Abstract
β-Blockers are widely used in the prophylaxis of migraine and have been described as very effective drugs in many studies. Some investigators have demonstrated that the clinical improvement of migraine corresponds to the normalization of the contingent negative variation (CNV), a slow cortical potential measuring cortical information processing. However, most of these studies have contained a variety of methodological pitfalls, which we attempted to address in the current study. Twenty patients suffering from migraine without aura were randomly divided into two groups. The groups were treated either with controlled-release metoprolol or placebo for 3 months, using a double-blind design. Twice before and once after each month of the treatment the CNV was recorded. After 3 months, a significant reduction of migraine frequency, duration and intensity was demonstrated for the metoprolol compared with the placebo group. The CNV was characterized by a marked reduction of the amplitude of the total CNV and postimperative negative variation and normalization of the eartly CNV habituation following treatment. Therefore, metoprolol may exert its prophylactic effect in migraine through the influence on cortical information processing and excitability represented by the CNV.
Introduction
β-Blockers are widely used as drugs of first choice in the prophylaxis of migraine (1, 2). A comparison of metoprolol with the most proven, standard drug propranolol, demonstrated few significant differences, but suggested that metoprolol may be better tolerated for the prophylaxis of migraine (3). Although the clinical efficacy of β-blockade is obvious, the mechanisms of the prophylactic effect of these drugs are still unknown and are mostly based on speculation.
The most likely action of β-blockers is central, mediated by inhibiting central β-receptors interfering with the vigilance-enhancing adrenergic pathway, interaction with 5-HT receptors and cross-modulation of the serotonin system. The central nervous system effect of β-blockers in migraine patients has been demonstrated. Metoprolol and propranolol caused a significant reduction of abnormally increased visual evoked potential (VEP) amplitudes (4, 5) and normalization of an enhanced intensity dependence slope of auditory evoked potentials (AEP) (6), demonstrating an influence on abnormal cortical information processing and excitability in migraine.
The influence of β-blockers on the contingent negative variation (CNV) has been investigated in several studies. The CNV is a slow cortical event-related potential that is recorded from the scalp between two stimuli while a subject is waiting for the second event and preparing for task performance (7). This potential is related to the level of cortical excitability following activation in the striato-thalamo-cortical loop and reflects different stages of information processing (8). Migraine patients are characterized by increased amplitudes and reduced habituation of the CNV, especially of its early component (9–11). The CNV may represent the attack anticipation because its amplitude and habituation change during the headache-free interval. There is a gradual increase of abnormalities a few days before a migraine attack, so that the maximal negativity and the most pronounced loss of habituation may be observed just before an attack (12).
Maertens de Noordhout et al. have shown CNV normalization in response to β-blockers, which is consistent with central adrenergic hyperactivity in migraine (13). Migraineurs with elevated CNV scores have a much better response to β-blocker therapy (80% effective) than migraineurs with a low or normal score (22% effective), suggesting that the CNV may predict the efficacy of treatment with β-blockers (14). Ahmed (15) replicated the results of Maertens de Noordhout and demonstrated a significant decrease of the CNV amplitude during treatment with β-blockers. The clinical efficacy of β-blockade corresponded to the normalization of the CNV amplitude.
However, most of the CNV studies listed above have a number of methodological shortcomings. First, none of the studies investigating the influence of β-blockers on the CNV or other neurophysiological parameters have employed randomized, double-blind, placebo-controlled designs. Placebo controls are especially important in CNV studies because this potential is sensitive to the psychological state of a subject (attention, motivation, emotional involvement, resource mobilization, etc.) (7). The same psychological parameters influence the placebo response. Therefore, without a placebo comparison it is very difficult to differentiate between the real central pharmacological effect of β-blockers and the consequence of, for example, pain release after treatment. Moreover, placebos have been shown to influence the CNV, so this has to be taken into account (16).
Second, lacking a placebo comparison and limiting analysis of CNV data to just two time points (before and after treatment), one cannot rule out regression to the mean as a possible cause for significance. This is especially so, as most patients have been selected on the basis of having deviant CNV values. Multiple recordings collected during treatment (along with the appropriate placebo comparison condition) can afford a more differentiated basis for analysing the relationship between the clinical course of migraine and changes in CNV parameters.
Finally, the above-mentioned studies did not analyse the effect of β-blockers on different CNV components. As pointed out previously, the amplitude and habituation of the early CNV component are typically abnormal in migraineurs (10, 11). The early CNV response is related to noradrenergic activity (7), so it can be hypothesized that normalization of the CNV under β-blockade may first and foremost be explained by the normalization of the early CNV. This remains to be investigated.
In the present study we tried to overcome the described problems and investigate the influence of a controlled-release (CR) form of metoprolol on the amplitude and habituation of the early and late CNV components using a double-blind, placebo-controlled, parallel-group design with systematic multiple CNV recordings during the treatment phase in order to provide more complete analysis of the treatment process. The CR formulation of metoprolol was chosen because it provides a plasma concentration–time profile with once-daily dosing, so that it is possible to achieve fairly constant and effective steady-state plasma concentrations throughout a 24-h period (17). Such properties should offer therapeutic and scientific benefits, for example, by minimizing peak plasma-related adverse effects and avoiding concentration-related fluctuations in neurophysiological parameters. The efficacy of metoprolol-CR in the prophylactic treatment of migraine has been demonstrated and well replicated (18, 19), so that this formulation seems to be more beneficial for neurophysiological studies than any conventional form.
Subjects and methods
Subjects
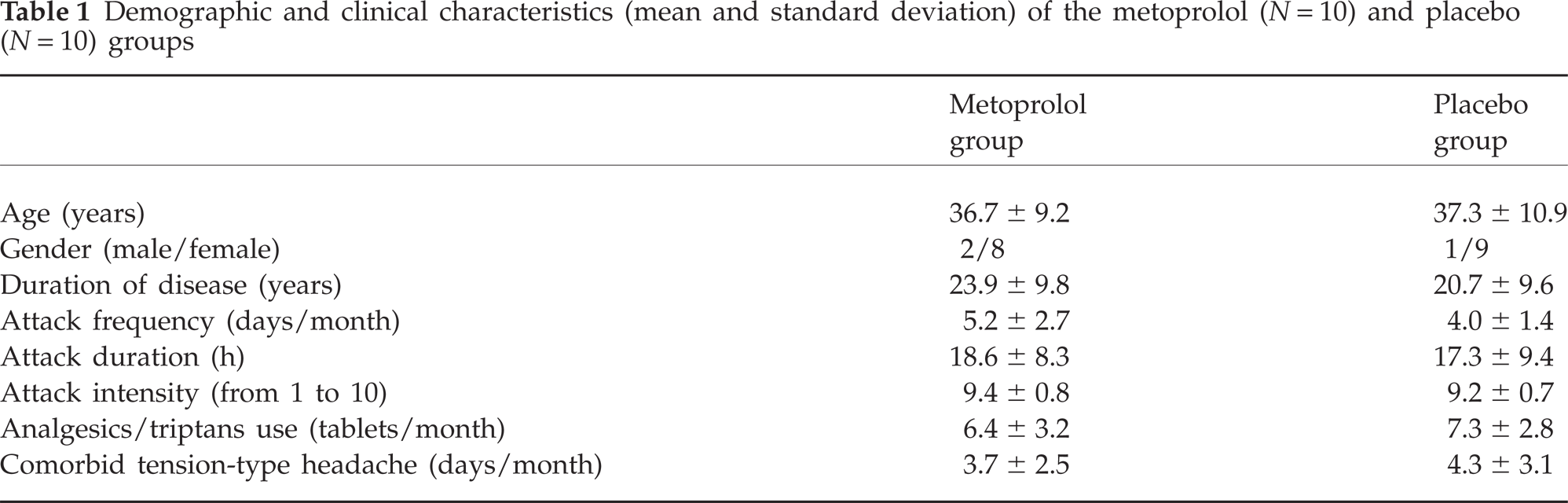
Twenty patients suffering from migraine without aura were recruited from the out-patient departments of the Clinic of Neurology (n = 5) of the Institute of Medical Psychology (n = 3) and by placing advertisements in the local newspaper (n = 12). None of the enrolled subjects dropped out during the study. The diagnosis was made according to the criteria of the International Headache Society (20) (code 1.1) by the first author (neurologist) trained in diagnosis and treatment of headaches. The patients were randomly divided into two groups. One group (n = 10) was treated with metoprolol-CR and the other (n = 10) with placebo. Table 1 presents demographic and clinical characteristics of the groups. No significant differences were found between the groups for any of the variables examined. Seventeen patients had no other experience with prophylactic antimigraine medications. The other three patients did not take any prophylactic medication during the 6 months before inclusion in the study.
Demographic and clinical characteristics (mean and standard deviation) of the metoprolol (N = 10) and placebo (N = 10) groups
Inclusion criteria consisted of age between 18 and 60 years, both sexes, migraine history of at least 12 months' duration and a mean number of 2–10 migraine attacks per month within the last 3 months prior to the study. Exclusion criteria were pregnancy or lactation; abuse of ergotamine, triptans or analgesics (intake on >12 days within a month); any prophylactic treatment of migraine (drugs, psychotherapy, acupuncture, and others) during the 6 months preceding the trial; neurological, psychiatric or internal diseases during the treatment and in the last year before the study; all specific contraindications for β-blockers (AV block, bradycardia, hypotension, diabetes mellitus, asthma, etc.); concomitant non-migraine headaches more than three times per month within the last 3 months; alcohol or drug addiction according to the Diagnostic and Statistical Manual of Mental Disorders-IV; or change in oral contraceptive therapy 3 months before or during the study. Prior to study entry, the patients underwent physical and neurological examination, including ECG and EEG (no abnormalities were observed).
The study was approved by the Ethics Committee of the Faculty of Medicine, University of Kiel, Germany. The subjects were informed about the course of the experiment and gave written informed consent according to the Helsinki convention.
Study design and treatment
The study was designed as a randomized, parallel-group, double-blind, placebo-controlled clinical trial. The first visit included recording of medical and migraine history, a physical examination and providing a detailed description of the study. Patients who fulfilled the inclusion criteria entered a 4-week baseline period without any prophylactic treatment. During the baseline phase all patients were qualified for randomization to metoprolol or placebo. The treatment period consisted of 3 months. During the first month the treatment began with the gradual increase of the metoprolol/placebo dose in steps of 50 mg (1/2 tablet) per week. Because of the controlled-release formulation of metoprolol, all patients took the medicine once in the morning. The maximal dose of 200 mg metoprolol (two tablets verum or placebo) was reached in week 4 of treatment. Treatment lasted an additional 2 months. After the third month of the treatment period a gradual decrease of the metoprolol dosage was performed, also in steps of 50 mg per week. During the baseline phase and the whole period of treatment the patients were required to keep a headache diary to record the following parameters: days on which a migraine attack occurred, duration of migraine attack in hours, intensity of headache (three assessment times per day using a visual analogue scale), dosage of all medications taken, and side-effects. Patients were allowed to use whichever other medication they found helpful for aborting migraine attacks. However, they were instructed not to change this medication or to try any new drug during treatment. During the baseline period the CNV was recorded twice: at the beginning and to the end of the baseline phase. Neurophysiological data were collected at the end of each treatment month. In general, five recordings (two baseline and three recordings under treatment) were performed for each patient.
Contingent negative variation
All participants were seated in an armchair with open eyes in an electrically shielded, sound-attenuated room. The auditory warning (S1) and imperative (S2) stimuli (75 dB(a)) were produced by a loudspeaker located behind the subject. The interval between S1 and S2 was 3 s. A CNV session consisted of 32 trials in which the subject had to react to the imperative stimulus (GO-response). Additionally, eight trials were randomly presented where no reaction was expected (NO-GO-response). The warning stimulus (S1) for the GO-response had a frequency of f = 1000 Hz and lasted 100 ms. The warning tone for the NO-GO-response had a frequency of f = 200 Hz. The imperative stimulus (S2) had a frequency of f = 2500 Hz, lasted a maximum of 1500 ms and was deactivated by pressing a button. Reaction time was defined as the period between onset of S2 and the pressing of the button. S1 and S2 pairs were offered at random intervals of 10–15 s. The duration of one recording was 6 s (the recording began 1 s before S1 and ended 2 s after S2). The period between recording onset and S1 was taken as the baseline for all measurements.
The EEG was recorded using non-polarizable Ag/AgCl electrodes over Cz according to the International 10–20 System with linked mastoids as the reference. The electrode site on the scalp was prepared with alcohol and abraded with rough paper, resulting in an electrode impedance of <7 kΩ. The EEG signals were amplified using a Nihon Kohden amplifier with a time constant of 5 s (equivalent to a high-pass filter of 0.03 Hz) and a low-pass filter of 35 Hz digitized at a rate of 100 Hz for each channel. Vertical eye movement artefacts were excluded by a parallel recording of the electro-oculogram (EOG) with electrodes (Ag/AgCl) positioned 1–1.5 cm above and below the right eye. The trial was rejected if EOG deflections >20 µV interfered with 5 s of the EEG recording. A protocol listed the number of rejected trials for each recording. Approximately two to six (4.05 ± 2.16) of the EEG trials were rejected due to eye movement artefacts. There were no significant differences between groups according to the number of rejected trials.
The GO-trails were averaged and the amplitudes of the total CNV, early CNV, late CNV and postimperative negative variation (PINV) were calculated. The total CNV was assessed between 500 and 3000 ms following S1. The early CNV component was defined as the mean amplitude in a window of 200 ms duration around the maximal amplitude of the expectancy wave between 550 and 750 ms after S1 (21). The late CNV component was the mean amplitude during the 200 ms preceding S2. PINV was the mean amplitude of CNV between 500 and 2000 ms following S2. Each recording was divided into eight blocks of four consecutive trials to determine the course of habituation and trends in the early CNV amplitudes. Only the habituation of the early CNV was considered because of its special relevance to migraine (10–12).
Statistical analysis of the CNV
Because the CNV data were normally distributed, characterized by homogeneous variances and correlated with each other, analysis of variance was used as the main data analytic tool. Two sets of analyses were employed. In the first, separate 2 × 4
The course of the early CNV habituation was estimated using a second
Statistical analysis of clinical efficacy
The efficacy profiles of metoprolol-CR and placebo were evaluated using a single-case time–series autoregressive-integrated-moving-average (ARIMA) methodology, which provides an appropriate statistical model for long courses of daily self-evaluations (23). This method employs a modified product–moment correlation in order to determine the serial dependency of sequential data. It is particularly suited for clinical serial data, and therefore also for headache studies, and its success has been demonstrated (24). It is well known that the population of migraine patients is rather heterogeneous. Therefore, single-case statistics are an alternative to the construction of the ‘arithmetic mean patient’, normally used by inference statistical methods such as group t-tests or
Results
Clinical efficacy of metoprolol-CR
Figure 1 presents the clinical efficacy of metoprolol-CR compared with placebo. A significant reduction was found in the number of days with migraine/month, attack intensity and duration of headache under metoprolol-CR compared with placebo. Concerning side-effects, four patients treated with metoprolol-CR reported tiredness (n = 2), dizziness (n = 1), and cardiovascular problems (n = 1) compared with three patients treated with placebo, who developed gastrointestinal disturbances (n = 2) and tiredness (n = 1).

Clinical efficacy of metoprolol-CR. Aggregated z-values of autoregressive-integrated-moving-average (ARIMA) analyses (headache diary, baseline vs. treatment), z-values ≤−1.96 indicate a significant reduction in the number of days with migraine, attack intensity and duration of a migraine attack. ▪, Metoprolol; □, placebo.
For the validation of results of ARIMA statistics, Wilcoxon signed ranks tests were performed for each group (metoprolol-CR vs. placebo) comparing the attack frequency during the baseline period and the frequency of migraine during the last months of treatment. The frequency of migraine attacks was significantly reduced under metoprolol-CR (baseline 5.2 ± 2.7 attacks/month, third month of treatment 2.2 ± 1.3 attacks/month, difference Z = −2.68; P = 0.01), but not under placebo (baseline 4.0 ± 1.4 attacks/month, third month of treatment 3.1 ± 1.66 attacks/month, difference Z = −1.67; P = 0.094).
Contingent negative variation
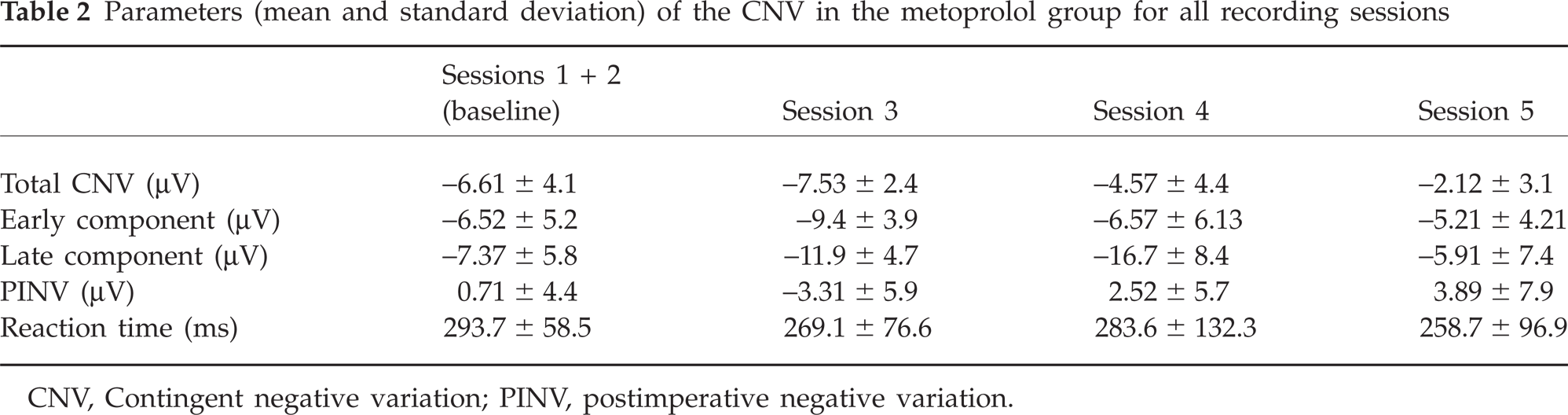
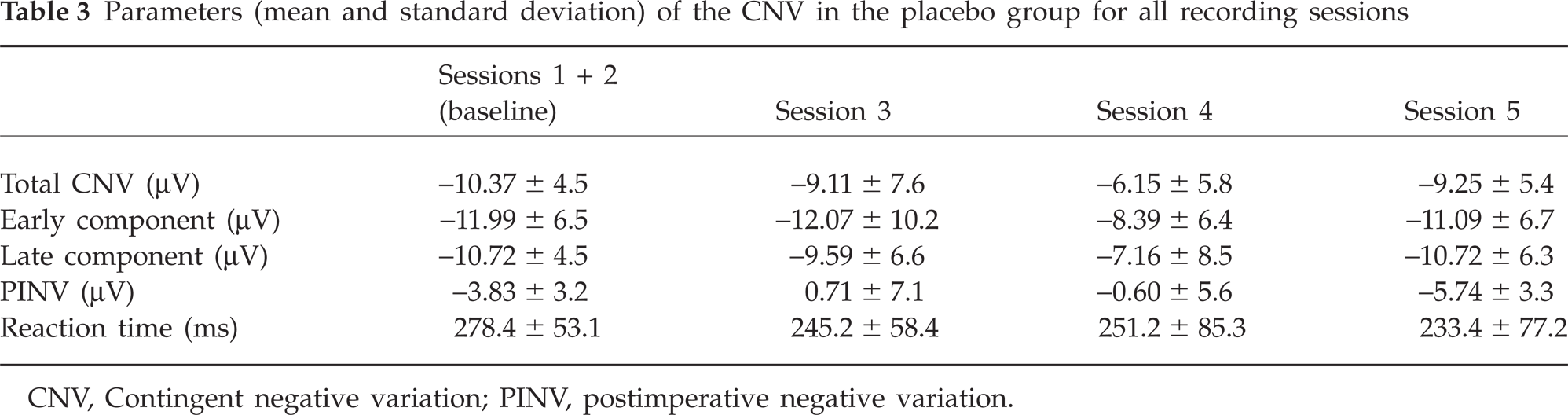
Table 2 presents CNV data for the metoprolol group of migraineurs for the pretreatment period as well as the treatment phase. Table 3 shows the same CNV parameters recorded in the placebo group. Table 4 summarizes results of between-group comparisons performed for each point of investigation. Figures 2 and 3 demonstrate habituation of the early CNV component for each group. Although the amplitudes of the CNV in the baseline phase were larger in the placebo group compared with the metoprolol group, the differences between the groups were non-significant. The analysis of variance demonstrated a significant main effect for ‘Session’ for the total CNV (F(3, 51) = 5.335; P = 0.003) and the early CNV component (F(3, 51) = 2.884; P = 0.045). No main effects emerged for ‘Group’. The interaction effect (‘Session × Group’) was significant for the total CNV (F(3, 51) = 2.440; P = 0.05) and PINV (F(3, 51) = 6.708; P = 0.001). Indeed, the total CNV and PINV demonstrated a more pronounced reduction of the amplitude from session to session in the metoprolol group compared with placebo group. This was confirmed by post hoc tests. During the treatment, the differences according to CNV parameters became significant only in the last session (the third month of treatment) and only for the total CNV and the PINV (see Table 4). The amplitude of the late CNV component as well as the reaction time did not show any changes over time and did not differ between the groups.

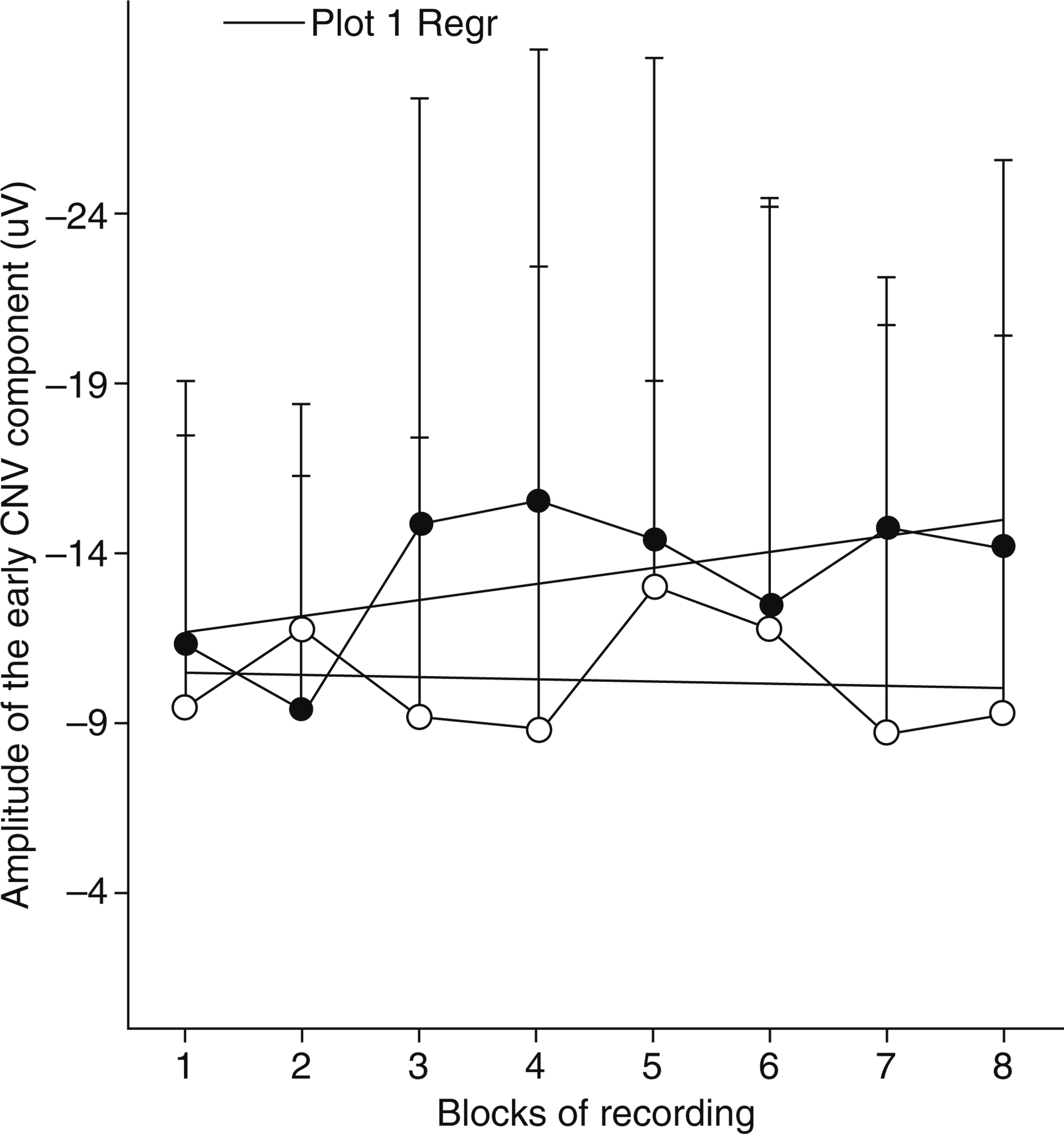
Habituation of the early contingent negative variation (CNV) component (amplitudes of the early CNV in eight blocks of recordings and slopes of the linear regression for each course of habituation) presented for session 1 + 2 (•) and the last session 5 (○) in the metoprolol group.
Habituation of the early contingent negative variation (CNV) component (amplitudes of the early CNV in eight blocks of recordings and slopes of the linear regression for each course of habituation) presented for session 1 + 2 (•) and the last session 5 (○) in the placebo group.
Parameters (mean and standard deviation) of the CNV in the metoprolol group for all recording sessions
CNV, Contingent negative variation; PINV, postimperative negative variation.
Parameters (mean and standard deviation) of the CNV in the placebo group for all recording sessions
CNV, Contingent negative variation; PINV, postimperative negative variation.
Results of comparison (t-values/P as significance of difference, degree of freedom is d.f. = 18 for all comparisons) between metoprolol and placebo groups concerning CNV parameters for each session
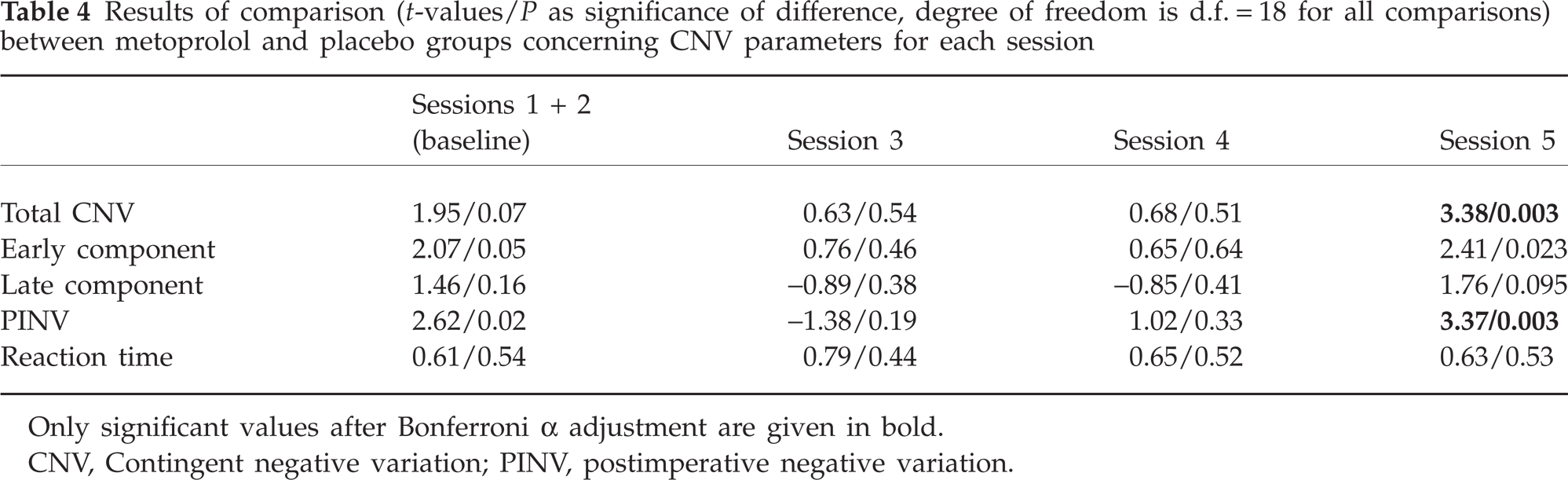
Only significant values after Bonferroni α adjustment are given in bold.
CNV, Contingent negative variation; PINV, postimperative negative variation.
In examining the habituation of the early CNV component, only a significant three-way interaction ‘Group × Session × Habituation’ (F(21, 336) = 1.603; P = 0.046) was observed. Therefore, the habituation of the early CNV amplitude increased significantly from session to session and this increase was more pronounced in the metoprolol group compared with the placebo group (see Figs 1 and 2).
The more marked decrease of the amplitude of total CNV and PINV and the increase of the early CNV habituation under metoprolol-CR compared with placebo can not be related to differences between the groups according to the distance to the previous or next migraine attack (Table 5). The analysis of variance revealed neither any significant main effect nor a significant interaction between ‘Group’ and ‘Session’ according to the time passed from the last migraine attack or time between the recording and the next attack.
Time between a CNV recording and a (previous or next) migraine attack (mean and standard deviation) given for each session and each patient's group
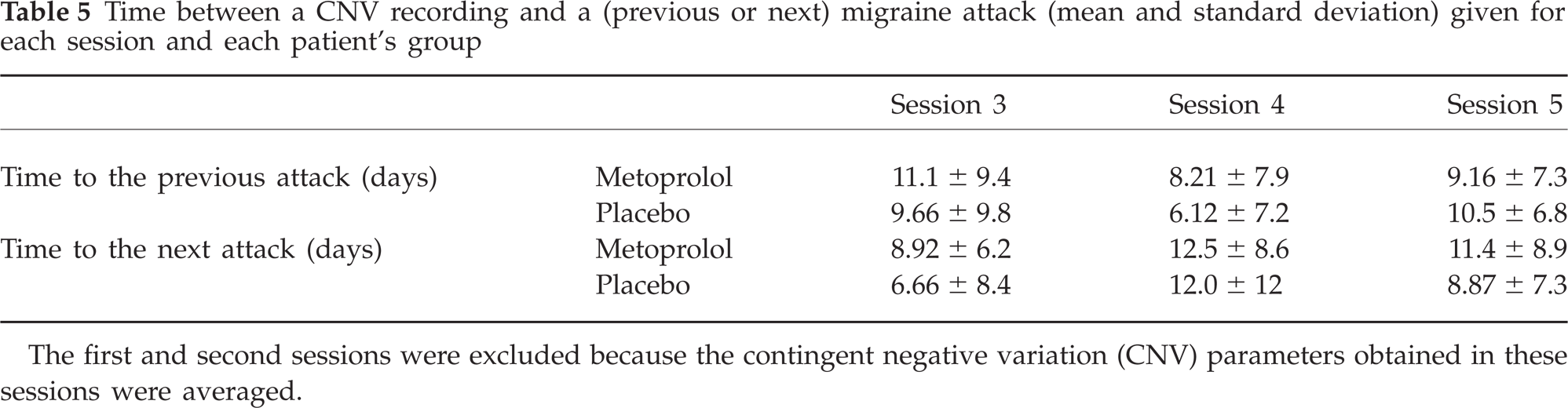
The first and second sessions were excluded because the contingent negative variation (CNV) parameters obtained in these sessions were averaged.
Discussion
This study has demonstrated that the efficacy of metoprolol may be related to changes in cortical excitability and information processing, because during treatment a significant reduction in the amplitude of the total CNV and PINV and an increase in the habituation of the early CNV component were observed in the metoprolol group, but not in the group treated with placebo. This study confirms previously published results concerning the influence of β-blockers on the CNV (13–15).
It seems likely that the prophylactic efficacy of metoprolol is due to its central mechanisms (14). It has been demonstrated in a number of studies that the amplitude of the CNV and of VEPs normalizes and an abnormal amplitude/stimulus intensity function slope of AEPs reduces under metoprolol (4–6, 13–15). The intensity of CNV, VEP and AEP play a specific role in the pathogenesis of migraine and characterizes pathological processes that are closely related to the initiation of a migraine attack. Amplitudes, habituation and the intensity dependence slope of these potentials change during the headache-free interval, representing attack anticipation and increased susceptibility of the brain to precipitating agents (12, 25, 26). For example, the CNV shows its maximal negativity and the most pronounced loss of habituation just before a migraine attack, and this has a predictive value: the larger the CNV amplitude, the greater the probability of a migraine attack (12, 27). It could be suggested, therefore, that metoprolol-CR induces changes in cortical excitability and information processing reflected by the CNV and, in such a way, elevates the threshold of a migraine attack and reduces the attack anticipation. This could help explain the clinical efficacy of the drug.
Surprisingly, only the total CNV and PINV were reduced during treatment with metoprolol-CR. As demonstrated in previous studies, the early CNV component was characterized by a sufficient specificity and sensitivity to migraine (12, 16, 27). The early CNV is related to the noradrenergic activity in the brain and mechanisms of the orienting response, the function mainly influenced by the activity of locus coeruleus (7). Therefore, the most pronounced changes in CNV under β-blockers could be expected for the early CNV component. Our expectations, however, were not realized. The amplitude of the early CNV demonstrated a tendency to change only under metoprolol-CR. How may these results be explained? First, the non-significant changes of the early CNV may be a consequence of type II error, i.e. of an insufficient number of patients in groups. A replication of this study employing a larger number of patients would possibly solve this problem. Second, the CNV represents a measure of cortical excitability in the underlying neural tissue (7, 8). This depends on the state of cortical neurons (extent of activity) and their environment (lactate level, pH, ion concentration), the mechanisms regulating the threshold of excitability via the striato-thalamo-cortical loop, and the activity of subcortical structures (locus coeruleus, raphe nuclei, midbrain reticular formation), which are responsible for the occurrence and maintenance of central arousal (28). β-Blockers could influence cortical excitability either through direct noradrenergic projections to the cortex or through non-direct noradrenergic influences on cortical neurons via the striato-thalamo-cortical loop or hippocampus (29). Therefore, the changes in voltage of slow cortical potentials (total CNV and PINV) independently on a particular component may be explained by a non-specific broader influence of β-blockers on central structures and biochemical systems.
However, as expected, the habituation of the early component normalizes under metoprolol-CR. Because the loss of habituation in response to external stimuli represents one of the most important features of the migraineous brain (30), its normalization during metoprolol treatment additionally underlines the special role of this phenomenon in the pathogenesis of migraine. Habituation of the early CNV component bridges the gap between noradrenergic activity, sensory information processing and migraine (31). Noradrenaline is broadly distributed in areas concerned with the representation of the world and conjunction of sensory and motor inputs. It is released in times of novelty and uncertainty. The most effective and reliable stimulus for eliciting a noradrenergic response is that which disrupts behaviour and evokes the orienting response (32). Therefore, by influencing the habituation of the early CNV component, metoprolol-CR normalizes cortical information processing related to orienting activity, changes cortical preactivation and excitability, and decreases the vulnerability of the brain to migraine precipitants.
In summary, this is the first randomized, double-blind, placebo-controlled study to focus on neurophysiological effects of migraine prophylactic agents and use a longitudinal design with multiple measurements in order to avoid pitfalls associated with past research. Because its main topic was the influence of metoprolol-CR on different CNV components, most attention was paid to repeated CNV recordings in individual cases (resulting in 100 observations) and not to the investigation of clinical efficacy in large groups of patients. For small group sizes, as in this study, single-case analysis is preferred to the traditional statistical method, which is mostly insensitive to effects in small groups. Therefore, ARIMA statistics were carried out, which demonstrated a significant clinical effect of metoprolol-CR compared with placebo. The validity of these results is supported by a number of other studies, providing strong evidence for clinical efficacy of metoprolol (for review see (1, 2)).
In conclusion, our results confirm the central mechanisms of metoprolol-CR and underline the role of adrenergic activity and the orienting response in the pathogenesis of migraine.
