × 3 cm at the time of surgical excision.
Tissues were fixed in 10% buffered formalin, routinely processed, embedded in paraffin, and sectioned at 5 μm, followed by routine staining with hematoxylin and eosin. On histologic examination, there was minimal variation among the neoplasms. Samples consisted of sheets of invasive neoplastic cells within a finely fibrillar, occasionally vacuolated to dense fibrous connective-tissue stroma. Cells were spindloid to polygonal with indistinct cell borders and abundant eosinophilic cytoplasm. Many of the specimens (9/21) demonstrated liposarcomatous change characterized by variably sized, clear, well-demarcated intracytoplasmic vacuoles (Fig. 1). There was variable anisocytosis and anisokaryosis. Nuclei were round to oval, often indented, with finely stippled-to-vesiculated clumped chromatin, and single to multiple small prominent nucleoli. The number of mitotic figures varied among samples, with the majority (16/21) demonstrating an intermediate mitotic index of 1–3 mitotic figures per 400X microscopic field. Interspersed among the neoplastic cells was a second abundant population of multinucleated cells. Multinucleated cells were polygonal with distinct cell borders, abundant eosinophilic cytoplasm, and up to 100 nuclei in some tumors (Fig. 2). Nuclei were round, with finely stippled to vesiculated clumped chromatin and small prominent nucleoli. The majority of tumors (14/21) demonstrated varying degrees of necrosis and hemorrhage. All neoplasms demonstrated varying numbers of hemosiderin-laden macrophages. In addition, occasional specimens (4/21) demonstrated moderate-to-marked numbers of associated lymphocytes and plasma cells.
Immunohistochemical evaluation of the neoplasms was performed for cytokeratin, vimentin, a CD18 b (histiocytic marker), smooth-muscle actin, a desmin, a CD31 c (endothelial cell marker), CD3 d (T-cell marker), and CD79a e (B-cell marker). Immunohistochemical staining was performed on a NexES autostainer a by using standard techniques. Briefly, 4-μm sections were cut and mounted onto positively charged slides. The sections were deparaffinized and then rehydrated with descending alcohol concentrations to buffer. Heat-induced epitope retrieval for 20 min was used for CD3 and CD79a immunostaining. Protease I epitope retrieval was performed for CD18 evaluation. A 2-hr incubation was performed with the primary antibody. Immunohistochemical staining for the CD3 antigen used a polyclonal rabbit anti-human CD3 d at a dilution of 1:300. CD79a immunostaining used a monoclonal antibody (mAb) at a dilution of 1:20. e CD18 staining used a mAb at a dilution of 1:20. b A V-Red basic alkaline phosphatase red detection kit a was used to detect the immunoreactive complexes. The slides were then counterstained with the Mayer hematoxylin. Positive control slides consisted of equine lymph node. Negative control slides consisted of the sample tissue, without addition of the primary antibody. Eighteen of 21 cases strongly expressed vimentin within the mesenchymal population (Fig. 3), and 19 of 21 cases expressed CD18 within the multinucleated giant cells (Fig. 4). Immunohistochemical stains were negative for cytokeratin, smooth-muscle actin, CD3, CD79a, CD31, and desmin. These results support a mesenchymal origin for the neoplastic-cell population, which is consistent with their histologic appearance. Potentially, the histogenesis is of a primordial or mesenchymal stem cell origin with retained capacity for differentiation, which could potentially explain the presence of multinucleated giant cells and liposarcomatous change. In addition, because of the CD18 positivity of the multinucleated giant cells, this population may represent a secondary non-neoplastic population recruited by the mesenchymal neoplastic-cell population.
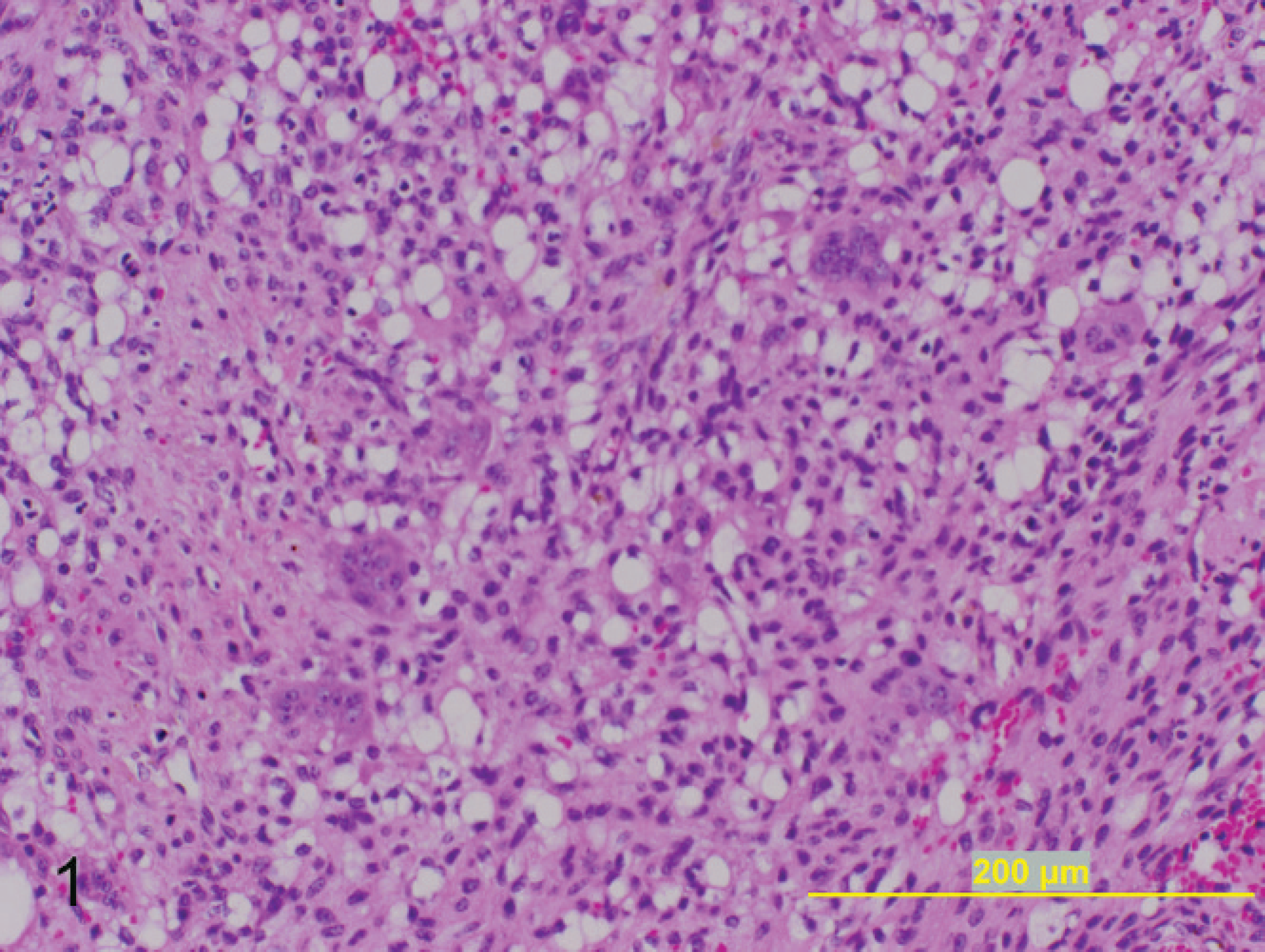
Subcutaneous mass on left lateral hind limb; 8-year-old Arabian stallion. The tumor consists of sheets of swirling neoplastic spindloid cells with marked liposarcomatous change interspersed with numerous multinucleated giant cells. Hematoxylin and eosin. Bar = 200 μm.
In 19 of 21 cases, a surgical excision was performed. A biopsy was performed on the remaining 2 cases. Surgical excision was deemed clinically complete in 18 of 19 cases. Surgery resulted in almost (often within a few millimeters) complete surgical excision in 7 of 19 cases, with incomplete surgical excision in 12 of 19 cases, as assessed by histologic evaluation. Despite incomplete surgical excision, a recurrence after surgery occurred in only 2 cases, one within 2 months and one within 5 months. The first case was treated with additional surgery on 3 separate occasions, each separated by approximately 1 month. Surgical reexcision was performed each time because of rapid recurrence of the neoplasm at the surgical site. Final surgical treatment addressed the neoplasm similar to a sarcoid with removal of the mass flush with the skin and topical application of Dermex, f an experimental herbal drug derived from a plant native to Wyoming. There was no further recurrence of this mass in the next 8 months, at which time the horse was lost to follow-up. The second case was treated with multiple cisplatin injections after the initial incomplete surgical excision. Local recurrence occurred within approximately 5 months of the initial excision, at which time a second surgical excision was performed. The second surgical excision was histologically incomplete, and a second course of cisplatin injections was performed. A primary difference in the first case of recurrence demonstrated a relatively high number of mitotic figures (5–10/400X field) and ulceration. The second case demonstrated 1–2 mitotic figures per 400X field with ulceration and extensive invasion into the external and internal abdominal oblique muscles. One case was not treated because of the horse's age and the thoracic location of the neoplasm, which would not permit primary closure after wide surgical excision. The neoplasm grossly reduced in size by half within the 1.5 years after surgical biopsy and diagnosis, and the horse continued to thrive as of September 2007.
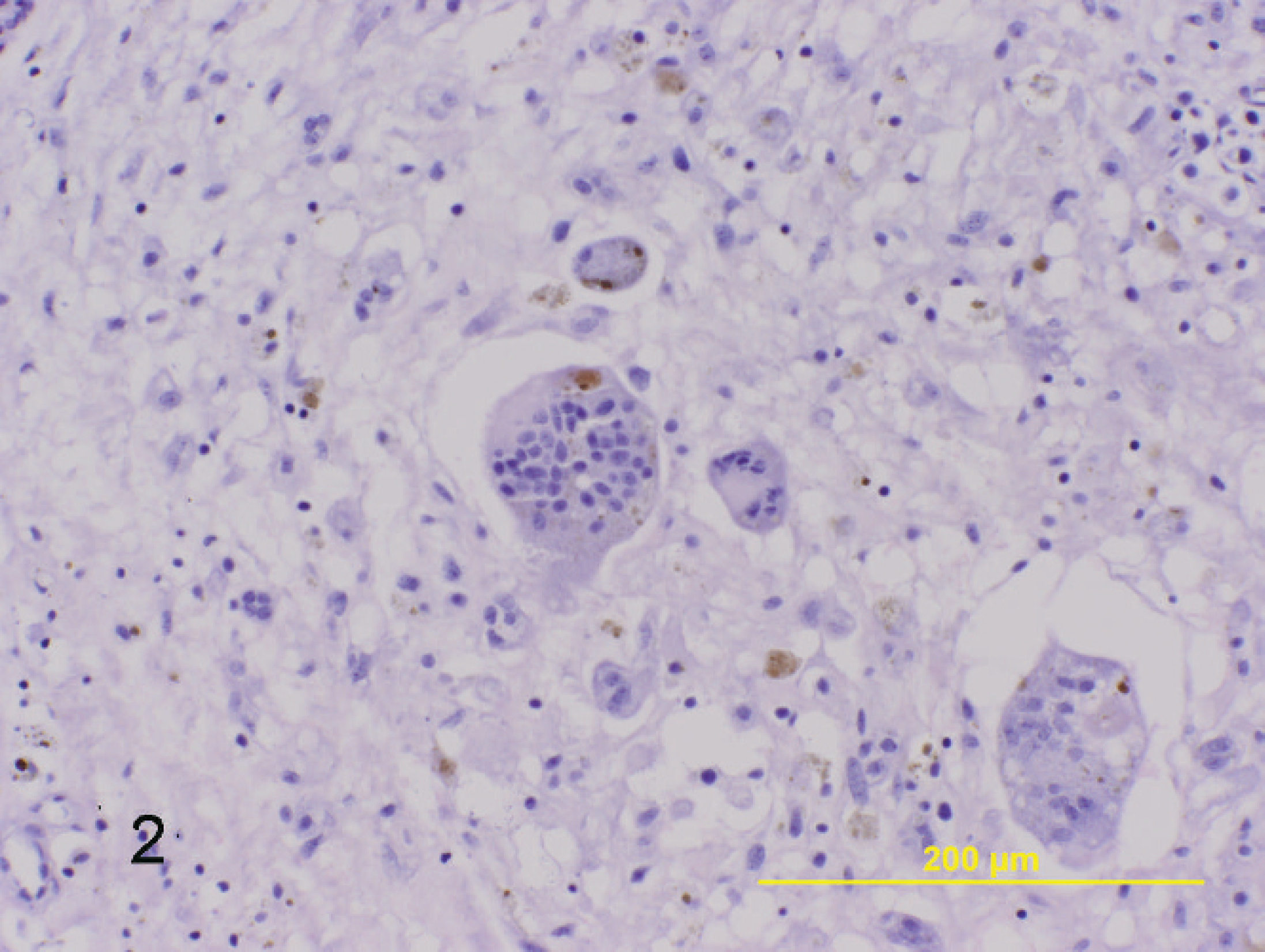
Subcutaneous mass on left lateral hind limb; 8-year-old Arabian stallion. Characteristic finding within these neoplasms are numerous multinucleated giant cells that often contain 40 or more nuclei, as well as hemosiderin-laden macrophages. Hematoxylin and eosin. Bar = 200 μm.
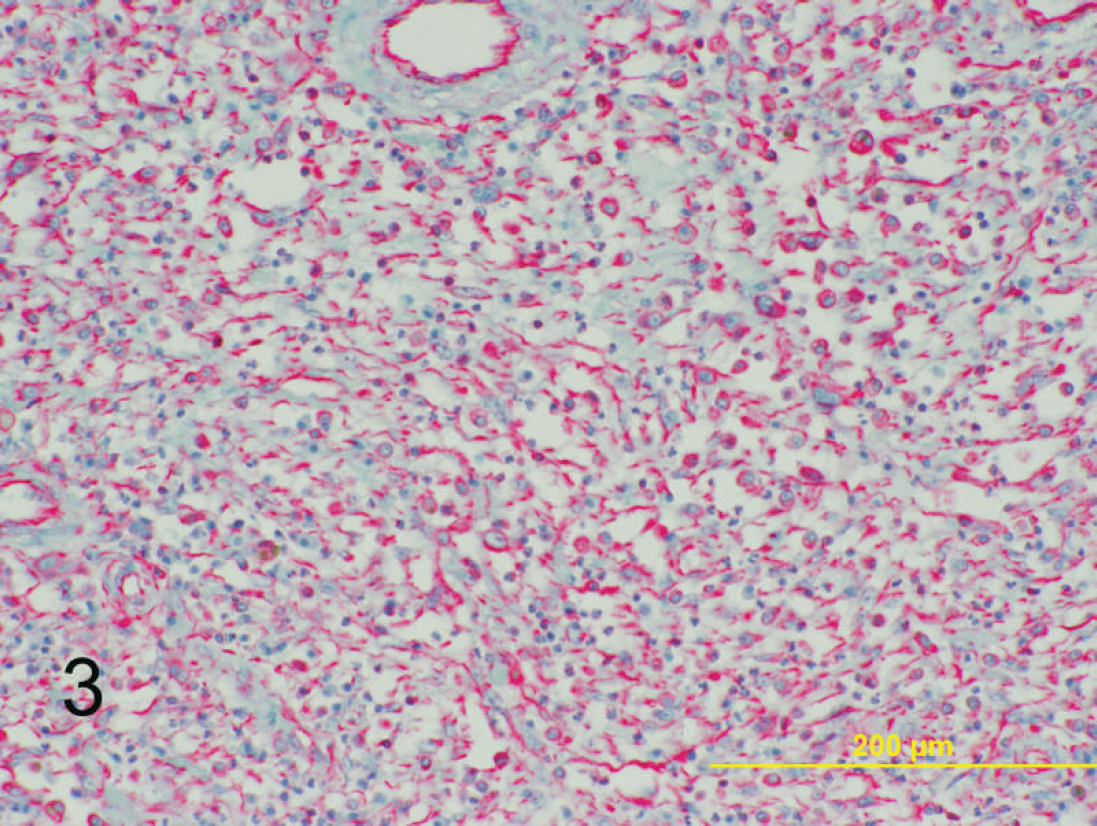
Dermal mass on left hind limb; 6-year-old Quarter horse mare. Strong positive cytoplasmic immunoreactivity is identified for vimentin within the neoplastic mesenchymal population. Basic alkaline phosphatase red and hematoxylin. Bar = 200 μm.
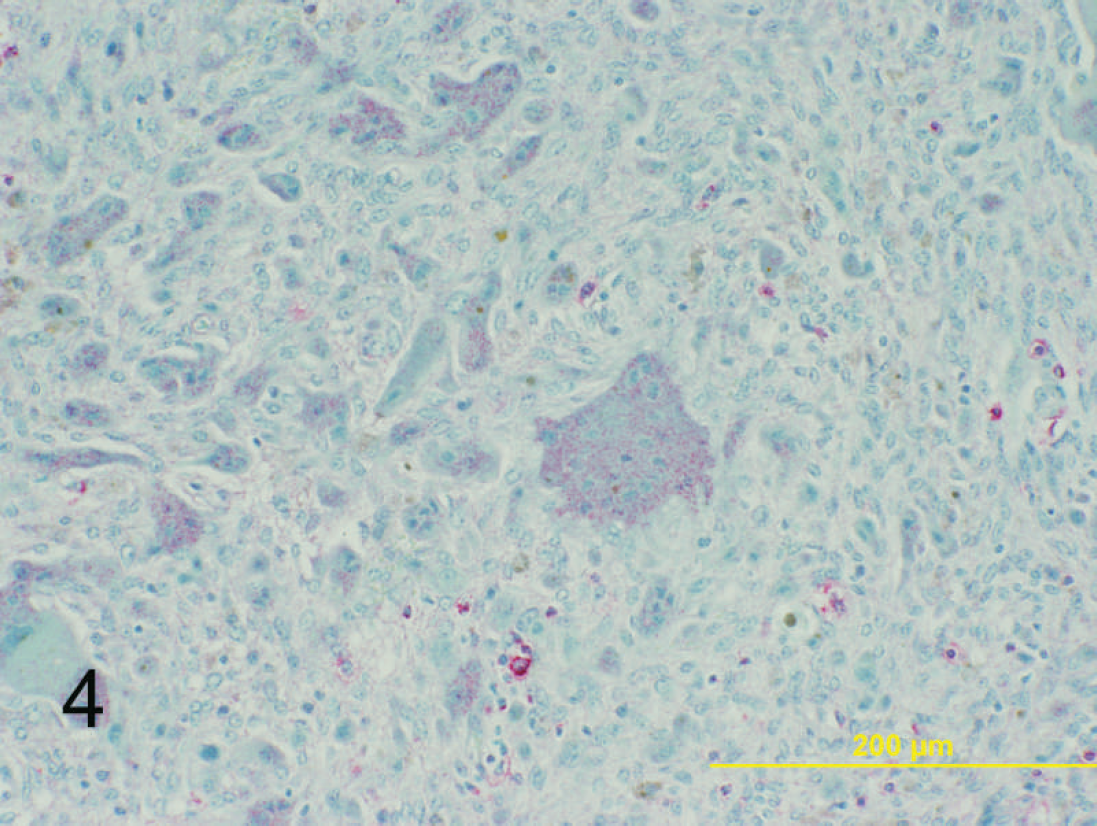
Dermal mass on left hind limb; 6-year-old Quarter horse mare. Moderate positive stippled cytoplasmic immunoreactivity is identified for CD18 within the multinucleated giant cell population. Basic alkaline phosphatase red and hematoxylin. Bar = 200 μm.
As of September 2007, 11 of the horses remained alive and healthy, although follow-up information could not be obtained on the remaining 10 because of changes of ownership. These 11 horses had, approximately 3 months to 6 years 7 months after surgery, no clinically evidence of metastasis.
To the authors' knowledge, the current case series presented the largest published series of equine GCTSP. In addition, this case series indicated that there may be a predisposition for male and older horses, as well as a tendency to occur on the hind limbs. The majority of cases were presented as firm, rapidly growing, dermal masses that were histologically similar in appearance. Common histologic characteristics included numerous multinucleated giant cells with hemosiderin-laden macrophages (21/21), liposarcomatous change (9/21), a modest number of mitotic figures (16/21), and variable areas of necrosis and hemorrhage (14/21). Immunohistochemical results demonstrated 2 distinct cell populations: vimentin-expressing neoplastic mesenchymal cells and CD18-expressing multinucleated giant cells. These results suggest a mesenchymal origin of the neoplasm, with possible recruitment of the secondary histiocytic population.
Within the 11 horses that were available for follow-up evaluation with a median time course of 3 years 3 months, there was no evidence of metastasis, despite the integrity of the surgical margins, and all 11 horses continued to do well. Local recurrence only occurred within 2 cases, and both cases were clinically ulcerated. There did not appear to be a histologic indicator useful for prognosis, because even incomplete surgical margins did not typically result in tumor recurrence. However, because many cases were lost to follow-up, further studies are required to assess the prognosis in cases of equine GCTSP.
Footnotes
a.
Ventana Medical Systems Inc., Tucson, AZ.
b.
VMRD Inc., Pullman, WA.
c.
Lab Vision Corp., Fremont, CA.
d.
Dako Denmark A/S, Glostrup, Denmark.
e.
Biocare Medical, Concord, CA.
f.
Dermex Pharmaceuticals, Parker, CO.
