Abstract
Toxoplasmosis was diagnosed in a woodchuck (Marmota monax) and 2 American red squirrels (Tamiasciurus hudsonicus). The woodchuck was euthanized by a wildlife rescue organization in New York after progressive clinical signs of head tilt, circling, and rapid weight loss. Necropsy examination revealed acute subdural hemorrhage over the right cerebral hemisphere. Histologic lesions included meningoencephalitis, myocarditis, and hepatitis. Protozoal cysts were present in affected and unaffected neuroparenchyma. The squirrels were found dead, emaciated, and moderately infested with fleas near a park in northern Indiana. In both squirrels, the lungs were consolidated with numerous nodules up to 2 mm in diameter. Histologically, pneumonia and encephalitis were associated with intracellular and free protozoa. Additional histologic lesions included multifocal lymphoplasmacytic encephalitis with intralesional protozoa in both squirrels. The protozoa were positive with Toxoplasma gondii-specific immunohistochemistry and had ultrastructural features consistent with T. gondii in both squirrels. A real-time polymerase chain reaction test using T. gondii–specific probes demonstrated protozoal DNA in the lung, brain, and kidney of the squirrels and in the brain and heart of the woodchuck. To the authors' knowledge, this is the first report of toxoplasmosis in woodchucks or American red squirrels. Because rodents are common near urban settlements, this finding underscores their role as important intermediate hosts for T. gondii.
Toxoplasmosis is a prevalent, systemic parasitic zoonosis caused by the coccidian protozoan Toxoplasma gondii. The life cycle of T. gondii is facultatively heteroxenous with felids, including the domestic cat, as the definitive hosts and a range of vertebrates, including domestic and wild mammals, birds, and humans, as intermediate hosts. 9,12,23,24 Infections are usually acquired by ingestion of uncooked or undercooked meat containing infective tissue cysts, ingestion of food or water contaminated with oocysts from feces of infected cats, or transplacental transmission when infection is acquired during pregnancy. 6,12 Cats become infected by ingesting tissues of intermediate hosts containing T gondii tissue cysts. The organism replicates within the gastrointestinal tract and numerous environmentally resistant, infective sporulated oocysts are shed in their feces.
This report describes 3 cases of toxoplasmosis: 1 case in a woodchuck (Marmota monax) submitted from a wildlife rescue facility in New York and 2 cases in American red squirrels (Tamiasciurus hudsonicus) from Indiana. The diagnosis of toxoplasmosis was confirmed by molecular diagnostics in the woodchuck and by immunohistochemical, ultrastructural, and molecular diagnostic methods in the squirrels. To the authors' knowledge, this is the first report of toxoplasmosis in woodchucks or American red squirrels.
Necropsy examinations of all animals were performed at the Purdue University Animal Disease Diagnostic Laboratory (ADDL). Tissue specimens from each case were collected in 10% buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). For immunohistochemistry (IHC), formalin-fixed, deparaffinized sections were incubated with mouse monoclonal antibody against cytoplasmic antigen of T. gondii tachyzoites. a The streptavidin-biotin detection system (DAKO LSAB2 peroxidase kit) b was used according to the manufacturer's instructions. For ultrastructural studies, 1 cubic mm fragments of fresh, unfixed lung from each of the 2 squirrels were fixed in 3% glutaraldehyde, post-fixed in 1% osmium tetroxide, dehydrated in ethanol, and embedded in Epon. Sections were cut at 70–90 nm, contrasted with lead citrate and uranyl acetate, and examined under a Philips 201 electron microscope.
To confirm the identity of the protozoa, formalin-fixed (woodchuck) or frozen (squirrels) tissue samples were analyzed by a previously described T. gondii-specific TaqMan®-type real-time polymerase chain reaction (PCR) assay. 10 DNA from the brain, lungs, and kidneys from the squirrels and from the brain, heart, and liver from the woodchuck was extracted using a commercial kit (DNeasy Tissue Kit) c according to the manufacturer's recommendations. Real-time PCR was performed in a total volume of 25 μl, using 5 μl of the extracted DNA as template, 0.4 μM of each primer, and 0.3 μM of the TaqMan probe. DNA extracted from the brains of mice experimentally infected with T gondii was used as a positive control; DNA extracted from tissues of mice infected with Neospora caninum was used as a negative control. The assay was performed using the Smart Cycler II instrument. d Thermal cycling was carried out with 1 cycle of 95°C for 300 seconds, followed by 50 cycles at 95°C for 15 seconds, 58°C for 30 seconds, and 72°C for 15 seconds. Data acquisition and analysis were done with Cepheid Smart Cycler software (version 2.0b). d The threshold cycle (Ct) values were determined by a primary curve analysis program with the threshold limit set at 30 fluorescent units.
In the first case, an adult male woodchuck was admitted to a New York wildlife rescue center in September 2005 with clinical signs of head tilt and circling. After a 1-month clinical course of rapid weight loss and persistence of neurological signs, the woodchuck was euthanized and submitted to ADDL. At necropsy, the woodchuck weighed 2.0 kg and was emaciated, with severe depletion of fat stores and reduction in skeletal muscle mass. Acute subdural hemorrhage, measuring 2.5 cm × 1.2 cm in greatest dimensions, was present over the right cerebral hemisphere. Overlying calvaria and soft tissue, and underlying leptomeninges, revealed no gross evidence of trauma, hemorrhage, or inflammation. All other organ systems were unremarkable.
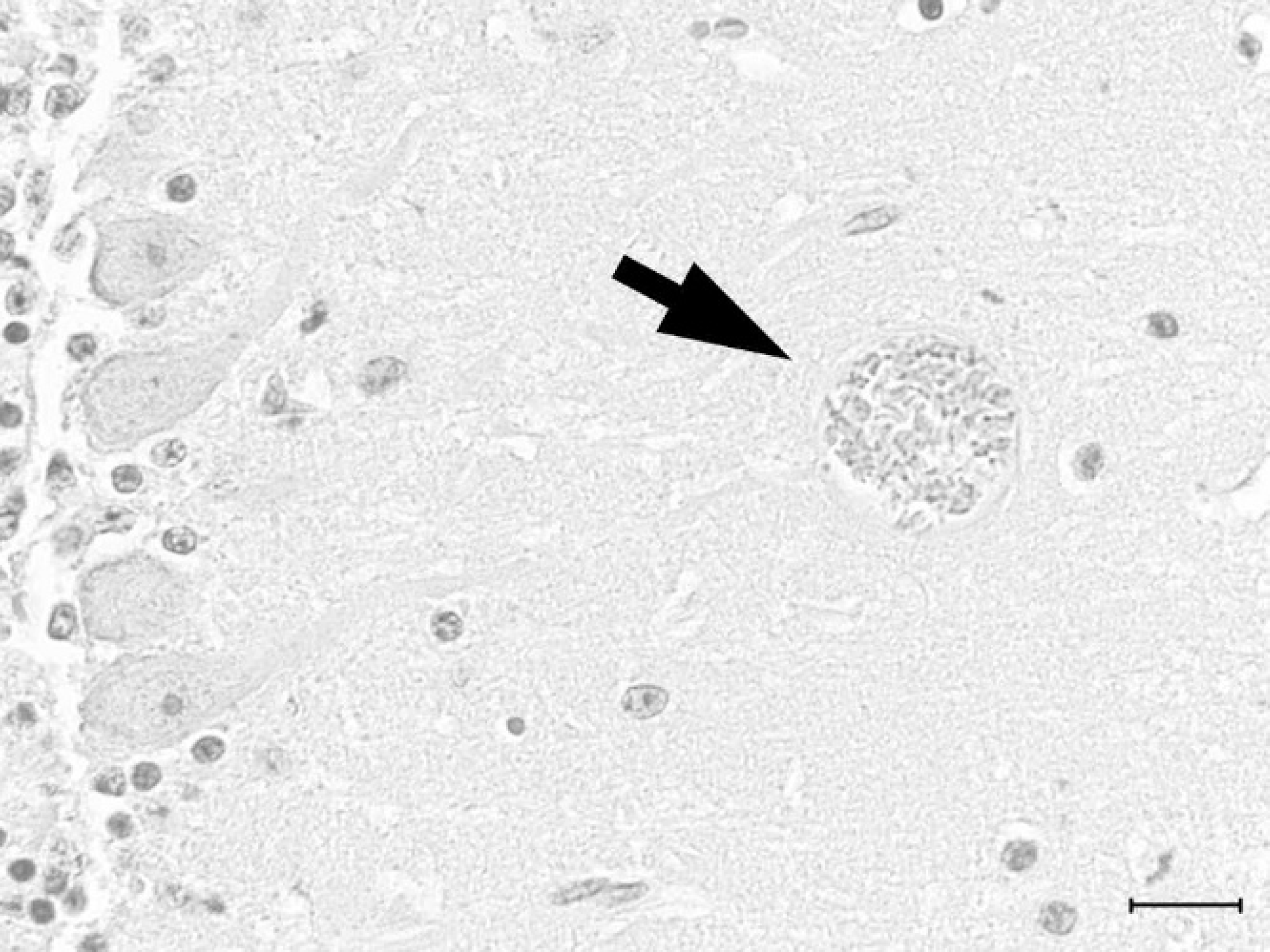
Cerebellum; woodchuck. Toxoplasma gondii protozoal cyst (arrow) in the molecular layer of the cerebellum. Hematoxylin and eosin. Bar = 20 μm.
Histologically, the woodchuck had necrotizing encephalitis with loss of neurons, vacuolization of neuroparenchyma, hemorrhage, and randomly distributed microglial nodules in the cerebellum, medulla oblongata, and pons, with the cerebral cortex and hippocampus less severely affected. Lymphocytes and plasma cells cuffed several small vessels within brain sections and moderately infiltrated the leptomeninges. No hemorrhage was identified in the cerebral cortical leptomeninges or parenchyma to correspond to the subdural hemorrhage observed grossly. Protozoal cysts, ranging from 15–40 μm in diameter and filled with crescent-shaped bradyzoites measuring approximately 5 μm in length (Fig. 1), were associated with necrotic foci and were present in cerebral and cerebellar neuroparenchyma without inflammation. The protozoa failed to react with T. gondii antibody using IHC. Additional histopathological findings included lympho-histiocytic interstitial myocarditis, with rare cardiac myocyte necrosis. Occasional, random foci of hepatocellular necrosis were infiltrated by lymphocytes, plasma cells, and histiocytes. The same types of leukocytes surrounded multiple bile ducts. Other than in the brain, protozoal cysts were not observed in HE-stained tissue sections, including stomach, intestine, liver, spleen, kidney, urinary bladder, testis, tongue, heart, and lung. Based on the presence of histologic lesions, brain, heart, and liver were selected for molecular diagnostic testing. Toxoplasma gondii-specific DNA was detected in all tissues tested (cerebrum, cerebellum, and heart) except liver using real-time PCR; the cerebrum contained the highest DNA content (Ct = 29.22) and the heart contained the least (Ct = 43.06). No DNA was amplified in the negative controls.
In the next cases, 2 male American red squirrels were found dead in Elkhart County, Indiana, in late October 2005. The squirrels weighed 262 g and 250 g, and both were in poor body condition with moderate flea infestation. In both squirrels, the lungs were incompletely collapsed, diffusely wet, mottled dark red, and contained numerous pale tan to yellow, slightly raised, oval to spherical nodules measuring up to 2 mm in diameter (Fig. 2). Sections of the lung sank in formalin.
Histologically, both squirrels had diffuse, moderate to marked, necrotizing lymphohistiocytic interstitial pneumonia with numerous foamy macrophages and intracellular tachyzoites and tissue cysts in alveolar spaces (Fig. 3A, Fig. 3B). There was mild hyperplasia of type II pneumocytes. Protozoa in the lung and brain stained positive for T gondii by IHC (Fig. 3C). In addition to intracellular tachyzoites and tissue cysts, extracellular tachyzoites were also demonstrated by IHC. Multifocal lymphoplasmacytic encephalitis with intralesional protozoa was present in both squirrels. Toxoplasma gondii cysts, surrounded by glial cells, were present mostly in the gray matter. Some cerebrocortical vessels were cuffed by lymphocytes and plasma cells. In the heart, there was multifocal myocardial necrosis with mild infiltration of lymphocytes. A few cardiac myocytes not associated with the necrotic foci contained intracellular protozoa. Additional histologic lesions included mild lymphoplasmacytic periportal hepatitis and interstitial nephritis and mild lymphoid depletion of the splenic white pulp.
Ultrastructurally, intracytoplasmic club-shaped protozoan cysts, tachyzoites (Fig. 4), and rare binucleated endodyozoites with apicomplexan morphological features 21 were noted in the lung of both squirrels. Tachyzoites measured approximately 5 μm × 10 μm, were surrounded by a 3-layered pellicle, had a caudally placed nucleus with marginated heterochromatin, and contained numerous mitochondria. Protozoa were noted extracellularly as well as within parasitophorous vacuoles in alveolar macrophages. Autolysis and necrosis obscured ultrastructural details of the host cells harboring these organisms. Toxoplasma gondii-specific DNA was detected in the lung, brain, and kidney from both squirrels tested by real-time PCR. Threshold cycle values for the cerebral, pulmonary, and renal tissues were 23.23, 17.97, and 23.81, respectively. The Ct value for the positive control sample was 24.09. No DNA was amplified in the negative controls.
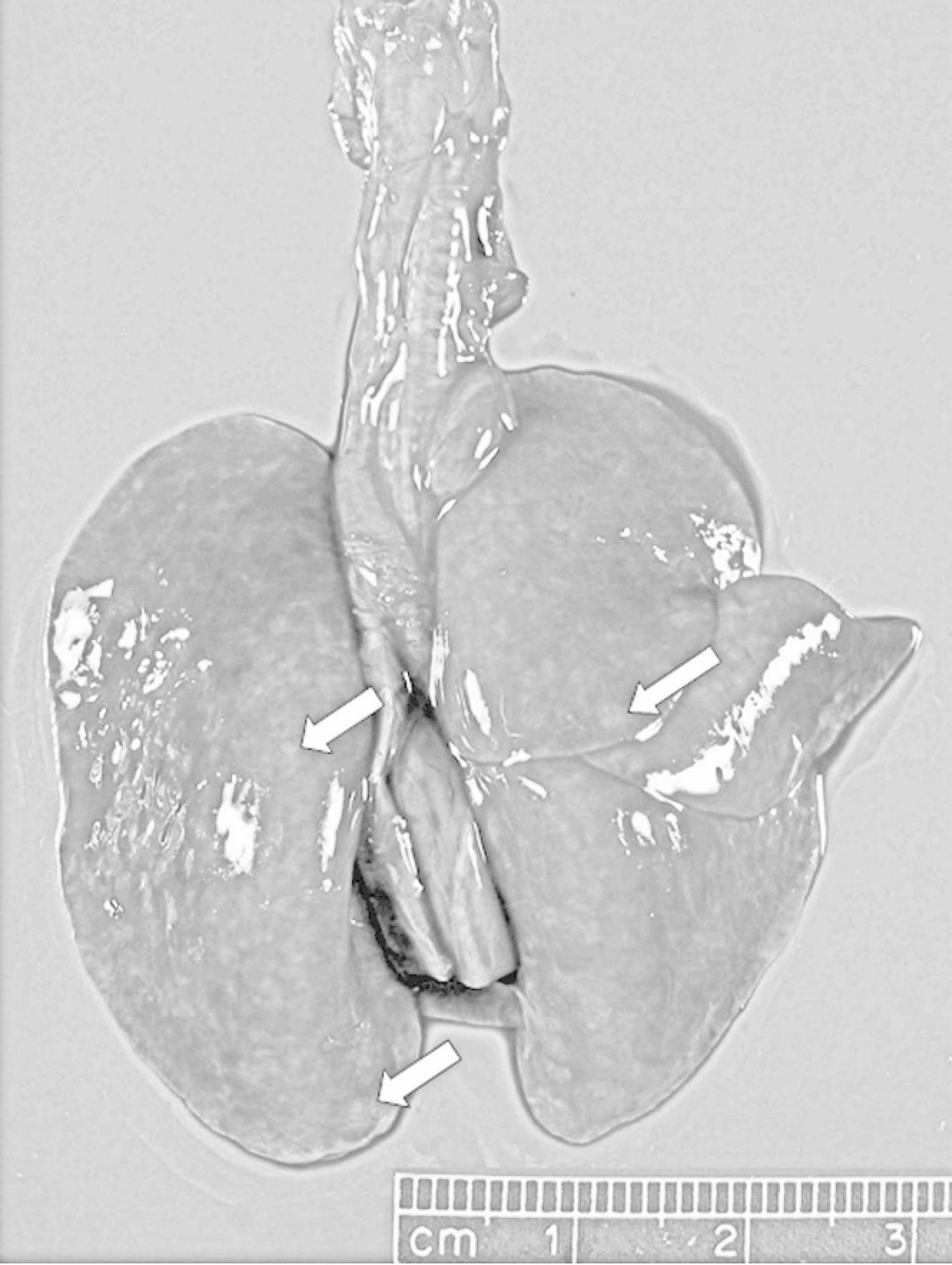
Lung; American red squirrel. Both lungs are diffusely mottled, dark pink to red, and wet with numerous irregular to roughly oval, up to 2 mm in diameter, pale tan nodules (arrows).
The diagnosis of toxoplasmosis was confirmed by IHC, ultrastructure, and real-time PCR in the squirrels and by real-time PCR in the woodchuck. The negative IHC immunoreactivity in the woodchuck tissue samples with T. gondii antibody created a diagnostic challenge and may reflect a species variation in host influence on antigenic expression, resulting in inadequate binding by the monoclonal antibody. The inability to detect T. gondii-specific DNA in sections of woodchuck liver by real-time PCR may reflect sampling variation, DNA degradation due to formalin fixation, or a lack of association between the mild hepatic lesions and toxoplasmosis.
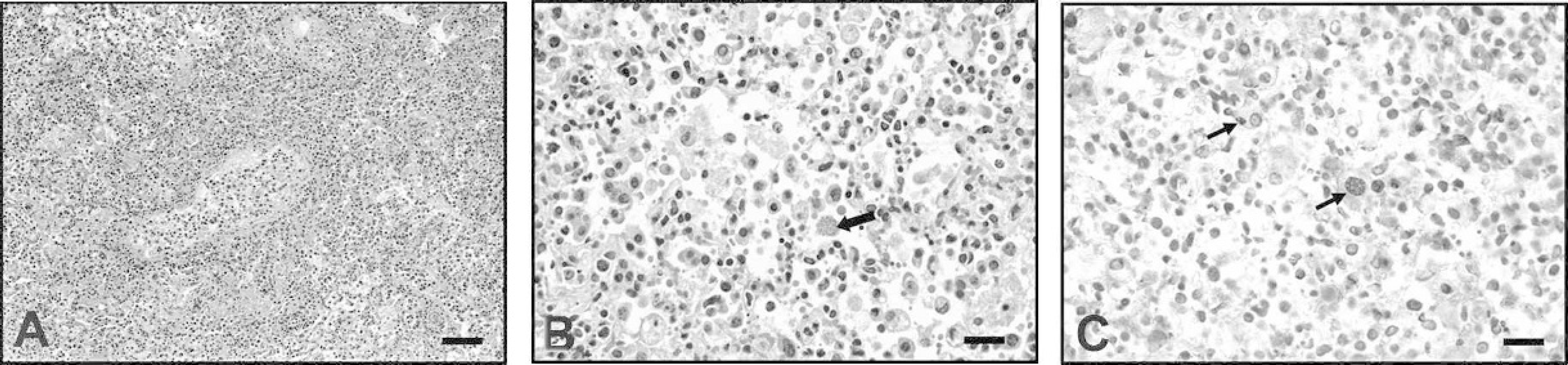
Lung; American red squirrel.
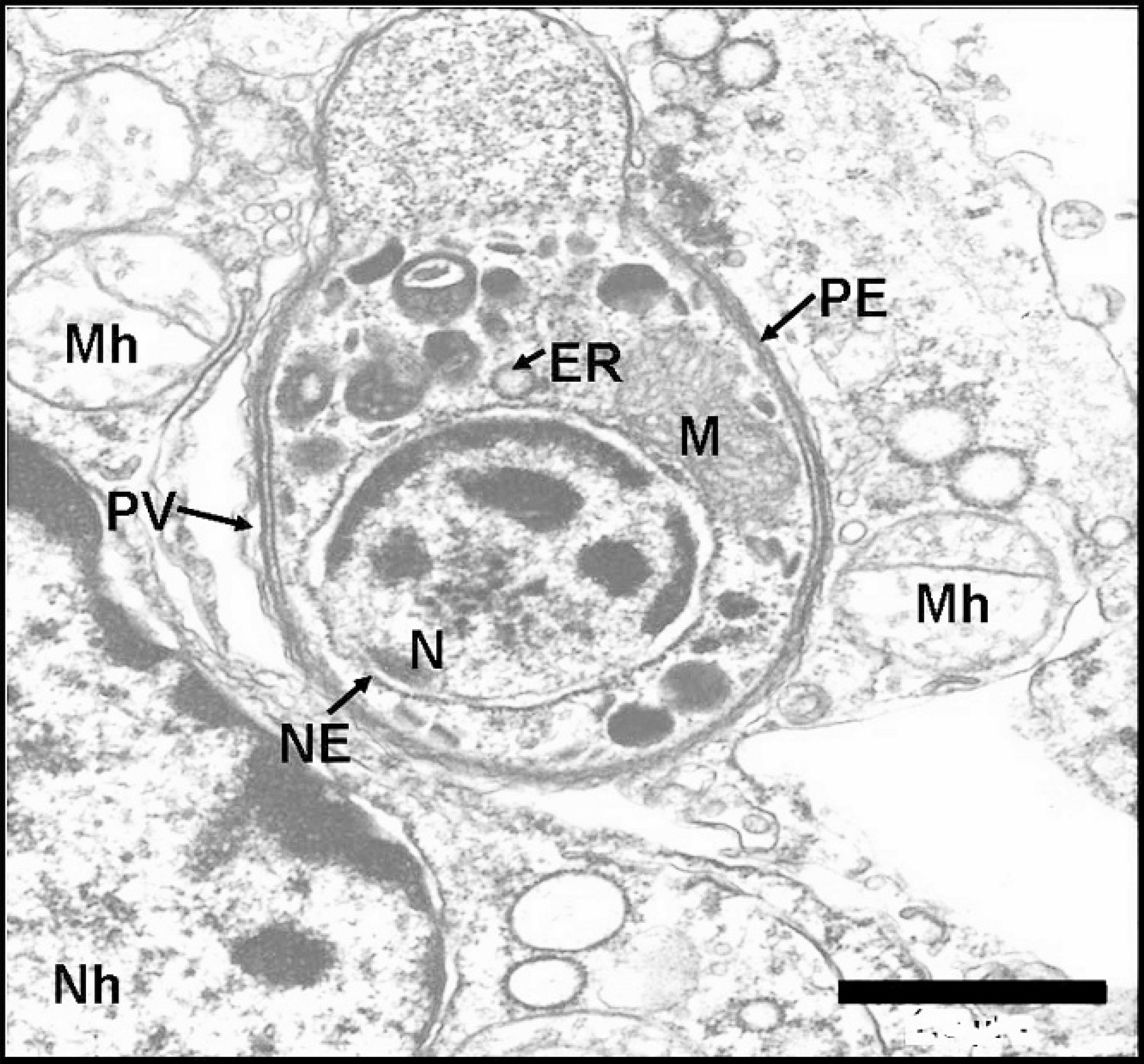
Transmission electron micrograph; lung; American red squirrel. Transmission electron micrograph showing an intracytoplasmic club-shaped protozoan tachyzoite. N = nucleus, M = mitochondria, ER = endoplasmic reticulum, NE = nuclear envelope, PE = pellicle, PV = parasitophorous vacuole, Mh = host cell mitochondria, Nh = host cell nucleus. Uranyl acetate and lead citrate stain. Bar = 1 μm.
Woodchucks and American red squirrels are rodents of the squirrel family, Sciuridae. 17 Natural infections with T. gondii have been reported from various sciurids, including the English red squirrel (Sciurus vulgaris), 5 eastern gray squirrel (Sciurus carolinensis), 7,20 13-lined ground squirrel (Citellus tridecemlineatus), 26 California tree squirrel (Sciurus griseus), 22 Himalayan flying squirrel (Peaurista inoratus), 18 and Korean squirrel (Tanias sibericus). 4 Gross and histopathological lesions in the present cases were similar to those previously described in other squirrel species 4,8,20,26 as well as other domestic and wild animals. 9,13,16 In most of these cases, T. gondii infection was associated with fatal disease. Recently, Dubey et al. 8 reported acute fatal toxoplasmosis in eastern gray squirrels with necrosis and inflammation associated with tachyzoites and bradyzoites in multiple tissues, as reported here in American red squirrels. They also noted numerous tissue cysts in the lungs of gray squirrels.
Consistent with the known predilection of T. gondii for nervous tissue, protozoal cysts and histologic lesions were present in the brains of the woodchuck and squirrels. Toxoplasma gondii tissue cysts in the brain can impair memory and learning and alter behavior in laboratory rats and mice. 2,27 Toxoplasmosis has also been associated with schizophrenia in humans. 25 Animals with altered behavior due to toxoplasmal cysts in the brain may be more susceptible to predators, including cats, thus increasing the chances of completing the parasite's life cycle. Sciurids with affected behavior may approach human settlements or become easy targets for hunters and thus present a potential public health risk. Clinical signs of central nervous system (CNS) infection by T. gondii in squirrels may resemble those of rabies. 20,22 Similarly, CNS disease in woodchucks, allowing their capture by humans, often merits rabies testing, although one study 11 illustrated that woodchucks rarely test positive for rabies. Of the 3 woodchucks submitted to ADDL for rabies testing since 2000, including the case described in this report, all were negative. It is therefore important to develop alternative differential diagnoses for neurological signs including ataxia, vestibular signs, lack of wariness to humans, or aggression in this species. Cerebrospinal nematodiasis was diagnosed in multiple woodchucks from New York 11 and in 1 wood-chuck from Indiana. 14 All animals exhibited behavior suggestive of rabies, but all tested negative. In 2 reports of individual woodchucks with vestibular signs of ataxia, nystagmus, head tilt, circling, and/or rolling, pathologic examination revealed cysticerci within the third and lateral ventricles of one case 3 and a meningioma of the brainstem in a captive 11-year-old woodchuck in the second case. 19 Spongiform encephalopathy associated with severe renal disease was identified in 2 young laboratory woodchucks, although only 1 displayed neurological signs of head-pressing before euthanasia. 1
Toxoplasma gondii infections in sciurids and other wild animals suggest contamination of the environment with protozoal oocysts. 7 The source of infection in the present cases was not determined. Because these species are often abundant near urban settlements, exposure to soil and water contaminated with oocysts shed in feces of infected domestic cats is suspected. Immunocompromised animals are likely to develop acute infection as may be seen in dogs and raccoons with canine distemper virus infection. 9 The immune status of the sciurids in the present case is unknown but histologic examination of tissues in the squirrels revealed lymphoid atrophy.
In summary, toxoplasmosis was diagnosed in a woodchuck and 2 American red squirrels. Because these rodents are common near residential areas and environments cohabited by cats, they may serve as important intermediate hosts for T. gondii. Toxoplasmosis in red squirrels has public health significance as these squirrels are hunted for their fur in some parts of Canada. 15 Toxoplasmosis should be included in the differential list for neurological symptoms in woodchucks, and potential for human exposure should be considered when handling sick red squirrels and woodchucks.
Acknowledgements. The authors would like to thank Dawn Slessman for submitting the squirrels, Phyllis Lockard for transmission electron microscopy, and the staff of the histopathology laboratory of Purdue University Animal Disease Diagnostic Laboratory for their technical help.
Footnotes
a.
Chemicon, Temecula, CA.
b.
DAKO, Carpinteria, CA.
c.
Qiagen, Valencia, CA.
d.
Cepheid, Sunnyvale, CA.
