Abstract
Introduction:
Acute pulmonary embolism is a confirmed cause of up to 5% of out-of-hospital cardiac arrest and 5%–13% of unexplained cardiac arrest in patients. However, the true incidence may be much higher, as pulmonary embolism is often clinically underdiagnosed. Thrombolytic therapy is a recognized therapy for pulmonary embolism–associated cardiac arrest but is not routinely recommended during cardiopulmonary resuscitation. Therefore, clinicians should attempt to identify patients with suspected pulmonary embolism. Many point-of care ultrasound protocols suggest diagnosis of pulmonary embolism for cardiac arrest patients.
Case presentation:
We describe two male patients (60 years and 66 years, respectively) who presented to the emergency department with cardiac arrest within a period of 1 week. With administration of point-of care ultrasound during the ongoing cardiopulmonary resuscitation in both patients, fibrinolytic therapy was initiated under suspicion of cardiac arrest caused by pulmonary embolism. Both patients had return of spontaneous circulation; however, only the second patient, who received fibrinolytic therapy relatively early, was discharged with a good outcome. In this report, we discussed how to diagnose and manage patients with cardiac arrest–associated pulmonary embolism with the help of point-of care ultrasound. We also discuss the different clinical outcomes of the two patients based on the experience of the clinicians and the timing of thrombolytic agent application.
Conclusions:
If acute pulmonary embolism is suspected in patients with out-of-hospital cardiac arrest, we recommend prompt point-of care ultrasound examination. Point-of care ultrasound may help identify patients with pulmonary embolism during cardiopulmonary resuscitation, leading to immediate treatment, although the clinical outcomes may vary.
Introduction
Pulmonary embolism (PE) is a potentially reversible cause of shock and cardiac arrest. 1 Acute PE is a confirmed cause of up to 5% of out-of-hospital cardiac arrest (OHCA) and 5%–13% of unexplained cardiac arrest in patients.2,3 However, in the clinical setting, PE is often not suspected and is underestimated as a cause of acute cardiopulmonary collapse4,5
In many cases, the diagnosis of acute PE in the situation of cardiac arrest is difficult, and PE is established only during autopsy after unsuccessful cardiopulmonary resuscitation (CPR). One post-mortem study reported that the clinical diagnosis of PE was missed in 84% of all PE cases. 6
Recently, many emergency physicians have been using point-of-care ultrasound (POCUS) to evaluate patients with cardiac arrest and identify reversible causes of pulseless electrical activity (PEA). Furthermore, many POCUS protocols suggest diagnosis of not only PE but also pericardial effusion, tension pneumothorax, and hypovolemia in patients with cardiac arrest.7–14
In this report, we present two cases with opposite prognoses based on the timing of thrombolytic agent application during cardiac arrest secondary to PE and assisted by POCUS; thus, we suggest the potential benefits of using POCUS to guide the administration of thrombolytic agents (Table 1).
Patient characteristics.
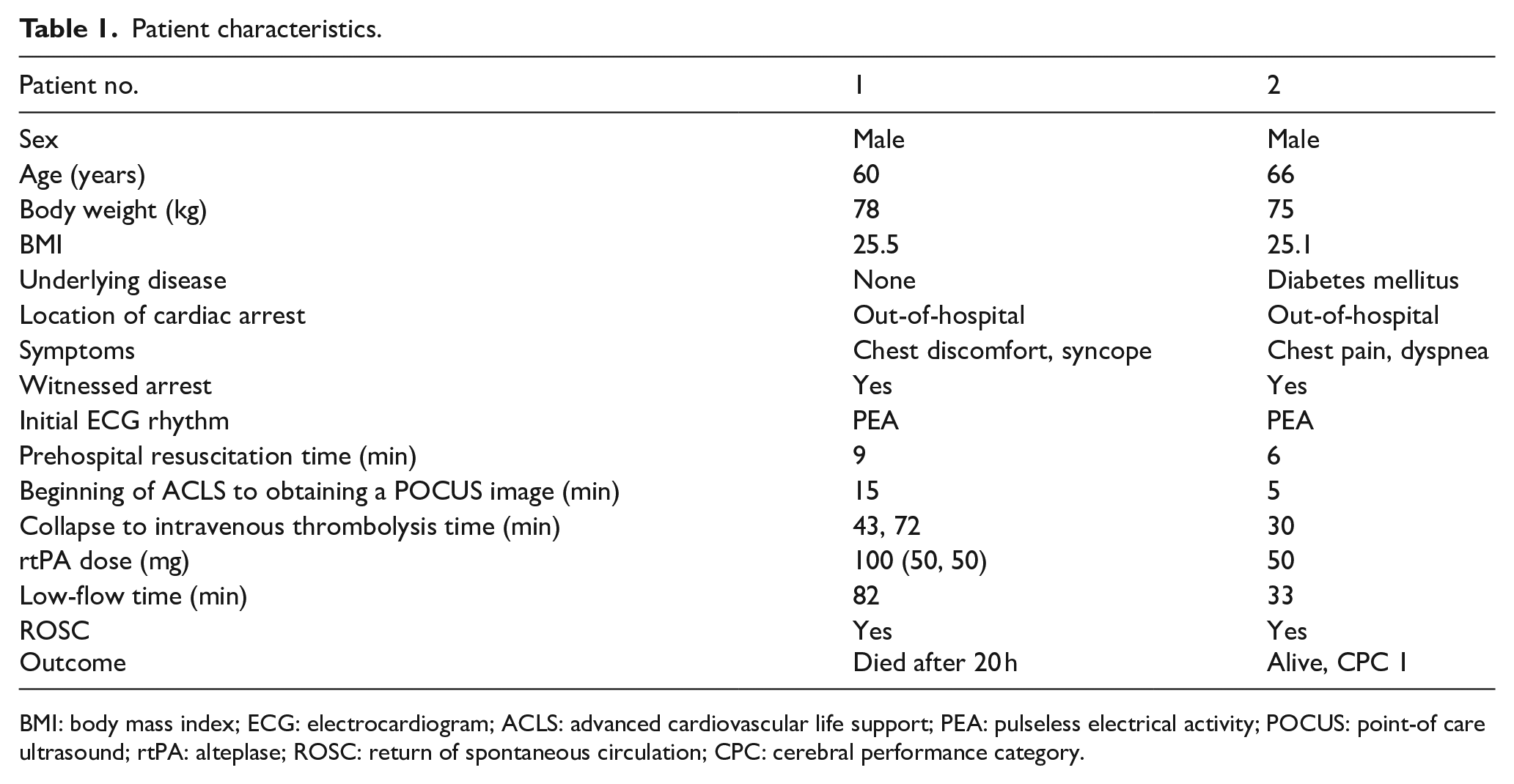
BMI: body mass index; ECG: electrocardiogram; ACLS: advanced cardiovascular life support; PEA: pulseless electrical activity; POCUS: point-of care ultrasound; rtPA: alteplase; ROSC: return of spontaneous circulation; CPC: cerebral performance category.
Case presentations
Case 1
A 60-year-old man presented to the emergency department (ED) with cardiac arrest. He was previously healthy but suddenly felt chest discomfort and syncope while waiting for physical therapy at the local orthopedic clinic. During the transit through emergency medical service (EMS), he fell into PEA arrest and basic life support (BLS) was administered for 9 min by emergency medical technicians (EMTs). On arrival at the ED, the patient was in a comatose mental state, his femoral pulse was not palpable, but electrocardiogram (EKG) monitoring showed PEA. Endotracheal intubation was performed immediately, and advanced cardiovascular life support (ACLS) was initiated simultaneously. Arterial blood gas (ABG) before endotracheal intubation showed severe respiratory acidosis with severe hypercapnia (pH 6.8, PaO2 5.2 mm Hg, PaCO2 90.6 mm Hg, HCO3 −13.4 mmol/L), despite 100% oxygen support using a bag valve mask. Along with conventional ACLS after ED arrival about 15 min, portable echocardiography revealed right ventricle (RV) enlargement with a flattened left ventricle as well as mechanical activities. Bedside views were performed at interval of “pulse-checks” during ACLS to avoid interruption of the ACLS protocol. Return of spontaneous circulation (ROSC) and cardiac arrest occurred 6 times each during the 34 min of ACLS implementation. Thrombolysis was determined to be due to cardiac arrest caused by massive pulmonary thromboembolism (PTE), and 50 mg of alteplase (rtPA; Actilyse; Boehringer Ingelheim Pharma GmbH & Co., Ingelheim, Germany) was administered immediately via intravenous (IV) bolus. After the first administration of rtPA, ROSC and PEA arrest occurred 3 more times, and a second dose of 50 mg of rtPA was administered by bolus, 29 min later from first administration, and sustained ROSC occurred after additional 10 min of ACLS. A
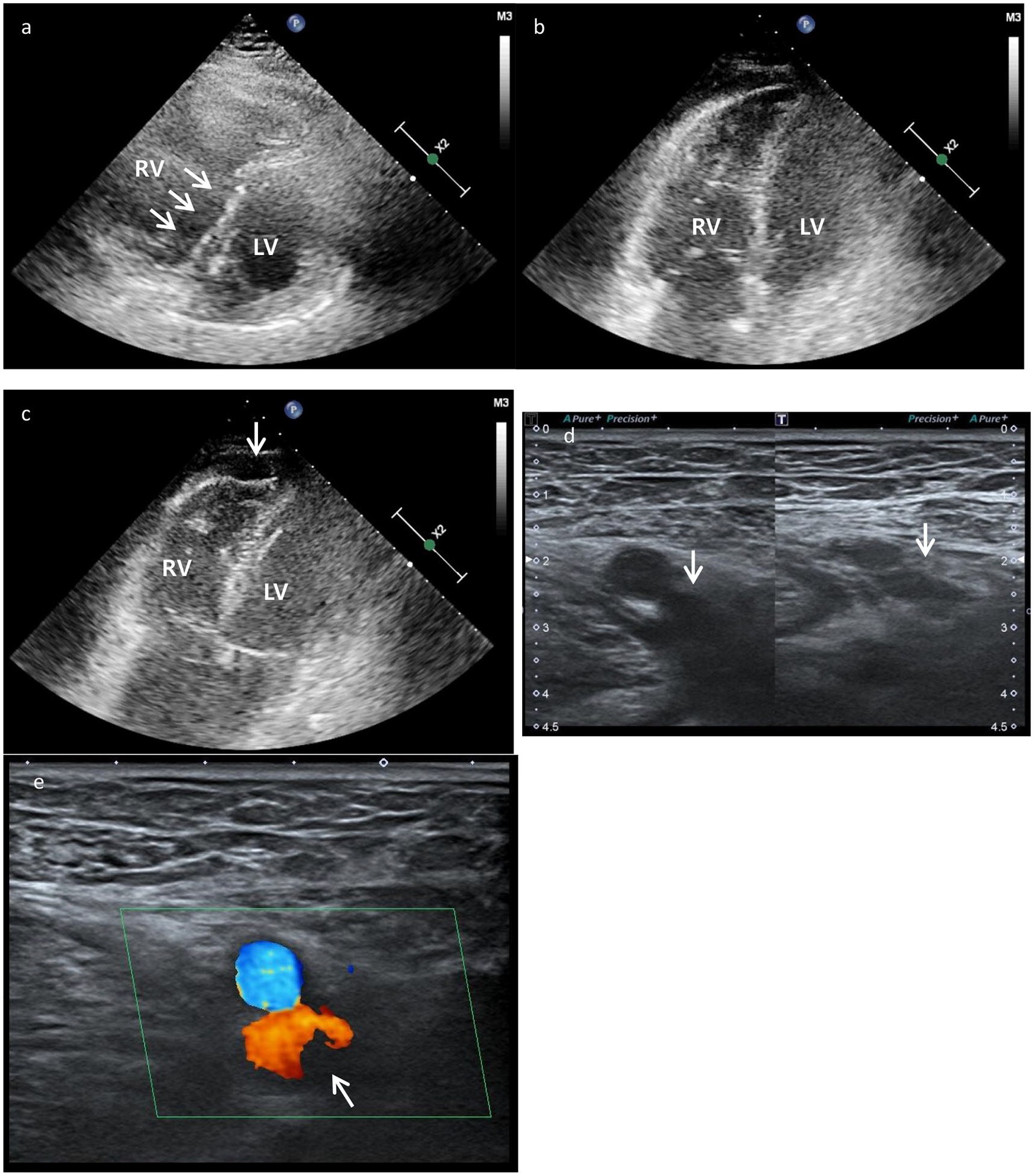
Point-of-care ultrasound (POCUS) findings after thrombolytic therapy and return of spontaneous circulation (ROSC) in the second case. POCUS imaging showing (a, b) right ventricle (RV) enlargement with a flattened left ventricle (arrow); (c) acute RV dysfunction—akinesia of the mid-free wall and hypercontractility of the apical wall, known as McConnell’s sign (arrow); (d) non-compressed (left) versus compressed (right) images at the left common femoral vein (arrow); and (e) non-compressible venous segment with suspicious focal Doppler signal voiding at the left common femoral vein (arrow), suggesting deep vein thrombosis (arrow).
Initial laboratory findings.
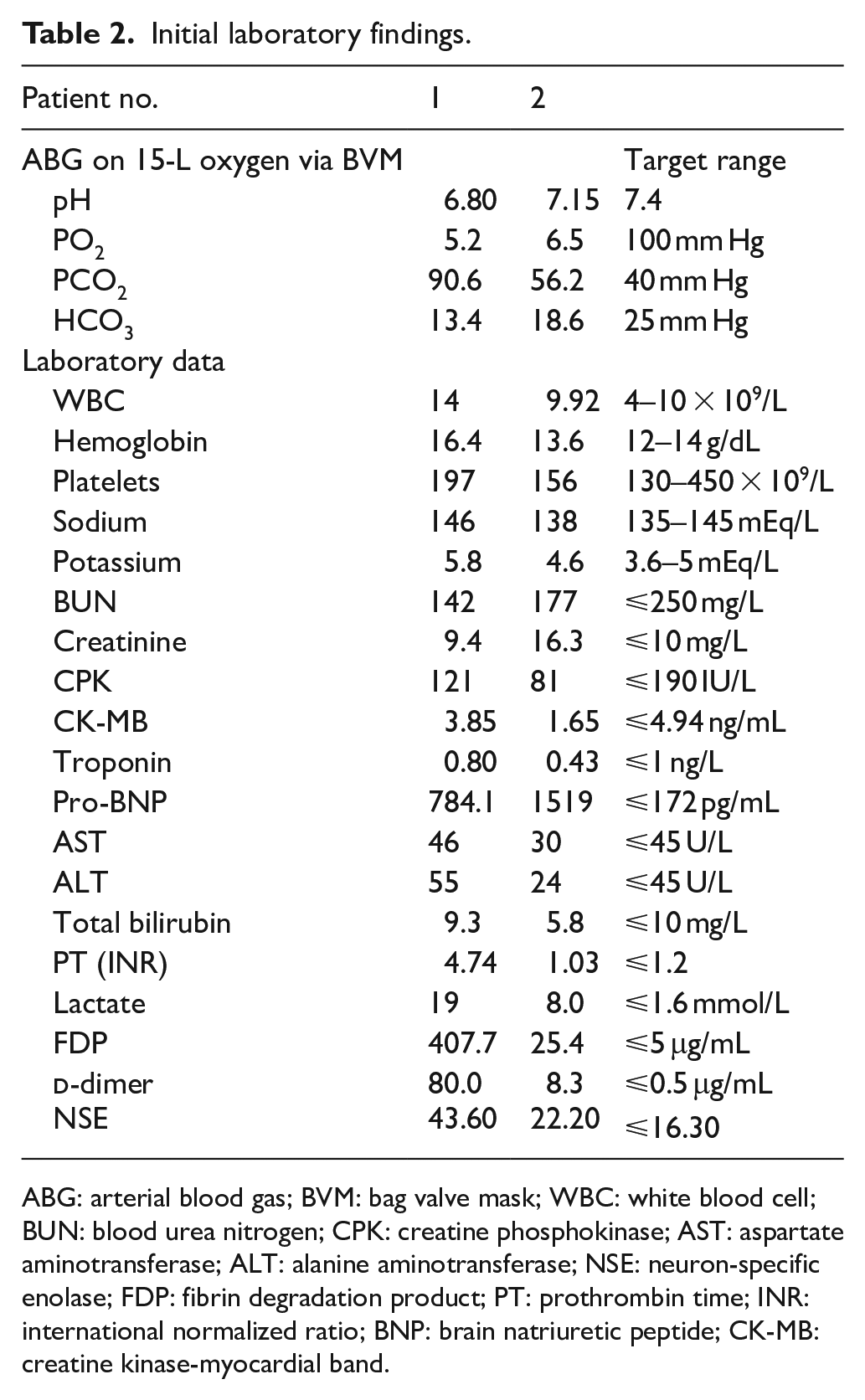
ABG: arterial blood gas; BVM: bag valve mask; WBC: white blood cell; BUN: blood urea nitrogen; CPK: creatine phosphokinase; AST: aspartate aminotransferase; ALT: alanine aminotransferase; NSE: neuron-specific enolase; FDP: fibrin degradation product; PT: prothrombin time; INR: international normalized ratio; BNP: brain natriuretic peptide; CK-MB: creatine kinase-myocardial band.
Case 2
A 66-year-old man presented to the ED with cardiac arrest. He had a history of diabetes mellitus as an underlying disease and he was undergoing rehabilitation due to cervical spine injury from a traffic accident 3 years before. Recently, he could barely walk. He had been taking analgesics for intermittent chest pain and back pain over the last 5 days. He suddenly felt chest discomfort and dyspnea before transfer to the ED. PEA arrest occurred during transport through EMS and BLS was performed for 6 min. On arrival at the ED, the patient was in a comatose state, his femoral pulse was not palpable, but electrocardiogram (EKG) monitoring showed PEA. ACLS was initiated with immediate intubation. ABG before endotracheal intubation showed a pH of 7.141, PaO2 of 6.5 mm Hg, PaCO2 of 56.2 mm Hg, and HCO3 of −18.6 mmol/L, despite 100% oxygen support using a bag valve mask. Along with conventional ACLS, immediate portable echocardiography revealed RV enlargement with a flattened left ventricle as in case 1. Moreover, the venous Doppler ultrasound was positive for deep venous thrombosis (DVT) in the left femoral veins (Figure 2). The cause of cardiac arrest was considered to be due to massive PE. Although conventional ACLS during 24 min, ROSC and PEA arrest were repeated. Thrombolysis was determined, and 50 mg of IV rtPA was administered bolus and sustained ROSC occurred after additional 3 min of ACLS. A
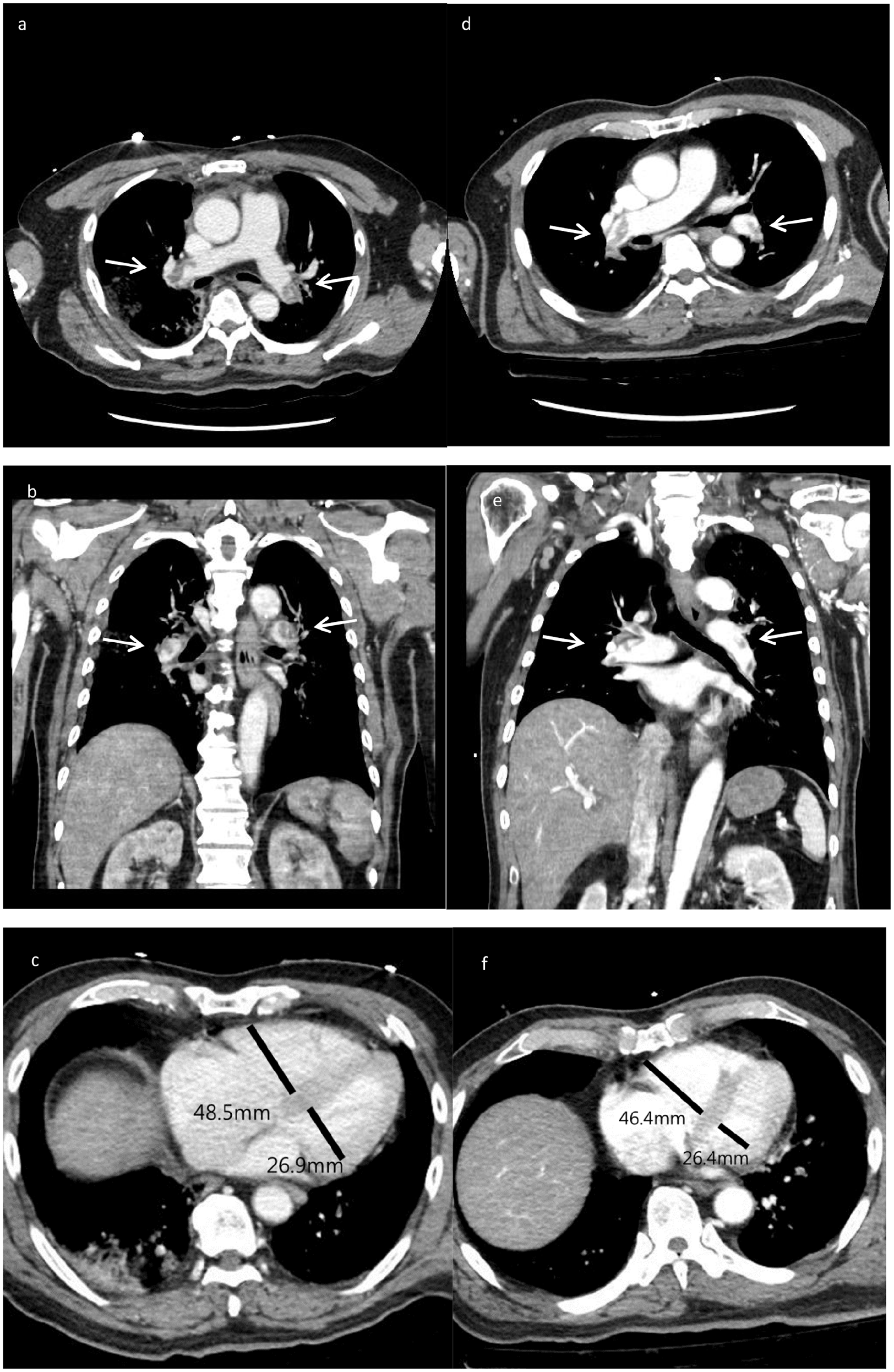
Computed tomography (CT) findings after thrombolytic therapy and return of spontaneous circulation (ROSC). CT images from case 1 showing (a, b) diffuse pulmonary thromboemboli (PTE; arrow) in both the main pulmonary arteries and (c) RV enlargement; CT images from case 2 showing (d, e) diffuse PTE (arrow) in both the main pulmonary arteries and (f) RV enlargement.
Discussion
Mortality rates due to cardiac arrest following massive PE range from 65% to 95%. 15 Because of persisting mechanical obstruction by PE, routine CPR is often ineffective. In these cases, thrombolytic therapy is associated with significantly higher ROSC (81% vs. 43%, P = 0.03). 2 Recent resuscitation guidelines advocated using rtPA in PE-related cardiac arrest.16,17,18 However, the authors of the Thrombolysis in Cardiac Arrest (TROICA) study (only randomized controlled trial that compared fibrinolytic therapy to placebo in patients with cardiac arrest of all causes) did not recommend thrombolytic therapy as a routine procedure in unselected patients with OHCA. 19 In this study, there was no benefit (P > 0.05) in clinical outcomes on comparison with the placebo group. However, the incidence of intracranial hemorrhage was significantly higher in the intervention group. 19 Considering the poor prognosis of patients with cardiac arrest, the possible benefits of thrombolysis outweigh the potential risks. 20 Therefore, during CPR, clinicians need to select patients in whom PE is highly suspected if considering thrombolysis.
Massive PE causes a sudden increase in pulmonary vascular resistance (PVR) and the mean pulmonary arterial pressure (mPAP), which is proportional to the degree of obstruction in patients without preexisting pulmonary vascular disease. 21 This leads to increased RV wall tension and RV failure. 22 Because of this pathophysiological nature, echocardiographical evidence of an enlarged RV with a flattened interventricular septum supports the diagnosis of PE, not only in hemodynamically unstable patients but also in cardiac arrest victims.23,24 Although echocardiography has limited sensitivity and specificity for the diagnosis of acute PE, bedside POCUS including cardiac scan and lower extremities scan can be helpful in identifying acute PE-related RV dysfunction and deep vein thrombosis during CPR.23,25
The various point of care ultrasound (POCUS) protocols that have been described to assess cardiac arrest patients in the ED also include a search for the presence of deep vein thrombosis and PE26–30. ED-based limited compression ultrasonography for the detection of the presence of lower limb DVT has been associated with a diagnostic sensitivity and specificity of 90% and 95%, respectively, when conducted by trained ED clinicians.31,32 In our cases, neither patients had a unilateral swollen leg; however, the second patient had left-sided femoral vein thrombus in the POCUS examination performed after ROSC.
In a recent study by Er et al., 33 earlier initiated thrombolysis after cardiac arrest onset (13.6 vs. 24.6 min) was associated with a higher rate of eventual ROSC for cardiac arrests caused by PE in in-hospital patients. This study suggests early administration of rescue thrombolysis in patients with cardiac arrest–associated PE because of highly significant association between the delay of rtPA administration and mortality. 33 However, in our cases, thrombolysis was somewhat delayed as the victims were OHCA patients and clinicians avoid using early thrombolysis because of the relative inexperience of junior emergency physicians. In contrast, in the second case, thrombolysis was applied earlier due to the early POCUS application and previous experience of the clinician.
The thrombolytic agent application time could be reduced with early application of POCUS during resuscitation of cardiac arrest victims. One recent study showed that the average time from the beginning of CPR to obtaining a suitable echo image was 3.9 min (15 s–10 min). 34
Thrombolytic therapy has been shown to improve pulmonary artery pressure, arteriovenous oxygenation, and pulmonary perfusion, thereby relieving symptoms, preventing recurrent PE, and reducing mortality in fulminant PE patients.35,36 However, in clinical practice, systemic thrombolytic therapy is less frequently used for a variety of reasons. These reasons include (a) limited evidence, (b) lack of specific guidelines about dosing and timing, and (c) risk of bleeding with high-dose thrombolytics. 37
The treatment dose of IV rtPA in non–cardiac arrest patients is largely agreed upon to be 100 mg over 2 h.38,39 However, during CPR, there is not enough time and infusion of rtPA over 2 h is impractical; therefore, a bolus has been proposed.40–42 Representative examples of emergency thrombolysis dosing regimens for fulminant PE include rtPA 50 mg via IV bolus with an option of a repeat bolus after 15 min, or single-dose weight-based tenecteplase.40–42 In our study, a 50-mg bolus injection of rtPA with an option for a repeat bolus 15 min later was applied and no complications of massive bleeding occurred.
Other therapeutic options include surgical embolectomy and percutaneous mechanical thrombectomy, especially when thrombolysis is contraindicated, in patients with a history of recent major trauma or surgery.16,43 In a small study by Fava et al., 44 six of the seven patients who had underwent percutaneous pulmonary thrombectomy during CPR survived, but the included patients of this study had experienced in-hospital cardiac arrest and three of the seven patients had already been diagnosed before cardiac arrest. To date, the evidence for surgical embolectomy and percutaneous mechanical thrombectomy during CPR is weak. 16 The European Resuscitation Council (ERC) recommends that consideration should be given to surgical embolectomy or mechanical thrombectomy when PE is the known cause of cardiac arrest; however, it does not recommend these interventions when PE is only suspected. 43
The use of extracorporeal life support (ECLS) techniques in specialized facilities has also been associated with treatment success in patients with cardiac arrest following PE45–47. ECLS provides an opportunity to restore systemic perfusion and oxygenation rapidly and, if necessary, may be used in combination with other therapies. However, in our relatively small hospital, extracorporeal cardiopulmonary resuscitation (E-CPR) is not always available; another limitation is that there are few doctors experienced in implementing extracorporeal membrane oxygenation (ECMO) during CPR.
Both patients had something in common about EMS-witnessed OHCA, PEA in the first monitored rhythm and relatively short prehospital BLS time, even though the prehospital BLS time of the first patient was a little longer and the blood test showed a difference. Despite all these common features, the prognoses of the two patients were vastly different for many reasons. The differences in the time to POCUS after visiting the ED, the timing of thrombolytic agent administration based on POCUS, and the experience of the physician in terms of the use of thrombolytic agents during resuscitation are thought to be the major reasons. Earlier application of POCUS and thrombolytic agents will improve patient prognosis.
In conclusion, if acute PE is suspected in patients with OHCA, we recommend prompt POCUS examination. POCUS may help identify patients with PE during CPR, leading to immediate treatment, although the clinical outcomes may vary.
Footnotes
Author contributions
All authors made a substantial contribution to the concept or design of the work, or acquisition, analysis, or interpretation of data; drafted the article or revised it critically for important intellectual content; approved the version to be published; and participated sufficiently in the work to take public responsibility for appropriate portions of the content. The manuscript has been read and approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the institutional review board (IRB) committee of Chungbuk National University Hospital (CBNUH).
Informed consent
Patient 1 and the family of patient 2 provided consent for publication of the medical course described in this report.
Human rights
This work was conducted according to the Declaration of Helsinki.
Prior publication
This manuscript has not been published or presented elsewhere in part or in entirety and is not under consideration by another journal.
Availability of data
The data presented in this case report are available from the corresponding author.
