Abstract
Objective
This study examined the rates and persistence of clozapine-induced tachycardia and heart-rate differences in patients treated with β-blockers in the largest sample of patients with a psychotic disorder to date.
Method
An audit of medical files for 101 patients who attended a clozapine community clinic and analysis of monthly measurements of resting heart rates.
Results
51% met the clinical criteria for tachycardia. Heart rates were stable over time. β-blockers were associated with small but significant reductions in heart rates.
Conclusion
The cardiovascular risks of clozapine are often overlooked. β-blockers are useful in lowering heart rates but they may be insufficient to reduce cardiac risk.
Despite long-standing concerns about the cardiac risks associated with clozapine,1–3 tachycardia often remains undiagnosed and untreated in patients with schizophrenia and related psychotic disorders. 4 Tachycardia is an abnormally rapid heart rate (HR) often defined as a HR >100 beats per minute (bpm). 5 Clozapine-induced tachycardia is hypothesized to occur because of anti-muscarinic effects on cholinergic activity blockade and might contribute to cardiac complications and potentially fatal outcomes such as cardiomyopathy. 6
There are inconsistent reports about the prevalence and persistence of clozapine-induced tachycardia, with large variabilities in frequency estimates ranging between 3% and 70%.1,3,7–10 There are also questions about the temporal course of tachycardia,5,11,12 with some studies suggesting it is a transient entity temporally linked to dose titration.13,14 Study limitations include small heterogenous samples and narrow operational threshold for tachycardia. Given the cardiovascular risks in people with psychotic disorders, expert recommendations recommend repeated measurements of heart rate to better characterize cardiac risk and increase data reliability. 15 Finally, information is lacking about the extent to which β-blockers can normalize heart rates in this clinical group. 16
Using a longitudinal retrospective design, repeated monthly measurements of resting HR were collected for 6 months in a large group of stable patients with psychotic disorders. This study examined HR measurements and rates of tachycardia, and factors contributing to differences including clozapine dose/duration, plasma serum levels, and β-blocker treatment.
Methods
Participants
101 people with a psychotic disorder attending a clozapine community clinic in 2020. Inclusion criteria included continuous treatment with clozapine for at least 6 months, past the 18-weeks initiation period, and clozapine and norclozapine steady-state levels. Exclusion criteria included an inpatient admission within the audit period. Standard mandatory assessment for all patients included a monthly examination, 6 monthly clozapine levels, full blood analyses, HR measurements, and a side-effect checklist. Missing data were rare (0.8%). Other causes of tachycardia such as systemic illness (including myocarditis and cardiomyopathy) were excluded during clinical review.
Data extracted from medical databases included sociodemographics (age, gender, diagnosis, and smoking habits), clinical characteristics (diagnosis, medication type/dose/date), and metabolic parameters (supine HR and blood pressure, clozapine plasma level). Resting HR was measured with a blood pressure cuff after a rest period. Data integrity measures included inter-rater reliability of HR measurements against BP cuff. The most recent measurement of blood plasma level was utilized for analyses.
Statistical analyses
Analyses in Jamovi 23 17 included t test and correlational analyses with Spearman’s rho. Mixed models (General Analyses for Linear Models, GAMLj, Restricted Maximum Likelihood, REML) 18 examined HR variations and longitudinal effects using HR (bpm) as a dependent variable and time as a fixed factor (6 observation points). The fit of distribution first tested a homogenous group on time alone, followed by a model in which β-blocker (rated 0/1 categorical) was added as an independent and fixed factor after LRT analysis determination, before and after the addition of other factors.
Ethics
The project was conducted as part of a health service clinical quality improvement project with Institutional governance review approval and exemption from ethics review (#39941).
Results
Baseline demographics and clinical characteristics of all patients on clozapine, and subgroups comprising patients treated with β-blockers and patients not treated with β-blockers
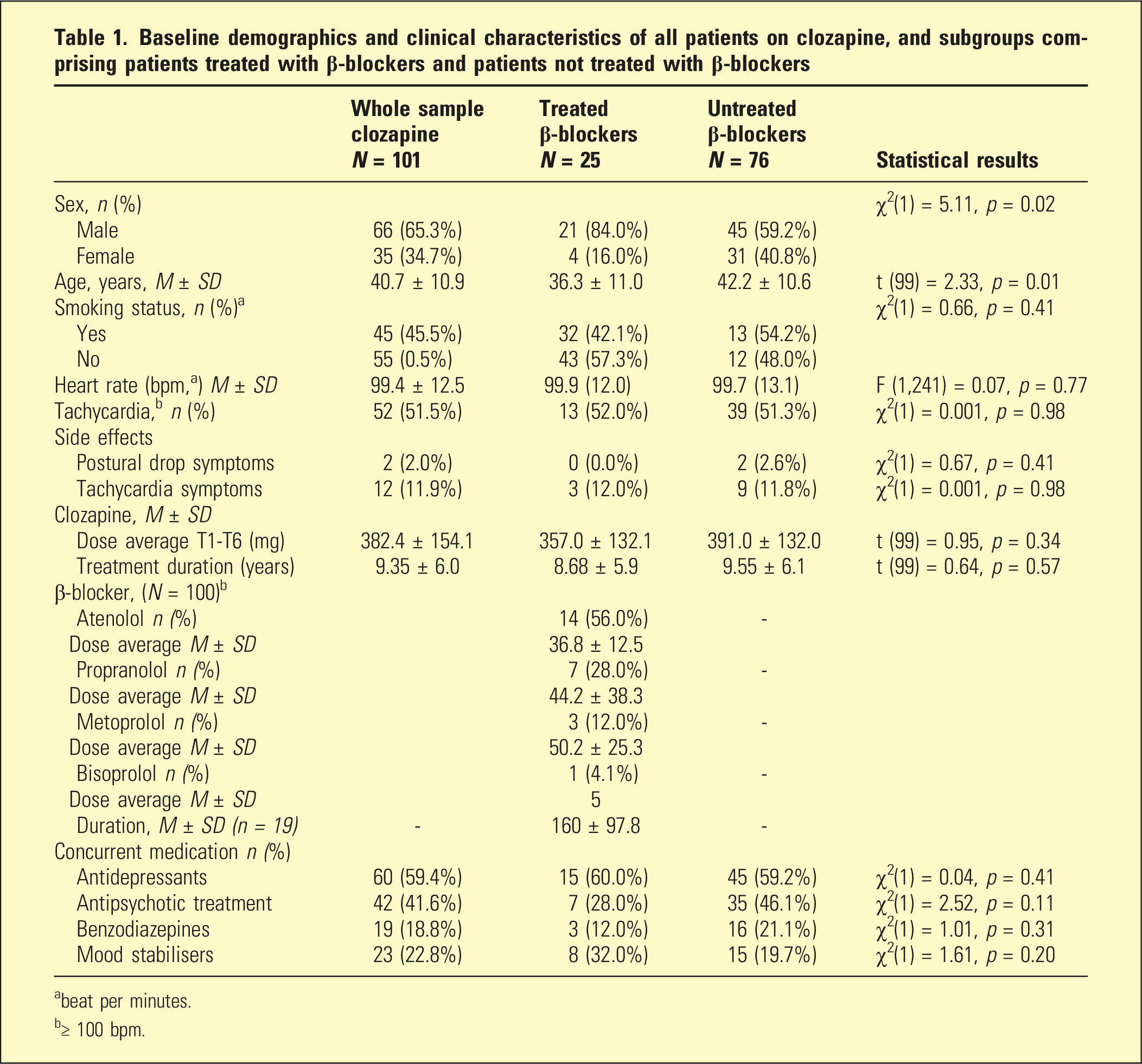
abeat per minutes.
b≥ 100 bpm.
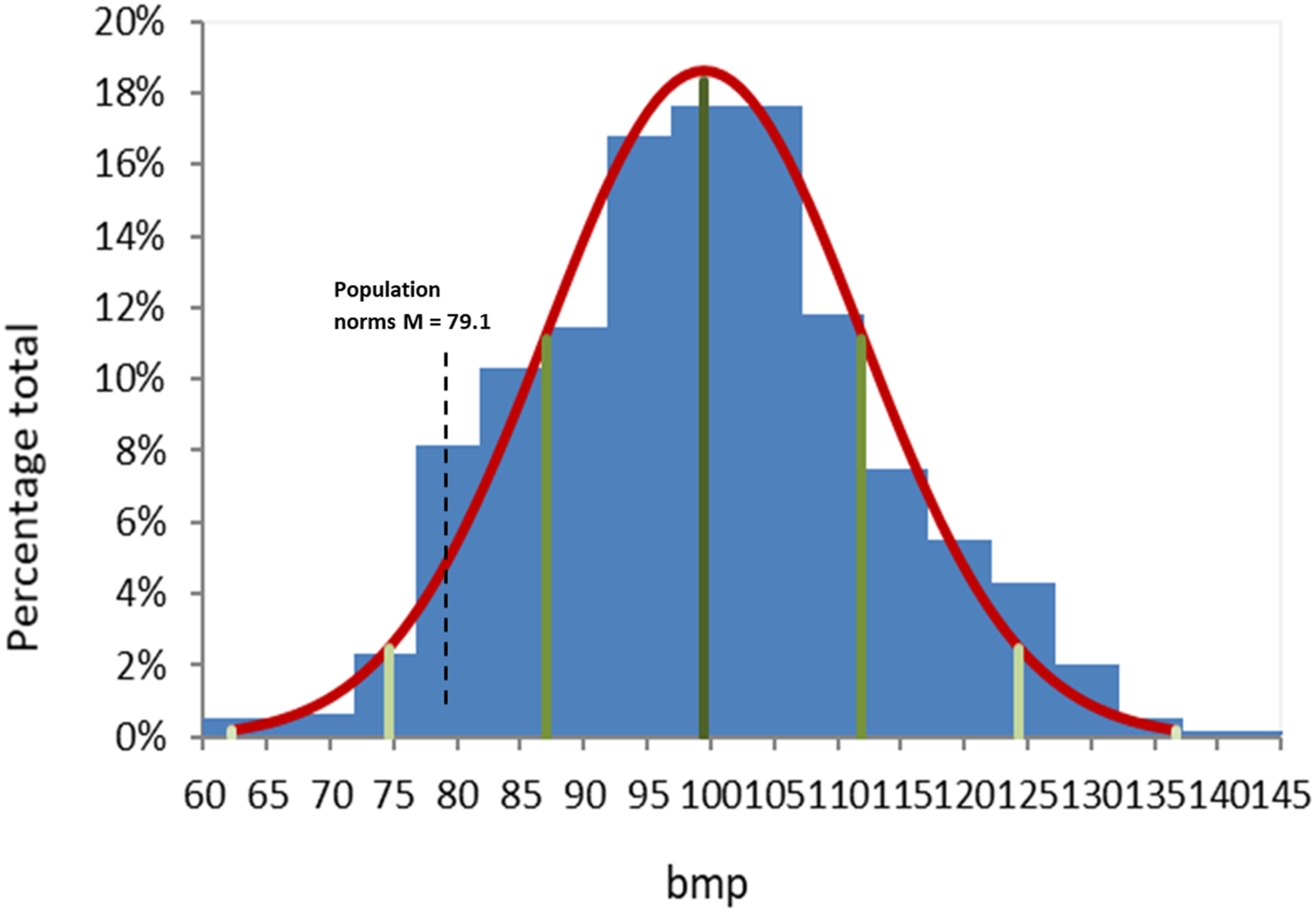
Histogram of heart rate with mean and quartiles (n = 101, 601 data points), with average general population norm for comparison 23 (black dotted line, average 79.1 bpm ± 14.5 STD).
Mixed model analyses demonstrated non-significant effects of time, F (5, 495) = 1.07, p = 0.37 (Model 1, Supplemental Material 2), but significant effects of β-blockers, showing that patients treated with β-blockers had significantly lower HR (bpm) than non-treated patients (Model 2, F (1, 233) = 8.13, p < 0.01, LRT = 313, p < 0.001). There were non-significant time x β-blockers interactions, F (5, 489) = 0.38, p = 0.86, suggesting that HR measurements remained constant within individual patients and treatment groups (Figure 2). As expected, the addition of covariates (Model 3) produced a slight decline in the variance of the intercept but the fit of the final model was good. The final model captured 61% of the variance (R2 Conditional = 61). After the inclusion of covariates, the only significant factors contributing to HR variations were the treatment group and age, F (1, 232) = 10.3, p < 0.01 and F (1,89) = 3.83, p < 0.05, respectively. Even holding other variables constant, the β-blockers treatment group contributed the largest variance compared to other factors and was associated with a predicted reduction of 6.3 bpm per individual patient. There were non-significant effects noted of clozapine (dose, plasma levels) or concurrent antipsychotics (all p > 0.25). Effects Plots of Resting Heart Rate over time: Observed score, with 95% Confidence Interval (95% CI).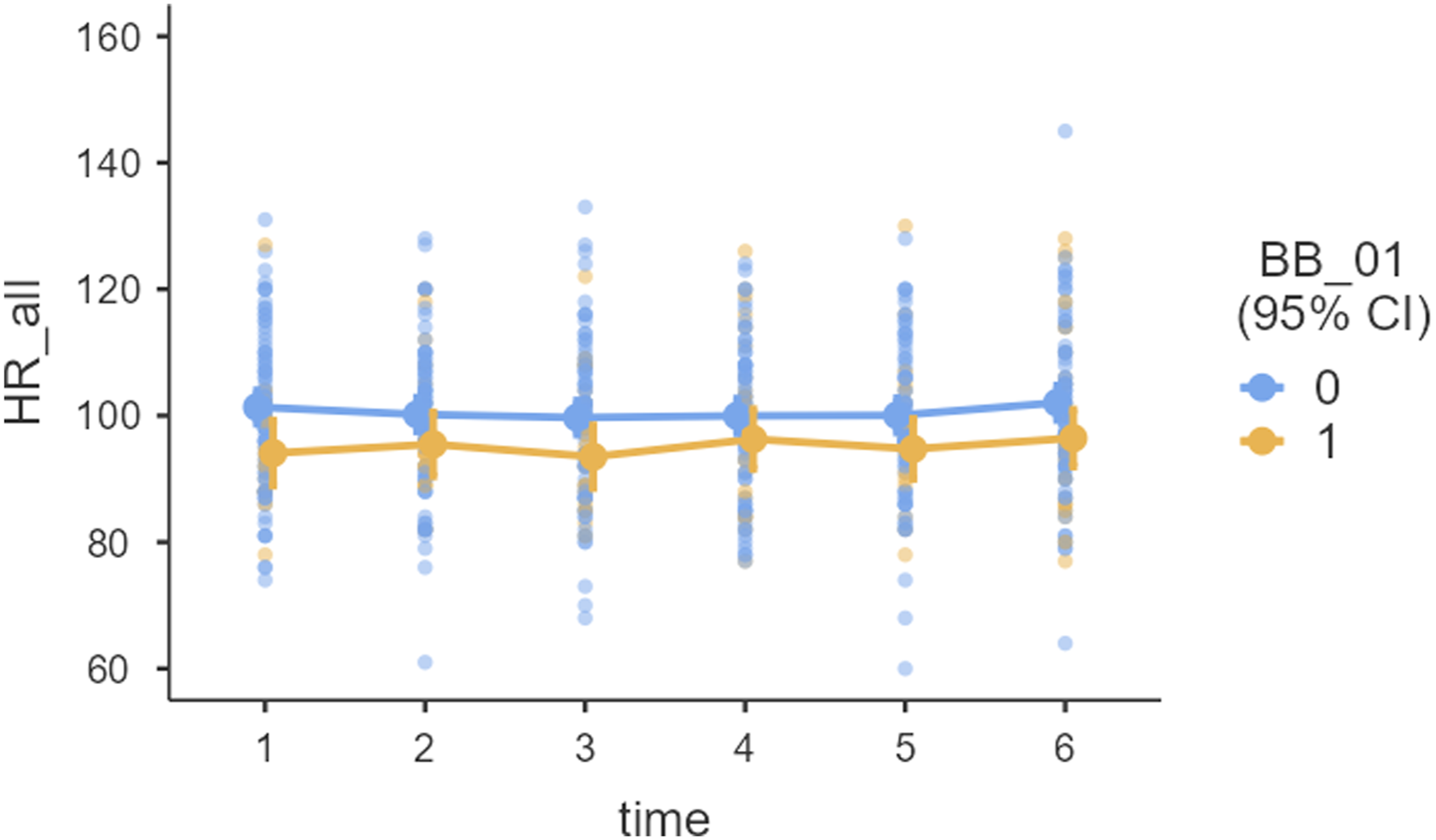
Discussion
This study examined the rates and persistence of clozapine-induced tachycardia and differences in patients treated for tachycardia. Substantially high rates of tachycardia were identified. Half (52%) of all participants had HR greater than 100 bpm, which is larger than the 25% reported by some1,3 but in line with other studies,8,19,20 and higher than 13% possibly accounted for by white-coat tachycardia. This suggests that, despite existing guidelines,1,7,21 many patients still are under-treated for tachycardia. Furthermore, HR values showed that the group average was approximately higher than population norms by 20 bpm.22–24 Given that the risk of adverse outcomes increases with higher HR, heart-rate parameters are increasingly used as a marker with prognostic significance (Norman et al., 2017). This has led experts to suggest a review of the “textbook” threshold of 100 bpm for tachycardia in some population groups to avoid incorrect assumptions about levels of risk. 15
In this study, regression analyses showed that effects on HR were specific to clozapine and not other antipsychotic medication, replicating existing findings 4 . It is worth noting that Martini 8 suggested that antipsychotic augmentation was a predictor of tachycardia, hence, this area requires further investigation. We found significant positive correlations between HR and clozapine levels, replicating previous results suggesting dose-dependent effects linearly related to pharmacokinetics.4,25 The effects are thought to occur via clozapine’s anti-muscarinic and chronotropic properties on increasing noradrenaline.4,25,26 Muscarinic m2 blockade leads to inhibition of both vagal action and presynaptic α2 adrenoreceptors, with indirect activation of β adrenoreceptors.
There was a positive correlation between elevated HR and plasma levels of clozapine and norclozapine. 27 The clozapine to norclozapine ratio was also significantly associated with HR suggesting that serum clozapine level might be more potent in its effect on HR than norclozapine. 25 This is of interest given the role of agents such as fluvoxamine in mediating these metabolites in clozapine augmentation. 28 However, the association was no longer significant in the mixed model analysis after the inclusion of all the variables, hence, caution is required in interpretation.
Our results showed tachycardia with significantly elevated HR even after almost 10 years of treatment and with minimal variation with repeated measurements across 6 months. These findings are contrary to classical teaching that would suggest that clozapine-induced tachycardia is a transient phenomenon,1,7,21 and that tachycardia is temporally related to the introduction or rapid titration of clozapine.13,14,21 Other studies have confirmed enduring tachycardia, albeit in a proportion of patients (10% to 50%)4,10,20 suggesting the contribution of individual differences. Interestingly, the current results showed that age made a more substantial statistical contribution than the duration of treatment when all covariates were combined (treatment duration was no longer significant). This would suggest that advancing age contributed to a greater degree than did tolerance to clozapine’s effects.
Pharmacokinetic interactions such as changing concomitant medication or smoking cessation are unlikely as these variables did not change during the study period. Nonetheless, smoking, clozapine levels, and tachycardia likely have complex interactions which this study was underpowered to delineate. Importantly for clinical practice,4,16 our findings demonstrated that β-blocker treatment was successful in reducing HR. However, the reduction was estimated to average 6.3 bpm, and a limited decrease would not be sufficient to return HR to normal ranges (from 99 to 79 bpm) or to make a difference statistically when using 100 bpm cut-off values. The lack of normalization of HR with β-blockers in patients with higher levels of tachycardia is a phenomenon previously reported in the general population. 29
Limitations in this study included an observational design and a lack of matched non-clozapine controls. In addition, we lacked data before treatment with β-blockers, so causal effects could not be measured. While our sample size was large compared to others on the topic, this study was underpowered to explore within group effects, including dosing differences or sex differences. Pharmacokinetic interactions such as changing concomitant medication or smoking cessation were unlikely as these variables remained stable during the study period.
From a clinical perspective, clozapine remains underutilized in resistant psychotic illness but it is also associated with unique side effects. Given the known risks of untreated tachycardia on cardiac remodeling and cardiomyopathy, in addition to the already increased cardiovascular burden present in patients with psychotic illness, the early identification and treatment of clozapine-induced tachycardia should be a strong consideration.
In conclusion, this study highlights the high prevalence and enduring nature of clozapine-induced tachycardia, indicating potential undertreatment despite existing guidelines. While β-blockers showed some effectiveness in reducing heart rate, the reductions were modest and did not bring rates back to normal. The study emphasizes the need for reevaluating assessment and management approaches. Recommendations include more systematic referrals to specialists should initial therapy not normalize heart rate. Rate-reduction strategies should be initiated as soon as the clinical threshold of risk is reached4,23 similar to the approach to hypertension and dyslipidemia for psychiatric patients. 21
Supplemental Material
Supplemental Material - Clozapine-related tachycardia: A conundrum to identify and treat
Supplemental Material for Clozapine-related tachycardia: A conundrum to identify and treat by Dermot Brennan, Sweta Lal, Frans Hugo, Flavie Waters and Gordon Shymko in Australasian Psychiatry
Footnotes
Acknowledgements
The authors would like to thank Dr Rooprai for his early feedback and Dr Siyaguna-Kosgodage for assisting with data collection.
Author contributions
DB, SL, and FH contributed to the concept, design, acquisition, and wrote the first draft; FW conducted the analyses and redrafted the manuscript; All authors contributed to the interpretation of data and provided critically intellectual content; All authors approved the version to be published.
Disclosure
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
The project was conducted as part of a quality improvement activity with institutional governance approval (Armadale East Metropolitan Mental Health Service, Approval number 39941).
Informed consent
It involved the retrospective analyses of de-identified data and did not require a consent process additional to that elicited as part of the patient’s routine clinical care.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
