Abstract
Objectives
To examine the literature for evidence of adverse events associated with the use of intranasal oxytocin in older adults (60+).
Methods
A systematic review was undertaken according to PRISMA guidelines. Peer-reviewed literature was searched for studies involving intranasal oxytocin use in older populations. The Joanna Briggs Institute’s (JBI) critical appraisal tool was used to assess the quality of included studies.
Results
The search identified nine randomized controlled trials (RCTs) that investigated the effects of intranasal oxytocin on a total sample size of 331 older participants. Adverse effects associated with oxytocin administration were predominantly mild and varied inconsistently between studies. Compared to placebo intranasal oxytocin was not significantly associated with severe adverse outcomes in doses ranging from 24 to 72 IU after single dose and or repeated doses in the short-term.
Conclusion
In a population of older adults, intranasal oxytocin is devoid of serious adverse events. Although this review offers valuable insights, it may not fully reflect the potential adverse events associated with the long-term administration of intranasal oxytocin such as would be expected in its clinical application if approved for the treatment of dementia.
Beyond its established role in facilitating childbirth and lactation, the nine-amino acid neuropeptide oxytocin has been shown to exert significant modulatory effects on pro-social behaviours such as enhancing empathy. 1 There is a growing body of evidence supporting the potential therapeutic applications of oxytocin to treat several mental health disorders, 2 including autism spectrum disorders, 3 first-episode psychosis, 4 and post-traumatic stress disorder. 5 Oxytocin also shows promising neuroprotective potential in Alzheimer’s disease by restoring cognitive function and decreasing β-amyloid and Tau accumulation, as well as preventing neuronal death. 6 Additionally, oxytocin plays a crucial role in modulating social cognitive processes and has been implicated in building trust and relationships. 7 Given that social isolation and loneliness were found to be significant risk factors for dementia, there is increasing interest in exploring oxytocin’s potential benefits for conditions such as frontotemporal dementia (FTD), 8 Alzheimer’s disease,6,9 and vascular dementia. 10
Intranasal delivery of oxytocin presents several advantages over traditional systemic delivery: non-invasive; rapid onset of action; no first-pass metabolism; and fewer side effects due to a more targeted central nervous system delivery. 11 Most research concerning the safety of intranasal oxytocin has been conducted in young healthy male populations. A review of 38 controlled human trials testing intranasal oxytocin concluded that acute administration of a dose between 18 and 40 IU in both healthy and clinical populations, which were primarily composed of young men, is generally well-tolerated and not significantly associated with adverse effects. 12 A review of three open-label studies and eight RCTs, in which intranasal oxytocin was administered to 261 children and adolescents found that adverse events reports were mostly mild, including dizziness, drowsiness, dry throat, nasal irritation, runny nose, stomach ache, headache, and anxiety. 13
The risk of adverse events in older adults is likely to be higher than in children and younger adults due to the likelihood of the presence of chronic conditions coupled with the potential of interaction with medication regimens. 14 The three D’s of old age psychiatry – dementia, delirium, and depression – represent some of the most complex diagnoses in the older population. Dementia impacts a considerable number of older adults, and its treatment is complicated by associations with delirium, depression, frailty, and failure to thrive. 15 Therefore, rigorous investigation into the safety of oxytocin is essential to understand the effects of intranasal oxytocin administration on the vulnerable ageing brain.
The primary objective of this study was to systematically review the safety of using intranasal oxytocin in the older population and to compile a comprehensive list of adverse reactions observed during the administration of intranasal oxytocin to older adults. This is the first literature review to focus exclusively on the safety of intranasal oxytocin in individuals 60 years and over.
Methods
Details of search strategy

All published literature was included, encompassing qualitative, quantitative, and mixed-methods studies that examined the use of intranasal oxytocin. Studies were selected for inclusion if they reported on adverse events in older adults, defined for this study as those aged 60 years or over. However, studies comparing the safety between young and older adults were also included. Additionally, any studies with a particular focus on FTD were considered, as this type of dementia is most often diagnosed between the ages of 45 and 65 years. 17
Eligibility criteria

After eliminating duplicates, titles and abstracts were screened against the eligibility criteria. Full papers were obtained, and a final stage of screening occurred to ensure relevance. References of secondary studies were used to search for additional articles not captured by the databases search. Two reviewers conducted all stages of screening, and an independent reviewer was available to mediate if uncertainty regarding inclusion was identified.
The quality of the included studies was assessed by the authors using Joanna Briggs Institute (JBI) critical appraisal tools. 18 Each criterion of the assessment was assessed as ‘yes’, ‘no’, ‘unclear’, or ‘not applicable’. Details are available on request from the first author.
Data pertaining to the following variables was extracted: authors, year, country, study design, population, setting, sample size, age range, oxytocin dose, route, duration, brand/manufacturer, and any reported adverse effects, dropouts, and assessment measures. Conducting a meta-analysis was not feasible due to the limited quantity and quality of the collected data, inconsistencies in oxytocin dosage, frequency, and duration of use, and the diverse methodologies used to screen and exclude participants. Additionally, there were inconsistent measures used to monitor side effects and participant reactions.
Ethical considerations
There were no human participants in this article and informed consent is not required.
Results
Studies retrieved
Search of the databases yielded 150 records. After duplicate records were removed, 98 unique studies were screened for eligibility. After applying the inclusion and exclusion criteria to the title and abstracts, 44 full-text articles were further assessed against the eligibility criteria. The excluded studies either lacked information on adverse events, were non-empirical research, did not meet the age criteria for study participants, or used a non-intranasal route of administration. The reference lists of secondary studies were reviewed, but no new eligible studies were identified. After applying the eligibility criteria nine studies were included in the review for data extraction and quality assessments.19–27 The studies were conducted in three different countries, with five completed in the United States, two in Australia, and two in Canada. The PRISMA chart is reported in Figure 1. PRISMA flow diagram for systematic review.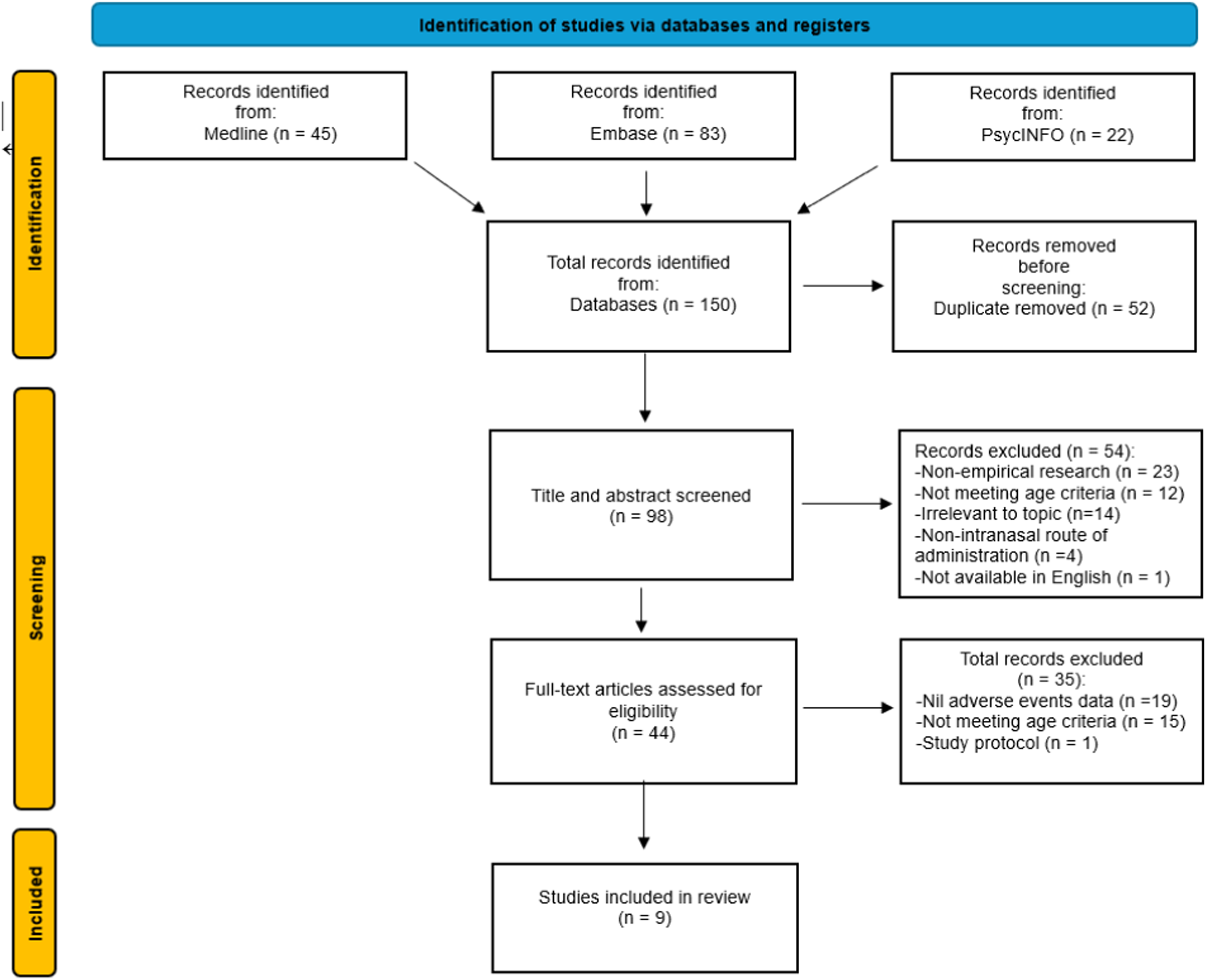
Trial designs
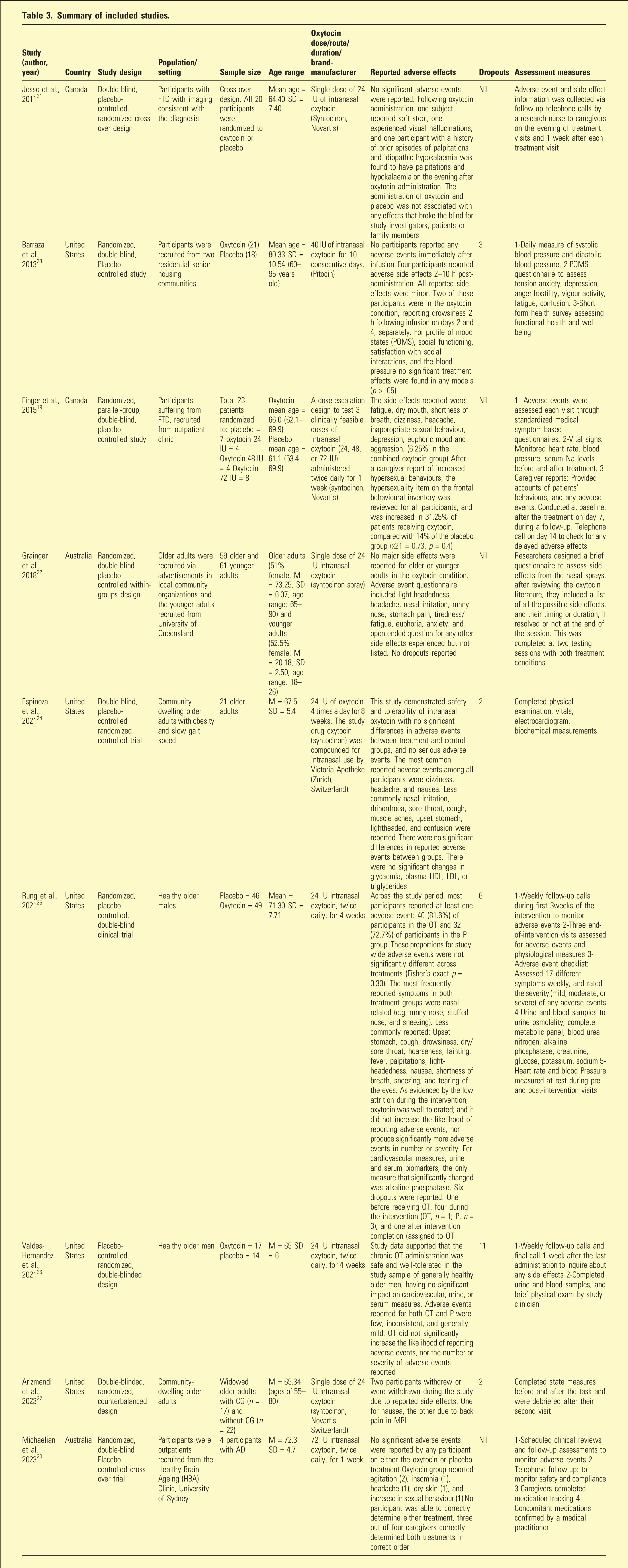
Summary of included studies.
Doses of oxytocin
Single 24 IU doses of oxytocin were studied in three trials, while five trials examined repeated administration over a period of 1 to 8 weeks in the dose range 24 to 72 IU. One examined the effects of three escalating doses of intranasal oxytocin (24, 48, or 72 IU) administered twice daily for 1 week.
Subject characteristics
These trials included in the review utilized diverse methodologies for participant screening, exclusion criteria, and monitoring of side effects and participant responses. Studies mostly included healthy participants, except for three studies; one focused on individuals with Alzheimer’s disease and two studies on individuals with FTD. Studies included both male and female subjects for the most part, but one study was specific to male participants, while another did not disclose the sex of participants, mentioning only that due to sample size limitations, sex was not included as a factor in the primary analysis. For studies that included females, no statistically significant sex-specific findings were reported.
Severity of adverse effects
Adverse effects associated with oxytocin administration were predominantly mild and varied inconsistently. Individual studies reported no statistically significant difference in the incidence of adverse effects between the oxytocin and placebo groups. For individuals with FTD, intranasal oxytocin was not significantly associated with adverse events or significant changes in the overall neuropsychiatric inventory. 19 The low dropout rates among participants across the nine studies (24 out of 331) suggests a favourable tolerability profile of intranasal oxytocin in older adults, indicating a promising potential for further clinical applications on safety considerations.
Quality of studies
The findings from the JBI’s critical appraisal tools indicated that all studies demonstrated true randomization, blinding, and concealed allocation to treatment groups. Of the nine RCTs, only five studies had intranasal oxytocin safety and or tolerability as one of their primary outcomes. The reporting of adverse effects in the studies was not standardized and the methods for assessing adverse effects were neither consistent nor clear. Only three studies employed a systematic approach by designing an assessment tool that listed all potential side effects based on previous literature reports. Additionally, only five out of the nine studies included biochemical testing and cardiovascular measures. These limitations led to the reduced reliability of measuring the safety of intranasal oxytocin, impacting the relevance of these findings for clinical practice in old age psychiatry.
Findings
As noted in Table 3, following administration of intranasal oxytocin to older adult subjects, commonly documented mild adverse effects included nasal irritation, sneezing, rhinorrhoea, sore/dry throat, hoarseness, cough, tearing of eyes, and muscle aches. Gastrointestinal disturbances such as soft stool, nausea, and abdominal pain were also commonly reported. In addition, headaches, fatigue, drowsiness, dizziness, light-headedness, and fainting were reported by several studies.19,20,23–25
In contrast, moderate-to-severe adverse effects, although infrequent, included visual hallucinations, 21 euphoria, 19 insomnia, 20 confusion24,, agitation, 20 and aggression. 19 Furthermore, instances of increased inappropriate hypersexual behaviours were observed in two different studies.19,20 Only one study reported metabolic impacts comprising hypokalaemia, 21 and another reported a decrease in alkaline phosphatase. 25 Additionally, cardiovascular changes were documented, including changes in heart rate21,25 and shortness of breath.19,25
Only two adverse events were statistically significant compared to the placebo: tearing of the eyes and a decrease in alkaline phosphatase levels. 25 Notably, one study 20 reported that only caregivers correctly identified the treatment condition. However, this may be attributed to bias associated with an anticipatory effect.
Discussion
This review aimed to evaluate the existing literature to assess the safety of intranasal oxytocin use in older adults. Studies included were all RCTs, which were completed in three different countries: United States, Australia, and Canada. To the best of our knowledge, this is the only study specifically focused on the older population. Intranasal administration of oxytocin to older subjects, although resulting in several side effects, was found to be safe with no serious adverse events reported in the studies conducted to date. Such side effects as were reported tended to be of mild to moderate severity and transient. Some of the events noted (e.g., throat and nose irritation) might well be associated with the vehicle in which oxytocin is presented rather than oxytocin per se. None of the serious adverse events which have been reported with intravenous administration of oxytocin for induction of labour, for example, anaphylaxis or water intoxication due to a diuretic effect, have been reported with intranasal administration in this population. Oxytocin doses were generally in the range of 24 to 72 IU, whereas the doses during intravenous infusions for labour are more often than not far higher. On the other hand, any proposed therapeutic use of oxytocin as a nasal spray for psychiatric conditions is likely to use doses within the range canvassed in the present review.
The extended repeated administration of the hormone was rarely addressed in the existing studies. Most studies involved single dose or short-term daily dosing, ranging from 1 to 8 weeks, thereby constraining the current safety findings.
The inconsistent adverse effects reporting hindered the ability to compare findings across different studies. Notably, only three19,22,25 of the nine studies utilized a comprehensive approach, designing a questionnaire to assess adverse effects based on the intranasal oxytocin literature, which included a list of all possible side effects, and one of the three studies added an open-ended question about any additional side effects experienced that were not listed. 22 To improve data quality in future studies, it is recommended to unify adverse events questionnaires and safety measures assessments and possibly adding cognitive and neurological assessments and mental state examinations. Additionally, there were inconsistencies in oxytocin dosage and duration, as well as variations in the methodologies used for screening and excluding participants, and in monitoring side effects and participant reactions.
It is crucial to critically evaluate the classification of severity of symptoms. For example, describing dizziness or light-headedness as mild side effect can be imprecise, considering that older adults with comorbid mental and physical health conditions may have a reduced tolerance for these symptoms, posing a greater risk compared to younger individuals. Another challenge is determining whether the cognitive impairment in people with dementia affects the accuracy of their accounts of adverse events. Furthermore, although rarely reported, it is essential to investigate potential causal links between oxytocin use and hypokalaemia, as well as decreases in alkaline phosphatase, particularly in the context of prolonged administration over a few months in comparison to current studies of a few weeks.
Some of the rare but serious adverse effects of intranasal oxytocin, such as hypersexual behaviours,19,20 confusion24,, agitation, 20 and aggression, 19 can be interpreted as symptoms of delirium. 28 However, it can be challenging to determine cause and effect, as older participants are generally more susceptible to delirium, due to high rates of comorbidity, altered pharmacokinetics, and polypharmacy. 29
The literature suggests sex differences in response to oxytocin. For example, oxytocin has been shown to improve emotion-recognition abilities in older men compared to older women.30,31 The number of females included in the studies reviewed was insufficient to confidently assess sex differences in regard to adverse events.
Finally, oxytocin enhances in-group measures of trust, love, and favouritism but increases negative opinions and fear of the out-group. 32 Predicting the impact of fear of out-group members on individuals with dementia, who may have difficulty recognizing familiar faces and perceive them as strangers, is challenging. This could explain reports of behavioural changes following intranasal administration. Viewing oxytocin solely as a pro-social hormone is overly simplistic. A study investigating oxytocin serum levels in abstinent heroin-addicted patients challenged this notion, corroborating previous evidence that links the peptide to aggressive behaviour and mood disorders. 33 This suggests a more complex mechanism where oxytocin enhances sensitivity to social salience cues, influenced by contextual or inter-individual factors. Consequently, oxytocin may promote pro-social behaviour in perceived ‘safe’ conditions. In contrast, in contexts perceived as ‘unsafe’, it may trigger more defensive and ‘anti-social’ emotions and behaviours, which might be associated with the reports of adverse effects of agitation 20 and aggression. 19
In the aforementioned study, the latter response was linked to the clinical history of individuals with addiction. However, if the same concept of perceived ‘unsafe’ conditions triggering aggressive behaviour is applied, then the older population suffering from dementia may be at increased risk. This is due to the nature of their illness, which is characterized by frequent misperceptions and misidentifications. People with dementia may perceive ‘safe’ conditions as ‘unsafe’. Consequently, the report of rare adverse events of agitation or irritability should not be minimized, but rather, warrant closer examination and further investigation.
Relevance for future research
The observations from the extant research on intranasal oxytocin administration in older adults provide a crucial baseline for future therapeutic inquiries. By demonstrating its general safety and tolerability across a range of doses (24 to 72 IU) in single dose and short-term trials, these studies establish a solid platform for exploring the safety of prolonged use in ageing populations. This baseline is pivotal for designing further investigations into the specific clinical applications and efficacy of oxytocin therapy.
It is imperative that future research efforts prioritize several critical areas to enhance our understanding of intranasal oxytocin’s safety profile. Firstly, there is a need to establish the optimal dosage for therapeutic efficacy while minimizing potential adverse effects. This includes investigating dose-response relationships and identifying the minimal effective dose. Secondly, future studies should involve rigorous and comprehensive monitoring of adverse events, with particular attention to rare but serious potential adverse events that can mimic common presentations in the older population such as delirium. Given the vulnerability of the older adult population, detailed standardized safety assessments are essential. This includes long-term follow-up to identify any delayed or cumulative adverse effects, and incorporating comprehensive periodic biochemical testing, cardiovascular measures, mental state examination, and cognitive and neurological assessments.
Additionally, future research should consider stratified analyses to understand the differential impacts of demographic factors such as age, gender, and pre-existing health conditions, on intranasal oxytocin use. Such analyses can help tailor oxytocin administration to individual patient profiles, enhancing efficacy and safety. Furthermore, studies should incorporate robust methodological designs, including standardized outcome approaches for assessing adverse events and the objective safety of intranasal oxytocin.
Given oxytocin’s role in modulating social behaviours, emotional responses, and potentially cognitive functions, it may be worthwhile to investigate its effects in people with dementia. More rigorous research is needed to thoroughly investigate adverse events associated with long-term use, especially concerning potential medication interactions in the older populations where polypharmacy is common.
Limitations
The findings from this literature review should be interpreted with caution due to several limitations. Firstly, the review did not include grey literature, such as theses, dissertations, conference papers, or other unpublished research. This exclusion may have led to the omission of high-quality studies that could provide additional insights and a more comprehensive understanding of the topic. Consequently, the results may not fully capture the breadth of existing evidence.
Secondly, the review was confined to studies published in English, which may limit the generalizability of the findings to non-English-speaking regions. Furthermore, the research was primarily conducted in Australia and North America, thereby restricting the relevance of the results to Western societies and particular ethnic groups.
Thirdly, the complexity of clinical presentations and the heightened vulnerability of older adult patients likely contributed to the scarcity of studies specifically focused on safety and tolerability within this population. To address this gap, the review included all studies where adverse events were reported as either primary or secondary outcomes. By broadening these inclusion criteria, the review aimed to capture a wider spectrum of data regarding adverse events, thereby offering a more nuanced understanding of the safety profile for this demographic. However, the inherent difficulties in researching this demographic emphasize the necessity of tailoring study designs to specifically consider their unique mental and physical health needs.
Conclusion
The current review presents evidence that suggests that in comparison to placebo intranasal oxytocin administration in older adult recipients: (1) it is associated with side effects that were predominantly mild and varied inconsistently and (2) it is not significantly associated with adverse outcomes when administered in doses ranging from 24 to 72 IU for in both single dose and short-term trials, in controlled research settings. These findings identify the general safety and tolerability of intranasal oxytocin within the specified dosage range, with the longest experimental period being 8 weeks. Although the results from these trials are valuable, they should be tempered by the consideration that they do encompass long-term administration of intranasal oxytocin over several months, as would be anticipated in its practical use if approved for the treatment of dementia.
Footnotes
Acknowledgements
The authors of this review would like to acknowledge Mina Nichols-Boyd for assisting with the search strategy.
Author contributions
A.S. confirms responsibility for study conception and design, data collection, analysis, and interpretation of results. A.S. and M.M. conducted all stages of screening. T.N. and J.O. were available to mediate if consensus could not be reached. A.S. took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research and manuscript.
Disclosure
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Antoine Sawares does not have any conflicts of interest to declare. James Olver does not have any conflicts of interest to declare. Marco Morcos does not have any conflicts of interest to declare. Trevor Norman does not have any conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of the paper.
