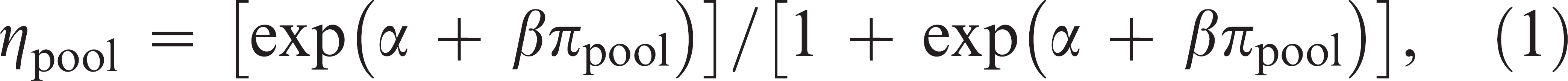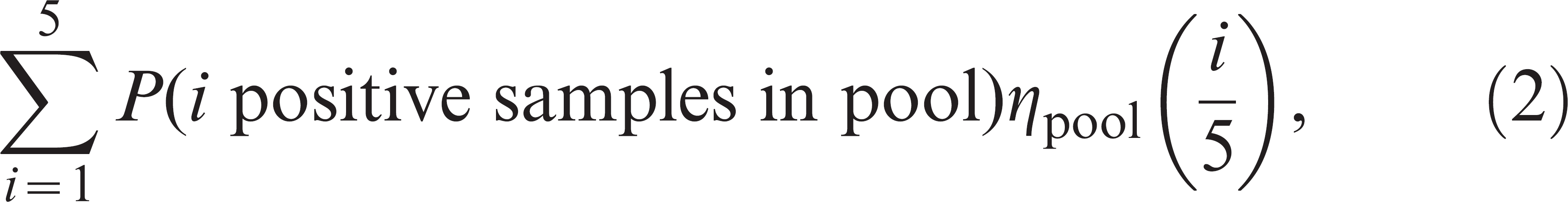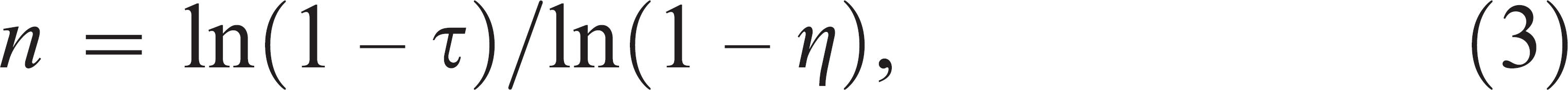Abstract
Verocytotoxin-producing Escherichia coli O157 (VTEC O157) may cause severe illness in people. Cattle are regarded as an important source of VTEC O157, and in an outbreak investigation, there is a necessity to establish whether or not the putative contact herd shares infection with the human case. The effectiveness of a herd investigation is impacted by the number of samples required, which will influence the time taken to collect samples and then process these in the laboratory. The objective of this study was to investigate the effectiveness of pooled sampling for detecting VTEC O157 in cattle herds in the United Kingdom. On farm 1, 150 individual fecal samples were collected during the course of a VTEC O157 outbreak investigation. One-gram and 10-g subsamples were tested from each individual sample. Once the culture results of the individual sample were known, pools comprising 5 and 10 individual samples were formed, with each pool containing a known number of positive samples. This data showed that the sensitivity of pooled sampling depended upon the proportion of positive samples in the pool. Further samples were collected from 2 more infected farms (2 and 3). Each individual sample was tested in duplicate. Pools of 5 feces were formed on-farm, and half the number of pooled feces were tested as individual feces. There was no significant difference between the number of cultures required for pooled sampling, as was the same for individual sampling, and therefore pooling did not improve the effectiveness of detection of VTEC O157.
Introduction
Verocytotoxin-producing Escherichia coli O157 (VTEC O157) cause human disease outbreaks in the United Kingdom every year (http://www.hpa.org.uk/infections/topics_az/ecoli/O157/data.htm). Symptoms range from mild diarrhea to hemolytic uremic syndrome, which can be fatal especially for very young children. Cattle are the main reservoir for VTEC O157; they are asymptomatic carriers and shed the bacteria in feces. 7 In England and Wales, the Health Protection Agency (HPA) can request assistance from the Veterinary Laboratory Agency (VLA) to identify possible animal sources of human outbreaks of VTEC O157 using comparative molecular typing (mainly phagetype and pulsed-field gel electrophoresis). 16 The standard procedures adopted by VLA investigators use epidemiological groups rather than individual animals, because VTEC O157 excretion in individual cattle is intermittent, whereas group-level status remains stable for longer (http://www.defra.gov.uk/science/project_data/DocumentLibrary/OZ0138C/OZ0138C_3750_FRP.doc). Verocytotoxin-producing E. coli O157 is usually widespread in fecally contaminated environments of infected animals and tends to circulate between individual animals within close proximity of each other or indirectly via other epidemiological routes. 4
Currently, during outbreak investigations, a large number (often 100 to 200 or more) of fecal samples are examined for VTEC O157 based on a 95% confidence of detecting at least 1 positive at a minimum estimated within-group prevalence of 10% within each distinct epidemiological group. Adopting a pooled approach for sample collection from epidemiological groups is potentially much more practical, cost-effective, and safer for the sampler as it does not require handling of individual cattle, provided it offers adequate statistical confidence and test sensitivity. Use of efficient sampling methods to minimize the cost and duration of an outbreak inquiry would facilitate an outbreak investigation. If costs are high, then this may discourage investigators from examining a potential farm source unless circumstantial evidence is compelling or other inquiries have failed to yield results. Thus, we consider that to be effective, an outbreak investigation should be epidemiologically robust, based on sample-size estimates being 95% confident of detecting infection if it were present at or above a threshold value in any epidemiological group (e.g., 10%). This demands that the sensitivity of the detection method be known. Furthermore, the minimum number of samples should be collected in the interests of not only economy, but also timeliness.
Pooling of fecal samples for the determination of the infection status of groups has been shown to be more efficient than the use of individual samples for Salmonella in pigs 1 and Johne's disease in cattle. 15 If validated for VTEC O157 in cattle, it offers considerable potential cost savings, and the approach could be adopted for surveys and intervention studies. One investigation into pooling fecal samples for detection of VTEC O157 in cattle has been performed; 12 however, the details of the test procedures in that study for the culture of VTEC O157 differed from those used routinely for outbreak investigations in the United Kingdom. Furthermore, in the previous study, pooling was restricted to samples pooled in the laboratory, whereas in this study samples were pooled on-farm for 2 of the 3 farms from which samples were taken, as would be carried out in practice. The objective of this study was to determine the sensitivity of pooled and individual-level sampling to determine whether pooled sampling offered reductions in the number of samples required to detect VTEC O157 on cattle farms in Great Britain.
Materials and methods
Testing of pools containing known number of positives
Samples were collected during the course of a VTEC O157 outbreak investigation undertaken by VLA at the request of the HPA. One hundred and fifty individual fecal samples were collected. On arrival at the laboratory, the samples were well-mixed and 1-g and 10-g subsamples were inoculated into 9-ml and 90-ml volumes of buffered peptone water (BPW) and incubated at 37°C for 6 to 8 hours. After incubation, the volumes of BPW were subject to automated immunomagnetic separation (AIMS) using Dynal O157 Dynabeads. a The Dynabeads were plated out onto cefixime tellurite sorbitol MacConkey's (CTSMAC) agar plates and incubated at 37°C for 18 hours. After incubation, the plates were examined for typical nonsorbitol fermenting colonies. Up to 10 typical colonies were tested by latex agglutination test b for the presence of O157. Once the individual sample results were known, a series of 1-g pools was created from the individual samples as follows:
Pools of 5 individual feces with 0, 1, 2, 3, 4, and 5 positives per pool—10 replicates per pool for the pools of 0, 1, 2, and 3 positives; 5 replicates of 4 and 5 positives. This results in a total of 50 pooled samples.
Pools of 10 individual feces with 0, 1, 2, 4, 6, 8, and 10 positives per pool—10 replicates per pool for the pools of 0, 1, 2, 4, and 6 positives; 5 replicates of 8 and 10 positives. This results in a total of 60 pooled samples.
Positive and negative samples were all selected at random from the set of test-positive samples (i.e., positive for at least 1 of the 1-g and 10-g cultures by AIMS) and testnegative samples, respectively, from the 150 samples from the outbreak farm, according to predetermined tables created using random numbers generated by Microsoft Excel. c While awaiting the result of the VTEC O157 culture, the samples were stored at 4°C, and all the pooled samples were formed and tested within a week of collection. All VTEC-positive individual samples were spiral plated 11 at the same time as the pooled samples were formed to determine the number of VTEC O157 organisms in the individual (and pooled) samples. A previous study on the survival of E. coli in naturally occurring cattle feces 11 had shown that E. coli counts start to die off in some of the pats after about 7 days, which would suggest that there would be no die-off in the interval between collection of the samples and the formation of the pools in the present study, since the pooled samples were all formed within a week of collection. The farm from which the samples were taken will be referred to as farm 1.
Testing of pools formed in the field
In order to provide data on the sensitivity of pooled samples as would be carried out in the field, samples were collected from 2 more infected farms (2 and 3). From each farm, the number of individual samples required to detect a positive at 10% prevalence with 95% confidence from each of the 3 groups of cattle was taken in accordance with the established VLA outbreak investigation protocol based on published tables for sample-size requirements. 2 Each group was a distinct epidemiological unit on the farm; that is, each group was housed separately, and all animals sampled were between 6 and 18 months of age. For each group sampled, n/2 pooled samples were collected in addition to the individual samples, where n was the number of individual samples collected. In total there were 3 sets of 22 individual samples and 3 sets of 11 pooled samples from farm 2, and 3 sets of 16 individual samples and 3 sets of 8 pooled samples from farm 3. Each pooled sample comprised approximately equal quantities (approximately 3 to 5 g) of 5 randomly selected freshly voided feces. These were collected into the same 150-ml plastic pot with a wooden spatula (the same for all samples comprising the same pool). In terms of practicality, some repeat sampling of feces from the same animals within a pen or group was considered acceptable. The pen from which the pooled samples were taken was recorded.
At the laboratory, each pooled sample and 2 1-g subsamples from each individual sample were well-mixed and 1-g removed for culture and examined for E. coli O157 using the same method for the individual samples as described above and previously. 9 Representative O157 latex-positive isolates were sent to the Laboratory of Enteric Pathogens (HPA Colindale, Colindale, UK) and the Department of Food and Environmental Safety (VLA Weybridge, UK) to determine whether they were verocytotoxigenic strains. Spiral plating quantification of VTEC O157 in 1 set of individual samples from each farm was performed at the same time as the IMS to provide data that could potentially explain any farm-level differences in sensitivity that might be found.
Statistical analyses
Testing of pools with known number of positives. Logistic regression models were fitted to the data on the pools formed in the laboratory to determine whether the best-fitting model was one that depended on 1) the proportion of positives in the pool or 2) the colony-forming unit (CFU)/g of VTEC O157 in the pool (estimated by summing up the counts from the individual samples comprising the pool). For this analysis it was assumed that all positive samples were detected by at least 1 of the 1-g and 10-g samples tested by AIMS. For the proportion of positive samples in the pool as the explanatory variable, this leads to the probability of a pool testing positive, ηpool, being equal to:
where πpool was the proportion of samples in the pool that are true positives, and α and β were parameters determined by maximum likelihood, with a similar formula for the CFU/g model. The adequacy of the fit of each model to the data was assessed by a Hosmer-Lemeshow chi-square test 3 (implemented in Limdep 7.0 d ). A likelihood ratio test was carried out to determine whether there was a significant difference between the parameters α and β for the pools of 5 and 10, respectively, and thus determine whether the data could be pooled for a combined estimate depending only on the proportion of pools positive, rather than taking into account the number of samples in the pool (i.e., a pool of 5 with 1 positive has the same sensitivity as a pool of 10 with 2 positives).
Testing of pools formed in the field. The results from the pools formed on-farm provided data on the sensitivity of pooled samples as would be carried out in the field, relative to individual samples. In this case the number of positive samples in each pool was not known but was assumed to follow a binomial distribution dependent on the number of samples in the pool and the prevalence of VTEC O157 infection in the group of cattle from which the pooled sample was formed. The best-fitting model from the previous section was used to describe the sensitivity of the pool according to the proportion or count of VTEC O157 in the pool. It was assumed that the tests had 100% specificity.
It was assumed that the individual-level test sensitivity was equivalent to a pooled sample containing all positive samples. The log-likelihood of the data from the 2 outbreak farms was maximized with the group-level prevalence on each farm and the parameters α and β in Equation (1) as unknowns, resulting in 8 unknown parameters. The optimization was then repeated with the parameters α and β, allowed to vary between the 2 farms, and a likelihood ratio test carried out to determine whether a significantly better fit was obtained. The motivation behind this is that data from a recent study, 8 in which multiple subsamples were tested from individual samples, suggested that the sensitivity of detection in individual-level samples could vary considerably between herds; central estimates of sensitivity of 24%, 55%, and 82% for prevalence equal to 15%, 46%, and 91%, respectively, were obtained.
The effectiveness of pooled versus individual sampling was compared by calculating the number of samples that would be required in order to detect VTEC O157 with 95% probability in each group of cattle on farms 2 and 3 for pooled and individual sampling. For individual sampling, the probability that a single test would be positive was given by the product of the group-level prevalence and the individual-level test sensitivity. For pooled sampling it was given by
where the summation is up to 5 since there are 5 samples per pool, and the probability of i positive samples in the pool follows a binomial distribution with parameters given by the pool level prevalence and the number of samples in the pool. Sample sizes were then calculated according to the formula 2
where τ is the statistical power required and η the probability that each test would be positive. Equation (3) relies on the assumption that the number of fecal samples would be much larger than the number of samples required. When this is not true, one would be required to take multiple samples from some individual fecal pats. This would tend to result in slightly smaller sample sizes (for both individual and pooled sampling).
Results
Pools formed in the laboratory
Of the 150 individual samples collected from the outbreak investigation (which were to be used to create the pools), 137 were positive for at least 1 test, of which 121 were positive at 1 g and 133 were positive at 10 g. Assuming that all true positive samples were positive for at least 1 of the 2 tests, this gives 88% sensitivity for 1-g culture and 97% sensitivity for the 10-g culture.
As the number of positive samples in the pool increased, an increased proportion of the pools tested were positive (Table 1). The logistic regression provided an adequate fit to the data according to the Hosmer-Lemeshow chi-square test (P = 0.40), and showed that the dependence on the proportion of positive samples in the pool and the probability that a pool would test positive for VTEC O157 was significant (P < 0.0001), thus showing that there was a dilution effect of mixing true-positive with negative samples on the test sensitivity. There was no significant difference between the estimated parameters for the pools of 5 and 10 (P = 0.24), respectively. This indicated that the model depended only on the proportion of positives in the pool and not the size of the pool. The parameter values from the logistic regression were α = −3.69 (95% confidence interval [CI]: −5.03, −2.35) and β = 8.20 (95% CI: 5.30, 11.10).
The proportion of positive pools with various proportions of positive samples included in the pool.
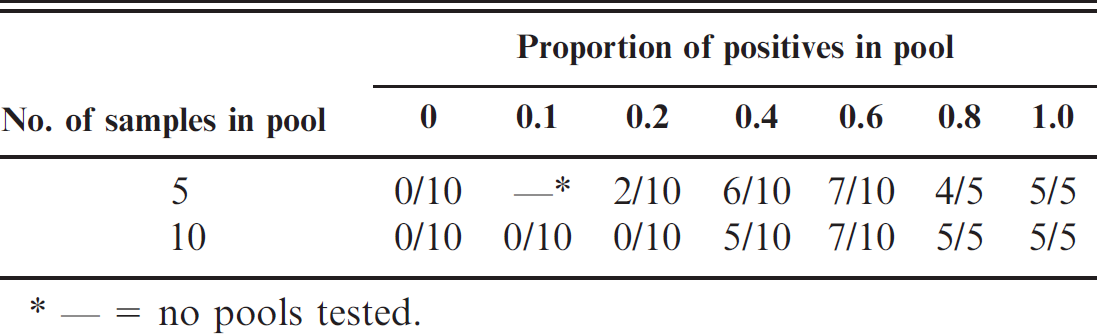
— = no pools tested.
The logistic regression model that used the count of VTEC CFU/g as the explanatory variable also provided an adequate fit to the data according to the Hosmer-Lemeshow chi-square test (P = 0.57), but showed a lower positive predictive value and negative predictive value than the model that used the proportion of positive pools as the explanatory variable, largely a result of some of the pools with very high counts testing negative (Fig. 1b). As a consequence, the proportion positive model was preferred.
Pools formed in the field
Table 2 gives the results for the individual-level and pool-level samples collected from 2 further known infected farms. Individual-level test sensitivity was estimated to be equal to 84% if assumed not to vary between farms 2 and 3. The values of α and β, taking into account the data in Table 2, were (with 95% confidence intervals) α = −3.2 (−3.7, −2.8) and β = 4.8 (4.3, 5.4). However, a likelihood ratio test showed that α and β varied significantly between the farms (P = 0.01), and that the test sensitivity was lower in farm 3 than farms 1 and 2; the sensitivity of the individual-level test was estimated to be approximately 89% on farm 2 and 47% on farm 3, and the corresponding estimates for the pool-level test sensitivity are shown in Fig. 1.
The distribution of VTEC O157 CFU/g within the AIMS-positive fecal samples was compared for each of the farms in the study (Table 3). There was a wide distribution of CFU/g obtained and a similar proportion of samples (approximately 75%) with <200 CFU/g in each of the farms.
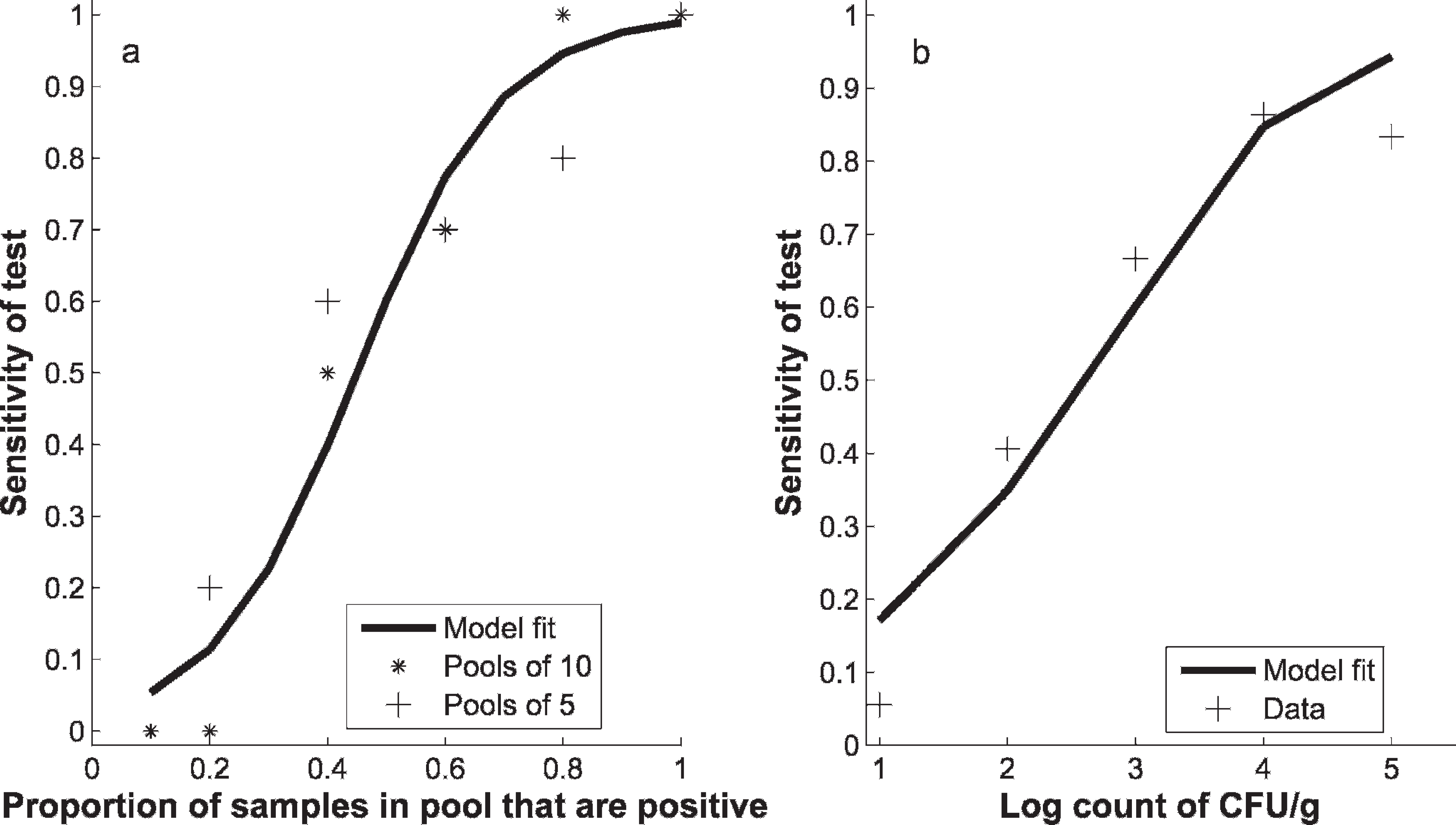
The fit of the logistic regression models to the sensitivity of laboratory-formed pooled samples when the dependent variable is
Summary of results for farm-level sampling of verocytotoxin-producing Escherichia coli O157 using individual and pooled samples.
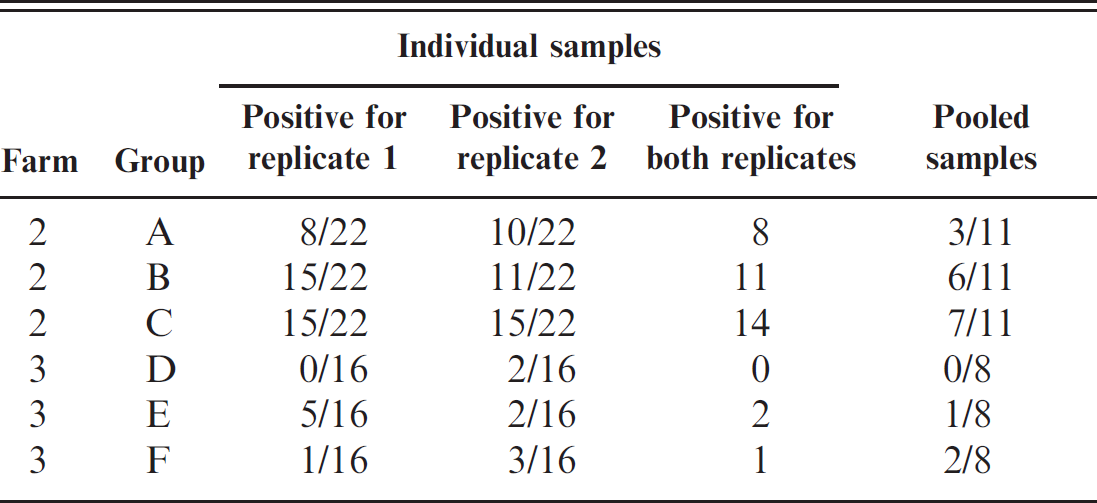
There were several samples from both farms that tested positive for only 1 of the 2 duplicate samples taken from each individual-level sample (Table 2), suggesting a heterogeneous distribution of the organisms within the fecal sample. There was a far greater difference in prevalence between the 2 farms than between the groups on each farm (estimates of 45%, 69%, and 75% for groups A through C, respectively, from farm 2; 14%, 41%, and 33% for groups D through F, respectively, from farm 3).
The resulting relationship between the number of positives in a pool and the probability of a test-positive is shown in Fig. 2 for each of the 3 farms in the study. The reducing probability of detection as the proportion of positives in the pool decreases indicates a dilution effect of mixing positive and negative samples on the probability of detection.
Comparison of pooling and individual-level sampling
The number of samples required to obtain at least 1 positive test for each group on farm 2 was 9, 4, and 4 for pooled sampling for groups A through C, respectively, and 6, 4, and 3 for individual sampling. For farm 3, the sample sizes for groups D through F were 48, 17, and 21, respectively, for pooled sampling, and 46, 15, and 19 for individual level sampling. These results indicate that pooled sampling does not result in a reduction in the required number of cultures to detect VTEC O157. The individual sampling required the same or fewer samples in each case. A sign test 17 was employed to determine whether the observed differences between pooled and individual-level sampling (Table 2) were significant. Of the 12 individual-level sample results, there were 7 instances when the proportion of individual-level positives were greater, 3 when the pooled samples were greater, and 2 ties, which resulted in no significant difference between pooling and individual-level sampling (P = 0.34). This shows that the sample-size requirements for pooled sampling were either the same as for individual sampling, or too small a difference from individual sampling to be detected by the study.
Comparison of the verocytotoxin-producing Escherichia coli count distribution in individual samples identified as positive between the 3 farms in the study.
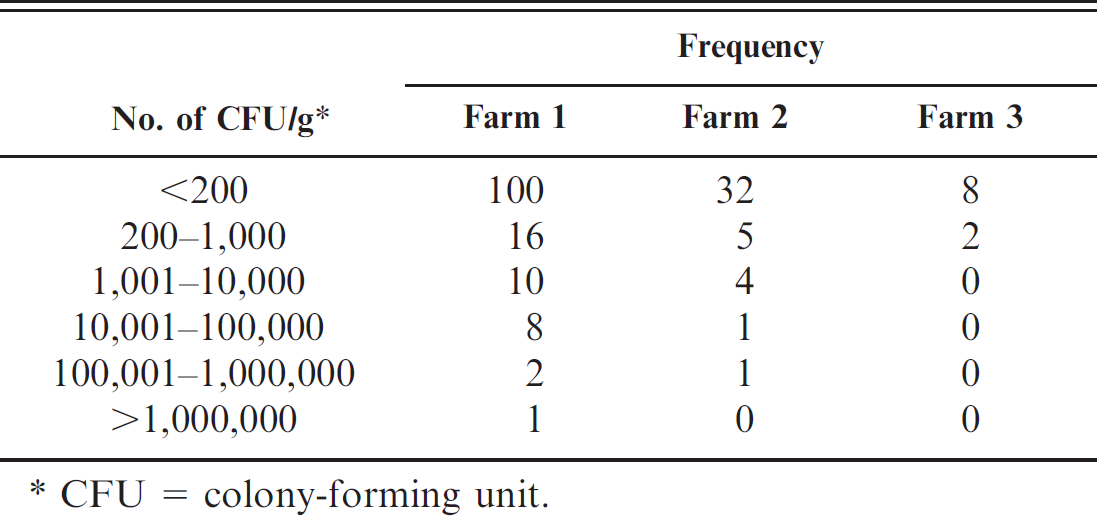
CFU = colony-forming unit.
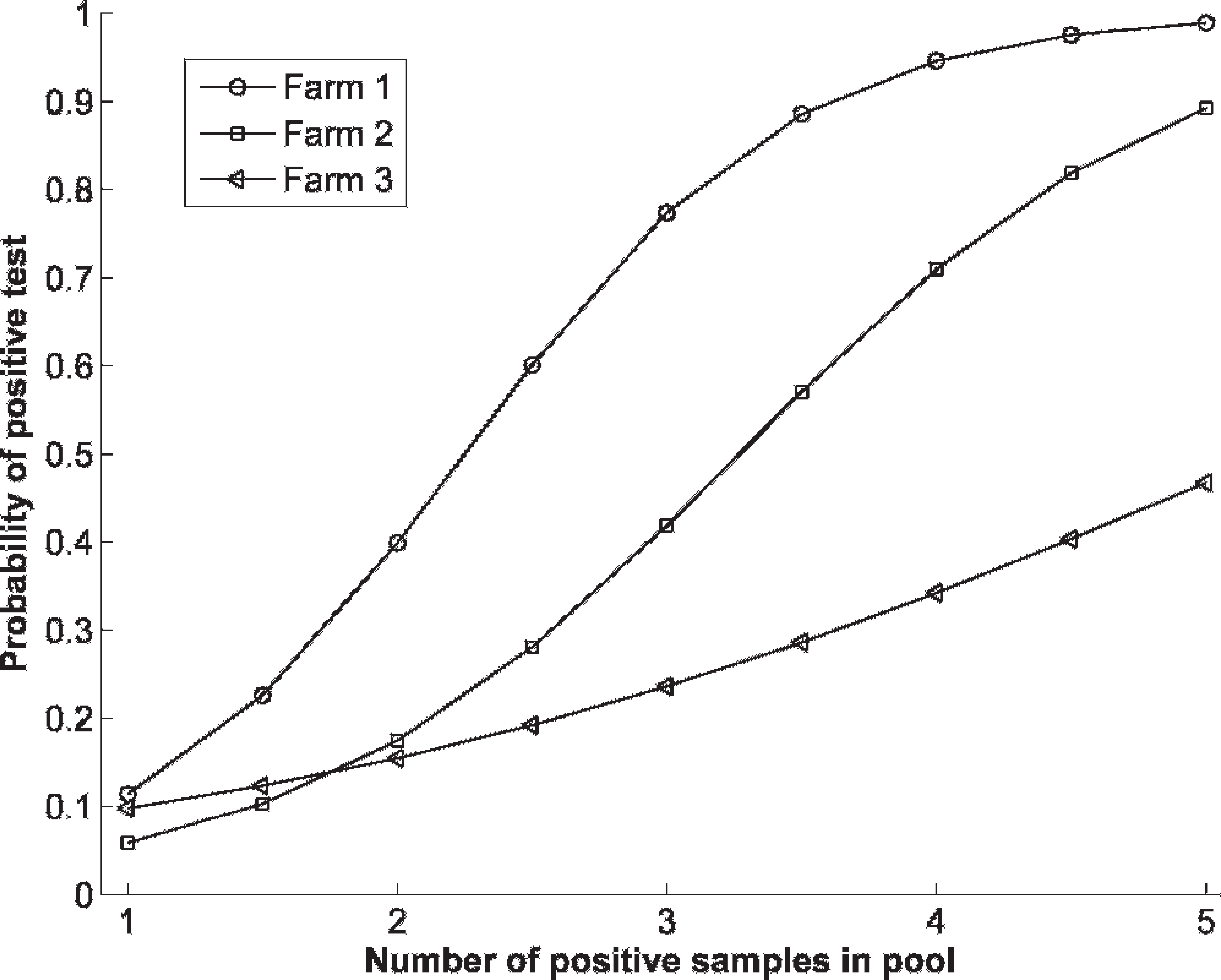
The estimated probability of obtaining a positive result from a pooled sample according to the number of true positives in the sample for each of the 3 farms.
Discussion
The results from this study suggest that pooling of 1-g fecal samples is no more efficient than testing individual 1-g samples during outbreak investigations. This is caused by the reduced probability of detecting VTEC O157 when true-positive samples are diluted with true-negative ones, which cancels out the increased probability of including VTEC O157 in the pooled sample compared with the individual sample. Reduced sensitivity for the pooled samples compared with individual samples caused by the dilution effect was also described in a previous study. 12 However, the expected proportion of pooled samples that tested positive with 1/5 or 2/5 positive samples in the pool was much higher for that study, at around 50% for both cases, than was found in the present study, with 7% and 20% for 1/5 and 2/5, respectively. The differences between the present study and the previous one could have arisen as a result of differences in the experimental protocols for the bacterial culture. For example, in the previous study a stomacher was used prior to culture, whereas in the present study samples were mixed by hand, and it is possible that the difficulties of effectively homogenizing the pooled samples led to the small estimated difference between the sensitivity of pooled and individual samples observed in this study. This would be a key difference between pooling samples for VTEC O157 in cattle and Salmonella in pigs; in the latter case, pooling was shown to be more efficient than individual sampling without the use of a stomacher. 1 There were also other differences between this study and the previous study 12 that could cause differences in results. First, in the present study feces were pooled on-farm, as would be done in practice if pooling was routinely adopted for outbreak investigations, rather than mixing sets of individual fecal samples in a laboratory. Second, the distribution of VTEC O157 CFU/g in individual fecal samples in the present study was markedly lower than those obtained in the previous study, with only 10% of samples >5,000 CFU/g in the present study, compared with approximately 50%. The CFU/g is likely to affect the rate of positives in diluted pools as there is a correlation between the probability of successful culture and the number of CFUs/g. The large difference between the distributions of CFU/g of this study and the previous study could have been caused by differences in the sampling ages of the animals between the studies (as there is an age-association on shedding patterns 6 ), the method of enumeration of the VTEC O157, the fact that the previous study used inoculated calves rather than naturally infected ones, or merely the considerable natural variability of shedding. A comparison of the shedding distribution of cattle in the present study showed no significant difference (as measured by a chi-square test) from 2 other studies in the United Kingdom, 5,8 and a similar range of CFU/g to a third study. 10 Overall, therefore, while the CFU/g distribution would have an impact on the performance of pooling, there is nothing to indicate that the farms sampled in the present study had atypical counts of CFU/g.
The estimated individual-level test sensitivity was highly variable between the farms, with a lower estimate of 52% in farm 3 and approximately 90% for farms 1 and 2. This is in accordance with the findings of a recent study 8 where the estimated test sensitivity was also higher where the estimated prevalence was higher. The most likely reason for the lower test sensitivity in farm 3 is lower VTEC shedding rates in the individual animals compared with the other 2 farms. While the proportion of positive samples with a count of less than 200 CFU/g was similar for each farm (Table 3) at approximately 75%, 2 factors should be borne in mind. First, the low prevalence for farm 3 results in a small number of positive samples and a large uncertainty in the VTEC CFU/g distribution. Second, taking into account the expected number of true-positive samples that were missed by the duplicate testing of individual-level samples, the proportion of samples with <200 CFU/g for farm 3 rises from 8 to 12, which then gives 85%. The authors cannot easily quantify the effect on the AIMS of such an increase in the proportion of samples with a low CFU/g since the relationship between CFU/g and the probability of detection of the AIMS for low CFU/g for naturally infected samples is not accurately known. The actual CFU/g in samples below the detection limit for the spiral plating technique is also not known in the present study. However a lower level of shedding on farm 3 remains a possible explanation for both the lower prevalence and the lower test sensitivity. External factors have been reported to have an effect on shedding rate or infectious burden of VTEC O157 in groups of cattle. Clean animals, dry bedding, avoiding movement between epidemiological groups, and clean water troughs have been reported to reduce VTEC O157 within groups, which supports the importance of environment in the transmission dynamics (http://www.defra.gov.uk/science/project_data/DocumentLibrary/OZ0138C/OZ0138C_3750_FRP.doc). Individual animals have been found to excrete different concentrations of CFU, and heterogeneity in floor pats was described and modeled recently. 4 However, little is known about the reason for “super-shedding” and factors that may reduce the CFU/g excretion, but various feeds and the presence of other animal species have been suggested. 13,14
The superior fit of the proportion of positives dependent logistic regression model over one that depended on the VTEC CFU/g was unexpected; the VTEC CFU/g is likely to have a large influence over the probability of successful culture, although clearly there will be a correlation between the 2 models. There is, however, uncertainty in the measurement of the CFU/g by the spiral plating method, 11 which could have influenced the fit of the model to the data. It is also possible that background flora could have influenced the culture of VTEC 0157, leading to occasional test failures even for high VTEC loads.
The primary use of sampling for VTEC O157 in human outbreak investigations is to determine whether and where in the farm VTEC O157 identical to human cases is present, rather than to determine the actual prevalence of infection. The results of this analysis are important to determine the number of samples required to show whether the prevalence is less than a design prevalence, currently set at 10% for outbreak investigations in the United Kingdom. The basic message from the on-farm sampling is that the pools of 5 did not reduce the sample-size requirements compared with individual-level sampling. However, the 10-g samples were found to be more sensitive than the 1-g samples in farm 1, and it may be that testing larger samples would be a better route to increase the efficiency of sampling schemes than pooling. Another factor to take into account is that, if the test sensitivity were influenced by the prevalence of VTEC O157, this would have an impact on the sample sizes required, especially for detection of VTEC O157 on low prevalence farms. Therefore it is vital that further investigation into the variation of test sensitivity between farms is carried out. This could be done by multiple testing of individual-level samples collected during outbreak investigations. If farm-level factors were also noted, then it may be possible to determine whether there are any farm-level factors that affect the test sensitivity in a predictable way.
The present study has shown that pooling of samples requires at least as many cultures as the testing of individual samples for outbreak investigation. This study has also found that the individual-level test sensitivity for VTEC O157 was highly variable between farms and possibly correlated with the on-farm prevalence. Further work would be useful to investigate the between-farm variability of test sensitivity for VTEC O157, as it is crucial for the appropriate design of sampling protocols. It would also be useful to explore whether more sensitive methods of VTEC detection (e.g., culturing 10 g rather than 1 g) for pooled samples or individual samples would be worthwhile.
Acknowledgements
The authors thank Bob Futter, Alan Garnett, and Claire Scott at VLA Preston for the collection of samples and laboratory work. Funding for this work came from the Department for Environment, Food and Rural Affairs projects FZ2100 and ED1039. Copyright held by British Crown.
Footnotes
a.
Invitrogen, Paisley, Renfrewshire, UK.
b.
Oxoid Ltd., Basingstoke, Hampshire, UK.
c.
Microsoft Corporation, Redmond, WA.
d.
Econometric Software, Plainview, NY.